.
American Weasels, E. Raymond Hall
University of Kansas Publications
Museum of Natural History
Vol. 4, pp. 1-466, plates 1-41, 31 figures in text
December 27, 1951
AMERICAN WEASELS
BY
E. RAYMOND HALL
University of Kansas
Lawrence
1951
University of Kansas Publications, Museum of Natural History
Editors: E. Raymond Hall, Chairman, A. Byron Leonard, Edward H. Taylor, Robert W. Wilson
Vol. 4, pp. 1-466, plates 1-41, 31 figures in text
December 27, 1951
University of Kansas
Lawrence, Kansas
PRINTED BY
FERD VOILAND, JR., STATE PRINTER
TOPEKA, KANSAS
1951
23-3758

Coloration of head and foreparts in ten subspecies of long-tailed weasel, Mustela frenata. All figures are of males, approximately × 1/2.
In regions of heavy rainfall (see figs. 2 and 3) there is an increase in pigmentation and extent of blackish color backward over the neck and a decrease in extent of the white facial markings. In regions progressively more arid (see figs. 3 to 7) there is a decrease in pigmentation and extent of blackish color and an increase in extent of the white facial markings.
As shown by rearing mammals from humid regions in arid regions, and vice versa, the color is not visibly altered in one or a few generations; the color is an hereditary character. Beginning with the southernmost subspecies (fig. 1) and continuing northward to the northern subspecies (fig. 10) there is a darkening, next a lightening, and finally a darkening closely conforming to amounts of precipitation in the geographic regions concerned. A fuller discussion of this correlation is given on page 51.

Fig. 1. Map showing localities of capture of specimens depicted in plate 1.
American Weasels
BY
E. RAYMOND HALL
CONTENTS
| PAGE | |
| Introduction | 7 |
| Paleontological History | 10 |
| Skeleton and Dentition | 12 |
| Disparity in Numbers of Males and Females | 19 |
| Materials, Acknowledgments and Methods | 21 |
| Variation | 24 |
| Variation with Age | 24 |
| Secondary Sexual Variation | 26 |
| Individual Variation | 28 |
| Seasonal Variation | 30 |
| Variation in Coloration and Molt | 30 |
| Variations of Taxonomic Worth | 44 |
| Distribution and Speciation | 54 |
| History of Classification | 69 |
| Chronological List (annotated) of Specific and Subspecific Names Applied to American Weasels | 71 |
| Check-List of American Species and Subspecies of the Genus Mustela | 81 |
| Artificial Key to American Species of the Genus Mustela | 83 |
| Diagnosis of the Genus | 83 |
| Explanation of Systematic Treatment | 84 |
| Systematic Accounts of Species and Subspecies | 87 |
| Mustela erminea | 87 |
| Mustela rixosa | 168 |
| Mustela frenata | 193 |
| Mustela africana | 406 |
| Explanation of Cranial Measurements | 417 |
| Table of Cranial Measurements | 418 |
| Literature Cited | 442 |
| Index | 461 |
American Weasels
By E. Raymond Hall
INTRODUCTION
The weasel's agility and speed take it in and out of retreats, over obstacles and across open places in amazingly rapid fashion and are responsible for the animal's actions being described as "quick as a flash." The common long-tailed weasel of the United States measures approximately a foot and a half in length, of which the tail comprises a third; but the round, slender body is scarcely more than an inch and a half in diameter. Brown above and whitish below in summer dress, the animal is sleek as well as lithe and graceful. It is easy to understand, therefore, why the Bavarian name Schönthierlein (pretty little creature) and the Italian name donnola (little lady) were bestowed upon it. The Spanish name is comadreja (godmother).
In the winter, in temperate and northern regions, the coat becomes pure white except for the black tail-tip. In this dress the correct name for the animal is ermine, a mammal whose fur is known to all and justly esteemed, especially for its luster in artificial light, where it is scarcely excelled in enhancing the beauty of gems and their feminine wearers.
In relation to its weight, the weasel is thought to be unsurpassed, and perhaps it is unequalled among mammals, in the effectiveness with which it exercises its carnivorous heritage; it kills with speed and strength a wide variety of animals including many much larger than itself; and it has been known to attack even man himself when he stood between the weasel and its intended prey. In structure and temperament it is so highly specialized for offense that, when opportunity affords, it sometimes kills, for storage in its larder, far more than enough to meet its immediate needs. After speaking of this tendency, Elliott Coues (1877:129) has said:
"A glance at the physiognomy of the weasels would suffice to betray their character. The teeth are almost of the highest known raptorial character; the jaws are worked by enormous masses of muscles covering all the side of the skull. The forehead is low and the nose is sharp; the eyes are small, penetrating, cunning, and glitter with an angry green light. There is something peculiar, moreover, in the way that this fierce face surmounts a body extraordinarily wiry, lithe, and muscular. It ends in a remarkable long and slender neck in such a way that it may be held at right angle with the axis of the latter. When the creature is glancing around, with the neck stretched up, and flat triangular head bent forward, swaying from one side to the other, we catch the likeness in a moment—it is the image of a serpent." Although Coues' colorful description more closely links the weasel with the symbol of evil than pleases me, his description does emphasize the raptorial character of the weasel.
Even though most weasels are intractable as pets, they have a value to man, as, for instance, when he is plagued by mice. In a field where mice and other small rodents are so abundant as to damage cultivated crops, the weasel is the farmer's best friend. A weasel may inhabit one den until the rodents thereabouts are almost exterminated in an area two or three hundred yards across; in this way the weasel acts as a control, locally, as well as a check more widely, on the increase in size of populations of kinds of rodents upon which it preys. The smaller species are mousers of remarkable efficiency and can, if necessary, follow a mouse to the end of the mouse's burrow. The slender body allows the weasel to pass through any burrow or hole into which it can thrust its head. This ability in an organism as highly specialized for killing other animals as is the weasel, has earned for it a bad name in connection with poultry yards. Authentic instances are recorded in which a weasel, gaining entrance through a knot-hole to a coop of young chickens, killed several dozen of the fowls. In other instances, however, weasels have lived under buildings close by a poultry yard without even molesting the birds in the slightest; in the latter instances the weasels probably were present because there was an abundant supply of rats and mice. At least three poultry raisers (see page 214) have encouraged weasels to live in their poultry yards feeling that the good they do by destroying rats outweighs the damage caused by the occasional weasel which turns to the fowls; the idea is that the individual weasel can be eliminated if he becomes destructive.
Although tending to be nocturnal, weasels are almost as active by day as by night. Their young, numbering 4 to 9, are born in a nest in a burrow and as with other members of the Order Carnivora, are blind, and incapable of looking after themselves at the time of birth. In Mustela frenata of Montana, breeding occurs in July and August, and the young are born in the following April and May. Wright (1948A:342) showed that the gestation period could not have been less than 337 days in one individual and that it averaged 279 (205-337) days in 18 instances. Findings of the same author (1942B:109) showed that the embryos are implanted only 21 to 28 days before the young are born. In the preceding part of the "long gestation period, the embryos lie dormant in the uterus as un-implanted blastocysts. The young female weasel [of M. frenata] mates when 3 or 4 months old." Consequently, in the spring, all females of this species may produce young (Wright, 1942A:348). The circumboreal species Mustela erminea likewise has been shown to have a delayed implantation of the ova. Each of these two species, M. frenata and M. erminea, has only one litter per year; but the weasel, Mustela nivalis, of the Old World seems to lack the delayed implantation, in this respect resembling the ferret (subgenus Putorius) as it does also in its ability to have more than one litter per year (see Deanesly, 1944). The manner of reproduction in the South American species M. africana and the circumboreal species M. rixosa at this writing is unknown.
The genus Mustela includes the true weasels, the ferrets and minks. The ferrets commonly are treated as a subgenus, Putorius, along with the Old World polecat. The minks usually are accorded subgeneric distinction under the name Lutreola, and the true weasels comprise the subgenus Mustela, the three subgenera together, along with some other subgenera which are mostly monotypic, comprising the genus Mustela. Considered in this way, the group of true weasels, subgenus Mustela, has a geographic range roughly coextensive with that of the genus Mustela. This range includes Asia and Europe, Northern Africa, North America and northern South America. Java has its weasel. Australia and nearly all the oceanic islands lack weasels, and the animals are absent from roughly the southern half of Africa and the southern half of South America. Other small mustelids, weasellike in shape and with corresponding habits and dentition, take the place of true Mustela in the southern half of Africa and in the corresponding part of South America.
In America the subgenus Mustela occurs from the northernmost land in Arctic America southward to Lake Titicaca in the Andes of South America, a distance of approximately 6900 miles. Felis, I think, is the only other genus of land mammals in the western hemisphere that has a geographic range as extensive from north to south. Felis does not range so far north but does range farther south. The one species, Mustela frenata, ranges from Lake Titicaca northward to about 57° N in British Columbia or for approximately 5000 miles in a north to south direction and from within the Alpine Arctic Life-zone through the Tropical Life-zone. In North America, weasels occur in almost every type of habitat, being absent only in the extremely desert terrain of western Arizona and western Sonora and in adjoining parts of California and Baja California. Even this area, along the Colorado River, may support some weasels; evidence suggesting that it does so is given in the account of Mustela frenata neomexicana.
PALEONTOLOGICAL HISTORY
The paleontological record fails to show the precise ancestry of Mustela. The genus has been found in deposits of Pleistocene age, but, so far as I can ascertain, not in deposits of earlier times. The Pleistocene remains are not specifically distinct from Recent (living) species, and in only a few instances (see M. f. latirostra and M. e. angustidens) are they even subspecifically distinct from the Recent weasel living in the same area today. It is true that fossil remains from deposits of several stages of the Tertiary beds have in the past been identified in the literature as Mustela, but most of these identifications were made many years ago when the generic name Mustela was used in a far broader and more inclusive sense than it is today and much of the fossil material was so fragmentary that the generic identity could not be ascertained, at least at that time. Because the generic identity could not be ascertained, the fossil material was tentatively assigned to the genus Mustela, the "typical" genus of the family Mustelidae instead of to some other more specialized or less well-known genus of the family. To satisfy my curiosity about these species of "Mustela" of a geological age earlier than the Pleistocene I have personally studied nearly all of the original specimens from North America and have found each to be of some genus other than Mustela. Also, such study as I have been able to make of the Old World fossils themselves that have been referred to the genus Mustela up to 1938, and my study of the illustrations and descriptions of the others from there lead to the same conclusion; that is to say, none that is true Mustela is known up to now from deposits older than the Pleistocene.
When, in 1930 (pp. 146-147), I wrote about the taxonomic position of three American genera of fossils (known only from lower jaws), each of which had been previously referred to the genus Mustela, I said that they pertained "to that section of the weasel family (Mustelidae) which comprises the polecats, true weasels, ferrets, minks and martens. The fossil specimens . . . are smaller than any other later Tertiary members of the group yet described, and are more primitive than any of the above mentioned Recent relatives. Of the three extinct genera . . . Miomustela [Lower Pliocene or Upper Miocene of the Lower Madison Valley, Montana] is the most primitive and Martinogale [Pliocene, 18 mi. SE Goodland, Sherman County, Kansas] is the most advanced. This view rests largely on the character of M=1 which in Miomustela has a deeply basined, short, narrow talonid with a thick, high metaconid situated partly posterior to the protoconid. In Martinogale the talonid is incipiently trenchant, long, broad, and it has a lesser developed metaconid which is situated more anterior [ly]. Pliogale [Lower Pliocene, Humboldt County, Nevada] is intermediate in this respect.
"These three forms are of special interest as possible ancestors of the subgenus Mustela, true weasels. No members of this subgenus, nor related forms which can with any degree of certainty be regarded as directly ancestral to them, have yet been described from Miocene or Pliocene deposits. Palaeogale of the Old World and Bunaelurus of North America, each of Oligocene age, have been placed by Schlosser (1888, p. 116) and Matthew (1902, p. 137) as members of the primitive group of mustelids ancestral to Mustela. This course seems logical; and with no truly intermediate links between these forms of the Oligocene on the one hand, and Mustela which first appears in the Pleistocene, on the other, more definite statements about ancestral positions of the small Oligocene forms can hardly be made. The deciding considerations for authors who placed Palaeogale and Bunaelurus as ancestral to Mustela were the absence of a metaconid on M1 and the trenchant talonid of that tooth. These characters are found also in Mustela. On the other hand certain structures in the basicranial region of Palaeogale and more especially of Bunaelurus indicate that these genera possibly are not close to the ancestral form of Mustela . . . Martinogale may stand near the ancestral form of Mustela and . . . Pliogale may be ancestral to Martinogale. Pliogale, in turn, may have had an ancestor similar to Miomustela. If this should prove to be the case, Palaeogale and Bunaelurus might be regarded as an independent branch which displays merely a parallelism to Mustela in the loss of the metaconid on M1 and the development of a trenchant talonid on that tooth. The writer would make it clear that he does not hold such to be the case. The ancestral relation of Martinogale to Mustela is presented merely to show the possibility, and not the special probability, of such an origin for Mustela. Knowledge of the tympanic bullae and other structures of the basicranial region would go far toward answering the question and until these structures are known [in mustelids of the Later Tertiary,] some uncertainty will remain."
At the present writing I can add to the above statement only a few facts. The discovery of better material of Bunaelurus than was available to previous workers led Simpson (1946), correctly I think, to synonymize Bunaelurus with Palaeogale. Simpson figures the cranial foramina in Palaeogale. The differences, between Palaeogale and Mustela, in cranial foramina, possibly are only the result of the elongation of the tympanic bullae. The bullae of the subgenus Mustela are seen to be much elongated posteriorly if comparison is made with the bullae of earlier mustelids. Consequently, it might be concluded that there is nothing in the arrangement of the cranial foramina which would preclude the derivation of Mustela from Palaeogale. However, the anterior situation of the carotid foramen—well forward along the medial margin of the tympanic bulla—is a character typical of other mustelids and the posterior location of this foramen in Palaeogale might indicate that it was not ancestral to Mustela.
SKELETON AND DENTITION
The outstanding features of a weasel's skeleton are its length and slenderness. Whereas the length of the vertebral column measured from the atlas (the first cervical vertebra) to the last sacral vertebra is 175 per cent of the length of the hind leg (as measured from the head of the femur to the tip of the longest claw), the corresponding percentage is only 116 in the raccoon. Stated in another way, the vertebral column and the hind leg are of approximately equal length in a raccoon, but in a weasel the vertebral column is one and three-fourths times as long as the hind leg.
VERTEBRAE
The vertebral column consists of 7 cervicals, and ordinarily 14 thoracics, 6 lumbars, 3 sacrals and, depending on the species, 11 to 23 caudals. For the three species of which skeletons were examined, variations from the normal number of vertebrae are noted in the following table:
Table 1
Data on vertebrae in three species of the subgenus Mustela
(Numerals in parentheses indicate number of specimens)
| Mustela erminea | Mustela rixosa | Mustela frenata | |
| Number of cervical vertebrae | 7(75) | 7(12) | 7(65) |
| Number of thoracic vertebrae | 14(71) | 14(12) | 14(54) |
| 15(4) | 15(13) | ||
| The dorsal vertebraconstituting the anticlinal | 11th(18) | 11th(12) | 11th(40) |
| 12th(7) | 12th(27) | ||
| Number of lumbar vertebrae | 5(2) | 5(11) | |
| 6(73) | 6(12) | 6(54) | |
| Number of sacral vertebrae | 2(9) | 2(3) | |
| 3(65) | 3(10) | 3(67) | |
| 4(1) | 4(2) | ||
| Number of pseudosacral vertebrae | 0(73) | 0(12) | 0(57) |
| 1(2) | 1(6) | ||
| 11(1) | |||
| 14(3) | |||
| 15(2) | 15(7) | ||
| 16(3) | 16(1) | ||
| 17(9) | |||
| Number of caudal vertebrae | 18(28) | ||
| 19(11) | 19(6) | ||
| 20(14) | |||
| 21(14) | |||
| 22(7) | |||
| 23(1) |
Variation according to the species is evident in the number of caudal vertebrae, but in the other categories of vertebrae no consistent difference in number according to species was found in the material examined. Apparently there is also some geographic variation in the number of caudal vertebrae within a species. For example, the one skeleton seen of Mustela rixosa eskimo (no. 219036, U. S. Nat. Mus., from St. Michaels, Alaska) has only 11 caudal vertebrae, whereas in the 11 Mustela rixosa rixosa from Roseau County, Minnesota, the usual number is 15 with extremes of 14 and 16. Similarly specimens of Mustela frenata from Idaho and California almost always have 1 or 2 more caudal vertebrae than do individuals of the shorter-tailed subspecies of the same species from eastern Kansas.
Of the vertebrae, only the cervicals, of which there are 7, were found to be constant in number. In M. erminea, two of the seven individuals in which the anticlinal vertebra was the 12th (instead of the 11th) had 15 instead of the customary 14 thoracic vertebrae. In M. frenata, seven of the twenty-seven individuals in which the anticlinal vertebra was the 12th (instead of the 11th) had 15 instead of 14 thoracic vertebrae. The one M. erminea with a pseudosacral vertebra had only two instead of the customary 3 sacral vertebrae but the same individual had 15 thoracic vertebrae. Of the six M. frenata with a pseudosacral vertebra, two animals had only two instead of three sacral vertebrae. Conceivably, therefore, the pseudosacral vertebra in each of the three instances mentioned may represent merely an unfused sacral vertebra, instead of a true pseudosacral as occurs in four individuals of M. frenata.
TEETH
In American weasels, for example in Mustela frenata, the permanent dentition normally is
I 3 C 1 P 3 M 1 -, -, -, -, -, -, -, - i 3 c 1 p 3 m 2
or 34 teeth in all. In most respects the dentition is typical for post-Tertiary mustelids but in several parts is highly specialized for a diet of flesh, the degree of this specialization being second only to that of the cats, family Felidae. The outstanding specialization is in the first lower molar, in which, as in the cats, the internal cusp (metaconid) is completely suppressed and the heel (talonid) forms an elevated blade for cutting food rather than a basin for crushing it. In one sense the tooth is simplified since it owes its distinctive form to a reduction in number of parts; nevertheless, the distinctive form of the lower molar clearly is correlated with a diet of flesh, and the tooth is correctly to be thought of as the lower blade of a pair of shears; the upper blade is the fourth upper premolar. The reduction in size of the second (last) lower molar and small size of the inner lobe of the one remaining upper molar probably are additional modifications for a diet of flesh.
The absence of the last two upper molars and last molar in the lower jaw would be expected in any mammal as highly specialized for a diet of flesh as is the weasel, but these teeth are absent also in other Quaternary members of the family Mustelidae, many of which are substantially less specialized for a diet of flesh than is the weasel. Therefore, in the weasel, it is reasonable to regard the absence of these teeth more as a heritage than as an indication of a special adaptation. The absence of a first premolar above and below, as in the weasel, is to be expected in any carnivore that has the first lower molar and fourth upper premolar highly specialized for shearing, but the loss of these premolars and the small size of the second premolars may be as much the result of a slight shortening of the face as it is a result of a lengthening of the third and especially the fourth premolars. The lengthening of these more posteriorly-situated teeth would appear to be an adaptation to a diet of flesh. The cause of the lengthening of the mentioned teeth and the reason for the absence of the first premolars probably will be unknown until the fossil record is more complete.
The teeth of American species vary little except in size. The absence of P2 in Mustela africana is the only difference of a qualitative (presence or absence) nature that was detected. Also, the Central American subspecies of Mustela frenata exhibit a tendency to early loss of P2 and thus foreshadow the condition typical of M. africana.
As a whole the dentition of the weasel exhibits a high degree of specialization for a diet of flesh and this specialization is fully as evident in the deciduous dentition as in the permanent dentition.
The deciduous, or milk, dentition, of Mustela frenata, as known from immature specimens of Mustela frenata noveboracensis and Mustela frenata frenata available for this study, is comprised of canines, one on each side above and below, and 3 cheek teeth on each side above and below. See figures 2-9. The upper cheek teeth from anterior to posterior are: a minute peglike tooth in general similar to the first premolar of the permanent dentition; a shearing tooth in general similar to P4 of the permanent dentition; and an anteroposteriorly compressed tooth in general similar to M1 of the permanent dentition. In the lower jaw, behind the canine, there is first a minute peglike tooth, second a two-rooted tooth similar in general outline to a permanent third premolar, and finally a shearing tooth corresponding in function to m1 of the permanent dentition.
No postnatal specimens which show deciduous incisors have been examined.
Selected, outstanding differences between the permanent teeth and the deciduous teeth are as follows: In the deciduous teeth the canine above has on the posterior face a well-defined ridge extending from the tip to the cingulum. This ridge is absent or at most faintly indicated in the permanent tooth. The lower deciduous canine, in cross section is seen to have a marked indentation on the anteromedial border in the region of the cingulum; this indentation is lacking in the permanent tooth. The anterior one of the deciduous cheek teeth, both above and below, is single rooted and its crown-surface is only about one-fifteenth as much as that of the anterior premolar of the permanent dentition. The second deciduous cheek tooth below has two roots, usually fused, and differs from p4 of the permanent dentition in having the tip of the principal cusp more recurved, in having the anterior basal cusp better developed and the posterior heel less well developed.
The second deciduous cheek tooth above corresponds in function and general plan of construction to P4 of the permanent dentition but differs from that tooth in the more pronounced protostyle, longer tritocone, more posteriorly located deuterocone and as noted by Leche (1915:322) separation of the protocone and tritocone by a notch. The third upper deciduous tooth has a single cusp internally and two cusps laterally. Thus it reverses the relation of parts seen in M1 where the internal moiety is larger than the lateral or buccal moiety. The third deciduous tooth below differs from m1 in very much shorter talonid and separation of the paraconid from the protoconid by a deeper notch.
All the features in which the last two deciduous teeth, both above and below, are described as differing from their functional counterparts in the permanent dentition, are features found in the permanent teeth of primitive fossil mustelids and certain fossil and Recent viverrids. Even so, taking into account Leche's (1915) work, which shows that the milk teeth of some carnivores have structures lacking in the corresponding permanent teeth of the same individual animal and also in the teeth of genera that seem to be ancestral, a person suspects that some of the structural features mentioned above are not inheritances of ancestral conditions but rather specializations of the milk dentition.
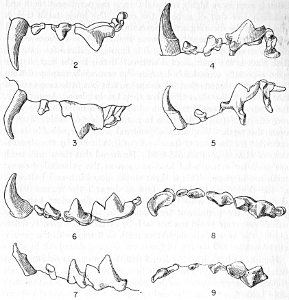
Figs. 2-9. Views of permanent and deciduous teeth of Mustela frenata nigriauris. Incisors not shown. In each instance teeth are of the left side.
Permanent dentition × 3. No. 32421, Mus. Vert. Zoöl., ♂, adult; Berkeley, Alameda County, California; obtained October 4, 1921, by D. D. McLean.
Deciduous dentition × 5. No. 132158, U. S. Nat. Mus., ♂, juvenile; Stanford University, Santa Clara County, California; obtained May 7, 1898, by W. K. Fisher.
Figs. 2-3. Lateral views of upper teeth, of adult and juvenile respectively.
Figs. 4-5. Occlusolingual views of upper teeth of adult and juvenile respectively.
Figs. 6-7. Lateral views of lower teeth of adult and juvenile respectively.
Figs. 8-9. Occlusolingual views of lower teeth of adult and juvenile respectively.
In other deciduous teeth there is clearer evidence of more specialization for a diet of flesh in the deciduous teeth than in the permanent teeth. For example, the upper carnassial of the milk dentition is even more highly sectorial than is the permanent tooth and strikingly like that of some of the cats. The lower tooth that is effective in the shearing action bears no more trace of the metaconid than does the permanent first lower molar. These features of the deciduous dentition suggest that it is more specialized for a diet of flesh than is the permanent dentition. If this be the fact, it may seem especially remarkable because the commonly employed term "milk teeth" suggests that the animal makes but little or no use of these teeth in the short time that they are in place. Accordingly, the student may credit the form of these teeth more to some indirect effects of inheritance than to natural selection acting directly upon the teeth. But, after all, natural selection probably is responsible for the form of these teeth as is indicated by the observations of Hamilton (1933:318-325). He found that these milk teeth are used for eating solid food as soon as the principal shearing teeth are in place. This is three weeks after birth and before all of the deciduous teeth have broken through the gums. These shearing teeth are used for almost two months before being replaced by the permanent teeth and it is, therefore, evident that natural selection could operate to fully as great a degree in determining the form of the deciduous teeth as it may with the permanent teeth.
Hamilton (1933:325-326) found that the permanent dentition was complete at 75 days after birth in captive specimens of Mustela frenata noveboracensis. In the same subspecies, he noted 28 days after birth that the canines and carnassial teeth [second deciduous cheek tooth above and third below] had erupted through the gums. Animals 45 days old, Hamilton found, were losing the milk dentition, and had the gums broken through by several of the permanent cheek teeth.
Study of the cleaned skulls available of juveniles indicates that the deciduous teeth which persist longest are, on each side of the mouth, the second cheek tooth above and the third cheek tooth below. These teeth persist until after the permanent P4 and m1 have come into use. These permanent teeth are situated immediately behind their functional counterparts of the milk dentition. P3 and p4 are the teeth of the permanent dentition which ultimately push out the last milk teeth to be lost. Accordingly, in the permanent dentition, P4 and M1 appear before P3 does, and m1 and m2 make their appearance before p4.
DISPARITY IN NUMBERS OF MALES AND FEMALES (IN ZOOLOGICAL COLLECTIONS)
The question has frequently been asked why twice as many male as female weasels are captured. This is the proportion in research collections, as may be seen from table no. 2, and I am convinced that the specimens in these collections are saved in approximately the same proportion as that in which they are caught. Although it might be assumed, upon first consideration, that there are twice as many males as females in nature, selective factors enter into the catch. For example, because a male weasel is approximately twice as heavy as a female, it may be necessary for him, in a given length of time, to travel twice as far as the female to obtain the required amount of food with the result that a given number of traps or snares will catch twice as many males as females. Indeed, Glover (1943B:8) shows that, on the average, in Mustela frenata noveboracensis in Pennsylvania, the male actually does travel slightly more than twice as far as the female (704 feet versus 346 feet). From table no. 2, it may be seen that in most winter months the ratio is 3 males to one female. This ratio is reasonable enough, in view of what has been said, if it is considered also that the lighter weight of the female permits her safely to step on the pans of traps that would be sprung by heavier males.
If in the breeding season, which is April through August in M. frenata, the female is passive and if the male is restlessly searching for her, he may thus increase still more his chances of being caught in traps set for weasels.
My own studies of live weasels in nature indicate that in the season when females are attending young which are half grown, or larger, the adult male weasels live singly in dens of their own, separate and apart from the females and their young (Hamilton, 1933:328, records adult males living with the female and her young, but possibly this was when the young were less than half grown). Perhaps these males at that time travel no farther than is necessary to obtain food for themselves. Females, at this time, forage not only to meet their own needs, but for food to supply their young as well. At this time, in May and June, as may be seen from table no. 2, almost as many adult females as adult males are caught. The reason why only relatively more females than in other months, instead of actually more females than males, are caught at this time probably is that the adult males also are extraordinarily active at this time because they are in breeding condition. Perhaps the explanation in part is to be found in the lesser weight of the female (approximately half of the male's weight) which, as indicated above, permits her to step on the pan of a steel trap without springing it whereas the heavier male does spring the trap and as a consequence is caught. Hamilton (1933:299-300), who mentions this selective factor, found an equal number of males and females in the three newly born litters that came under his observation.
Table 2
Specimens of Mustela frenata (north of the range of M. f. frenata) arranged by sex and under each sex by age
| Male | Female | total number of ♂ and ♀ | total number of adults, ♂ and ♀ | |||||||||||||
| adult ♂ | ♂ ad., % of total adults | subadult ♂ | young ♂ | juvenal ♂ | total number of ♂ | ♂, % of total | adult ♀ | ♀ ad., % of total adults | subadult ♀ | young ♀ | juvenal ♀ | total number of ♀ | ♀, % of total | |||
| May | 29 | 55 | 4 | 14 | 7 | 54 | 59 | 24 | 45 | 1 | 9 | 3 | 37 | 41 | 91 | 53 |
| June | 42 | 53 | 14 | 40 | 8 | 97 | 59 | 38 | 47 | 4 | 25 | 2 | 69 | 41 | 166 | 80 |
| July | 59 | 70 | 18 | 55 | 2 | 130 | 59 | 25 | 30 | 5 | 58 | 2 | 90 | 41 | 220 | 84 |
| August | 40 | 77 | 23 | 55 | .. | 113 | 74 | 12 | 23 | 2 | 25 | .. | 39 | 26 | 152 | 52 |
| September | 15 | 79 | 25 | 12 | 1 | 51 | 75 | 4 | 21 | 4 | 9 | .. | 17 | 25 | 68 | 19 |
| October | 11 | 58 | 46 | 7 | .. | 43 | 66 | 8 | 42 | 13 | 1 | .. | 22 | 34 | 65 | 19 |
| November | 41 | 70 | 48 | 1 | .. | 88 | 73 | 18 | 30 | 12 | 2 | 1 | 33 | 27 | 121 | 59 |
| December | 59 | 69 | 43 | 1 | .. | 108 | 73 | 26 | 31 | 15 | ... | .. | 41 | 27 | 149 | 85 |
| January | 80 | 69 | 32 | 2 | 1 | 126 | 72 | 36 | 31 | 14 | ... | .. | 50 | 28 | 176 | 116 |
| February | 45 | 66 | 19 | 5 | .. | 82 | 73 | 23 | 34 | 4 | 3 | .. | 30 | 27 | 112 | 68 |
| March | 38 | 72 | 2 | ... | .. | 57 | 70 | 15 | 28 | 8 | 1 | .. | 24 | 30 | 81 | 53 |
| April | 30 | 67 | 2 | 4 | 3 | 39 | 67 | 15 | 33 | .. | 2 | 2 | 19 | 33 | 58 | 45 |
| Totals | 489 | 67 | 281 | 196 | 22 | 988 | 68 | 244 | 33 | 82 | 135 | 10 | 471 | 32 | 1,459 | 733 |
I suppose that in nature there are approximately equal numbers of male and female weasels and further suppose that the selective factors which cause more males than females to be caught are the greater distances traveled by the males and their greater weight.
MATERIALS, ACKNOWLEDGMENTS AND METHODS
At a late stage in the preparation of this manuscript a total of 5,457 specimens had been examined. For the most part these were conventional study-specimens; that is to say, they were stuffed skins with the skulls separate and each was accompanied by the customary data as to locality of capture, date of capture, name of collector, external measurements and sex recorded on the labels by the collectors. Skulls unaccompanied by skins, nevertheless, comprised a large share of the total and a small proportion was made up of skins unaccompanied by skulls, mounted specimens, skeletons, and entire animals preserved in liquid.
It was the recognition of this need for specimens from extensive areas from which no specimens previously had been collected that influenced me, approximately a year after the study was begun, to allot for it a long span of time. The procedure adopted, in general, was to study the weasels of one species from a given geographic area in so far as the material warranted, then lay this aside until additional critical material could be obtained, and finally, some months or a year later, complete the account. In this fashion the manuscript of the American weasels received my attention in each of the past twenty-five years (September, 1926 to date of publication). This is a confession of fact rather than a recommendation of procedure. This type of procedure unduly delays the diffusion of knowledge and for a variety of reasons justifiably annoys other students of the subject. Nevertheless, many gaps have been filled that otherwise would have remained open. Although specimens to solve several problems still remain to be collected and studied, it seems that a point of diminishing returns has now been reached, which, in fairness to all concerned, calls for publication of the results so far obtained.
For assistance in the entire undertaking, I am more indebted to Miss Annie M. Alexander than to any other one person; she provided the means by which specimens from critical areas were obtained, made it possible to examine the European collections, and assisted in other ways. The late Professor Joseph Grinnell and Mr. Charles D. Bunker, among others, gave truly valuable encouragement and assistance.
Collections containing weasels which were examined in the study here reported upon were as follows:
- Acad. Nat. Sciences of Philadelphia
- American Mus. Nat. History
- Baylor University
- Berlin Zoological Museum
- Boston Society of Natural History
- Brigham Young University
- British Museum of Natural History
- California Academy of Sciences
- Carnegie Museum
- Charleston Museum
- Coe College
- Collection of J. Arnold
- Collection of Stanley C. Arthur
- Collection of Rollin H. Baker
- Collection of William Bebb
- Collection of R. H. Coleman
- Collection of Ian McTaggart-Cowan
- Collection of Stuart Criddle
- Collection of John Cushing
- Collection of Walter W. Dalquest
- Collection of William B. Davis
- Collection of J. M. Edson
- Collection of Ralph Ellis
- Collection of John Fitzgerald, Jr.
- Collection of Mr. Green
- Collection of Ross Hardy
- Collection of Donald V. Hemphill
- Collection of L. M. Huey
- Collection of R. W. Jackson
- Collection of Stanley G. Jewett
- Collection of E. J. Koestner
- Collection of J. E. Law
- Collection of A. H. Miller
- Collection of Lloye H. Miller
- Collection of R. D. Moore
- Collection of J. A. Munro
- Collection of O. J. Murie
- Collection of Robert T. Orr
- Collection of Arthur Peake
- Collection of Kenneth Racey
- Collection of William B. Richardson
- Collection Rocky Mt. Spotted Fever Lab.
- Collection of Victor B. Scheffer
- Collection of William T. Shaw
- Collection of O. P. Silliman
- Collection of W. E. Snyder
- Collection of Frank Stephens
- Collection of T. C. Stephens
- Collection of D. D. Stone
- Collection of Myron H. Swenk
- Collection of Joe and Dean Thiriot
- Collection of John Tyler
- Collection of Jack C vonBloeker
- Collection of Alex Walker
- Collection of Edward R. Warren
- Colorado Museum of Natural History
- Charles R. Conner Museum
- Cornell University
- Donald R. Dickey Collection
- Field Museum of Natural History
- Florida State Museum
- Fresno State Junior College
- Humboldt State Teachers College
- Illinois Natural History Survey
- Iowa State College
- Iowa Wesleyan College
- Kansas State Agric. College
- Leland Stanford Junior University
- Leningrad Academy of Science
- Los Angeles Mus. Hist. Art and Sci.
- Louisiana State University
- Mt. Rainier Nat'l Park Collection
- Museum of Comparative Zoölogy
- Mus. Polonais d'Hist. Nat., Warsaw
- Mus. Vert. Zoöl., Univ. California
- Museum of Zoölogy, Univ. Michigan
- National Museum of Canada
- Naturhistoriska Ricksmuseum, Sweden
- Neuchatel University Museum
- New York State Museum
- Ohio State Museum
- Oklahoma Agric. and Mech. College
- Ottawa University, Kansas
- Paris Museum
- Provincial Museum of British Columbia
- Royal Ontario Museum of Zoölogy
- San Diego Society of Natural History
- State Hist. and Nat. Hist. Soc. Colo.
- State Normal School, Cheney, Wash.
- Texas Cooperative Research Collection
- United States National Museum
- University of Arkansas
- Univ. California Mus. Palaeo.
- University of Idaho
- Univ. Kansas Mus. Nat. History
- University of Minnesota
- University of Notre Dame
- University of Oklahoma
- University of Oregon
- University of South Dakota
- University of Utah
- Univ. Washington Museum of Zoölogy
- University of Wisconsin
- Univ. Zool. Mus., Copenhagen
The largest single collection is in the United States National Museum, where the specimens of the National Museum proper and the United States Biological Surveys Collection, together, provide essential materials including a large share of the holotypes. Specimens in all of the North American collections including Canada and México have been made available, by loan, and in 1937 materials were examined in the principal collections of northern and central Europe. After the materials in North American collections were assembled, special effort, with considerable success, was made in each of several winters, to obtain specimens from areas not previously represented in collections.
To the many persons who were in charge of the collections consulted, to those who at my request sought critical specimens, and to those who assisted in various stages of assembling data and in preparation of the manuscript, I am grateful indeed. Likewise, I am deeply appreciative of the grants-in-aid received from the Carnegie Institution of Washington, the University of California Chapter of Sigma Xi, the John Simon Guggenheim Memorial Foundation and the Kansas University Endowment Association. I am mindful also of an obligation to those who appropriated funds, by legislative action, for research use by The University of California and The University of Kansas.
For assistance with the illustrations I am indebted to the late Major Allan Brooks for Plate 1 , to Mrs. Mary Blos for figures 25-31, to Miss Ann Murray for figures 11-13, to Mr. W. C. Matthews for all the photographs, to Mrs. Freda L. Abernathy for figures 2-9, 18-22, 24, and for retouching all the photographs except the following which were retouched by Mrs. Virginia Unruh: figs. d of plates 2, 3, 4, 9, 10, 11, 16, 17; figs. i of plates 5, 6, 7; figs. h, j, k of plate 7; figs. f and g of plates 12 and 13; and figs. c and d of plate 14. To Mrs. Unruh I am further indebted for figures 1, 16, 17 and 23 and for much terminal assistance with preparing most of the illustrations for the engraver.
The methods of study, after specimens were assembled, included first comparisons of specimens of like age and sex from each of several localities to ascertain the constant features by which full species were distinguishable, one from the other. For example, it was found that in every individual from Trout Lake, Washington, of the species here designated Mustela erminea, the postglenoidal length of the skull amounted to more than 47 per cent of the condylobasal length whereas it was less than 47 per cent in all individuals here designated as Mustela frenata, from the same locality. Testing of specimens from other localities by means of this and other selected characters permitted the outlining of the geographic ranges of the full "species-groups." By comparing specimens of other nominal species and by examining specimens from localities geographically intermediate between the nominal species, I found intergradation and therefore arranged the nominal species as subspecies of a single species. Intergradation here is understood to be the result of crossbreeding in nature between two kinds of animals in the area where the geographic ranges of the two kinds meet. Presence of intergradation between two kinds of weasels was basis for according them subspecific rank. Absence of intergradation in nature at every place where the geographic ranges of two kinds met or overlapped, and absence of intergradation by way of some other kind, or chain of kinds, was basis for according each of the two kinds full specific rank. By thus applying the test of intergradation, or lack of it, I found that there were four full species of weasels, of the subgenus Mustela, in all of the Americas.
Next, the specimens of one species were arranged in trays in a geographic sequence. The specimens from any one locality were segregated by sex and under one sex from one place were arranged from oldest to youngest, that is to say by age. The four series with the largest numbers of individuals of a given age were selected. Seventeen cranial measurements and three external measurements were recorded for each individual of each of these four series. For each measurement, the coefficient of variation, standard deviation and probable error were computed. The four samples subjected to such analysis were a series of adult males, one of adult females, one of subadult males and one of subadult females. Also, studies of each sex were made to ascertain seasonal changes in pelage. After data were obtained on ontogenetic (age) variation, secondary sexual variation, seasonal variation, and degree of individual variation by studying specimens in the manner described above, tests were made for subspecific (geographic) variation by comparing series of specimens of like sex, age and season, from different localities. For each one of several geographically variable features noted, a map was prepared for animals of each sex. When all the data thus obtained were codified, subspecific ranges were, in a sense automatically, obtained. On the resulting map showing geographic ranges of subspecies for a species, a type locality was accurately plotted for each name that had been applied to the species, and names then were applied in accordance with the international rules of zoölogical nomenclature.
VARIATION
Variation with Age
The kind of variation which results from increasing age has been dealt with extensively for the skull (of the Old World Mustela erminea) by Hensel (1881) and for the external features and to some extent for the skull by Hamilton (1933) in the North American forms M. erminea cicognanii and M. frenata noveboracensis.
The young of both erminea and frenata are hairless and blind at birth. In M. frenata noveboracensis, the eyes open on approximately the 37th day. When 2 to 4 months old, the tail is pointed at the tip. This is because the terminal hair of the tail, including the black tip, is short and lies flat on the tail. In subadults and adults the hair on the terminal part of the tail is as long as that on the basal part, and the tail appears to be of uniform diameter all the way out to the end.
In the western subspecies of M. frenata, and in its tropical subspecies, animals so young as to have pointed tails commonly have the underparts of the body more intensely colored than do adults. The young may have salmon-colored instead of yellowish fur on the underparts.
Otherwise, in animals that have attained approximately adult proportions—which appears to be at approximately 6 months of age in males—there are no variations which are ascribable to increasing age in the color-pattern or pelage that cause the systematist to confuse species or subspecies.
Of the several parts of the skull in juvenal animals, the braincase and width of the posterior part of the palate are most nearly of the size attained in the adult, the facial part of the skull at birth is the least developed, and the interorbital region is, in relation to its ultimate adult size, intermediate in stage of development. The permanent teeth are acquired when the animal is approximately eleven weeks old.
Four age groups, based on characters of the dentition and skull, have been recognized. They are:
Juvenile.—One or more deciduous (milk) teeth present. Birth to three months of age.
Young.—Sutures widely open between the maxillae and nasals and between the premaxillae and nasals. Three to seven and a half months of age.
Subadult.—Sutures between maxillae and nasals visible but indistinct. Seven and a half to ten months of age.
Adult.—Bones of rostrum coalesced with no traces of sutures visible to the naked eye. More than ten months old.
The skull as a whole increases in size until the animal is two-thirds of the way through the stage designated as young. After this time the width of the rostrum, as measured across the hamular processes of the lacrimals, increases until approximately a third of the way through adulthood. The interorbital breadth decreases from late subadulthood to adulthood and even in adults there appears to be a slight decrease in this part of the skull with increasing age.
The average zoölogist will readily distinguish skulls of juveniles and young from adults but usually fails to distinguish subadults from adults. Nevertheless, subadults must be distinguished from adults if geographic variation is to be measured accurately. The reason for this is that such differences in the form (not size) of the skull as result from increasing age equal and often exceed the differences of a geographic sort which serve for distinguishing subspecies that have adjoining geographic ranges. All sutures in the skull, except those between the tympanic bulla and the braincase, and those on the dorsal face of the rostrum, are obliterated while the animal is a subadult. Most kinds of mammals retain sutures throughout life or until the animals are well into adulthood. Therefore, skulls of weasels offer fewer features for estimating age than do those of most mammals and the skulls of weasels that are subadults or older are more difficult to classify accurately as to age than are the skulls of most other mammals. More reliance on shape of entire skull and less reliance on extent and shape of any individual bone is necessary in estimating the age of a weasel. Wright (1947:344) shows that the weight of the baculum (os penis) is a certain means of differentiating adults from males of lesser age. When approximately eleven months old, Mustela frenata oribasus of western Montana molts from the white winter coat into the brown summer coat. At that time spermatogenesis starts for the first time and the weight of the baculum increases from less than 30 milligrams to more than 52 milligrams.
In the autumn and early winter, most of the specimens are subadults. Ordinarily the few adults obtained in these seasons can easily be segregated from the subadults because ontogenetic development in the twelve additional months of life of each of the older animals has obliterated the sutures on the rostrum, heightened (vertically) and lengthened (anteriorly) the sagittal crest, widened the rostrum, and produced still other changes in form that are revealed by direct comparison of specimens of the two ages.
Secondary Sexual Variation
The secondary sexual variation, which has been detected, is in size of the animal, relative length of the tail and shape of the skull. The female is the smaller. In the small Mustela rixosa and apparently in Mustela africana the secondary sexual difference in size is relatively slight. In Mustela frenata and Mustela erminea, males are approximately twice as heavy as females, the degree of difference very definitely depending upon the subspecies. For example, in M. e. richardsonii the recorded weights are 175 and 69 grams as opposed to 81 and 54 grams in M. e. cicognanii. In general, within one species the greatest difference in size of males and females is in those subspecies in which the animals are of large size. The secondary sexual variation in size is much more than the individual variation in either sex. The same is not true of secondary sexual difference in length of the tail (relative to the length of the head and body), which in eighteen subspecies of M. erminea is from 1 to 7 per cent longer in males than in females. In two subspecies, M. e. haidarum and M. e. olympica, the tail is a fraction of a per cent the longer in females if we may rely upon the few specimens for which collectors' measurements are available.
In both M. erminea and M. frenata the skull of the female is approximately 45 per cent lighter than that of the male, or put in the opposite way, the skull of the male is 83 per cent heavier than the skull of the female. The difference in this respect varies greatly depending on the subspecies. For example, the skull of the male is 127 per cent heavier than that of the female in M. e. richardsonii but only 33 per cent heavier in M. e. anguinae. In Mustela frenata, the subspecies noveboracensis shows most sexual dimorphism in weight of skull (3.6 and 1.7 grams) and olivacea the least (5.3 and 3.8 grams). In general, the difference in this respect is less in subspecies the individuals of which are of small size.
Therefore, as might be expected, the secondary sexual variation in weight of the skull is less in M. rixosa, individuals of which are of small size, than in M. erminea or than in M. frenata, in general of larger size. Nevertheless, in M. africana, in which the individuals are of large size, there appears to be less sexual dimorphism in weight of the skull than in M. frenata or than in M. erminea, although it should be remarked that there are too few data for M. africana to allow of forming a trustworthy conclusion concerning the amount of secondary sexual variation in that species.
The secondary sexual variation in shape of the skull consists of a slenderness in the female. In relation to the basilar length the spread of the zygomatic arches is more in males and, except in the one subspecies M. f. altifrontalis, the rostrum is broader. Also the interorbital region is relatively broader in males of most subspecies. In most subspecies of both M. frenata and M. erminea the tympanic bullae are relatively (to the basilar length) longer in females. The maximum sexual dimorphism occurs in M. erminea arctica and the minimum dimorphism in M. e. haidarum, M. e. anguinae and M. e. muricus. Taking into account all of the subspecies of each of the North American species, the shape of the skull differs most in M. erminea and least in M. frenata. In the latter species the greatest difference in shape of the skull, as was true also of its weight, is in the subspecies M. f. noveboracensis. In these two subspecies, M. f. noveboracensis and M. e. arctica, in addition to the secondary sexual variation already mentioned in the skull, females have the braincase smoother and more rounded, the postorbital-, mastoid-, and lacrimal-processes relatively smaller, and the ventral face of the tympanic bulla at its anterior margin more nearly flush with the floor of the braincase.
In the weasels, subgenus Mustela, the disparity in size of the two sexes is almost or quite as much as in any other fissiped carnivore. It is because of this large degree of difference that the skulls of the two sexes are described separately in the following systematic accounts. The need for such treatment was recognized by Reinhold Hensel (1881:127) more than sixty years ago when he wrote in the introduction to his "Craniologische Studien," of Mustela, as follows: ". . . die Geschlechtsdifferenzen am Schädel vieler Säugethiere . . . so gross sind, dass man diese wie Schädel verschiedener species behandeln muss, während in anderen Ordnungen (Rosores, Edentaten) die Schädel solche Unterschiede nichtzeigen." In the past, failure to appreciate the large amount of secondary sexual variation has resulted in erroneous deductions as regards characters of certain geographic races and has been the cause of some nomenclatural confusion, as for example, in Mustela frenata macrura, where the female was named as a separate species (Mustela jelskii).
Individual Variation
Individual variation is here considered to be the variation in one species which can occur between offspring of a single pair of parents, after variation ascribable to differences in age, sex, and season is excluded. Individual variation, therefore, is a term here used in a composite sense; it includes variations which probably represent different genetic strains within certain populations and variations induced within one generation by environmental factors.
In skulls of weasels, the individual variation in size is more than it is in relative proportions. Hensel (op. cit.) has stressed that weasels, like other carnivores, produced "dwarfed" individuals more than do herbivorous mammals. I cannot vouch for the accuracy of this view, but can say that individual variation is not greater than in some other fissiped carnivores. Impressions to the contrary probably result largely from failure to recognize age-variation. When skulls of a large series from any one locality are arranged first by sex, and under each sex according to probable age on the basis of extension anteriorly of the sagittal crest and of degree of postorbital constriction, individual variation is seen to be less than a cursory examination, even of only one sex, would suggest.
Study of a large series of one age of one sex of one species from one locality shows that some parts, of the skull for example, vary more than other parts. In illustration, among 22 male topotypes of Mustela frenata washingtoni the least interorbital breadth varied 25 per cent (9.0 mm. to 12 mm.) whereas the length of the tooth-rows varied only 13.3 per cent (15.6 mm. to 18.0 mm.). In color the individual variation definitely is more in areas of intergradation between subspecies than in other areas. Details of one such instance of intergradation are given in the account of Mustela frenata spadix.
Statements to the effect that there is much individual variation in the color of weasels, were made mostly fifty years or so ago by writers who had but few specimens from widely separated localities. Where marked climatic differences exist between localities only a few miles apart, marked differences occur in coloration of the weasels from the different localities. Much of what formerly was mistaken for individual variation now proves to be geographic variation. Individual variation actually is of slight amount in comparison with that in mammals generally. Differences in size and relative proportions of parts usually are correlated with geographic differences in color. The color does fade slightly in the period between molts. Also as a result of the seasonal color change, in autumn along the upper margin of the Austral Life-zone, some individuals become white whereas others become white on only the underparts, the upper parts changing only to lighter brown. Probably it would be correct to say that this variation was a combination of seasonal and individual variation rather than either one alone.
As might be supposed, individual variation is not the same in all species or subspecies. For example, p2 is always absent in Mustela africana and always present in certain subspecies of M. frenata. In some other subspecies of M. frenata, p2 is absent approximately as often as present. In the writer's experience, when only a few specimens are available for comparison, individual variation is more difficult to distinguish from specific and subspecific (geographic) variation than is age-variation or secondary sexual variation.
Among the larger series of specimens examined, only one instance of what might be called a mutation in the old sense of a large, sudden change, was detected. That was the loss of the second lower molar in many (less than a third) of the specimens from Newfoundland. The six instances of abnormal coloration described on pages 41 to 43, might be regarded as mutations of large magnitude but no evidence was found of repetition of an abnormality in any one population. Otherwise, in every instance where plotted, the manifestations of a variation arranged themselves about the mean in such a way as to form a smooth, unimodal curve.
Seasonal Variation
When subspecific and specific variations are the objectives of study, seasonal variation must be understood, in order to be excluded from consideration, in the same way that variations ascribable to age, sex and individualism must be understood in order to be excluded from consideration. In weasels, change in color of the pelage is the seasonal variation most important for the systematist to understand. Other seasonal variations in the pelage are hairiness versus nakedness of the pads of the feet, length of the pelage on the body, and possibly the density of the pelage on the body. In the northern half of North America, roughly speaking, seasonal change in color is so pronounced (white in winter and brown in summer) as to be easily recognized. South of this area, in the Austral and Sonoran life-zones, the color of the winter pelage differs only slightly from that of the summer pelage. In these more southern latitudes the winter pelage in almost all subspecies is of lighter color than the summer pelage and has a smoky suffusion. With material of the two seasons in hand for comparison, close attention to the variation will permit the systematist to recognize the difference in shade of brown as seasonal variation and not geographic or specific variation. Farther south still, in the Tropical Life-zone, seasonal difference in color was not detected in the material studied. Seasonal change in color is discussed in the section immediately following.
Variation in Coloration and Molt
In all American weasels (subgenus Mustela) the color, at least in summer, is brown with more or less white or whitish on the underparts. In one species, Mustela africana, there is a longitudinal stripe of brown on the middle of the light-colored underparts; this stripe is absent in each of the other three American species. Two species, M. erminea and M. frenata, always have a black tip on the tail. Of the other two species, M. africana lacks the black tip and M. rixosa may or may not have a few black hairs in the tip of its tail. White or light yellowish facial markings occur in subspecies of M. frenata from the southwestern United Stated to Central America. Subspecies having the most extensive light-colored facial markings have the remainder of the upper part of the head black. In weasels without light facial markings the upper parts of the head all are brown. In the two species, M. erminea and M. frenata, the extent to which the light color of the underparts extends down the insides of the legs and out on the underside of the tail, or the absence of light color on these parts, is a matter of geographic variation. The same can be said for M. rixosa except that first its tail is unicolored and second individual variation as well as geographic variation accounts for the color pattern on the underparts and legs in animals from the southeastern part of the range of the species.
The most remarkable feature of the coloration of weasels is the winter whitening. This occurs in the northern part of North America in each of the three species of weasels found on that continent. The black tip of the tail in M. erminea and M. frenata remains black in winter. If an individual of M. rixosa has black hairs on the tip of its tail in summer, there are thought to be black hairs there also in winter. Otherwise the winter pelage is all white in northern areas in each of the three species. In this white winter coat the animal is known as ermine.
The underlying cause seems to be protective coloration. At any rate, weasels are always white in winter if they are from areas where snow lies on the ground all winter, every winter, or almost every winter; and they are always brown if from areas where there is never, or rarely, snow in winter. The changes in color are effected by molt, one in autumn and one in spring. Animals that are brown in winter undergo the same two molts as do those that are white in winter. The capacity to acquire a white coat or a brown coat in winter is an hereditary matter just as one man grows red hair and another grows black hair. In the weasels, however, all individuals in the north turn white in winter and if one that was born there is kept through successive winters in the warmer south where there is no snow, he will still turn white each winter. A weasel born in a southern area, where all are brown in winter, molts into a brown (not white) winter coat even when kept in a cold, snowy, northern area where native weasels of the same species all turn white. Obviously, therefore, neither snow nor temperature is an immediate cause and, as we have said, the color in winter is a matter of heredity. The time of the molt, we now know, is determined by the amount of light. When nights grow longer and days shorter, a point is reached at which the lesser light received through the eyes causes the pituitary gland to cease producing a gonadotropic hormone. Directly or indirectly, the lack of this hormone stimulates molt and, probably enzyme action, or the lack of it, causes the melanoblasts of the cells in the hair follicle to be without pigment. Hence the hair grown from a follicle under such conditions lacks pigment (melanin) and is white. In spring, as the days grow longer and the nights shorter, the increasing amount of light received day by day through the eyes stimulates the pituitary gland to produce the gonadotropic hormone which directly or indirectly, stimulates molt and, probably by enzyme action, the melanoblasts are caused to be present in cells of the hair follicle and the melanoblasts provide granules of melanin pigment which are incorporated in cells of the growing hair. These granules of pigment give the hair its color.
Evidence in support of this hypothesis is given below.
Along the Pacific Coast from British Columbia southward, M. erminea (see fig. 25 on page 95) is brown in winter. This is an area where snow rarely falls and the temperature in winter ordinarily is above freezing. In the remaining part of the American range of this species the temperature in winter is below freezing much of the time and snow remains throughout the winter or for long periods. In this colder part of the animal's range, only white coats occur in winter. M. frenata likewise has a white coat in winter in the part of its geographic range where snow and freezing temperatures prevail throughout most of the winter and a brown coat in warmer, snowless areas to the southward and along the Pacific Coast. The third species, M. rixosa, exhibits a corresponding correlation between coat color and climate. On the Asiatic continent, several species, including M. erminea, provide parallel correlations and nowhere are there any exceptions for the subgenus Mustela. These data are an important part of the material on which we have based the induction that the underlying cause of seasonal change in color is a need for protective coloration.
As regards molt, most naturalists who have written upon the subject regard it as responsible for the change from the white winter coat to the brown summer coat. However, the change from brown summer coat to white winter coat has been thought by several writers to be effected by change in coloration of the individual hairs. Among those holding this opinion there may be cited Bell (1874:197) in reference to Mustela erminea, and Coues (1877:123) in reference to American specimens to which he applied the same name. More lately Hadwen (1929) has taken this same view, and Gunn (1932) also discusses the possibility of the hairs changing color. Bachman (1839:228-232), Macgillivary (1843?:158), Audubon and Bachman (1851 (vol. 2):62), Schwalbe (1893:538), Pearson et al. (1913:447), Miller (1930, 1931A), Hamilton (1933:300) and Rothschild (1942), among others, have been inclined to the opinion, or positively affirm, that the color change in autumn is the result of a molt. The papers cited above contain, in turn, references to many other printed accounts dealing with this question.
To my mind, it has not so far been demonstrated that the change in color of weasels in autumn is accomplished without a molt. Also so far as I am aware, no explanation has been given of how the pigment may disappear from the hair of weasels. Metchnikoff's (1901:156) idea that the senile whitening of the hair in man is accomplished by phagocytes which remove the pigment granules would hardly seem to explain the relatively sudden and complete autumnal change occurring in weasels. Anyhow, Danforth (1925:108), and some other students have thought that the action of these phagocytes was at most a factor of slight importance in the whitening of hair. Whatever be the complete answer to the question of how the weasel changes color in autumn, at least one specimen of long-tailed weasel, which is in process of color change in autumn, presents clear evidence of molt of the overhairs. This specimen of M. f. longicauda is no. 188408, U. S. Nat. Mus., taken on November 12, 1897, at Rapid City, South Dakota. Other specimens of M. erminea which were taken in autumn similarly show molt to be in progress. For these and other reasons, I am inclined to the opinion that the autumnal change in color, like the one in spring, is effected by molt. During the period of the autumnal color change, Noback (1935:27) had a captive M. f. noveboracensis and, each morning, found clumps of brown hair on the floor of its cage; this was strong indication that molt was responsible for the color change in this instance.
However, I freely admit that the evidence does not prove that the change from brown to white can be accomplished only by molt; in the present state of knowledge it would be unscientific to deny that the change were possible of accomplishment by other means. Also, it is true that the fifteen specimens before me of Mustela frenata, subspecies included, in process of change from brown to white, with the exception of the one from Rapid City, South Dakota, if taken individually, do not, in macroscopic examination, show definite molt lines or other absolutely convincing evidence of molt. However, these same specimens, insofar as examined microscopically, do show overhairs all white, or overhairs pigmented throughout. The lighter color of the proximal parts of the overhairs in itself should not be accepted as evidence of color change, for in the fresh summer pelage, the same condition exists. Also, careful macroscopic examination suffices to show that in the transitional pelage of autumn, the brown overhairs generally are longer than the intermixed white overhairs.
Whether the underfur behaves in exactly the same way as the overhair, I have not myself definitely ascertained, but I assume that the underfur is molted twice each year, at least in the northern populations of Mustela frenata and in the other species of more northern distribution. Schwalbe's (1893) work, including sectioning of the skin and study of the hair follicles, led him to conclude that the underfur was molted twice each year in Mustela erminea.
In Mustela frenata noveboracensis, M. f. nevadensis, and M. f. nigriauris, measurements taken on adult males show the overhairs to be longer in the winter pelage than in the summer pelage of specimens from the same locality. For example, in M. f. nigriauris from Berkeley, California, the overhairs of the summer coat (July and August) average 8 millimeters in length on the hinder back and 7 mm. on the belly, but average 9.5 mm. and 8 mm. respectively in January-taken specimens possessing the full winter coat. At Ann Arbor, Michigan, in the summer coat, the longest hairs on the hinder back average approximately 12 mm., and those on the belly, 9.5 mm., against 13 mm. and 9.5 mm. respectively in winter. Although general observations initially led me to believe that the black, terminal hairs of the tip of the tail are longer in the winter pelage than in the summer pelage, actual measurements fail to show a difference in length.
The change from one coat to the other in the long-tailed weasel has been described among others by Miller (1930, 1931A), Hamilton (1933) and Glover (1942) on the basis of captive specimens. In a general way, the progress of the molt in their specimens agrees with that which I have been able to make out from examination of skins taken in the wild. There is, however, this difference: Their specimens show a more spotted pattern when in process of hair-change than do specimens taken in the wild. Probably the more or less unnatural conditions under which these captive animals lived modified the normal progress of molt.
In wild-taken specimens of the species Mustela frenata, subspecies included, the spring molt begins on the mid-dorsal line and proceeds laterally, producing, at almost any given time, a relatively sharp molt line separating the white winter hair from the incoming brown summer coat. However, in autumn the change takes place first on the belly, then on the sides, and finally makes its appearance over all the upper parts at about the same time, with the result that the upper parts have a salt-and-pepper appearance without at this time any sharply defined molt lines. In general, the molt pattern can be said to be reversed in the two seasons; in spring, it begins on the back and in autumn, on the belly. The difference in spring and autumn color pattern is better illustrated on plate 39 than by additional description. Swanson and Fryklund (1935:123) have observed that the "spring molt proceeds differently" than the fall one in Mustela rixosa, and Barrett-Hamilton (1903:309) in commenting on the European hare (and the stoat?) remarks, "In spring the moult, and with it the brown colour, progresses in exactly the opposite order . . ." as compared with the white color of autumn, which that particular writer thought resulted from removal of pigment from the hairs rather than from molt.
The tail, excepting the black tip, lags in the molt in many instances, with the result that, especially in spring, it may retain a few white hairs as late as does the belly. In autumn it is less tardy and so far as I have observed, becomes white at about the same time that the general area of the back changes color. On the tail, the black tip itself, as clearly shown in more than a score of specimens, is molted at approximately the same time in autumn as is the pelage of the body. However, the long black hairs, which appear in, say, November, appear to increase in length until January. In spring, the long black hairs of the tip of the tail seem not to be shed at the same time as the rest of the winter pelage, but remain approximately six weeks longer and then are replaced by long black hairs of the summer coat. At any rate, this is the picture presented by a half dozen specimens of M. f. nevadensis and M. f. longicauda which do show a spring molt to be in progress on the black tip of the tail. Schwalbe similarly (1893:536-537) has suggested that the black tip of the tail in Mustela erminea in spring is not molted until about two months after the pelage on the rest of the body is changed. Schwalbe (loc. cit.) thinks also that in M. erminea studied by him, the black tip of the tail in autumn is replaced approximately one month in advance of the pelage on the rest of the body. As indicated above, my specimens of Mustela frenata, subspecies longicauda and nevadensis, do not show this discrepancy in autumn. I have considered the possibility that the black tip of the tail, in some species of Mustela, is molted only once while the remainder of the coat was undergoing two molts. My inconclusive data lend but little support to this possibility.
The difference in pattern of color between specimens taken in autumn and spring is known to some fur-trappers of my acquaintance who have suggested that molt occurs in spring, whereas the individual hairs change color in autumn. Reference to plate 39 will show how gross comparisons might lead one to this erroneous explanation of the color change.
As to time of molt: In eight subspecies of Mustela frenata, namely, noveboracensis, occisor, primulina, spadix, longicauda, arizonensis, nevadensis and effera, material is available to indicate that the autumnal molt begins in October and is completed in November, and that the spring molt occurs in March or April. A condensed list of specimens providing basis for this statement is as follows:
M. f. noveboracensis: 26 specimens in transitional pelage taken in autumn and 14 taken in spring; M. f. occisor: One topotype has acquired one-fifth of the winter pelage on October 22, 1896; M. f. primulina: 2 in November, one in March, and 2 in April are in process of change; M. f. spadix: 6 autumnal specimens and one in April show pelage change; M. f. longicauda: 7 autumnal specimens and one in April show pelage change; M. f. arizonensis: 12 specimens in autumn and 3 in spring are in process of molt; M. f. effera: One November-taken male has acquired four-fifths of the winter coat and another taken on April 21 at Fort Rock, Oregon, is half finished with the spring molt.
It may be added that no marked difference in time of either autumnal or spring molt is apparent as between the more northern and more southern localities from which the mentioned specimens come. With more complete material I would expect to find a difference in this regard.
The material of the other, more southern, subspecies of Mustela frenata has not been adequate to show the time of molting or the number of molts which occur in one year.
Animals in the northern part of the range of Mustela frenata acquire a white winter coat, whereas those in the southern part acquire a brown winter coat, and in an intervening area the winter coat may be either brown or white. By plotting on a map the localities of capture of all specimens examined in the winter coat, it was possible to outline this intervening area as shown in figure 10 on page 37. However, Dearborn (1932:36) shows that in Michigan some animals have a brown coat in winter at places farther north than figure 10 shows to be the case. Hamilton's (1933-306) map for New York shows the same to be true in that state. Accordingly, the boundaries of the area shown in figure 10, in which both brown and white long-tailed weasels occur in winter, are known to be only approximate; with full information available the belt would be represented as wider.
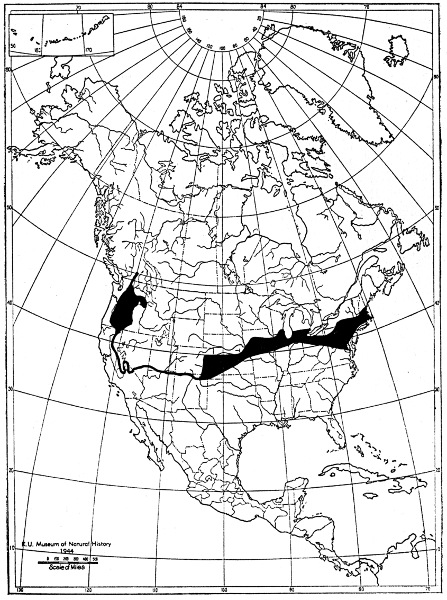
Fig. 10. Map showing the region (in black) where both the brown and white winter pelage is found in the long-tailed weasel, Mustela frenata.
Hamilton (1933:302) has pointed out that "Where half of the weasels remain brown, these brown winter specimens are always males." The results of my own examination of specimens not studied by Hamilton, in a general way provide confirmatory data. More exactly, my examination reveals that at the most northern localities where brown specimens occur, only males are in this coat. In explanation, it may be said that in plotting on a map localities of capture of specimens in the winter coat, thirteen places were found where both sexes were represented and where both brown and white winter coats were found. With the two sexes, it is theoretically possible to have nine different combinations of coat color. With males all brown, there might occur females (1) all brown, (2) all white, or (3) some brown and some white. In addition to these three combinations, we might have three more by finding the mentioned types of female coat color repeated where all males are white, and three more, or nine in all, by substituting a population of males some of which were brown and some of which were white. Seven of these possible combinations actually were found. The two combinations not found were all white males with all brown females, and all white males with females both brown and white. In the three instances where the males all were brown and the females all were white, the localities of capture were in the northern part of the variable area. This indicates that where the brown winter coat occurs at northern localities, the brown individuals are all males. Farther south, of course, the females, too, acquire the brown winter coat.
Stated in another way, there is a broad belt across North America from the Atlantic to the Pacific in which males of Mustela frenata at any one locality may be either brown or white in winter. Inside this broad belt there is a narrower one, approximately half as wide, in which females at any one locality may be either brown or white.
In support of the idea that color of the winter coat is an hereditary matter and that it is not dependent on temperature, the following evidence derived from my transplanting specimens of Mustela frenata supports the idea that color of the winter pelage is dependent on heredity and not on temperature or snowfall.
A male captured on June 24, 1937, in the brown summer coat in Salt Lake City, Utah, was received by me at Berkeley, California, five days later and kept in captivity almost six months. On November 17, 1937, half the pelage was white and on December 27, 1937, when next examined, the animal was in the full, white, winter coat as it was on January 25, 1938, when it died. Native weasels all turn white in winter in Salt Lake City, but in Berkeley native weasels always are brown in winter.
A juvenile or young animal, a male, captured in May, 1936, at Lafayette, Contra Costa County, California, was kept there until August 13, 1936, when transferred to Calneva at the north end of Lake Tahoe, California. The weasel was kept at Calneva until its death on December 23, 1937. In both the winter of 1936-'37 and in that of 1937-'38, the winter coat was brown as in animals from its place of origin (Contra Costa County) and unlike weasels of the Tahoe region nearly all of which turn white in winter.
Two females, each approximately two months old, captured on May 1, 1936, at James Landing, 4 miles northwest of San Pablo, Contra Costa County, California, were kept in Berkeley, California, until August 13, 1936, when they were transferred to the mouth of Blackwood Creek, on the west side of Lake Tahoe, California. On October 25, 1936, both weasels escaped. On December 25, 1936, the headless body of one of these was found approximately 300 yards south of the mouth of Blackwood Creek. The animal had been dead at most a few days when found and was in the brown winter coat. At the place of its origin all weasels are brown in winter but at the mouth of Blackwood Creek only 2 of 60 weasels caught there in the winter coat were brown; the other 58 were white. The headless weasel was identified, as one of the two formerly in captivity, by means of certain short toes, the ends of which had been clipped off when the animal was a captive. No trace of the second female was found.
A female of unknown age, in white winter pelage, captured 4 miles southeast of Tahoe City, California, and kept there until April 3, 1937, on which date it was brought to Berkeley, California, molted to brown in the spring. The first signs of the brown coat were noted on April 14. On May 24 or 25 she gave birth to 4 young which lived less than ten days. In the following winter this animal acquired a white coat. As previously noted, weasels native to the Berkeley area, where this female was kept, have brown coats in winter.
The weasels were in every instance kept in cages out-of-doors. The sides of the cages were open to the elements. A nest box in each cage provided shelter. All were of the species Mustela frenata.
The significant results, it seemed to me, were that the winter coat was the kind found in the area where the weasel originated instead of the kind found in weasels native to the areas in which the specimens were held in captivity.
That the time of molt is determined by the amount of light has clearly been shown by Bissonnette (1944:223) for American weasels of the two species Mustela erminea and M. frenata. In his words (op. cit.:246) "Reducing the daily periods of light induced molting and regrowth of new fur. . . . In the Bonaparte weasels [Mustela erminea], white replaced brown. . . . Increasing daily light-periods caused molting and change to dark brown. . . . Incomplete molts in both directions (toward white or toward brown) were produced as a result of early reversal of increase or decrease of daily light-time. . . . That this stimulus is received through the eyes and acts through the anterior pituitary gland is indicated by Bissonnette's [1935:159] studies on ferrets, a nearly related animal. That the thyroids and sex-glands are not essential is at least suggested . . . by Lyman's (1942) study on the varying hare [Lepus americanus]." It can be added that Lyman (1943:451) demonstrated in Lepus americanus that the effect of light is received through the eyes. He demonstrated this by masking the animals. To Wright (1942B:109) who studied the two American weasels, M. erminea and M. frenata, it seemed likely that the pituitary produced or released gonadotropic hormone at about the time of the spring molt and that this molt and the spring changes in the reproductive tracts of the weasels might be caused by a stimulus from a common source. Later, Wright (1950:130) injected a gonadotropic hormone into long-tailed weasels which had recently acquired their white winter pelage and thereby caused them to lose the white pelage and acquire the brown pelage. It is Lyman (1943:450) who says, in relation to Lepus americanus, "When in the physiologically white condition, the melanoblasts of the regenerating guard- and pile-hair follicles contain no melanin-forming enzyme (dopa-oxidase), which may be the reason for the lack of pigment." Schwalbe (1893) by sectioning the skin and microscopically examining the hair-follicles of M. erminea learned that the basal cells producing hairs lacked pigment granules in autumn when the European ermine (M. erminea) was acquiring its white winter coat and that the cells contained granules of pigment in spring when, as we know, the granules are incorporated in the growing hair and give it its color.
The above material, then, is basis for the account on pages 31 and 32 of what causes the weasel of northern areas to have a white coat in winter. The discerning student will instantly perceive that although some parts of the account on pages 31 and 32 are precisely accurate, other parts are the result of inferences which need to be proved. More careful work of the kind that Schwalbe (1893) and Wright (1942B) did is needed. The account on pages 31 and 32 is merely the best that can be given with the information now available.
Many writers have commented on the yellowish color, sometimes with a greenish tinge, found on the fur of weasels in the white winter coat. The stain is more often found on the tail and hinder-parts of the body than elsewhere. Possibly, partly on this account, some have ascribed this color to the smearing of the fur with urine. Still others have thought it resulted from the smearing of the fur with secretions from the anal scent glands. Schumacher (1928) takes this point of view, and while it may be that he has not proved his point, still his conclusions fit the known facts and seem sound to me. Schumacher points out that the same soiling of the fur is present in summer as well as in winter, but that on the summer pelage the stain can be detected only on the light-colored underparts. It is from this point of view that he criticizes the systematic worth of white versus yellowish-white underparts in the summer pelage of geographic races of Mustela erminea and Mustela nivalis. Although in the long-tailed weasels (Mustela frenata) the underparts of all the races are pigmented with some form of red, orange or yellow, it seems probable to me that the additional color resulting from the soiling effect of this glandular secretion explains the greater variation, found at a single locality, in the color of underparts than of upper parts in the summer pelage.
I have neither seen nor heard of a black weasel in any part of the New World or of the Old World. I have found only one albino among American specimens. It is an adult female, no. 121424, American Museum of Natural History, of Mustela erminea richardsonii, taken on August 30, 1935, at Hot Springs, Northwest Territory. This place, I am told by G. G. Goodwin who obtained the animal, is on the "Nahanni River where the rugged mountain ridges rise abruptly from the low mud flat lands, latitude 61, longitude 125." The shortness and coarseness of the hair corresponds to that of the summer pelage and not winter pelage. The pelage is everywhere white, even the tip of the tail. True, all except the nape and top and sides of the head has a faint yellowish-green tinge which has been supposed to result from staining by secretion of the anal scent glands but there is no pigment in the hair as in erythristic specimens. From the Old World, Farurick (1873:17) has recorded what he regards as an albino of Mustela vulgaris since it had no black hairs on the tip of its tail. Flintoff (1935:228, 229) records what may have been an albino Mustela vulgaris from Yorkshire and an albino M. erminea from an unstated locality. Jäckel (1873:459) mentions specimens of Mustela erminea and Mustela vulgaris, which were partly "albinistic" or "erythristic." Among the American specimens of M. erminea I have not recorded any which appeared to be either partly or wholly erythristic or only partly albinistic. Among the 1550 skins of M. frenata which were in summer pelage or brown winter pelage, five, described below, show marked abnormalities in color.
Two of these five are partly albinistic. One is an adult male, no. 223880, U. S. Nat. Mus., from Billy's Island, Okefinokee Swamp, Georgia, which has the nose as well as the area between the eyes white. Also there is a tuft of white hairs at the anterodorsal margin of each ear, scattering white hairs suggesting a postorbital bar on each side of the head, and a patch of white hairs on the mid-dorsal line behind the ears. Markings of this kind are not abnormal in M. f. peninsulae, the subspecies adjoining on the south, except for the white nose which clearly is an instance of partial albinism. The second specimen is a subadult male, of M. f. noveboracensis, no. 177679, U. S. Nat. Mus., in process of acquiring the brown winter coat, taken on November 27, 1911, at Gaylordsville, Connecticut. It has white markings on the nose, on the right side of the neck, on the right hind foot and right forefoot, and on the tip of the tail. The white area of the nose on the left side extends back to the eye, but on the right side barely encircles the nose-pad. On the right side of the neck, all that area between the foreleg and ear is white from the mid-dorsal line (including 7 or 8 millimeters to the left of the mid-dorsal line) down to the throat, which is white as it is also in normal individuals. The toes of the right hind foot are more extensively white than in normal specimens of noveboracensis, and all of the right forefoot as well as the wrist is white. The tail is of striking appearance because of its tricolor pattern. The proximal part is of the normal brown color. The black terminal part commences proximally at the usual place, but the distal 11 millimeters of the fleshy part of the tail bear only pure white hairs producing a terminal white pencil 35 millimeters long.
The three other specimens abnormally colored are erythristic individuals. An adult male of M. f. latirostra, no. 7574, coll. D. R. Dickey, taken on April 14, 1918, at Covina, Los Angeles County, California, has the color of the upper parts greatly restricted, and, in addition, has spots and blotches of the color of the underparts distributed over the back and rump. A spot of this same color occurs above each ear. Incidentally, this and other subspecies of Mustela frenata from the Pacific Coast of North America obviously have the factor for erythrism operating over a larger part of the body than it does in M. erminea or than in M. f. noveboracensis, where the underparts sometimes are white. In M. f. latirostra and in other subspecies from the Pacific Coast the light color of the underparts always is tinged with this reddish color.
Another erythristic specimen is a young male of M. f. nevadensis, no. 23493, U. S. Nat. Mus., taken on August 6, 1890, at Birch Creek, Idaho. It has all of each foreleg, the axillary regions, and a saddle-shaped area over the shoulders of the same buff-yellow color as the underparts.
The third erythristic specimen is a subadult female, of M. f. oregonensis, no. 47149, Mus. Vert. Zoöl., taken on December 20, 1930, at Carlotta, Humboldt County, California. This specimen appears to be white and initially was thought to be merely an individual in the white winter coat. Closer examination, however, shows that it has a light wash of ochraceous or faint reddish color. Also, other specimens taken in winter at Carlotta show that weasels there do not acquire a white winter coat. The only normally brown area is approximately three millimeters in diameter at the anterodorsal margin of the pinna of the right ear. The tip of the tail is black as in a normal specimen. The specimen in question is actually pure white only on top of the head from a short distance behind the ears on over the forehead nearly to the eyes, and on the inside of the ears. In a normally colored animal this area is the dark area of the head. In this freak, the other parts of the head, which, in individuals of normal coloration are the white or light orange facial markings, have the reddish cast of the remainder of the body, although the color is less intense than on the back. The collector noted that the specimen had eyes of normal color. A possible explanation for the coloration of this specimen is that this species has three factors for color, one for the black tail tip, one for the reddish color, and a third, missing in the specimen in question, for the blackish brown.
For some more exact knowledge concerning this erythristic type of coloration, we are indebted to Pitt (1921:99), who describes a population of polecats, Mustela putorius, in Cardiganshire, England, in which this erythristic variation is maintained in a state of nature. In ferrets, Mustela furo, Pitt (op. cit.:114) notes that ". . . erythrism is certainly dependent on a Mendelian factor, being dominant to albinism and recessive to the black-brown coloration. Both in the ferret and polecat, erythrism seems to be correlated with increased size, and certainly in the ferret is usually accompanied by a quick temper and general increase in vitality."
Variations of Taxonomic Worth
Variations of taxonomic worth usually are referred to as characters. For example, shortness of the tympanic bulla is a character, and the opposite condition, long tympanic bulla, is another character. Specific variations, that is to say specific characters, are provided by the color-pattern, length of tail, number of premolar teeth, shape of the tympanic bullae, and length of the braincase in relation to the length of the tooth-bearing parts of the skull. Subspecific characters are provided by color-pattern, color itself, size as measured by weight of the animal, and its linear measurements, size of the skull, and size and shape of parts of the skull. The characters distinguishing subspecies from one another are not of a different nature from those distinguishing species from one another.
Given any one of the above structural features, say, dorsal outline of the skull, several characters may be provided by it. For example, weasels of the species Mustela frenata have the dorsal outline of the skull convex in southern Louisiana, straight in Missouri and concave in North Dakota, thus providing three characters. This is geographic variation. These variations, characters in zoölogical parlance, when plotted on maps, reveal the geographic occurrence of, say, the convex shape of the skull. In combination with other characters, for example, dark color and short tail, basis is provided for recognizing a subspecies, in this instance Mustela frenata arthuri of Louisiana. Because the change from convex to flat skull takes place geographically at about the same place (in eastern Texas) as does the change from short tail to long tail, and the change from dark color to light color, it is easy to draw a line there marking the western geographic limit of occurrence of the M. f. arthuri. This same line marks also the eastern margin of the geographic range of the subspecies Mustela frenata frenata, the subspecies next adjacent to the westward. On this line and for several miles to either side of it weasels show varying combinations of these three characters or an intermediate condition as regards one or more of the characters, or both. For example, from a locality in eastern Texas a weasel may have (1) a facial pattern exactly intermediate between that of the unicolored face of arthuri and that of the bicolored face of frenata, (2) the long tail of frenata and (3) the convex skull of arthuri. In the sum of its characters this specimen is exactly intermediate between typical arthuri and typical frenata. Another specimen from the same place may differ from the first specimen only in having the tail slightly shorter. The total "score" for the two specimens is, therefore, by a very slight margin in favor of arthuri. Let us suppose that we obtain a third specimen from the same place and that it has the face marked like that of arthuri but the tail fully as long, and the skull as lacking in dorsal convexity, as in frenata. Now the score is definitely for frenata. For convenience of handling, the population is referred to frenata, providing that the average of specimens from a nearby locality to the westward is not in favor of arthuri. In event the average of specimens from a locality next adjacent to the westward is in favor of M. f. arthuri, the total evidence from the two localities may be weighed together and appropriate decision as to subspecific status of weasels from the area is made according to what the average is for the area as a whole.
The three individual animals of an intermediate sort are ordinarily termed intergrades. This implies that their characters are the result of mixed parentage—perhaps a female of M. f. arthuri and a male of M. f. frenata but probably each parent itself was an intergrade and the offspring, of which we examined three, owe their characters to reproductive processes operating in obedience to Mendelian laws of inheritance.
The two kinds of animals, Mustela frenata arthuri and Mustela frenata frenata, are identified as subspecies because of the intergradation between them. If at this and all other places where the geographic ranges of arthuri and frenata met there was no crossbreeding (no intergrades), the two kinds would be treated as distinct species. Intergradation, and the lack of it, are accepted as the criteria of subspecies and species, respectively.
These criteria suffice for animals, in this instance weasels, which have a continuous geographic distribution. Some kinds of weasels are confined to islands, as for example the islands off the coast of Alaska and British Columbia. Because weasels are land animals, crossbreeding in nature between the weasels of two islands is, of course, impossible. A modified test (used in the study here reported upon) in deciding on specific versus subspecific status in these instances can be made as follows: On the adjacent mainland, ascertain the degree of difference between two subspecies whose geographic ranges meet (for example, M. e. richardsonii and M. e. alascensis). Next ascertain the degree of difference between the insular kind of animal and the kind on the mainland. If the degree of difference is greater when the insular kind is compared than when only the kinds of the mainland are compared, the insular kind is to be regarded as a species. If the degree of difference is no greater between the insular kind and the mainland kind than it is between the two adjacent mainland kinds, the insular kind is to be regarded as a subspecies. In short, for insular kinds, the criterion is degree of difference, with the limitation of geographic adjacency, rather than intergradation.
The geographic variation (subspecific characters) found could be spoken of as two kinds: First, there is the variation which is expressed in a general trend for a long distance, producing, in general, a cline of even slope; and second, that of inconstant trend in any one direction. In his "The Rabbits of North America" Nelson (1909:34-35) has commented on the latter type of variation as follows: "While studying series of specimens from all parts of the vast range occupied by the geographic races of such species as Sylvilagus floridanus and S. auduboni, I have been impressed with evidences of fluctuation of both external and skull characters. These fluctuations are somewhat wavelike in character and rise to central points of extreme development and then sink away to intermediate borders beyond which new waves rise. Where the waves of differentiation are pronounced they mark recognizable geographic races. Within the area covered by the larger or geographically broader waves of differentiation (recognized as of subspecific value), smaller waves of differentiation are included, which may represent local variations in intensity of characters of the subspecies, or these characters may diminish and the variation tend in other directions, sometimes even closely reproducing the characters of another subspecies occupying a distinct area." In Mustela frenata, much of the geographic variation at first inspection appears to be of this nature. Closer scrutiny, however, reveals that the repetition, at geographic intervals, of several features of color and structure are closely correlated with environmental features which are repeated only at these same places.
In Mustela erminea, much of the variation is of the first kind, namely, that which can be expressed as long clines of relatively even slope. As several authors have said, zoölogical classification based on this kind of variation is like dividing the spectrum and depends largely upon the standards set, for, theoretically, the possibilities of subdivision are unlimited. Actually, however, none of the clines has an even slope and the possibilities for subdivision therefore are limited. Also, when several features are used, instead of only one feature, the classification is more satisfactory even if the basis is more complex.
Some features of structure which provide subspecific characters are mentioned below.
Total length, of males, ranges from 598 to 360 mm. in M. frenata and from 336 to 228 mm. in M. erminea. There is no cline of sustained slope in M. frenata but in M. erminea there is a progressive decrease in total length from north to south.
Length of tail varies from as little as a half to as much as seven-tenths of the length of the head and body in M. frenata, the subspecies neomexicana having the long tail and the two subspecies arthuri and primulina having short tails. The geographic ranges of primulina and neomexicana are contiguous. In M. erminea there is likewise no variation of a clinal nature in length of tail and furthermore the variation is much less than in M. frenata.
In length of hind foot, which in males varies from 49 mm. in northern populations of M. erminea to 28 mm. in southern populations, the same cline is seen as in the total length of animals of this species. In M. frenata, however, there are several decreases and increases along any straight line which can be drawn through the geographic range of the species. The range of variation in males is 41 mm. (M. f. arizonensis) to 59 mm. (M. f. macrophonius).
Weight of the entire animal is an excellent measure of size but weights are unavailable for many subspecies. In M. frenata, the two subspecies texensis and macrophonius probably are the heaviest and effera, arizonensis and helleri probably are the lightest. Geographically the variation in weight behaves in approximately the same way as does the measurement of total length. In M. erminea the variation in weight of males is from 206 grams in northern animals to 58 grams in southernmost populations, there being a relatively constant gradient geographically.
Degree of hairiness of the foot-soles in M. frenata clearly is linked with the temperature; in regions of high average temperature the hairiness is least and in regions of low average temperature it is most. The decrease in hairiness is accomplished in two ways, namely, smaller breadth and decreased length of individual hairs and decrease in number of hairs on a given area of dermal surface. This correlation holds throughout the entire north to south range of the species. Corresponding differences are found on the same latitude where topographic diversity in an east to west direction produces northern conditions at high altitudes and southern conditions at low altitudes. The conclusion seems unavoidable that climate, directly or indirectly, determines the degree of hairiness. Less careful observations were made on the hairiness of the soles of the feet in other species but it is clear that the northern species M. erminea has the most hair on the foot-soles and that M. africana, the tropical weasel, has the least. In this regard, M. frenata is intermediate as it is also in geographic position.
Figs. 11-15. Dorsal views of adult skulls of each sex of five subspecies of the ermine, Mustela erminea, to show secondary sexual variation and geographic variation in size of the skull. Males on the left and females on the right. All × 1.
Note especially the geographic variation in decreasing size of the skull from north to south in each sex, and that the secondary sexual variation in size of skull is less in ermines with small skulls than in those with large skulls.
Fig. 16. Map showing the localities where the skulls, represented in figures 11-15, were obtained.
The maximum length of facial and carpal vibrissae is attained in M. erminea in the far north. In weasels from north of the Arctic Circle the longest facial vibrissae extend posteriorly beyond the posterior border of the ear. In the tropical weasel, M. africana, the facial vibrissae do not extend posteriorly beyond the ear and the carpal vibrissae are not so long as the distance between their bases and the apical pad of the first digit. The correlation of long vibrissae with low temperature, is mentioned here merely because length and density of pelage were under consideration.
The most obvious and most exact correlation between change in climate and change in the animal is furnished by color. This is well shown in the one species, Mustela frenata, to which the following remarks apply unless indication is given to the contrary. The color of the upper parts varies from bay (blackish brown) in M. f. panamensis to buckthorn brown (light brown) in M. f. neomexicana. The color of the head varies from solid brown (white chin excepted) to contrasting black and white markings.
Dark color of the upper parts is associated with a large area of this color; the enlargement of this area is at the expense of the area of light color on the underparts. In the weasels of darkest color the upper parts occupy four-fifths of the circumference of the body (as measured in the anterior lumbar region) but in the lightest-colored weasels the upper parts comprise only two-thirds of the total circumference. In these light-colored animals the color of the underparts extends onto the underside of the tail and down the insides of the legs and over the feet whereas in the animals with the darkest upper parts the entire tail, feet, and legs below the knees ordinarily are of the same dark color as the upper parts. The length of the black tip on the tail varies inversely with the length of the tail, probably because the lightest-colored weasel has the longest tail. In some subspecies the black brush is almost half as long as the tail-vertebrae but in others is less than a fourth as long as the tail-vertebrae.
The extent of the color of the head, as well as the intensity of the color there, varies markedly and is correlated with climatic conditions. The extent and intensity of this dark color is greater in weasels inhabiting regions of heavy rainfall than in those inhabiting regions of sparse rainfall. Considering the geographic range of each subspecies of Mustela frenata, that of M. f. panamensis has the maximum of rainfall. Reference to the colored plate (1) will show that in M. f. panamensis (2) the black of the head is extended over all of the upper parts. M. f. macrura (1) of Perú, to the southward, is from an area of lesser rainfall and is correspondingly lighter colored. Returning to panamensis (2) as a starting point and proceeding northward to the range of nicaraguae (3), which also has lesser rainfall, thence another step northward to Guatemala, which has still less rainfall, the weasel there, M. f. goldmani (4) has the black extending posteriorly only to the shoulders. M. f. leucoparia (5) from Michoacán, and M. f. frenata (6) from Tamaulipas are from progressively more northern and also progressively drier regions. In M. f. frenata (6) the dark color extends posteriorly only to the ears and is blackish rather than black. In M. f. neomexicana (7) of the extremely arid parts of Durango, Arizona, and New Mexico the dark marking of the head is confined to a brown spot on the nose. Its geographic range is the most arid of those of all of the subspecies. The contrast between neomexicana (7) and panamensis (2) illustrates the great range of geographic variation in color which occurs in the one species. Continuing from the geographic range of neomexicana (specimen from Safford, Arizona) northwesterly 480 miles to Riverside, California (see 8, latirostra), 430 miles north to Point Reyes, California (see 9, munda), and finally 570 miles north to Tillamook, Oregon (see 10, altifrontalis), each place with more rainfall than the one farther south, another correlation of increasingly dark coloration with increasing amount of rainfall is illustrated.
This geographic variation, it should be remembered, is all within one species. It is the more significant still when we remember that the same correlation, with never an exception, occurs at hundreds of places within the geographic range of the species. A particular feature of climate, namely rainfall, and possibly therefore humidity, is concerned in this correlation. The same correlation, heavy rainfall and dark color, is shown also in the other species of North American weasels. The conclusion is unavoidable that climate, directly or indirectly, determines or influences the color of weasels.
The light facial markings appear in American weasels in two separate geographic areas. One is the southwestern United States, México and northern Central America. The second area is in the same latitude, in Florida and adjoining parts of Georgia and Alabama. In the western weasels the markings are white south of latitude 32° N. North of this latitude, the facial markings, if at all extensive, usually are of the same yellowish color as the underparts of the body. Weasels of southern California and its interior valley usually have these yellowish instead of white facial markings. The light facial markings, in this instance, white markings, attain their maximum extent in M. f. leucoparia of the southwestern margin of the tableland of México, at latitude 19° N. A gradual decrease in area of the light facial markings occurs both to the north and south; they disappear at 10° N in M. f. costaricensis and at 35° N at approximately the southern limits of range of M. f. arizonensis and M. f. nevadensis. In the mild climate of California the light (yellowish) facial markings are found at still higher latitudes. These light facial markings crop up as vestiginal remnants, consisting of a few white hairs, in some individuals of nearly all races of weasels.
In certain parts of the skull there are trends, in size and shape, which continue for long distances geographically. In other words, clines can be recognized. Changes in size and shape in some other parts of the skull are wavelike; change toward narrower rostrum, for example, is not progressive in a given geographic direction for any great distance. Length of the upper tooth-rows and zygomatic breadth, when expressed as percentages of the basilar length, and also the actual length of individual teeth vary geographically in the same wavelike fashion as does the width of the rostrum.
Size of the skull, on the other hand, shows a sustained trend for a long distance; it becomes progressively smaller from the southern United States southward to Columbia, South America. This clinal variation can be demonstrated by plotting on a graph, the basilar length, the zygomatic breadth, or the weight of the skull. Beginning at Mérida, Venezuela, and proceeding southward to increasing elevations in the mountains of South America, there is a reversal of the direction of the variation in this cline; weight of skull, for example, increases to the southward from Mérida for a considerable distance. A cline of decreasing width of the postorbital constriction of the skull is evident from Panamá north into Texas.
Variations in the tympanic bullae provide many characters useful in distinguishing weasels from different localities. Most of these characters have to do with degree of inflation of the bullae. Indirectly correlated with degree of inflation is first the extent of removal of the anterior margin of the bulla from the glenoid fossa and foramen ovale, and second the form (convex, flat, or concave) of the part of the squamosal bone between the foramen ovale and the anterior margin of the tympanic bulla. As one proceeds southward from, say, southwestern Kansas through the geographic range of the species Mustela frenata, there is a progressive deflation of the bulla, an increase in length of the space between its anterior margin and the foramen ovale, and the floor of the braincase in front of the bulla changes from ventrally concave to ventrally convex. (See figs. e and h of pl. 24 and figs. e and f of pl. 27.)
One extreme of this variation in bulla is shown in Mustela frenata neomexicana (fig. e of pl. 24), in which the anterior margin of the bulla (viewed from the ventral side) rises vertically from the floor of the braincase to form a 90-degree angle. The other extreme, the uninflated bulla, is in Mustela frenata panamensis (fig. e of pl. 27), in which the anterior margin of the bulla is not raised above the floor of the braincase. This variation is remarkable because it occurs within a single species. Otherwise, in the family Mustelidae, differences in the tympanic bullae as great as that between the two subspecies M. f. neomexicana and M. f. panamensis, occur only between genera. The need for caution in inferring the limits of variation for a particular structure in one species or genus, on the basis of variation in another group, is therefore obvious.
Speaking now of full species, the most inflated tympanic bullae in American weasels are in Mustela frenata, and more restrictedly in those subspecies of it which occur in the temperate region. Subspecies of M. frenata in Central and South America, as already noted, have less inflated bullae. The tropical weasel, Mustela africana, of the Amazon drainage of South America has the bullae still less inflated (see fig. i of pl. 39 and fig. f of pl. 40). The bullae are less inflated even than in the mink, subgenus Lutreola. In M. africana the cleidomastoideus, omotrachelian, levator scapulae, and rhomboideus profundus muscles take origin from a fossa on the mastoid bone, whereas in the forms with greatly inflated bullae these muscles take origin from a raised ridge or tubercle. Using Mustela frenata of the temperate region as a starting point and proceeding northward, a reduction in inflation of the tympanic bulla is seen also in that direction in that Mustela erminea has less inflated bullae. The bullae are less inflated in southern than in far northern (arctic) populations of Mustela erminea. In erminea the lesser inflation is real enough but at the same time there appears to be less inflation than actually exists, for the squamosal floor of the braincase is "pushed down." This places the anterior end of the tympanic bulla farther in the braincase than it otherwise would be. Although the anterior end of the bulla is flattened to the extent that it resembles the sharp edge of a splitting-wedge, inspection of the lateral and medial edges shows that in its central part the bulla is more inflated than it is in the weasels of Central and South America.
For reasons set forth later, M. erminea is judged to resemble the ancestral stem form more closely than does any one of the other three American species of weasels. If this judgment is correct, the shape of the tympanic bullae of the American weasels may be explained as follows: In the subspecies of Mustela frenata of the temperate regions of North America the bullae have most nearly been pushed out of the braincase and at the same time have undergone some enlargement. The subspecies of this same species in Central and South America represent an earlier stage in the evolution of American weasels and retain less inflated bullae—less inflated even than those of the southern subspecies of erminea. M. africana probably separated from the stem form at a still earlier time if we may judge by the lesser inflation of its tympanic bullae. There are other reasons for thinking that africana separated from the stem form earlier than M. frenata did. During the time that elapsed since the separation of M. frenata from the stem form, the tympanic bullae of M. erminea probably increased slightly in size, as probably also did the brain but without shoving the auditory complex forward from its former position.
DISTRIBUTION AND SPECIATION
Weasels of the subgenus Mustela are known from the Pleistocene but not from deposits laid down at an earlier time (see page 10). The Pleistocene weasels from Rancho La Brea of southern California and from Potter Creek Cave and Samwel Cave, both of northern California, are subspecifically indistinguishable from the weasels living in those same localities today. The other notable occurrence of weasels in the Pleistocene is in the Conard Fissure of Arkansas. Brown (1908:181, 182, pl. 17) names two kinds from the Fissure. One is an extinct subspecies (Mustela frenata gracilis) possibly of the species which occurs in the same region today and the other, Mustela erminea? angustidens, is an extinct subspecies of a species which occurs only farther north today. M. erminea came south, probably in front of one of the ice sheets, as did several other species of American mammals, now of more northern distribution, that left their remains in Conard Fissure. Mustela rixosa is not recorded as a fossil in America although it is known from the "Diluvial" deposits of the Old World; see Woldrich (1884:1000), who employs the name "Foetorius minutus n. sp.," and see also Zimmerman (1943:295-296).
The ermine, Mustela erminea, is the most generalized of the full species. For example, the number of teeth is as large as in any other species and greater than in certain species. The teeth are sharp-pointed, uncrowded, and individually less specialized than in any other American weasel. M1 has the inner half, or lobe, of approximately the same size as the outer lobe instead of much larger than the outer lobe (the outer lobe is the larger in several other species). The tympanic bullae are less inflated and less protruded from the braincase. The skull is rounded, and has no marked crests and ridges whereas the skulls of the other species are more pronouncedly modeled and sculptured. Therefore, it is possible to think of these other species as derived from M. erminea. A derivation in the reverse direction would be more difficult. From the foot soles of an ermine, or a weasel closely resembling an ermine, the more complex soles of Mustela africana could have been derived by a decrease in hairiness, although it would be necessary to suppose that the thenar pad has been retained in africana and has been lost in the living erminea. The alternate possibility, namely, that the thenar pad was a relatively recent acquisition in the africana line seems less probable. The tail of erminea is of "average" length and in size of entire animal erminea is intermediate between the other American weasels. Structurally, Mustela erminea appears to be nearest the stem form from which all of the living weasels ascended. Its present holarctic distribution is in harmony with the view that it is a direct descendant from the stem form because the stem forms of most of the known kinds of mustelids appear to have lived in the holarctic region. To be sure, Mustela erminea is regarded as having undergone some progressive change in structure, but less than the other weasels, in the period of time when the weasels were evolving from the stem form.
The least weasel, Mustela rixosa, seems to be an ancient type and to judge from the size and proportions of its parts, was differentiated from the erminea stem at a time earlier than were the other American Recent species of weasels. In size, in reduction of the tail, and in proportions of the skull, M. rixosa is, in each instance, the most aberrant of all the weasels, Mustela nivalis of Europe and western Asia included. This aberrancy results from the retention of certain primitive features, in the teeth and basicranial region, and from specialization in proportions of the skull. The skull is long, deep, and narrow. These proportions probably are adaptations permitting the animal to follow the smaller kinds of mice into their burrows. In most of that part of North America where erminea and rixosa occur together, erminea is a much larger animal and takes as prey almost all kinds of land vertebrates that it is powerful enough to kill. These include varying hares and ptarmigans. The least weasel, rixosa, can hardly manage such large prey and lives on the smaller rodents. Mustela rixosa may eat numbers of insects (see page 176 beyond),—a kind of food which Mustela erminea is not known to eat. Apparently the two species are able to live in the same areas because each eats a somewhat different kind of food than does the other and hence they do not compete to the point where one is crowded out by the other. This is the case in the latitudes where the two species of weasels are of different bodily size, but in the southernmost latitudes where these two species occur, erminea becomes almost as small as rixosa and only one of the species, to the exclusion of the other, occurs in a given area. All through the Rocky Mountains, south of Montana and in the territory west of these mountains all the way to the Pacific Coast, only the small subspecies of erminea is to be found. In the Alleghenies of the eastern United States only rixosa occurs. In New England where erminea approaches the size of rixosa, the latter is unknown. Probably this exclusiveness results from competition for food, although competition for dens, safe breeding places and other requirements of life may be involved.
The species erminea invaded the western United States and in the process of invasion probably developed there the small size appropriate to permit erminea to live in that latitude before it could do the same thing in the Appalachian region. Later than erminea, the least weasel, Mustela rixosa, which was small to begin with, also spread southward from the holarctic region, stopped short in the western United States at the northern boundary of the area in which erminea was of small size, but in the Appalachian region of the eastern United States continued on southward to the limits of temperature tolerant for it because erminea had not yet penetrated into that region and no other small carnivore was there to offer competition.
The long-tailed weasel, Mustela frenata, occurs mostly south of the regions inhabited by the ermine, and mostly south of the region inhabited by the least weasel which appears to live as well with frenata as with erminea. It is true that erminea and frenata occur in the same region, but this is a relatively narrow belt across the United States; and from within it a person cannot go far either north or south without reaching a region in which only one of the two species occurs. Exception has to be made for the Rocky Mountains and the Sierra Nevada, where erminea is of exceptionally small size. In these mountains and in the boreal mountainous parts of the intervening region of the United States, erminea and the large-sized frenata occur together over a wide area. Presumably the two occupy different ecologic niches, much as rixosa and frenata probably do where they occur together.
Most of the geographic range of the long-tailed weasel, M. frenata, is in the temperate region. Structurally, this species is the most advanced of the American weasels. Its dentition is the most highly specialized for cutting. M1 is relatively small and the inner lobe is slightly larger than the outer lobe. The skull, throughout, is more modeled than in the other species; the rostrum, the lower jaws and the teeth—all parts of the offensive equipment—are well developed relative to the corresponding structures in other weasels; the basicranial region exhibits an advanced stage of development in that the tympanic bullae show the maximum degree of inflation. Also, they are thrust far out of the braincase, thereby providing more room for the relatively larger brain which is protected by a more solidly built braincase than in erminea.
Several subspecies of Mustela frenata occur in the tropics, that is to say, south of the Mexican tableland and on the coastal plain to the east of it. Each is structurally more primitive than subspecies of the temperate region. As compared with Mustela frenata frenata of the temperate Mexican tableland the size in these tropical subspecies is smaller; the tail is shorter; the braincase and entire skull are less modeled; the postorbital breadth is more; the teeth are smaller; the deuterocone of P4 is not so far anterior to the protocone; the tympanic bullae are less inflated, are farther removed from the foramen ovale, and a larger proportion of each bulla is contained within the braincase. These features serve to set off from northern races of frenata all those subspecies of frenata which occur from southern México southward to the northern and western limits of the Amazon drainage of South America. The Amazon Basin is inhabited by another species, Mustela africana, having more primitive characters.
In the species frenata, the explanation for this abrupt change in characters between the animals of the temperate highlands and those of the tropical lowlands may be this: In the early Pleistocene, after the emergence of much or all of Central America took place, weasels distributed themselves over the Isthmus and into South America. These weasels were more generalized in structure than those now inhabiting the uplands of México. Failure of this stock of weasels often to cross some still-persisting water barrier, or failure of this stock to cross some water barrier that was widened or reformed because of a rise in sea level in some one of the interglacial periods of the Pleistocene cut the frenata stock into two or more parts. After the land connection was established or re-established and when the necessary precedent plants and rodents again had established themselves, the two groups of weasels, one from the northern tableland of México, and the other from the southern area of tropical complexion, met. The weasels of the frenata stock that reinvaded the area from the north probably did so by following along the chain of high volcanic cones and narrow uplifts. If and when a subsequent inundation occurred in some part of Central America, weasels were stranded on the adjacent mountains—converted into islands—only the higher parts of which were above water. Mustela frenata costaricensis and Mustela frenata goldmani may be examples of a northern stock of weasel that pushed southward in the highlands and became stranded for a short time. Following the latest emergence of land to provide a continuous highway between the two continents, weasels from the south and the insular populations, as for example, M. f. costaricensis, were the first to invade the low tropical areas most recently under water. When the Pleistocene history of Central America is better known, the facts will provide a useful means of testing the hypothesis that has been outlined immediately above.
As explained above, fossil specimens of M. frenata from deposits of the last half of Pleistocene time show that no appreciable change occurred in some areas, for example, in the vicinity of Hawver Cave and Samwel Cave of California, and that but slight change occurred in other areas, for example, in southern California (fossils from Rancho La Brea) and probably in the central United States (fossil from Conard Fissure). It is possible to imagine, therefore, that the two groups of weasels, one occurring southward only as far as the highlands of Central America and the other occurring in northern South America, had not differentiated sufficiently in the period of their isolation to prevent crossbreeding when they last came into contact. If the separation of the two groups had been maintained for a longer period, the two groups, tropical weasels and austral weasels, probably would have been so different when the two met as to prevent crossbreeding and they would have constituted two full species instead of only one.
Mustela africana is the most primitive of the American weasels. Some of the most important structural features that mark it as such are in the basicranial region. The tympanic bullae are less inflated than in other weasels, are pointed anteriorly and posteriorly, and do not have the lateral margins carried outward to the outer margins of the braincase. The mastoid sinus is not involved, by inflation or marked modification in the production of the auditory complex. Between the alisphenoid and the squamosal there is a clear demarcation posteriorly from a point directly lateral to the foramen ovale. This demarcation permits a transverse rounding of the alisphenoid to form a longitudinal ridge between the anterior margin of each bulla and the base of the pterygoid of the same side. Nevertheless, there is no such specialization of this primitive, structural feature such as occurs in some African and Asiatic mustelids in which the tympano-pterygoid part of the alisphenoid fuses with the tip of the hamulus of the pterygoid. However, the tympano-pterygoid eminence has not been obliterated in M. africana as it has in the other American weasels. Another primitive feature in the basicranial region of M. africana is the tendency toward separation of the paroccipital processes from the tympanic bullae. The thenar pad of the foot probably is an inheritance from a primitive ancestor since the pad is present in the viverrids and in a majority of mustelids judged to be more primitive than Mustela.
Some specializations are obvious in Mustela africana. One is the reduction in number of premolars; p2 is absent whereas it is normally present in the other weasels; P2 has one instead of two roots; and, in relation to the other teeth, m2 is smaller. The shortness of the preorbital part of the skull in relation to the length of the skull as a whole may reflect the mentioned reduction of the premolars or retention of a primitive shape of skull, or both. Also, certain features which denote immaturity in other weasels are retained in adults of this species, as for example, sutures on the dorsal face of the preorbital region of the skull.
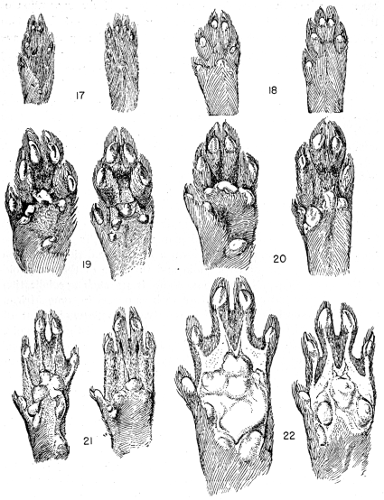
Figs. 17-22. Views of the feet of American weasels (subgenus Mustela) to show differences in number and arrangement of the pads and variation in degree of hairiness of the soles. × 1-1/2. In each figure, left-forefoot on left, and left hind foot on right.
Fig. 17. M. rixosa rixosa, Halifax, N.S.; juv., ♀, 7425 U.S.N.M.
Fig. 18. M. erminea richardsonii, Ft. Chimo; ad. ♀, 14866 U.S.N.M.
Fig. 19. M. frenata noveboracensis, Mich., July 7, 1913; ad. ♂, 44689 M.Z.
Fig. 20. M. f. frenata, Brownsville, June 1, 1892; yg. ♂, 34043 U.S.N.M.
Fig. 21. M. frenata panamensis, Panamá, February 17, 1911; sad. ♀, type.
Fig. 22. M. a. africana, Pará, Brazil, Sept., 1908; yg. ♂, 37475 A.M.N.H.
Figs. 17, 18 and 19. Drawn from specimens preserved in alcohol.
Figs. 20, 21 and 22. Drawn from relaxed feet of dried skins.
Mustela africana, all characters considered, is the most aberrant of the American weasels. That is to say, greater difference prevails between M. africana and any other American weasel than exists between any other two American weasels. The distinctive cranial and dental characters, excepting the reduction in number of premolars, are of a primitive nature. For example, the relatively wide postorbital region, the large braincase that is inflated anteriorly, and the flattened tympanic bullae are points of resemblance to the holarctic Mustela erminea, the species which is regarded as most closely resembling the stem form. Also, the mentioned characters in adults of M. africana resemble ontogenic stages passed through by other weasels. Consequently, it is thought that M. africana crossed the filter-barrier from North America to South America, remained isolated from the original stock for a length of time sufficient to permit africana to differentiate from North American weasels and vice versa to such a degree that crossbreeding with the frenata stock was prevented when frenata, at a later time, pushed southward over the, then zoölogically less-effective, water barrier, or continental bridge if it was by this time in existence.
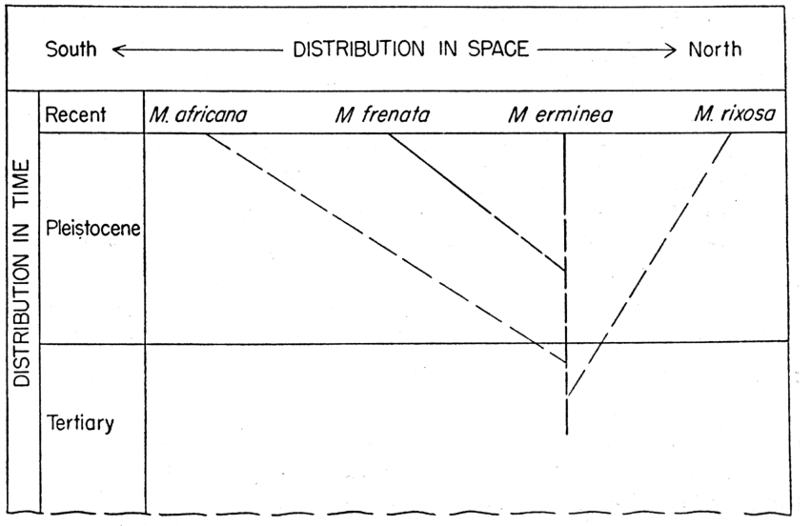
Fig. 23. Diagram indicating probable relationships of the species of American weasels.
The four full species of American weasels may well be thought of as having the same stem form of which erminea is the most nearly direct descendant. Geographic and climatic changes may have operated to isolate, and then to foster morphologic differentiation of, first rixosa in Eurasia, next africana, third the tropicalis section of M. frenata, and finally M. frenata itself, leaving M. erminea as a modern version, somewhat altered to be sure, of the stem form. Some of these ideas are expressed in figure 16. The climate is different in the ranges of the several species and the climate has changed through time in the ranges of at least many subspecies. Natural selection of morphological features best adapted to a particular kind of climate probably has altered some species more than others. M. erminea in almost every one of its characteristics is generalized and potentially progressive whereas africana retains more characters which are truly primitive along with a few which are specializations. M. africana is potentially the least progressive of any of the American weasels. The most specialized weasels are the North American races of Mustela frenata. A progressive series of increasing specialization is comprised in (1) M. africana, (2) the M. tropicalis (Central American, lowland) section of M. frenata, and (3) the races of M. frenata in North America.
Considering now features of the environment which have obviously influenced the distribution and speciation of weasels, water barriers are important. Bering Strait, Carquinez Strait (along with San Francisco Bay) which opens through the Golden Gate, and the channels between the islands of southeastern Alaska, have contributed to the formation of subspecies. The difference is really slight on the two sides of Bering Strait and San Francisco Bay and is slightly more on two sides of each of several of the channels between the islands of southeastern Alaska. The differences between the weasels on the two sides of one of these water barriers supposedly result from the preservation in animals on one side, or on one island, of small mutations, which would be swamped by crossbreeding if the water barrier were not present. The effect of this isolation is easily seen if ermines from the Queen Charlotte Islands are compared with those of the opposite mainland. The degree of morphological difference is great. Isolationwise, the Queen Charlotte Islands are the seaward end of a chain, beginning with Admiralty Island in southeastern Alaska, and are farther from the mainland, zoölogically, than the distance in actual miles across the water channel would suggest. Between any two islands that are geographically consecutive, however, and between the mainland and the first island of the chain, the difference in the ermines is small. In other places, water barriers of equal or greater width have contributed little if anything to the differentiation from one another of weasels on the two sides of the water barrier. The strait between eastern Canada and Newfoundland is an example.
The absence of water, or scarcity of it to a degree that closely approaches absence, in any large area appears to prevent weasels from living there. At any rate, the one sizeable region of North America from which weasels are unknown is the desert of the southwestern United States and adjoining part of northwestern México. More precisely, in western Arizona, the Mohave Desert and the desert of northwestern Sonora, collectors of mammals have repeatedly sought small carnivores without ever finding any weasels.
Degree of moisture is closely correlated with color in weasels. Humidity and cloudiness as well as actual precipitation seem to be involved. Even if we take into account average annual rainfall alone, the darkest-colored weasels are found in the areas of heaviest rainfall and the lightest-colored weasels in areas of lightest rainfall (extreme type of desert where no weasels occur being excepted). In any large region where there is a geographic gradient in rainfall, the transition from light to dark color almost exactly parallels the increase in amount of rainfall. Within a given species the same color reappears in widely separated areas that have the same amount and seasonal distribution of rainfall. This correlation is repeated so often that one can almost certainly say that heavy rainfall, or the associated phenomena of high humidity and cloudiness, acting separately or together, causes an increase in intensity of color. Relative extent of the color of the upper parts and underparts and presence and absence of light facial markings seem also to be correlated, in a more general way, with differences in rainfall. A fuller discussion of the nature and amount of the variation in color is given on page 51.
Temperature seems not to be an important factor in directly limiting the distribution of weasels, since M. frenata occurs from the hottest to some of the coldest parts of the Americas. Do M. erminea and M. rixosa range no farther south, than they do at present, because high temperatures constitute a barrier? No evidence is known to me which provides an answer, one way or the other, to this question. Granting that temperature is unimportant in limiting the distribution of weasels, it seems to cause geographic variation. Increase in mean annual temperature is correlated with decreased size in M. erminea and with increased size in M. rixosa. Temperature, it seems, causes the hair to vary; the pelage is harsher and sparser in weasels from tropical regions than in those from boreal regions. Difference in number of hairs is especially well shown on the soles of the feet. In the weasels from the far north, the pads are concealed by hair and in the weasels from the tropical regions the soles are mostly bare. Also, the hair on the soles of the feet is longer in northern than in southern weasels. Furthermore there is seasonal change in length of the hair on the soles of the feet; at a given locality in southern Canada the hair of the white winter coat is so long on the soles of the feet as to obscure completely the palmar and plantar pads whereas the hair of the brown summer coat is shorter and leaves these pads boldly exposed to view. This seasonal change, as would be expected, is most marked in animals of northern regions and is not perceptible in those from the tropics; it is correlated with increase in seasonal change as the distance from the equator increases.
Temperature and moisture acting together may cause extensive white facial markings, that neither alone would cause. In Mustela frenata these markings occur where there is heavy rainfall and high mean annual temperature. Where there is heavy rainfall and a low mean annual temperature they do not occur and where there is high mean annual temperature and light rainfall the markings are not pure white but are of the same color as the underparts. Plate 1 and the description of color on page 51 may be consulted in this connection. Extremely high mean annual temperature together with extremely heavy rainfall may inhibit the development of light facial markings. M. f. meridana, panamensis and costaricensis are cases in point. In either direction, north or south, from the territory inhabited by these three subspecies a similar combination of temperature and rainfall is found and similar light facial markings appear there.
Considering the delicate response of structure to climate, a person naturally questions whether or not natural selection accounts for all of the differences between subspecies. To show that natural selection determines the color of Mustela frenata, it would be necessary to assume that climate, color, and utility of color are positively correlated. Although climate (rainfall) and color are correlated in such a manner that three subspecies of weasel in places as far apart as New England, Perú, and the state of Washington are colored alike, other features of the three environments are unlike. Kinds of animals which the weasel catches for food, and flora in which the weasel finds concealment, are dissimiliar. If natural selection alone determined the color, some difference in color would be expected between the weasel which needed to be obliteratively colored, that is camouflaged, the better to catch a Phyllotis in Perú and the weasel in Washington which needed nature's aid in catching Microtus. Mustela frenata goldmani of the highlands of southern México, which is known to attack the huge pocket gophers, Orthogeomys and Cratogeomys, has a weaker dental armature than Mustela frenata texensis which does not have to overcome prey so formidable as does goldmani. Equally formidable enemies endanger M. f. goldmani and texensis. Examples of this nature could be multiplied. Without actually proving anything concerning selection, these examples give reason for us to suppose that some characters are not determined by natural selection.
Another question upon which data obtained from a study of Mustela has some bearing, is this: Where the geographic ranges of two subspecies meet, why does not the swamping effect of crossbreeding cause one subspecies to disappear? Although swamping may have occurred in some instances, it does not occur in the majority of instances. Witness the long-continued existence of the living subspecies Mustela frenata nevadensis of which skulls are available from Pleistocene deposits. Therefore, its distinctive characters, cranially at least, have been maintained for a long time. Furthermore, these characters are maintained over a large geographic region more than a thousand miles across. On the eastern margin of its range, at the eastern base of the Rocky Mountains in Colorado, M. f. nevadensis intergrades in a relatively narrow belt with the lighter-colored, longer-tailed and cranially different Mustela frenata longicauda, which has a geographic range almost equally extensive. M. f. longicauda also is uniform in its characters over a large area but at approximately 400 miles east of the base of the Rocky Mountains, it begins to intergrade with the darker-colored, shorter-tailed and cranially different Mustela frenata primulina and does so over a belt of 100 miles or more in width. At any given locality within this wide belt of intergradation the range of individual variation ordinarily does not exceed that in animals from a given locality well within the geographic range of M. f. longicauda. In the narrow belt of intergradation along the eastern base of the Rocky Mountains, the range of individual variation at several places is greater than in animals from a given locality well within the geographic range of M. f. longicauda or for that matter from well within the geographic range of M. f. nevadensis.
Considering the dominance and recessiveness of genes and the genetic mechanism in general by which characteristics of offspring are inherited from their parents, it would seem that M. f. longicauda and for that matter M. f. nevadensis and M. f. primulina would lose their distinctive characteristics because of the crossbreeding that is every year going on between longicauda and nevadensis on the one hand and between longicauda and primulina on the other hand.
Sumner (1932:84) suggests that homogeneity is prevented by population pressure. Applying his suggestion to the species Mustela frenata we could say that the subspecies longicauda pressing westward meets strong pressure from the subspecies nevadensis pressing eastward and that the width of the zone of intergradation between the two subspecies varies inversely with the strength of the population pressure from the two sides. Sumner recognizes that according to his hypothesis the two contiguous races would remain distinct only so long as there was a preponderance of centrifugal movement from both of the centers of dispersal. Sumner (op. cit.:85) recognizes that an abrupt change of environmental conditions could account in part for the boundaries of the ranges of the two subspecies and finally that his hypothesis does not certainly answer the question of why crossbreeding does not result in homogeneity between two subspecies with contiguous geographic ranges.
The hypothesis of harmoniously stabilized complexes of genes was offered by Timofeeff-Ressovsky (1940:124) to explain why the swamping effect of crossbreeding does not obliterate subspecies. The hypothesis takes into account that any one of several characters of a subspecies may be caused by several genes. Some characters of this kind may be favored by natural selection more than others. In the belt of intergradation between two subspecies, where two of these favored characters meet, a "biological tension" as Huxley (1939:415) terms it "will result, which will produce partial discontinuity between the two groups. Each group will evolve a gene-complex which is not only broadly adapted to the external environment of the central area of its range, but is also harmoniously stabilized, in adaptation to the internal genetic environment, by the selection of modifiers." Crosses, that is to say intergrades, between the two subspecies will lack this stabilization and will therefore be at a selective disadvantage. The zone of intergradation will therefore remain narrow; intermediates are constantly being brought into existence there by crossing but are as constantly being extinguished by selection.
These two hypotheses are the best that geneticists yet have offered. Neither has been tested and both, as originally proposed, would hardly apply everywhere because there are some contradictions.
I can offer no better explanation—in fact no original one as good—but would emphasize that under similar climate, weasels remain constant in character, or at most do not vary beyond certain limits. Crossing at the margins of ranges of two subspecies does not result in homogeneity of weasels. There is, therefore, some stabilizing influence, or influences, that maintain, and even develop, structural characteristics of weasels in opposition to the contrary tendency of crossing.
That this influence not only maintains uniform characters over areas of large extent, but also permitted their development over large geographic areas, must logically be supposed, for otherwise, considering the swamping effect of crossing, such variations would not have made their appearance in more than a few individuals. Also, if the races had been formed in response to some kind of physiological differentiation, or other non-climatic cause, the characters of the population in the belt of intergradation probably would disappear in a short time. In any event the close correlation between degree of change in weasels and degree of change in climate, at once makes one suspect that climate has been the deciding factor. Finally, when one recalls that in certain parts of the animal, certain characters invariably appear under similar climates and never under dissimilar climates, the evidence is almost conclusive that, given long enough time, the animals vary in response to climate. The variations (characters) may be induced indirectly, but are no less exactly reproduced than if they can be shown to be induced directly.
In considering how the species and subspecies of American weasels were formed and in attempting to account for some of the individual characters, it is profitable to view the facts in the light of some of the theories of species-formation—theories that are accessory to that of organic descent and that are concerned with the modus operandi of organic descent.
In any group of closely related species some of them, by the laws of chance, are almost certain to be more primitive than others. Mustela is no exception and the more primitive species closely match, in several characters, ontogenetic stages passed through by more advanced species. Jaeckel's (1902) theory of metakinesis, therefore, is to be considered since it postulates that many cases of epistasis occur; that is to say, that many sexually adult animals are arrested in development in early otogenetic stages and undergo no further development. Although this theory is appealing upon initial consideration, it is less so when we recall that in Mustela there is a direct correlation of increasingly primitive structure with decreasing latitude as one proceeds from the steppe of North America southward to the equator. It follows that the conditions seen in Mustela can be explained even better than by metakinesis, by assuming that the several species have differentiated from a stem form at different times, have developed at different rates, have developed in different directions and that ontogeny recapitulates phylogeny.
The theory of Age and Area (see Willis, 1922) holds that the species of widest distribution are, on the average, the oldest, and that the species which are distributed over small areas are, in general, of recent origin. So far as the weasels are concerned, little support is given to this theory. The same can be said of any one of the teological theories, including the orthogenesis of post-Darwinian writers. All of these imply a determinate line of variation controlled by the inherent qualities of the organism. The idea that the several species of Mustela result from mutations of large degree and sudden appearance is contrary to the evidence accumulated. In fact the evidence rather clearly indicates that the mutations which may have occurred were of small degree and in most instances owe their preservation to natural selection.
The data obtained by the study of weasels accords almost exactly with the theory of species-forming embodied in Matthew's (1915) "Climate and Evolution." Although the essential features of this theory were made out from a study of families and orders and therefore would not be expected to apply to members of only a genus or subgenus, the facts known about the present distribution of American Mustela, nevertheless, are strikingly in accord with the ideas advanced by Matthew. In the first place, climate is an important factor in the evolution of the weasels. In the second place, the line of migration seems to have been outward from the holarctic region. In the third place, the geographic changes necessary to explain the present distribution of the species of Mustela are not extensive and do not affect the permanency of oceans as defined by the continental shelf. These three statements are, almost verbatim, those made in the first three of the five points of Matthew's (1915:172-173) thesis. The remaining two points of Matthew's thesis have to do with generalizations based on evidence obtained from sources outside the scope of the present study.
Furthermore, the relative degrees of specialization of the different species and subspecies in relation to their geographic distribution are in accord with the ideas elaborated by Matthew. For instance, the most primitive species is farthest south from the probable center of dispersal, the holarctic region. Also the full species become progressively more primitive as one proceeds southward from the holarctic, or at least from the northern half of the nearctic, region. Although, in view of the known geological changes that have occurred in the Caribbean region, we cannot say that the more primitive species owe their positions entirely to having been pushed farther south from the center of dispersal by actual and continuous contact and competition with the more advanced species, this seems to have been the case in a general way. At any rate the more primitive kinds seem to have been prevented from pushing northward by the more advanced kinds which developed there and the latter have actually pushed southward.
Additionally and in review: There is strong indication that the American species of weasels were formed by gradual and slow change. Much of this change probably is the result of natural selection operating on fortuitous variations of a minor nature, but, also, particular features of the environment, especially climate, and more especially amount of rainfall, seem to compel variations that differentiate subspecies and that characterize full species—compel some of them without the direct operation of natural selection, or at least compel them within limits so wide that natural selection exerts no exact control.
HISTORY OF CLASSIFICATION
In the earlier accounts of American weasels, from the time of Linnaeus and before, up until 1890, names then in use for European weasels frequently were applied also to those in North America. For the next 50 years, and almost without exception after 1896, the American weasels were regarded as specifically distinct from those in the Old World. In this 50-year period many new names were proposed, usually as full species, although now that material from more localities has been brought together and studied, geographic intergradation is evident between many of the named kinds and most of these names now therefore take only subspecific rank. In 1933 Glover M. Allen showed that Mustela rixosa occurred also in the Old World, and in 1943 I emphasized that a second American species, Mustela erminea, was circumpolar in distribution. In neither rixosa, nor erminea, however, were the subspecies the same in the two continents. To this general outline of the nomenclature, exception must be made for weasels of the southwestern United States, México and Central America, and South America, because as early as 1813 a distinctive name was given to one of these and weasels from the three areas mentioned were, so far as I know, never given names of Old World kinds.
The first paper that could be regarded as revisionary in nature was "Remarks on the species of the genus Mustela" by the zoölogist and world-traveler, Charles L. Bonaparte, in Charlesworth's Magazine of Natural History, for 1838. In that paper three new names, Mustela cicognanii, M. richardsonii and M. longicauda, all still valid, were proposed for American weasels.
Audubon and Bachman in their "Quadrupeds of North America," which appeared in parts from 1845 to 1853, recognized 5 species. Actually they were dealing with only 3 taxonomically valid kinds. For one of these, Mustela frenata noveboracensis, they were misled by the difference in size between males and females, and in the males by the presence of a brown coat in some and a white coat in others. The male that was white in winter they regarded as Putorius ermineus of the Old World; the male that was brown in winter they designated by their earlier proposed name P. fuscus, and the female they named P. agilis. The ermine, subspecies M. erminea cicognanii, they called P. pusillus. Their fifth name, P. frenatus, included at least some animals that today are assigned to the subspecies M. frenata frenata. Each of three and perhaps four of the five names employed by Audubon and Bachman embraced individuals of more than one species and in that sense the names were composite.
Only five years later, in 1858, Professor Spencer Fullerton Baird's great work, "The Mammals of North America," made it clear that no American weasel was identical (in the modern subspecific sense) with any Old World weasel, and he applied most of his names in a correct zoölogical sense. It is true that he thought that the female weasel of the eastern United States was specifically different from the male, misapplied to it the name richardsonii, and did not correctly allocate every one of the few poor specimens available to him of the little ermine (M. e. streatori) of the Pacific Coast; but he did recognize that the least weasel was a distinct kind and his treatment in general was excellent.
After Baird came a period of great confusion in which most writers did no better than had Audubon and Bachman, ordinarily confusing the two sexes as different species, and, in 1877 in his "Fur-bearing Animals," Elliot Coues went rather to the other extreme and allowed only 4 kinds to all of the Americas, regarding two of these, for purposes of zoölogical nomenclature, as identical with the European species.
But, in 1896 Outram Bangs published "A Review of the Weasels of Eastern North America" in which he correctly recognized eight kinds. Although some of these were treated by him as full species, whereas the material accumulated since 1896 has shown that subspecific status is in order, his names, still in use, were correctly applied in every instance, save probably one. This was his use of Putorius richardsonii for the animal now known as M. e. arctica. Unlike the earlier, excellent treatment by Baird, this accurate one by Bangs was heeded and followed by subsequent writers. For example, Dr. C. Hart Merriam in the same year, 1896, accepted Bangs' conclusions except for correcting the application of the name richardsonii. The principal contributions of Merriam's paper "Synopsis of the Weasels of North America" were first, the wider geographic scope and second, the naming as new of several kinds outside the geographic area studied by Bangs. Otherwise the work was not up to Dr. Merriam's usual standard and the internal evidence of haste in its preparation and the superficial study of some of the material at his disposal explain why the weasels of North America since that time have been but little better understood than in 1896. Baird and Bangs, then, unquestionably did the best systematic work on the American weasels.
In 1916 Dr. Joseph A. Allen published a valuable paper on the South American weasels. The material available to him was inadequate and prevented a thoroughly satisfactory treatment. There are too few specimens even today to permit of a thorough treatment of the South American weasels in the present paper; nevertheless the material today is more nearly adequate than it was in 1916 and it is hoped that the systematic arrangement is correspondingly improved.
Chronological List (annotated) of Specific and Subspecific Names Applied to American Weasels
At least eighty-seven specific and subspecific names have been proposed for American weasels. Of these sixty-nine are now regarded as valid designations of recognizable subspecies. The average is 1.2 names per subspecies. Some names in the following chronological list were a second time applied wholly or in part to some other kind of weasel. In general, mention of the second or any other later application is omitted from the following list but two usages of agilis (1844 and 1853) and of americana (1865) are recorded.
1734. javonica (Mustela) Seba, Locupletissimi Rerum naturalium Thesauri ..., 1:77, 78, pl. 48, fig. 4. The weasel to which this name was applied was said to have come from Java. Since no animal answering to the description has again been found in Java, and because specimens from Central America or possibly some from northern India, may do so, it is conceivable that Seba was the first to distinguish by name an American weasel from those in the Old World. My attempts to locate the specimen concerned in places where it might have been preserved along with some of the other specimens thought to have belonged to Seba have been fruitless. Since it is impossible positively to link Seba's description with any known weasel, no further use is made of the name javonica in the present account.
1772. erminea (Mustela) Forster [= Mustela erminea richardsonii], Philos. Trans., London, 1772:373. Forster's use of the name is one of the earliest applications of it to American animals. The name dates from Linnaeus, Syst. Naturae, (10th ed.) 1:46, 1758, with type locality in Europe. In the subspecific sense the name applies to the ermine which occurs over most of the Scandinavian Peninsula, if Miller (1912:387) be followed in regarding the type locality as Upsala, Sweden. If, instead, Cabrera (1913A:394-396) be followed in regarding the type locality as in Switzerland, the name, in the subspecific sense, will apply to the ermine of continental Europe. As the earliest available name applied to the circumpolar species concerned, it is used now as the name of the species in the New World as well as in the Old World. From the time of Forster until approximately 1890 the name erminea by many, but not by all, authors was applied to the American weasels in the belief that they were zoölogically indistinguishable from those in the Old World. From 1896 to 1943 the name was not used by American authors at all because the ermine of America was in 1896 treated nomenclaturally by Merriam as specifically distinct from the animal in the Old World. Since 1943 erminea has been used in the specific sense for American animals in recognition of the circumpolar distribution of the species. Some of the early allocations of American specimens to erminea probably resulted in a composite use of the name in that one or another subspecies of the American species Mustela frenata may also have been included with individuals truly of the species erminea.
1772. nivalis (Mustela), Forster, Philos. Trans., London, 1772:373. This is one of the early applications of this name to American weasels of small size, made in the belief that they were taxonomically the same in America and Europe. Linnaeus, Syst. Nat. (12th ed.) 1:69, 1766 is the authority for the name [Mustela] nivalis, and the Province of Vesterbotten, Sweden, is regarded as the type locality. The name is in use today for the common weasel of Europe and parts of Asia. Animals of the species nivalis are intermediate in size between Mustela erminea and Mustela rixosa. The name as used for American animals by some authors who wrote later than Forster did, probably was composite in that these authors may have applied the name to the small weasels of North America and thus may have intended it to apply not only to Mustela erminea cicognanii but also to females of Mustela frenata noveboracensis, and conceivably to both sexes of Mustela rixosa of any American subspecies.
1813. Brasiliensis (Mustela) Sevastianoff, Mem. Acad. Imp. Sci. St. Petersburg, 4:356-363, table (= plate) 4. This name was proposed for a weasel brought to St. Petersburg by Capt. Krusenstern on his return from a voyage around the world. The animal was said to have come from Brazil, but to judge from the description, came instead from México, Central America, or west of the Andes in South America, and was based on some one of the subspecies of Mustela frenata. Although the name was in use for more than 60 years it was shown by Merriam (1896:27) to be unavailable because it was preoccupied by Mustela brasiliensis, a name earlier used by Gmelin (Syst. Nat., ed. 13, p. 93, 1788) for a South American otter.
1815. vulgaris (Mustela), Ord, Guthrie's Geography as reprinted by Rhoads in 1894, vol. 2, p. 291. This use by Ord is one of the earliest applications of this name to American weasels, in the belief that the smaller weasels of North America and Europe were zoölogically the same; [Mustela] vulgaris seems originally to have been proposed in 1777 by Erxleben on p. 471 of vol. 1 of his Syst. Regni Anim., for the weasel of the temperate part of Europe and to be a synonym of Mustela nivalis Linnaeus (1766). Probably the name as used by Ord was composite in the sense that he may have intended it to apply to females of Mustela frenata noveboracensis as well as to one or both sexes of Mustela erminea cicognanii and, if he ever saw them, to the two sexes of Mustela rixosa (one or several subspecies).
1818. africana (Mustela) Desmarest [= Mustela africana africana], Nouv. Diction. d. Hist. Nat., 19:376. In 1808 E. Geoffroy St.-Hilaire visited Portugal and was given several African primates and the specimen of Mustela named by Desmarest in 1818 who wrongly supposed that it, like most of the primates, came originally from Africa. After the name had been misapplied for 95 years Angel Cabrera showed that it pertained instead to the tropical weasel of Brazil. Of distinctive names applied to American weasels today, this is the one first proposed.
1832. frenata (Mustela) Lichtenstein [= Mustela frenata frenata], Darstellung neuer oder wenig bekannter Säugethiere, pl. 42 and corresponding text unpaged. This name is the first one available for the long-tailed weasel and therefore applies to the species as a whole.
1838. Cicognanii (Mustela) Bonaparte [= Mustela erminea cicognanii], Charlesworth's Mag. Nat. Hist., 2:38. The name erroneously spelled Cigognanii was correctly spelled on page 39. For a detailed consideration of this name see the account of the subspecies cicognanii on page 120.
1838. Richardsonii (Mustela) Bonaparte [= Mustela erminea richardsonii], Charlesworth's Mag. Nat. Hist., 2:39. Until 1896 the name sometimes was applied to the subspecies now known as M. e. arctica and sometimes to part of the subspecies now designated as M. e. cicognanii under the principal treatment of which see (page 120) for a detailed account of the basis of the name richardsonii, and the reasons for regarding Fort Franklin as the type locality.
1838. longicauda (Mustela) Bonaparte [= Mustela frenata longicauda], Charlesworth's Mag. Nat. Hist., 2:39. The type locality appears to be Carlton House, Saskatchewan, and the name always seems to have been applied to the long-tailed weasel of the Great Plains, although in some earlier accounts the name was used in a more inclusive sense to refer also to animals now of subspecies closely allied to longicauda. As with the two preceding names, a detailed consideration of the basis for, and application of, this name is given on pages 120-123 in the account of Mustela erminea cicognanii.
1840. Noveboracensis (Putorius) Emmons [= Mustela frenata noveboracensis], Quadrupeds of Mass., p. 45. This name was credited by Emmons to De Kay who in the same year published it in his report on the "Zoology of New York" but without a description and De Kay's name is a nomen nudum. Emmons' was the first use of the name accompanied by a recognizable description and therefore the name must date from Emmons although this obviously was not his intent since he credited the name to De Kay.
1842. fuscus (Putorius) Audubon and Bachman [= Mustela frenata noveboracensis], Jour. Acad. Nat. Sci., Philadelphia, 8: (pt. 2) 288.
1842. pusilla (Mustela) De Kay [= Mustela erminea cicognanii], Nat. Hist. of New York, Zool., Pt. 1, Mammalia, p. 34. This name was proposed for small weasels of 12 to 13 inches in length of which the tail amounted to a fourth of the same and although obviously applying in considerable part to the earlier named M. e. cicognanii seems to have included some individuals of the also earlier named M. f. noveboracensis.
1843. xanthogenys (Mustela) Gray [= Mustela frenata xanthogenys], Ann. and Mag. Nat. Hist., 11:118, February, 1843, was applied to all of the long-tailed weasels of California that had light-colored facial markings. Merriam in 1896 suggested that San Diego was the type locality and in 1899 Bangs proposed the name mundus for the California weasel north of San Francisco Bay thus restricting the application of the name xanthogenys. In 1936 Hall further restricted the application of the name and applied it to the long-tailed weasel of the big interior valley of California, pointing out that the name was correctly applied to this weasel of the big interior valley or possibly instead to the race named munda.
1844. agilis (Mustela) Tschudi [= Mustela frenata agilis], Untersuch. ü. die Fauna Peruana, p. 110, is a name applied today to the race of weasel of the Temperate Zone of the western Andes and intermountain valleys of Perú.
1851. nigripes (Putorius) Audubon and Bachman [= Mustela nigripes], Quadr. N. Amer., 2:297, 1851, applies to the black-footed ferret of North America.
1853. agilis (Putorius) Audubon and Bachman [= Mustela frenata noveboracensis], Viv. Quadrupeds N. Amer., 3:184, pl. 140. This name was proposed for the female in the mistaken belief that it was specifically distinct from the larger male for which several names already were available. Also Tschudi in 1844 had already used the name Mustela agilis for a South American weasel.
1864. aureoventris (Mustela) Gray [= Mustela frenata aureoventris], Proc. Zoöl. Soc. London, 1864:55, pl. 8, February 9, 1864, is the name applicable to the dark-colored weasel of the Pacific coastal region of Ecuador and Columbia.
1865. americana (Mustela erminea Var. 3) Gray, Proc. Zoöl. Soc. London, 1865:111. The larger individuals of American weasels of both Mustela erminea and Mustela frenata from the Atlantic Coast to as far west as Carlton House, Saskatchewan, were lumped under this name because Gray desired more information than he then had before recognizing as different from one another several species proposed for America up to the time concerned. The name is unavailable because it is preoccupied by Mustela americana Turton (1806) the name for the American marten.
1865. americana (Mustela vulgaris Var.) Gray, Proc. Zoöl. Soc. London, 1865:113. Under this name the smaller weasels of the northern and northeastern part of North America were lumped by Gray but the name is preoccupied and can be ignored.
1874. affinis (Mustela) Gray [= Mustela frenata affinis], Ann. and Mag. Nat. Hist., 14 (ser. 4):375, 1874, from New Granada [= Colombia], had the type locality restricted to Bogotá, Colombia, by Allen in 1916, and is applied to the long-tailed weasel of the tropical and temperate zones of the eastern Andes of Colombia.
1874. macrura (Mustela) Taczanowski [= Mustela frenata macrura], Proc. Zoöl. Soc. London, for 1874, p. 311, pl. 48, May 19, 1874, applies to the long-tailed weasel of central Perú and northern Ecuador.
1877. culbertsoni (Putorius) Coues [= Mustela frenata longicauda], Fur-bearing animals ..., p. 136, 1877, is based on specimens from Fort Laramie, Wyoming. In the past the name has been regarded as a nomen nudum but there is some reason for regarding it as having nomenclatural status. In either event it is here arranged as pertaining to the long-tailed weasel of the Great Plains which takes the prior name longicauda. See the account of longicauda for a more detailed account of the name culbertsoni.
1877. aequatorialis (Putorius (Gale) brasiliensis) Coues [= Mustela frenata aureoventris], Fur-bearing animals ..., p. 142. Proposed "merely as a substitute for Gray's [supposedly] preoccupied name," aureoventris.
1881. stolzmanni (Mustela) Taczanowski [= Mustela africana stolzmanni], Proc. Zoöl. Soc. London, for 1881, p. 835, November 15, 1881, is applied to the tropical weasel of the Upper Amazon Basin.
1881. jelskii (Mustela) Taczanowski [= Mustela frenata macrura], Proc. Zoöl. Soc. London, for 1881, p. 647, May 17, 1881, was proposed for the female in the mistaken opinion that it was specifically distinct from the larger male which the same author previously had named macrura.
1891. arizonensis (Putorius) Mearns [= Mustela frenata arizonensis], Bull. Amer. Mus. Nat. Hist., 3:234, June 5, 1891, until 1936 was applied to long-tailed weasels of most of the western United States west of the Great Plains but by restriction since 1936 has been applied only to the animals in parts of Arizona and New Mexico.
1894. peninsulae (Putorius) Rhoads [= Mustela frenata peninsulae], Proc. Acad. Nat. Sci. Philadelphia, 1894:152, June 19, 1894, applies to the weasel of central and southern Florida.
1896. alascensis (Putorius richardsonii) Merriam [= Mustela erminea alascensis], N. Amer. Fauna, 11:12, June 30, 1896, with type locality at Juneau, Alaska, has been used for the ermine of southeastern Alaska ever since it was proposed. In 1944 separate subspecific rank was accorded ermines on several of the islands of southeastern Alaska which proportionately restricted the range assigned to alascensis.
1896. streatori (Putorius) Merriam [= Mustela erminea streatori], N. Amer. Fauna, 11:13, June 30, 1896, applies to the ermine of the Pacific Coast from Puget Sound, Washington, south nearly to the Golden Gate of California.
1896. arcticus (Putorius) Merriam [= Mustela erminea arctica], N. Amer. Fauna, 11:15, June 30, 1896. Ever since it was proposed, this name has been applied to the subspecies of ermine of Alaska and the northern parts of Canada.
1896. kadiacensis ([Putorius arcticus]) Merriam [= Mustela erminea kadiacensis], N. Amer. Fauna, 11:16, June 30, 1896, is a valid name applied to the ermine of Kodiak Island, Alaska.
1896. washingtoni (Putorius) Merriam [= Mustela frenata washingtoni], N. Amer. Fauna, 11:18, June 30, 1896, applies to the long-tailed weasel of the southern Cascades of Washington and the northern Cascades of Oregon.
1896. saturatus (Putorius) Merriam [= Mustela frenata saturata], N. Amer. Fauna, 11:21, June 30, 1896, was little used until 1936 but applies to long-tailed weasel of limited region in northern California and southern Oregon.
1896. alleni (Putorius) Merriam [= Mustela frenata alleni], N. Amer. Fauna, 11:24, June 30, 1896, applies to weasel of Black Hills region.
1896. oregonensis (Putorius xanthogenys) Merriam [= Mustela frenata oregonensis], N. Amer. Fauna, 11:25, June 30, 1896, applies to long-tailed weasel of parts of western Oregon and northern California.
1896. goldmani (Putorius frenatus) Merriam [= Mustela frenata goldmani], N. Amer. Fauna, 11:28, June 30, 1896, applies to the long-tailed weasel of Chiapas, and parts of Guatemala and Salvador.
1896. leucoparia (Putorius frenatus) Merriam [= Mustela frenata leucoparia], N. Amer. Fauna, 11:29, June 30, 1896, applies to the long-tailed weasel of Michoacán and Nayarit.
1896. tropicalis (Putorius) Merriam [= Mustela frenata tropicalis], N. Amer. Fauna, 11:30, June 30, 1896, applies to the long-tailed weasel of the Tropical Life-zone of Veracruz.
1896. spadix (Putorius longicaudus) Bangs [= Mustela frenata spadix], Proc. Biol. Soc. Washington, 10:8, February 25, 1896, applies to the long-tailed weasel of Minnesota and adjoining areas.
1896. rixosus (Putorius) Bangs [= Mustela rixosa rixosa], Proc. Biol. Soc. Washington, 10:21, February 25, 1896, applies to the least weasel of Saskatchewan and adjoining areas and as the first available name for the species has been used as the specific name for the species in America since 1896.
1897. paraensis (Putorius (Mustela) braziliensis) Goeldi [= Mustela africana africana], Zool. Jahrb., abt. f. systematik, geogr. u. biol., 10:560, pl. 21, September 15, 1897, a synonym for the weasel of the lower Amazon area.
1898. neomexicanus (Putorius frenatus) Barber and Cockerell [= Mustela frenata neomexicana], Proc. Acad. Nat. Sci. Philadelphia, p. 188, May 3, 1898, applies to the long-tailed weasel of New Mexico, Arizona, Durango and adjoining areas.
1898. haidarum (Putorius) Preble [= Mustela erminea haidarum], Proc. Biol. Soc. Washington, 12:169, August 10, 1898, applies to the ermine of the Queen Charlotte Islands, British Columbia.
1899. notius (Putorius noveboracensis) Bangs [= Mustela frenata noveboracensis], Proc. New England Zoöl. Club, 1:53, June 9, 1899, was applied to the long-tailed weasel of the Carolinas until 1936 since which time it has been regarded as a synonym of noveboracensis.
1899. occisor (Putorius) Bangs [= Mustela frenata occisor], Proc. New England Zoöl. Club, 1:54, June 9, 1899, applies to the long-tailed weasel of central and northern Maine. Until 1936, occisor was ordinarily used as the name of a full species but since then has been arranged as a subspecific name under Mustela frenata.
1899. mundus (Putorius xanthogenys) Bangs [= Mustela frenata munda], Proc. New England Zoöl. Club, 1:56, June 9, 1899, is now applied, and generally has been since 1899, to the long-tailed weasel of the coastal district of California north of San Francisco Bay.
1899. muricus (Putorius (Arctogale)) Bangs [= Mustela erminea muricus], Proc. New England Zoöl. Club, 1:71, July 31, 1899, applies to the diminutive ermine, often erroneously designated least weasel, of the western United States.
1899. oribasus (Putorius (Arctogale) longicauda) Bangs [= Mustela frenata oribasus], Proc. New England Zoöl. Club, 1:81, December 27, 1899, applies to the long-tailed weasel of the Rocky Mountains northward from Yellowstone National Park.
1900. eskimo (Putorius rixosus) Stone [= Mustela rixosa eskimo], Proc. Acad. Nat. Sci. Philadelphia, 1900:44, March 24, 1900, is applied to the least weasel of Alaska and adjacent parts of boreal North America.
1901. allegheniensis (Putorius) Rhoads [= Mustela rixosa allegheniensis], Proc. Acad. Nat. Sci. Philadelphia, 1900:75, March 25, 1901, applies to the least weasel of the eastern United States.
1902. perdus (Putorius tropicalis) Merriam [= Mustela frenata perda], Proc. Biol. Soc. Washington, 15:67, March 22, 1902, applies to the long-tailed weasel of the Lower Tropical Life-zone from southern Veracruz into Guatemala.
1903. microtis (Putorius) Allen [= Mustela erminea richardsonii], Bull. Amer. Mus. Nat. Hist., 19:563, October 10, 1903, is a name applied to an individual ermine of small size from Shesley, British Columbia, which Allen thought was specifically distinct from the ermine of the Hudsonian Life-zone and adjacent territory. Now the name is arranged as a synonym of richardsonii.
1904. audax (Putorius) Barrett-Hamilton [= Mustela erminea arctica], Ann. and Mag. Nat. Hist., ser. 7, 13:392, May, 1904. In the original description the type locality, Discovery Bay, was erroneously stated to be in Greenland and the name audax until 1945 was applied to the kind of weasel occurring in northern Greenland whereas the type specimen was taken instead in northern Ellesmere Island and because the weasel there is subspecifically indistinguishable from ermines from farther west, audax is a synonym of Putorius arcticus.
1904. imperii (Putorius arcticus) Barrett-Hamilton [= Mustela erminea richardsonii], Ann. and Mag. Nat. Hist., ser. 7, 13:392, May, 1904, based on an animal from Fort Simpson, Mackenzie, Canada, proves to be inseparable from richardsonii which has priority.
1904. polaris (Putorius arcticus) Barrett-Hamilton [= Mustela erminea polaris], Ann. and Mag. Nat. Hist., ser. 7, 13:393, May, 1904, is the name used for the ermine of eastern Greenland and since 1945 has been used for the weasel of Greenland as a whole.
1905. macrophonius (Putorius) Elliott [= Mustela frenata macrophonius], Proc. Biol. Soc. Washington, 18:235, December 9, 1905, applies to the long-tailed weasel of the mountains along the eastern border of Veracruz.
1906. leptus (Putorius streatori) Merriam [= Mustela erminea murica], Proc. Biol. Soc. Washington, 16:76, May 29, 1903, until 1945 was applied to the diminutive ermine of the Rocky Mountains from Wyoming south to northern New Mexico but proves to be a synonym of muricus with type locality in the Sierra Nevada of California.
1908. angustidens (Putorius cicognanii) Brown [= Mustela erminea angustidens], Mem. Amer. Mus. Nat. Hist., 9(pt. 4):181, pl. 17, is applied to an extinct subspecies known from fossil remains of Pleistocene age from northern Arkansas.
1908. gracilis (Putorius) Brown [= Mustela frenata gracilis], Mem. Amer. Mus. Nat. Hist., 9(pt. 4):182, 1908, applies to a Pleistocene weasel known from a single skull from northern Arkansas.
1912. costaricensis (Mustela) Goldman [= Mustela frenata costaricensis], Proc. Biol. Soc. Washington, 25:9, January 23, 1912, applies to the long-tailed weasel of Costa Rica.
1913. primulina (Mustela) Jackson [= Mustela frenata primulina], Proc. Biol. Soc. Washington, 26:123, May 21, 1913, applies to the long-tailed weasel of the central part of the United States in eastern Kansas and adjoining areas.
1913. campestris (Mustela) Jackson [= Mustela rixosa campestris], Proc. Biol. Soc. Washington, 26:124, May 21, 1913, applies to the least weasel of the Great Plains region.
1913. olivacea (Mustela peninsulae) Howell [= Mustela frenata olivacea], Proc. Biol. Soc. Washington, 26:139, May 21, 1913, applies to the long-tailed weasel of the southeastern United States excepting most of Florida.
1914. meridana (Mustela) Hollister [= Mustela frenata meridana], Proc. Biol. Soc. Washington, 27:143, July 10, 1914, applies to the long-tailed weasel of northern South America.
1916. nicaraguae (Mustela tropicalis) Allen [= Mustela frenata nicaraguae], Bull. Amer. Mus. Nat. Hist., 35:100, April 28, 1916, applies to the long-tailed weasel of Nicaragua.
1927. arthuri (Mustela noveboracensis) Hall [= Mustela frenata arthuri], Proc. Biol. Soc. Washington, 40:193, December 2, 1927, applies to the long-tailed weasel of Louisiana and adjoining areas.
1932. semplei (Mustela arctica) Sutton and Hamilton [= Mustela erminea semplei], Ann. Carnegie Mus., 21(2):79, February 13, 1932, originally was applied to the ermine of Southampton Island but after 1945 was applied also to the ermine of Baffin Island, Melville Peninsula and the west side of Hudsons Bay as far south as Eskimo Point.
1932. panamensis (Mustela frenata) Hall, Proc. Biol. Soc. Washington, 45:139, September 9, 1932, applies to the long-tailed weasel of Panamá.
1932. anguinae (Mustela cicognanii) Hall [= Mustela erminea anguinae], Univ. California Publ. Zoöl., 38:417, November 8, 1932, applies to the ermine of Vancouver Island, British Columbia.
1935. labiata (Mustela arctica) Degerbøl [= Mustela erminea semplei], Rept. 5th Thule Exped., 1921-1924, vol. 2, no. 4, p. 25, 1935. When Degerbøl wrote his description and proposed this name he was unaware that Sutton and Hamilton had three years before based a new name on weasels from Southampton Island. Because the two names apply to the same subspecies, Degerbøl's name, labiata, must fall as a synonym of semplei which has priority.
1935. helleri (Mustela frenata) Hall, Proc. Biol. Soc. Washington, 48:143, August 22, 1935, applies to the long-tailed weasel of eastern Perú.
1936. nevadensis (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 91, November 20, 1945, applies to the long-tailed weasel of the western United States. For many years, animals of this subspecies were referred to longicauda and from 1891 until 1936 to arizonensis.
1936. effera (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 93, November 20, 1945, applies to the long-tailed weasel of the Blue Mountains region. From 1891 until 1936 this animal was referred to under the name arizonensis.
1936. altifrontalis (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 94, November 20, 1936, applies to the long-tailed weasel of the humid coastal district from Puget Sound southward into Oregon.
1936. nigriauris (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 95, November 20, 1936, applies to the long-tailed weasel of the coastal district of California from San Francisco Bay southward to Point Concepcion. Previous to 1936, xanthogenys was the name applied to this race of weasel.
1936. latirostra (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 96, November 20, 1936, applies to the long-tailed weasel of southern California which previously had borne the name xanthogenys.
1936. pulchra (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 98, November 20, 1936, is applied to the long-tailed weasel of the southern end of the San Joaquin Valley of California.
1936. inyoensis (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 99, November 20, 1936, is applied to the long-tailed weasel of Owens Valley, California.
1936. texensis (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 99, November 20, 1936, applies to the long-tailed weasel of central Texas which previously had been assigned to the subspecies frenata.
1936. perotae (Mustela frenata) Hall, Carnegie Inst. Washington, publ. no. 473, p. 100, November 20, 1936, applies to long-tailed weasel of the mountains along the Puebla-México boundary.
1938. boliviensis (Mustela frenata) Hall, Proc. Biol. Soc. Washington, 51:67, May 18, 1938, applies to the southernmost known long-tailed weasel which is in the Lake Titicaca region in Perú and Bolivia.
1944. salva (Mustela erminea) Hall, Proc. Biol. Soc. Washington, 57:35, June 28, 1944, applies to the ermine of Admiralty Island, southeastern Alaska.
1944. initis (Mustela erminea) Hall, Proc. Biol. Soc. Washington, 57:37, June 28, 1944, applies to the ermine of Baranof and Chichagof islands, southeastern Alaska.
1944. celenda (Mustela erminea) Hall, Proc. Biol. Soc. Washington, 57:38, June 28, 1944, applies to the ermine of Prince of Wales, Dall and Long islands, Alaska.
1944. seclusa (Mustela erminea) Hall, Proc. Biol. Soc. Washington, 57:39, June 28, 1944, applies to the ermine of Suemez Island, southeastern Alaska.
1945. invicta (Mustela erminea) Hall, Jour. Mamm., 26:75, February 27, 1945, applies to the ermine of the Rocky Mountains for several hundred miles both north and south of the United States-Canadian boundary.
1945. fallenda (Mustela erminea) Hall, Jour. Mamm., 26:79, February 27, 1945, applies to the ermine of the coastal mainland in southern British Columbia and northern Washington.
1945. olympica (Mustela erminea) Hall, Jour. Mamm., 26:81, February 27, 1945, applies to the diminutive ermine of the Olympic Peninsula, state of Washington.
1945. gulosa (Mustela erminea) Hall, Jour. Mamm., 26:84, February 27, 1945, applies to the diminutive ermine of the Cascades in Washington.
1945. bangsi (Mustela erminea) Hall, Jour. Mamm., 26:176, July 19, 1945, is the name applied today to the ermine of the western Great Lakes region.
In 1925 when this study was begun, the American weasels (subgenus Mustela proper) were arranged as belonging to 47 kinds (including subspecies) of 29 full species. In the present account a total of 68 kinds, belonging to 4 full species are recognized in the subgenus Mustela. The increase in number of subspecies and the decrease in number of species are the nomenclatural results ordinarily obtained in this decade from a systematic study of a genus of American mammals.
CHECK-LIST OF AMERICAN SPECIES AND SUBSPECIES OF THE GENUS MUSTELA
| Subgenus MUSTELA Linnaeus | |
| PAGE | |
| Mustela erminea | 87 |
| Mustela erminea arctica (Merriam) | 96 |
| Mustela erminea polaris (Barrett-Hamilton) | 103 |
| Mustela erminea semplei Sutton and Hamilton | 105 |
| Mustela erminea kadiacensis (Merriam) | 108 |
| Mustela erminea richardsonii Bonaparte | 110 |
| Mustela erminea cicognanii Bonaparte | 118 |
| Mustela erminea bangsi Hall | 124 |
| Mustela erminea invicta Hall | 128 |
| Mustela erminea alascensis (Merriam) | 131 |
| Mustela erminea salva Hall | 135 |
| Mustela erminea initis Hall | 136 |
| Mustela erminea celenda Hall | 139 |
| Mustela erminea seclusa Hall | 141 |
| Mustela erminea haidarum (Preble) | 142 |
| Mustela erminea anguinae Hall | 145 |
| Mustela erminea fallenda Hall | 148 |
| Mustela erminea olympica Hall | 153 |
| Mustela erminea streatori (Merriam) | 155 |
| Mustela erminea gulosa Hall | 159 |
| Mustela erminea muricus (Bangs) | 161 |
| Mustela erminea angustidens (Brown) | 165 |
| Mustela rixosa | 168 |
| Mustela rixosa eskimo Stone | 181 |
| Mustela rixosa rixosa Bangs | 184 |
| Mustela rixosa allegheniensis Rhoads | 187 |
| Mustela rixosa campestris Jackson | 190 |
| Mustela frenata | 193 |
| Mustela frenata noveboracensis (Emmons) | 222 |
| Mustela frenata occisor (Bangs) | 230 |
| Mustela frenata primulina (Jackson) | 232 |
| Mustela frenata arthuri Hall | 241 |
| Mustela frenata olivacea Howell | 244 |
| Mustela frenata peninsulae Rhoads | 250 |
| Mustela frenata spadix (Bangs) | 252 |
| Mustela frenata longicauda Bonaparte | 262 |
| Mustela frenata oribasus (Bangs) | 270 |
| Mustela frenata alleni (Merriam) | 274 |
| Mustela frenata arizonensis (Mearns) | 276 |
| Mustela frenata nevadensis Hall | 280 |
| Mustela frenata effera Hall | 291 |
| Mustela frenata washingtoni (Merriam) | 294 |
| Mustela frenata saturata (Merriam) | 297 |
| Mustela frenata altifrontalis Hall | 300 |
| Mustela frenata oregonensis (Merriam) | 304 |
| Mustela frenata munda (Bangs) | 309 |
| Mustela frenata xanthogenys Gray | 315 |
| Mustela frenata nigriauris Hall | 319 |
| Mustela frenata latirostra Hall | 323 |
| Mustela frenata pulchra Hall | 328 |
| Mustela frenata inyoensis Hall | 331 |
| Mustela frenata neomexicana (Barber and Cockerell) | 333 |
| Mustela frenata texensis Hall | 338 |
| Mustela frenata frenata Lichtenstein | 341 |
| Mustela frenata leucoparia (Merriam) | 347 |
| Mustela frenata perotae Hall | 351 |
| Mustela frenata goldmani (Merriam) | 355 |
| Mustela frenata macrophonius (Elliot) | 360 |
| Mustela frenata tropicalis (Merriam) | 363 |
| Mustela frenata perda (Merriam) | 366 |
| Mustela frenata nicaraguae Allen | 370 |
| Mustela frenata costaricensis Goldman | 372 |
| Mustela frenata panamensis Hall | 375 |
| Mustela frenata meridana Hollister | 379 |
| Mustela frenata affinis Gray | 384 |
| Mustela frenata aureoventris Gray | 387 |
| Mustela frenata helleri Hall | 391 |
| Mustela frenata agilis Tschudi | 393 |
| Mustela frenata macrura Taczanowski | 398 |
| Mustela frenata boliviensis Hall | 402 |
| Mustela frenata gracilis (Brown) | 404 |
| Subgenus Grammogale Cabrera | |
| Mustela africana | 406 |
| Mustela africana africana Desmarest | 409 |
| Mustela africana stolzmanni Taczanowski | 413 |
| Subgenus Putorius Cuvier | |
| (Black-footed Ferret-not treated in present work) | |
| Mustela nigripes (Audubon and Bachman) | |
| Subgenus Lutreola Wagner | |
| (Minks-not treated in present work) | |
| Mustela vison | |
| Mustela vison vison Schreber | |
| Mustela vison mink Peale and Beauvois | |
| Mustela vison lutensis (Bangs) | |
| Mustela vison evergladensis Hamilton | |
| Mustela vison vulgivaga (Bangs) | |
| Mustela vison letifera Hollister | |
| Mustela vison lacustris (Preble) | |
| Mustela vison energumenos (Bangs) | |
| Mustela vison evagor Hall | |
| Mustela vison aestuarina Grinnell | |
| Mustela vison nesolestes (Heller) | |
| Mustela vison melampelus (Elliot) | |
| Mustela vison ingens (Osgood) | |
| Mustela macrodon (Prentiss) |
ARTIFICIAL KEY TO AMERICAN SPECIES OF THE GENUS MUSTELA
| PAGE | |
| A Length of upper tooth-rows less than 20 mm. in males and 17.8 mm. in females. | |
| B Postglenoid length of skull more than 47 per cent of condylobasal length. | |
| C Tail without a black pencil and with at most a few black hairs at extreme tip; in both sexes mastoid breadth ordinarily exceeds breadth of braincase, | |
| Mustela rixosa, least weasel, p. | 168 |
| C' Tail with a black pencil; in females mastoid breadth ordinarily exceeded by breadth of braincase, | |
| Mustela erminea, ermine, p. | 87 |
| B' Postglenoid length of skull less than 47 per cent of condylobasal length. | |
| D Tail with distinct black tip; midventral line white, yellowish, orange, not same color as upper parts; p2 present; thenar pad on forefoot absent, | |
| Mustela frenata, long-tailed weasel, p. | 193 |
| D' Tail without black tip; midventral line same color as upper parts; p2 absent; thenar pad on forefoot present, | |
| Mustela africana, tropical weasel, p. | 406 |
| A' Length of upper tooth-rows more than 20 mm. in males and 17.8 mm. in females. | |
| E Abdomen all white; face with blackish mask; m1 lacking even a trace of a metaconid; distance between upper canines more than width of basioccipital as measured between foramina situated midway along medial sides of tympanic bullae, | |
| Mustela nigripes, black-footed ferret. | |
| E' Abdomen dark brown, like back; face uniformly brown without blackish mask; m1 with incipient metaconid; distance between upper canines less than width of basioccipital as measured between foramina situated midway along medial sides of tympanic bullae, | |
| Mustela vison, mink, American mink. |
DIAGNOSIS OF THE GENUS
Genus Mustela Linnaeus
Weasels, Ferrets, Polecats, Minks
Genotype.—Mustela erminea Linnaeus.
Diagnosis.—Legs short; body relatively long; adults 190 mm. to 700 mm. in total length; skull ranging in basilar length from 16 to 70 mm.; facial angle slight; tympanic bullae greatly inflated (moderately in Lutreola), cancellous, and with paroccipital processes closely appressed to bullae; palate behind upper molars; dental formula:
I 3 C 1 P 2-3 M 1 -, -; -, -; -, ---; -, -; i 3 c 1 p 3-2 m 2
inner moiety of M1 larger than outer; P4 with simple deuterocone; in m1 trigonid longer than talonid, metaconid absent (incipiently developed in Lutreola), and talonid trenchant.
For many years prior to 1911, the name Mustela was applied to martens, and Putorius was regarded as the first available generic name for the weasels. In 1911 Thomas (1911:139) showed that M. erminea (Mustela of Gesner) by tautonymy was the type of Mustela and subsequently the generic name Mustela has been used for the true weasels which include the American weasels to which we now apply the specific names erminea, rixosa and frenata. The mink, Mustela (Lutreola) vison, and the black-footed ferret, Mustela (Putorius) nigripes, since 1911 also have been referred by most American authors to the genus Mustela, the names Lutreola and Putorius being regarded by these authors as having no more than subgeneric status. European writers, on the other hand, accord greater taxonomic weight to the zoölogical differences between ferrets and weasels and, therefore, accord full generic rank to Putorius. Consequently, for the black-footed ferret, Europeans today write Putorius nigripes and Americans write Mustela nigripes. For the same reasons, the name of the mink is written by some European zoölogists Lutreola vison and by American zoölogists Mustela vison.
EXPLANATION OF SYSTEMATIC TREATMENT
For each full species there will be found under the account of it the following information: Type, statement of geographic range, selected characters for ready recognition, other characters of the species, a summary of geographic variation, and information on habits, in the order mentioned.
For each subspecies, information is presented in the following order: earliest available zoölogical name, synonyms, type, geographic range, zoölogical characters for ready recognition, description (mentioning size, certain external features including color, the skull and teeth) historical material when warranted, remarks which may elaborate on points made in preceding paragraphs, and other information thought to be useful, and finally a list of specimens examined.
In explanation of certain of these categories it should be said that in the synonymy no attempt is made to list every published reference to the subspecies concerned. It is aimed, however, to include at least one citation to each name-combination that has been applied, to the subspecies concerned, along with other especially important references. Mere records of occurrence are not regarded as especially important and citations to them ordinarily are omitted in the synonymy. No comma is placed between the zoölogical name and the name of the author who coined and first used the name in accordance with the rules of zoölogical nomenclature. Otherwise a comma is interposed between the zoölogical name and the name of the user (author). When the accepted (earliest available) name of a subspecies at the head of any one of the following accounts is combined with a generic name different from that with which it originally was placed, the authority for the name is set in parentheses. The same rule is followed with the name of a full species when it is written without any subspecific name following. Parentheses in such situations, therefore, denote that for the terminal part of the scientific name there has been a change in generic name with which the terminal part of the scientific name is here associated.
In the paragraph headed "characters for ready recognition," only a few characters, namely, those regarded as most useful for identification when the student has limited time, are mentioned. Other features useful for distinguishing the kind of animal in question from its near relatives are to be found in the description and comparisons.
In the description, external measurements, unless otherwise indicated, are those recorded by the collector on the label attached to the skin. Total length is the distance from the tip of the pad on the nose to the tip of the fleshy part of the tail when the relaxed animal is laid out straight, not stretched. This measurement does not include the hairs that project beyond the end of the fleshy part of the tail. Length of tail is the distance from the base of the tail, when it is bent at right angles to the long axis of the body, to the tip of the fleshy part of the tail excluding the hairs that project beyond the fleshy part of the tail. Length of tail and length of tail-vertebrae are synonymous. Length of hind foot is measured from the proximal end of the calcaneum to the tip of the longest claw.
Capitalized color terms, unless otherwise indicated, refer to Ridgway's (1912) Color Standards and Color Nomenclature. Some use is made of color terms taken from Oberthür and Dauthenay (1905) because those authors show a much larger number of shades between dark brown and black than does Ridgway (1912). The colors of the upper parts of most weasels are some shade or other of dark brown. Color terms that do not have the initial letter capitalized do not refer to any one standard and consequently are used in a general sense.
Relative extents of the color of the upper parts and underparts are computed from measurements of the circumference of the body at the place where the color of the underparts is narrowest. Ordinarily this place is in the lumbar region rather than in the thoracic region.
An explanation of how cranial measurements were taken is given on page 417. In designating teeth, capital letters are used for teeth in the upper jaw and lower case letters are used for teeth in the lower jaw. For example: I2 denotes the second incisor tooth in the upper jaw and i2 denotes the second incisor tooth in the lower jaw; C1 and c1 refer to the canine tooth of the upper jaw and lower jaw, respectively; P3 and p3 refer to the third premolar of the upper jaw and lower jaw, respectively, bearing in mind that the first (anterior) premolar is absent in the lower jaw and upper jaw of weasels (see fig. 31 on page 416), as also, in some kinds of weasels, is the second premolar; M1 and m1 refer to the first molar of the upper jaw and lower jaw respectively.
In describing the skull and teeth the two sexes are treated separately because differences in shape as well as size are the rule. Unless otherwise indicated, the skulls on which descriptions are based are of adults. Weights of skulls include the weight of the lower jaws. In general, every second subspecies is described. For a subspecies geographically next adjacent to the one described, only the differences between the two are enumerated. This method of description indicates also likenesses and is more economical of words than some other methods of description. Also, by use of this method, cross reference is reduced to one other subspecies. Following this formal description, there is a comparison of the cranial and dental characters with those of geographically adjacent subspecies.
In the paragraph headed "Remarks" the two words "character" and "structure" frequently appear. The word structure here is used to mean some part of an animal, as for example, a hair, a muscle, a bone, or an internal organ. A structure is not a system, as for example, the digestive system or osseous system. A character is some weight, linear dimension, volume, shape, color, or other perceptible attribute of a structure, of a system, or of an entire organism.
In recording the localities of capture of specimens examined, effort has been made to be exactly as precise as the locality data on the labels of the specimens permit. The word "County" is written out in full when the name of the county is written on the label of each specimen listed from that county. When one specimen, or more, here assigned to a given county lacks the name of the county on the label, then the abbreviation "Co." is used. The surprising frequency with which the same place name is repeated in a given state or province makes it desirable for the collector to write the name of the county, or corresponding minor political subdivision, on labels of study specimens at the time they are prepared.
SYSTEMATIC ACCOUNTS OF SPECIES AND SUBSPECIES
MUSTELA ERMINEA Linnaeus
Ermine
(Synonymy under subspecies)
Type.—Mustela erminea Linnaeus, Systema Naturae, 10th ed., p. 46, 1758.
Range.—From the British Isles and Atlantic Coast of Europe across Eurasia and North America including Greenland, from the northernmost land, south, in North America, to the lower margin of the Canadian Life-zone; geographically south to Connecticut, New York, Pennsylvania, northeastern Ohio, southern Michigan, Wisconsin, northern Iowa, Minnesota, North Dakota, in the Rocky Mountains to northern New Mexico, in the Sierra Nevada to Mono County, California, and on the Pacific Coast to the Golden Gate.
Characters for ready recognition.—Differs from Mustela rixosa in presence of black pencil on tail, tail-vertebrae more than a fourth of length of head and body, and in regions where the two species occur together, basilar length of skull more than 32.5 in males and more than 31.0 in females; from Mustela frenata, in regions where the two species occur together, by tail less than 44 per cent of length of head and body and by postglenoidal length of skull more than 46 per cent of condylobasal length in males and more than 48 per cent in females.
Characters of the species.—Size medium to small (total length 225 to 340 mm. in males and 190 to 290 mm. in females); tail 30 to 45 per cent of length of head and body, with distinct black pencil; caudal vertebrae 16 to 19; skull with long braincase and short precranial portion; postglenoidal length, when expressed as a percentage of the condylobasal length, more than 48 in females and ordinarily more than 46 in males; upper parts brown; underparts whitish, ordinarily continuous from chin to inguinal region but in subspecies in the humid region along the Pacific Coast interrupted in some individuals by brown of upper parts encircling body in the abdominal region. The soles of the feet in each of the subspecies are densely haired in winter and have only a relatively small area of the foot-pads exposed in summer, the intervening areas being well haired even at that season. The uniformity throughout the species as regards hairiness of the foot-soles and also the character of the vibrissae makes it unnecessary to describe these features in the accounts of the subspecies of erminea.
Geographic variation.—In the Old World 16 or more subspecies are currently recognized and there are 20 in North America. The features in which geographic variation is especially prominent are: First, size, as expressed by external measurements and weight, second, color pattern, depending on the extent, in relation to one another, of the dark-colored upper parts and light-colored underparts, and third, breadth and depth of the rostral region of the skull. Except in size, the variation in the skull is less than in M. frenata. Likewise in tone and shade of upper parts and hue or tint of underparts, erminea is less variable than frenata and has the face all of one color without the contrasting color-pattern of the face and head seen in many subspecies of frenata. M. erminea exceeds frenata as regards variation of the size of the area occupied by the light-colored underparts. At one extreme is the subspecies arctica in which the area of the light color extends well up on the sides of the body, down the insides of the legs, over the feet and far out on the lower side of the tail whereas at the other extreme are the races streatori and olympica in which the light-colored underparts are restricted to two areas, one on the chin, throat and chest, and the other on the inguinal region. These areas may or may not be connected by a thin line of white color along the midline of the underparts. In size of animal, erminea probably exhibits the maximum variation among American species of weasels; an average-sized male of the race arctica weighs 4 times as much as one of the race muricus, and in the species frenata I doubt that the difference is quite as great between individuals of the smallest race, effera, on the one hand, and either of the largest races, texensis or macrophonius, on the other hand although actual weights are not available for these races of frenata. As elsewhere indicated, the small-sized individuals of M. erminea are of the southern races and the large-sized individuals are of the northern races. This decrease in size southward occurs both in Asia and in America.
Natural History.—Habitat and numbers.—Along the International Boundary east of the Turtle Mountains, Soper (1946:136) found this species present only in timbered areas and absent from many untimbered areas. Of the same species to the westward he comments "so far as I know at present, there is no evidence to show that any short-tailed weasels inhabit a broad strip of treeless territory immediately north of the International Boundary in Canada from southwestern Alberta to southeastern Saskatchewan." The same author (1942) reports that in the general area of Wood Buffalo Park, Northwest Territory, south of Great Slave Lake, the ermine is uncommon on pine-grown sand ridge and rolling upland and common in lower spruce-aspen parklands, stream-side coniferous belts, and grassy, semi-wooded swamplands.
Nine ermines per square mile is the number that Soper (1919:46-47) estimated at Edmonton on the basis of the numbers that he trapped there in the winters of 1912-13 and 1913-14 and on the basis of the tracks of remaining ermines. From corresponding data he estimated the population in the winter of 1913 on the Hay River, north of Jasper Park, to be nine per square mile. In each of these instances he estimated ten weasels per square mile but he inclined to the view that one-tenth of the animals involved in his counts were long-tailed weasels (Mustela frenata). Osgood (1909B:30) and his field companion in the period July 31 to September 3, 1903, took a series of 42 specimens within a radius of 500 yards of their camp at the head of Seward Creek, Alaska, all caught in four traps, in one month. Of the 42 specimens, 28 are males and 14 are females.
Fluctuations of a multiannual nature are marked in this species. Bailey (1929:156) observes that in Sherburne County, Minnesota, when meadow mice are abundant for two or three years these weasels become abundant but that when the mice are scarce the weasels also become scarce. Manning (1943:56), on Southampton Island, noted "that the maximum and minimum points of the weasel cycle are much more sharply marked than those of the fox cycle and the increase and decrease are more rapid."
How far an ermine will travel in a given length of time has seldom been recorded but Hamilton (1933:293), on March 20, 1932, "followed the track of a small weasel, presumably a male cicognanii, for four miles in the fresh snow," and Ingles (1942) observed a diminutive ermine of the subspecies M. e. muricus, at Woods Lake, California, 286 yards from its den.
Behavior
As regards locomotion, Soper (1919:46), in reference to Mustela cicognanii, presumably in Ontario, Canada, writes that in the bounding gait the hind feet register almost, if not exactly, in the front-foot impressions, with the right front and hind feet lagging slightly behind. "The distance normally is about 19 inches, representing a regular rate of travel. . . . In traversing open spaces they resort to long, graceful leaps upwards of six feet in length. . . . I measured a record . . . of 8 feet, 2 inches."
Of M. e. arctica, Dice (1921:22) writes that when it runs "the tail is carried off the ground usually at an angle of about 45 degrees." Seton (1929 (2):598) states that "At Carberry [Manitoba] I have often seen this energetic little creature seeking for Mice in the deep, soft snow. Its actions are much like those of an Otter pursuing salmon. Sometimes it gallops along a log, or over an icy part of the drift; then plunges out of sight in a soft place, to reappear many yards away. . . ."
Little is recorded concerning swimming but on this score Seton (1929 (2):602) does quote J. W. Curran, who in July, 1899, at Lake Couchiching, Ontario, watched an ermine pursue a chipmunk into the water and for 100 yards before giving up the chase and wheeling around and making for shore. In swimming "The Weasel, I think, showed more of his body, and seemed to exert himself more" than the chipmunk.
As to voice, Dice (1921:22), at Tanana, Alaska, heard the ermine, when excited, bark somewhat like a mink but not so loud and Seton (1929 (2):606) quotes Manley Hardy to the effect that the species has a purring note.
Sense of smell was used by an M. e. muricus that Dixon (1931:72) watched as the ermine followed a three-fourths-grown pika. Concerning the ermine at Carberry, Manitoba, Seton (1929 (2):598-599) writes that "The smell of blood must be as far-reaching as it is attractive to these sanguinary little creatures. I have frequently hung new-killed Rabbits and partridges temporarily in trees, and, after an absence, in some cases of a few minutes only, have found an Ermine mauling the game, though there was no sign of such a visitor when the cache was made."
Enemies
George Measham, of Winnipeg, found sign in the snow indicating that a great snowy owl had killed an ermine and T. McIlwraith shot a bald eagle at Hamilton Bay which had the bleached skull of a weasel (probably of this species) clinging to the throat (Seton, 1929 (2):603).
A. B. Howell (1943:98) likens mustelid mammals to domestic cats in their manner of crossing roads and thinks that mustelids loiter at the side of the road until the stimulus of the approaching car causes them to make a dash whereupon they are caught by the wheels and killed. Three of four weasels seen to cross the road were killed, one even having apparently crossed the road before turning back and being killed under the car. One weasel killed was Mustela erminea cicognanii. Dalquest (1948:190) in writing of this species in the state of Washington, says "I have seen only one abroad in the daytime. It dashed from a roadside thicket . . . and was crushed beneath the wheels of a car."
Food
The killing of prey is described by Hamilton (1933:332) as follows: "A rapid dash, and the bird or mouse is grabbed over the back of the skull, the fore legs encircle the animal as though hugging it, and the hind legs are brought up to scratch wildly at the captive. . . . If [the prey is] a large animal, as a rat, the weasel usually lies on its side, while the diminishing struggles of the rodent continue, but if a mouse or a small bird [is the object of attack], the weasel is apt to crouch over its prey. Little time is lost over the first [mouse] . . . if two mice are present [;] a strong bite through the brain case . . . [is] sufficient. If only one animal is present, the weasel dawdles over its kill some time after life has departed."
Hamilton's (1933:333) study of the contents of the digestive tracts of bodies of ermines obtained from fur trappers and fur buyers in New York enabled him to publish the following "Frequency Indices of Mammal Genera in Fall and Winter Food of 191 Mustela cicognanii": Microtus, 35.7 per cent; mammals undetermined to genus but principally mice, 16.3; Blarina, 15.1; Peromyscus, 11.4; Sylvilagus, 9.0; Sorex, 4.9; Rattus, 4.4; Tamias, 3.6. Close correspondence is shown by the following data of Aldous and Manweiler (1942) for the ermine from Lake of the Woods, Minnesota: mice, 58.7 per cent by number and 54.5 by volume; shrews 22.5 and 21.8 per cent; birds, 2.7 and 5.0 per cent. Of the mice in stomachs, 40 per cent were microtines, 15 per cent were Peromyscus and 45 per cent were unidentified as to kind. Fragments of a small fish were found in one stomach. Summed up, the dominant winter foods were mice and shrews. Trapping of the mammal populations was done to see what the available food was and it was found that the small mammals were eaten in direct ratio to their relative abundance. Snowshoe rabbits and red squirrels were not eaten. The Minnesotan data were from 60 stomachs and 53 intestinal tracts recovered from 129 weasels trapped by use of scent (not bait) mostly from January 1 to February 7, 1939, although a few were trapped in 1938. Analyses of contents from stomachs gave approximately the same results as those from intestines. In 1939 at Lake of the Woods, weasels were concentrated where food was abundant but no such concentration was noted in the following winter.
Big short-tailed shrew (Blarina brevicauda).—In New York State, the ermine preys on Blarina as shown by Hamilton's (1933:330) seeing one being carried by a male ermine on May 6, 1931, and another being carried by a female on May 13, 1932. The same author (1928:249) found the remains of a Blarina in a small female from Malone, New York. Kirk (1921) observed, however, that the ermine (M. e. cicognanii) avoided the shrew, Blarina, caught in a trap and that Blarina avoided the weasel caught in a trap.
Chipmunk (genus Tamias).—Remains were found in a male ermine in New York on May 14, 1932 (Hamilton, 1933:330), and Seton (1929 (2):602) records a chipmunk at Lake Couchiching, Ontario, that was pursued into the water by an ermine.
Deer mice (genus Peromyscus).—As shown by Hamilton (1933:33) and Aldous and Manweiler (1942), Peromyscus was second only to microtines in numerical abundance among the food items of ermines in New York and Minnesota. Peromyscus and microtine rodents were brought to a den of the diminutive M. e. muricus in early August, in Fresno County, California, according to Ingles (1942). He observed that an Alpine chipmunk was active under and around the tree and that juncos reared young 40 feet from the den but that the chipmunk and juncos were unmolested by the ermines.
Lemming (genus Lemmus).—One was recovered from a female ermine (with milk in her glands) at Laurier Pass, British Columbia (Sheldon, 1932:201).
Red-backed mouse (genus Clethrionomys).—Criddle and Criddle (1925:146) record that on "May 31, 1921.—Saw a Bonaparte's weasel capture a Red-backed Vole after a long hunt during which the pursuer never once lost track of its victim."
Meadow mice (genus Microtus).—As shown by the data of Hamilton (1933:333) and Aldous and Manweiler (1942) recorded above, Microtus is the item of first importance in the diet of the ermine in New York and Minnesota. Criddle and Criddle (1925:146) write concerning the vicinity of Treesbank, Manitoba, that "October, 1918.—Following a severe outbreak of mice in 1916-17, Bonaparte's weasel increased enormously and very soon reduced the rodents to comparative rarity. This resulted in a scarcity of food for the weasels, which in their turn became greatly reduced in numbers."
Old World rat (Rattus).—Bishop (1923) found two headless rats near a nest of this species in Albany, New York.
Pika (Ochotona).—Dixon (1931:72) at Milner Pass, Colorado, on July 20, 1931, saw an ermine, of the subspecies muricus, following a three-fourths grown pika by scent and outrunning the pika. The pikas worked a relay system and the weasel abandoned the trail when the fourth pika became the object of the chase.
Cottontail (genus Sylvilagus).—Hamilton (1933:33), as noted above, found remains of cottontail in the digestive tracts of ermine that had been trapped for fur in winter. Possibly these remains were bait that had been placed at traps.
Snowshoe rabbit (Lepus americanus).—Morse (1939:210) in a study of predation on hares and grouse in the period of notable decimation of these two game species in 1935-1936 in the Cloquet Valley State Forest, in St. Louis County, Minnesota, found that "weasel predation on hares appeared to be of very low incidence or altogether lacking."
Wild birds (Class Aves).—Aldous and Manweiler (1942), as noted above, found that the remains of birds constituted five per cent by volume of the food of the ermine in winter in Minnesota.
Chicken (genus Gallus).—Criddle and Criddle (1925:145), who published relatively extensive data on the three species of weasels of Manitoba, write that: "We have no record of Bonaparte's weasel killing poultry, and we doubt whether it ever does so." However, Soper (1919:46) investigated the excited cackling of a hen brooding chicks at night and found a solitary ermine that had killed three chicks and that had the remainder under very active scrutiny.
Leopard frog (Rana pipiens).—One frog was found in a male ermine on November 20, 1931, in New York by Hamilton (1933:300).
Fish (Class Pisces).—Aldous and Manweiler (1942) found fragments of a small fish in one of 60 stomachs of ermine from Minnesota.
Earthworm (Phylum Annelida).—Osgood (1936:64), presumably at Rutland, Vermont, observed a pair of weasels from 2:15 P.M. to 5:00 P.M., in a barn and saw the female in that time make many trips for food for her young. Only earthworms were brought. Fifty traps in an adjacent, swampy field caught only one bull frog and no mice indicating that mice had been eliminated from the foraging territory of the ermine.
In handling food, Dice (1921:22) noted that the Alaskan ermine did not use the feet but only the mouth.
Reproduction
Litters of 4, 4, 7, 7, and 8, yielding an average of 6 young per litter have been recorded from the northeastern United States by Hamilton (1933:327). He (op. cit.:321-325) described animals one day old from New York State as being flesh-colored, having the long neck of the adult and a fine growth of white hair two milli meters in length, on the dorsal surface of the neck, that foreshadows the mane or pompadour that is prominent from the 14th to the 21st day of life. Six animals, when one day old averaged 1.7 grams in weight, which was three per cent of the weight of an adult female and one and one half per cent of the weight of an adult male. At two weeks of age the heavy brown mane stood out in marked contrast to the rest of the scantily, white-furred animal. The eyes opened on the thirty-fifth day of life.
Fig. 24. Mustela erminea richardsonii, adult female, Catalogue Number 14866, U. S. Nat. Mus., Fort Chimo, Ungava. × 1/2.
Ventral view of body of a pregnant female to show details of mastology. Note the five pairs of mammae characteristic of weasels, and the uneven arrangement of mammae of the two sides which is also common among weasels.
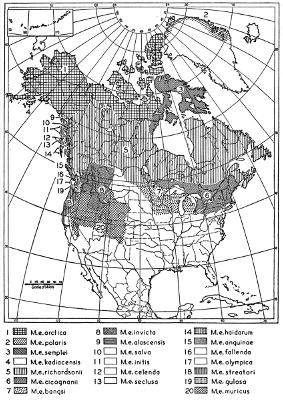
Fig. 25. Map showing geographic ranges of the subspecies of Mustela erminea in the New World.
For rearing their young, ermines live in burrows. Bishop (1923), in Albany, New York, found a burrow occupied by four young and a pair of adults. The burrow had many galleries and contained a nest constructed of rat fur, fine grass and fragments of leaves. At Woods Lake, Fresno County, California, in early August, Ingles observed (1942) some young and at least one adult at their den which was in a burrow beneath a hollow tree. The ermines used the hollow root and the hollow tree as well as the burrow beneath. Seton (1929 (2):591) quotes S. Eldon Percival, of Barretts Rapids, Ontario, as finding the living quarters of an ermine in unthreshed grain stacked in a barn and says (op. cit.:590) that John Burroughs dug out a nest, composed of leaves and the fur of mice and moles, two or three handfuls in bulk, from a cavity the size of a hat, arched over with a fine network of tree roots.
Four instances in which the male as well as the female was present at a den containing young are cited by Hamilton (1933:328) and he gives some evidence, although not at all conclusive, that "adults customarily pair, or at least run together, at times other than the breeding season." No other writers remark on this matter. I doubt that adult ermines are associated in pairs for most of the year but such may be the case.
Mustela erminea arctica (Merriam)
Ermine
Plates 2, 3, 4, 9, 10, 11 and 41
Putorius arcticus Merriam, N. Amer. Fauna, 11:15, pl. 2, figs. 1, 1a, and pl. 5, figs. 6, 6a, June 30, 1896.
Putorius (Gale) erminea, Coues, Fur-bearing animals, p. 109, 1877 (part).
Putorius richardsonii, Bangs, Proc. Biol. Soc. Washington, 10:16, pl. 1, figs. 3, 3a, pl. 2, figs. 3, 3a, and pl. 3, figs. 6, 6a, February 25, 1896 (part).
Putorius cicognanii alascensis, Osgood, N. Amer. Fauna, 19:43, October 6, 1900.
Putorius kadiacensis, Osgood, N. Amer. Fauna, 21:69, September 26, 1901.
Putorius audax Barrett-Hamilton, Ann. and Mag. Nat. Hist., 13(ser. 7):392, May, 1904, type from Discovery Bay, Ellesmere Island.
Putorius alascensis, Heller, Univ. California Publ. Zoöl., 5:345, March 5, 1910.
Mustela arctica arctica, Miller, U. S. Nat. Mus. Bull., 79:97, December 31, 1912; Dice, Journ. Mamm., 2:22, February 10, 1921.
Mustela arctica, Hall, Univ. California Publ. Zoöl., 30:420, March 19, 1929.
Mustela erminea arctica, Ognev, The mammals of U.S.S.R. and adjacent countries, 3:31, 1935; Hall, Proc. California Acad. Sci., 23:559, August 22, 1944; Hall, Journ. Mamm., 26:179, July 19, 1945.
Type.—Male, adult, skull and skin; no. 14062/23010, U. S. Nat. Mus.; Point Barrow, Alaska; July 16, 1883; obtained by John Murdock, original no. 1672.
The skull has a fracture, on the dorsal surface, extending from the anterior nares to the interorbital constriction and another fracture on the left margin of the nasal bone. The middle of the left zygomatic arch is broken away. Otherwise the skull is complete. Right incisor one, above and below, are missing. Otherwise the teeth are present and entire. The skin is in the brown summer pelage, well made, in a good state of preservation, and shows no obvious signs of fading.
Range.—Arctic regions of Alaska and western Canada from the Pacific Ocean to Smith Sound; from the northern limit of land south approximately to a line from Skagway through Ft. Goodhope, north shore of Great Bear Lake, south shore of Clinton Colden Lake, north shore of Baker Lake, west end of Wagner Bay to south end of Committee Bay. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. polaris in darker upper parts (Raw Umber rather than Buckthorn Brown) and less intensely colored underparts that are Sulphur Yellow, Colonial Buff or Primrose Yellow rather than Buff Yellow; from M. e. semplei, in males, in that hind foot more than 44 and basilar length more than 41 and in that females average larger, the skulls of females being only about 11 per cent heavier; from M. e. kadiacensis in hind foot more than 33 in females, zygomatic breadth amounting to more, rather than less, than distance between last upper molar and jugular foramen irrespective of sex; from M. e. richardsonii, alascensis, salva and initis, both sexes so far as known, by proximal two-thirds of under side of tail colored same as underparts rather than same as upper parts, and by zygomatic breadth amounting to more, rather than less, than distance between last upper molar and jugular foramen.
Description.—Size.—Male: Six adults from Tanana, Alaska, yield average and extreme measurements as follows: Total length, 336 (310-350); length of tail, 93 (84-105); length of hind foot, 49 (45-51).
Female: Five adults, one each from Alatna River, mountains near Eagle, Kamarkak in Alaska, Arctic Red River and Baillie Island in Canada, yield average and extreme measurements as follows: Total length, 285 (272-304); length of tail, 77 (68-95); length of hind foot, 39 (34-43).
Weight of 5 subadult males from Tanana is 206 (163-248) grams; adults would be heavier.
Color.—Winter pelage all white except tip of tail. Summer pelage with upper parts uniform in color and Raw Umber or darker (16n) of Ridgway and about tones 2 to 3 of Chocolate of Oberthür and Dauthenay, pl. 343, but in autumn some specimens have more light red than tones 2 or 3. Underparts Sulphur Yellow, Colonial Buff, or Primrose Yellow, often white on chin and insides of forelegs; color of underparts extends narrowly over upper lips, distally on posterior sides of forelegs onto antipalmar surface of forefeet, onto proximal two-thirds or three-fourths of underside of tail as length of tail is measured along tail-vertebrae, on medial sides of hind legs to a point between knee and ankle but reappears on antiplantar faces of toes and in some individuals is narrowly continuous onto toes; rim of ear in some specimens with short, white or pale hairs giving ears distinct whitish border; least width of color of underparts averaging, in adult males from Alaska, 65 (46-93) per cent of greatest width of color of upper parts. Black tip of tail in 5 males in winter pelage from Tanana averaging 84 (70-93) mm. which is 91 (75-107) per cent of length of tail-vertebrae.
Skull.—Male (based on 5 adult topotypes): See measurements and plates 2-4. As described in Mustela erminea richardsonii except that: Weight, 3.5 (3.1-3.9) grams; basilar length 42.5 (41.8-43.3); length of tooth-rows more than length of tympanic bulla; breadth of rostrum measured across lacrimal processes averaging more than a third of basilar length; interorbital breadth more than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth more than distance between last upper molar and jugular foramen.
Female (based on 2 adult topotypes and 2 adults and 4 subadults from central Alaska): See measurements and plates 9-11. As described in Mustela erminea richardsonii except that: Weight, 1.5 (1.2-2.0) grams; basilar length, 35.7 (34.5-37.0); length of tooth-rows more than length of tympanic bulla; breadth of rostrum more than 30 per cent of basilar length; interorbital breadth more than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth more than distance between last upper molar and jugular foramen (except in specimens from Ellesmere Island where two distances are approximately equal).
Cranial differences from Mustela erminea kaneii (which occurs on the Asiatic side of Bering Strait), in both males and females, are: larger size relatively as well as actually, broader except in mastoidal region where relatively (to basilar length) the width is less; preorbital part of skull broader as well as longer.
From kadiacensis differences in the skull of the male are: size less; 13 per cent heavier, relatively (to basilar length) narrower across interorbital region and zygomatic arches; tympanic bullae relatively as well as actually narrower. Judging by the single available adult female of kadiacensis, the skull of female arctica is larger in all parts measured, a fourth heavier, has tympanic bullae of almost twice the volume and the interorbital and preorbital regions, relative to the braincase, are much reduced in whatever plane measured.
Differences from richardsonii, additional to those noted above in the formal description of the skull, between the males, are: larger in all parts measured except length of tympanic bulla which is about the same; 42 per cent heavier; relative to basilar length, skull broader with preorbital part longer as well as broader; tympanic bullae more inflated posteriorly. The same differences prevail between females except that the skull is 36 per cent heavier and in arctica the length of the bulla is actually more (although relative to the basilar length less) and its greater inflation posteriorly is hardly perceptible. Differences from alascensis, additional to those indicated in the formal descriptions of the skulls of the two, in males, are: larger in every part measured; 95 per cent heavier; relative to the basilar length, skull broader with preorbital part longer as well as broader; measured at a point opposite the foramen lacerum anterius, the width of the pterygoid space is more, rather than less, than 40 per cent of its length. Excepting this difference in width of interpterygoid space, the same differences prevail between females, those of arctica being 56 per cent heavier.
Comparison with semplei is made in the account of that subspecies.
Skull indistinguishable from that of polaris.
Remarks.—The person who studies specimens of this subspecies finds labels inscribed with the names of naturalists well known to all readers of literature on the Arctic. Sir John Franklin, R. McFarlane, R. Kennicott, E. W. Nelson and R. M. Anderson are names which appear commonly. Of Alaskan specimens prepared according to modern methods, a large share was obtained by O. J. Murie and L. R. Dice.
The ermine was observed in the far north by early explorers and was mentioned in the literature, almost always under the name then used for the ermine of northern Europe and Asia. In 1896 Bangs misapplied to it the name richardsonii but Merriam in the same year corrected the application of this name and proposed as new for this weasel the name arctica, the name in use today. For almost 50 years after Merriam and Bangs wrote about it, arctica was treated, nominally at least, as a species distinct from its other relatives in both the Old- and New-World. The subspecific status of arctica was emphasized in 1944 (555) by the present writer in reporting in detail upon the specimens, of Mustela erminea, from Eastern Asia which were made available on loan by Professor B. S. Vinogradov and the late Anatol I. Argyropulo of the Leningrad Academy of Sciences. Specimens of Mustela erminea kaneii from the Asiatic side of Bering Strait and Mustela erminea arctica from the American side are distinguishable by slight cranial characters but in coloration and external measurements I can detect no differences. Merriam's (1896:16) mention of more golden-colored upper parts and darker underparts in American specimens than in erminea was the result of his comparison of Alaskan and northern European specimens. When Old World specimens from eastern Siberia, instead of from Europe, are used the differences mentioned by Merriam do not apply. Incidentally, many Siberian specimens have the white border, on the ear, which Merriam (loc. cit.) noted as a distinguishing feature of arctica. When Merriam named arctica he said (1896:15, 16) "Putorius arcticus . . . has heretofore been confounded with erminea or richardsonii. . . . It is interesting to find in this country an Arctic circumpolar weasel which, though specifically distinct, is strictly the American representative of the Old World erminea." Bearing in mind that Merriam's concept of species and subspecies (see Merriam, 1919:6) differed from that of nearly all modern systematists it is clear from his statement quoted above that he correctly understood the zoölogical relationship obtaining between the ermines of the Old and New Worlds.
Ognev (1935:31) seems to have been the first to use the name combination Mustela erminea arctica for Alaskan specimens. Thereby he expresses the view adopted here, namely that the American ermine is subspecifically but not specifically distinct from the Old World animal. Whether actual intergradation (crossbreeding) ever takes place across the narrow Bering Strait I do not know. I doubt that crossbreeding occurs but considering the Diomedes (islands), that might serve as a half way stopping point, and remembering Mr. Charles Brower's oral statement to me that he had seen tracks of ermine as far as 10 miles from the northern shore of Alaska out on the ice, the possibility must be granted of an occasional individual crossing from one side to the other of Bering Strait on the ice in winter or of being carried across when the ice broke up and drifted. If transfers of this kind occurred often one would expect ermines to occur also on Saint Lawrence Island where apparently they do not. The one skin (U. S. Nat. Mus. no. 259046) seen as labeled from there, my friend, Otto William Geist ascertained was imported as a skin with other furs from Siberia.
Ognev (op. cit.) who used the name combination Mustela erminea arctica for Alaskan specimens, applied it also to animals from Kamchatka. At the same time he recognized the animal from the eastern mainland of Siberia (as opposed to the peninsula of Kamchatka) under the name Mustela erminea orientalis Ognev 1928. Hall (1944:556) applied the earlier proposed name Putorius kaneii Baird 1857, to the animal on the eastern mainland of Asia and proposed the new name Mustela erminea digna for the ermine of Kamchatka. In comparing material of these two Asiatic races with topotypes and other specimens of M. e. arctica from Alaska, it seemed to me that the degree of relationship, one with the other, was about the same. M. e. digna has a slightly larger preorbital region than M. e. kaneii, and the skull is longer. In both of these particulars digna approaches closer to arctica. M. e. kaneii has longer tympanic bullae and a wider skull than digna and therein approaches more towards arctica than toward digna. As nearly as I can make out, digna and kaneii show a nearly equal degree of resemblance to arctica. Also the degree of difference between digna and kaneii is about the same as between either one of them and arctica. In view of the above considerations the ermines of the New and Old worlds are here regarded as only subspecifically distinct.
In the original description of Putorius audax (here regarded as inseparable from Putorius arcticus Merriam) Barrett-Hamilton erroneously designated the type locality as "Discovery Bay, North Greenland" whereas he should have written Grinnell Land [= Ellesmere Island of modern terminology] in place of Greenland. As reference to Nares (1877 and 1878) will readily reveal, Discovery Bay is near 65° W and 81° 40´ N, across Robeson Channel, to the west, from Greenland. The label on the type specimen and the specimen register in the British Museum of Natural History each designates the locality for this specimen, the type of audax, as Discovery Bay without mention of Greenland. The published accounts of Feilden (1878) and Nares (1877 and 1878) state that specimens of ermine were obtained at Discovery Bay. Probably H. C. Hart is the collector of the specimen; he was the naturalist attached to H. M. S. Discovery which wintered at Discovery Bay while H. W. Feilden was the naturalist attached to H. M. S. Alert which wintered a few miles southeast of Cape Sheridan, also on the eastern coast of Ellesmere Island.
It is true that from these ships a trip was made into Greenland and an ermine (only one individual it seems) was obtained there, but this individual was the type specimen of Mustela erminea polaris, in the account of which race something of the history of this specimen is given.
With the material available—and it is not entirely adequate—I can detect no features by which animals from the type locality of audax can be distinguished from typical arctica which latter name has priority.
Intergradation with richardsonii probably occurs completely across the continent. Intergrades here referred to arctica include those from Fort Goodhope. The one defective specimen from Lake Lebarge, Yukon, is not certainly identified as arctica and how far west of Teslin Lake the boundary-line between arctica and richardsonii should be drawn remains to be ascertained. The one specimen available from Hinchenbrook Island, no. 912 Mus. Vert. Zoöl., an adult female, is doubtfully referred to arctica because the damaged tympanic bullae appear to be no larger than in alascensis, and the size of the skull is more as in alascensis although intermediate between that race and arctica. Shape of the skull is more as in arctica. Possibly more nearly adequate material would show the existence on Hinchenbrook Island of an insular race differing in about the same degree from arctica of the mainland as does the insular kadiacensis. Nevertheless, the males from farther south at Cape Yakataga are in all respects arctica and this argues against near relationship to alascensis of the animal on Hinchenbrook Island. The three animals seen from Yakutat Bay are so young as not to display clearly the cranial characters of the subspecies but the extension of the color of the underparts onto the underside of the tail in them and also in the skin without corresponding skull from Glacier Bay, Alaska, is as in arctica, the race to which they are referred, and gives substantial basis for showing the geographic range of arctica as extending this far south along the Pacific Coast.
Specimens examined.—Total number, 281, arranged alphabetically by Districts and from north to south in each District. Unless otherwise indicated, specimens are in the collection of the United States National Museum.
Alaska. Point Barrow, 22 (1[1], 1[2], 1[75], 4[1], 7[60], 6[74]); Flaxman Island, 3; Collinson Point, 1[77]; Salirochet River, 1[77]; Hulahula River, 1[2]; 69°20´ & 141°, 1; Rampart House, 1; Yukon River, mouth of Porcupine River, 18; Alatna River, 30 mi. from mouth, 1; Koyakuk Riv., 16 mi. below Bettles, 4; Shelton, 1[75]; Kruzamepa, 1[75]; Tanana, 6; Boulder Creek, Chena River, 3; Fort Reliance, 4; Yukon River, 20 miles above Circle, 2; Mts. near Eagle, 42 (1[60]); Snake River, Nome, 1[9]; Nulato, 3; No[e]wikakat Riv., 1; Kantishna, 3; Fairbanks, 5 (1 20 mi. E and 1 33 mi. E); Richardson, 1; N. Fk. Kuskokwim R. at base of Mt. Sischo, 1; N. Fk. Kuskokwim R. at Junction with McKinley Fk., 1; Nenana Riv., mouth of Maurice Cr., 1; Ober Cr., trib. of Jarvis Cr., Delta Riv. region, 1; head of Savage Riv., near Jennie Cr., 1; Wonder Lake, 1[74]; Bear Cr., 3; Unlakleet, 3; St. Michaels, 11; 125 mi. E and a little N of Knik, Cook Inlet, on S side Matanuska Range, 1[60]; Hope, Cook Inlet, 1; Iak Lake, 1[68]; head of Behring Riv., 1; Bethel, 2; Kenai Lake, 8; Kenai Peninsula, 13 (2[2]); He[i]nchenbrook Island, 1200 ft., 1[74]; Sunshine Point, Kaliekh River, Yakataga Dist., 1[8]; Cape Yakataga, 3[8]; Yakutat Bay, 3[74]; Seward, 7; Seldovia, 22 (4[2]); Homer, 1[2]; Cape Elizabeth, 18; Akchookuk Lake, 1; Lake Weelooluk, 1; Kokwok Riv., 80 mi. up, 4; Nushagak, 1; Nushagak Riv., 1; Kolukuk, 1; Egooshik River at mouth, 1; Glacier Bay, 1; Becharof Lake, between Portage Bay and Becharof Lake, 1; Ugashik Riv., 4; Chignik, 7; East base Frosty Peak, 1; Pavlov Bay, 1[100]; Mt. Pavlof, 1[75]; Unimak Island, 2 (1[75]).
District of Franklin. Cape Sheridan, 1[2]; Discovery Bay, Ellesmere Island, 1[7] (type specimen of Putorius audax Barrett-Hamilton); Axel Heiberg Island, 1[95]; Bache Peninsula, Ellesmere Island, 1[77]; Bedford Pims Island, 4[75]; Craig Harbor, 2[77]; Cape Kellett, Banks Island, 1[77]; Franklin Isthmus, 1[95]; King William Island, 2[95].
District of Keewatin. Ualiak, Ogden Bay, 2[95].
District of Mackenzie. Baillie Island, 1[75]; Franklin Bay, 1; Langton Bay, arm of Franklin Bay, 15 mi. S of, 1[2]; Cockburn Point, 69°N, 115°W, 2[77]; Dolphin and Union Strait, 1[77]; Bernard Harbor, 2[77]; Kent Peninsula, 4[95]; Horton Riv., near Fort Anderson, 1; Fort Anderson, 6; Anderson River, 3; Barry Island, Bathurst Inlet, 1[77]; Fort McPherson, 1; Peels River, 2; Arctic Red River, 8[75]; Fort Good Hope, 6; Clinton Colden, 1[2].
Yukon. Kamarkak, 1[77]; Herschel Island, 1[75]; Lapierres House, 2; Forty Mile, L. T. Coal Cr., 4[74]; head of Coal Cr., 1; Macmillan River, Forks, 1; 20 mi. W. Ft. Selkirk, 1; Slims River, near Kluane, 1[75]; head of Lake Lebarge, 1.
Mustela erminea polaris (Barrett-Hamilton)
Ermine
Putorius arcticus polaris Barrett-Hamilton, Ann. and Mag. Nat. Hist., 13 (ser. 7):393, May, 1904.
Mustela erminea, Manniche, Meddelelser om Grønland, 45:80-85, 1 fig., 1910.
Mustela arctica polaris, Miller, U. S. Nat. Mus. Bull., 79:97, December 31, 1912.
Mustela erminea polaris, Hall, Journ. Mamm., 26:179, July 19, 1945.
Type.—Probably female, skin only; no. 78. 6. 19. 11, Brit. Mus. Nat. Hist.; Gap Valley, 7-1/4 miles northeast Cape Brevoort, 82° N, 59° 20´ W, Northwestern Greenland; June 15 or 16, 1876; obtained by Lewis A. Beaumont.
The skin is in full, fresh summer pelage, fairly well stuffed except for the tail which is unstuffed; the whole is in a good state of preservation.
Range.—North coast, and east coast as far south as Turner Sound (between 69 and 70 degrees) of Greenland. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. arctica in lighter upper parts (near [j] Buckthorn Brown rather than Raw Umber or darker) and more intensely-colored underparts that are Buff Yellow rather than Sulphur Yellow, Colonial Buff, or Primrose Yellow; from M. e. semplei in color in same fashion as from arctica and in larger size of skull.
Description.—Size.—Male: One subadult and two adults (one ad. from Scøresby Sound and other two from Ymer Island) measure as follows, the average being given first: Total length, 318 (301, 320, 315); length of tail, 72 (69, 70, 73); length of hind foot, 46.5 (44, 46, 47).
Female: No measurements taken in the flesh available but hind foot, measuring 33.5 in the dried state and therefore approximately 35 in life.
Color.—As described in Mustela erminea arctica except that upper parts in summer near (j) Buckthorn Brown and tone 4 of Dark Fawn of plate 307 to tone 1 of Raw Umber of plate 301 of Oberthür and Dauthenay. Underparts Buff-Yellow. Least width of color of underparts averaging, in 3 males, 66 (57-72) per cent of greatest width of color of upper parts. Black tip of tail in same males averaging 71 (70-72) mm. which is 99 (99-104) per cent of length of tail-vertebrae.
The lighter-colored upper parts and more intensely yellow underparts are the distinguishing features of the subspecies polaris in comparison with other races of American M. erminea.
Skull.—Male (based on 5 adults from eastern Greenland): See measurements. As described in Mustela erminea richardsonii except that: Weight more (not recorded); basilar length, 41.3 (39.0-42.4); length of tooth-rows more than length of tympanic bulla; breadth of rostrum measured across lacrimal processes averaging more than a third of basilar length; interorbital breadth more than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth more than distance between last upper molar and jugular foramen.
Female (based on 2 adults, Turner Sund and Kap Hoeg): See measurements. As described in Mustela erminea arctica except that basilar length 36.8 (35.9, 37.8), and length of tooth-rows not more than length of tympanic bulla. Skulls of females not in hand when this comparison is written; only the recorded measurements are available.
To me the skull of polaris is indistinguishable from that of arctica. Therefore the comparisons made of the skull of arctica with those of other subspecies will apply also for polaris.
Remarks.—In view of the heretofore erroneous assignment of the type locality of Mustela erminea audax to Greenland, pains were taken to verify the statement by Barrett-Hamilton (1904:393) relative to the type specimen of polaris. Taking pains thus seemed the more worthwhile because in the specimen register at the British Museum of Natural History, there is written to the right of catalogue numbers 78-6 = 19 nos. 1-11, "Discovery Bay Presented by Mr. Hart Arctic Collection." This refers to no. 78.6.19.1. There are no ditto marks below but by implication this data applies also to nos. 1-11, which include the holotype of polaris. A label attached to the specimen does however give the locality as "Hall Land" "N Greenland" and another label has on it "Ermine, procured by Mr. Beaumont Greenland Lat 89° Long W 59-20." The 89° is obviously a mistake (on the label or in my transcription of it) for 82°.
Reference to Nares (1877:385) reveals that Lieutenant Lewis A. Beaumont, under date of June 15 and 16, 1876, wrote in his field journal as follows: "I shot an ermine." In the daily accounts of his journey from Discovery Bay on Grinnell Land [= Ellesmere Island], across Robeson Channel and along the north coast of Greenland to the west base of Mount Farragut near 50° 30´ W he mentions the ermine only this once. For several other kinds of animals, Beaumont mentions individuals seen or shot, often with the notation that this is the second, or third seen. This mention of a kind of animal whenever seen was in accordance with orders. On page 39 of the Discovery Report (op. cit., 1877) in "General orders to sledging parties" by Captain G. S. Nares, Commanding the Expedition, we find ". . . note daily: IV State the animals seen and those shot." Reference to the map facing page 358 of the (op. cit.) report reveals that on the 15th and 16th, camps were made by Beaumont in Gap Valley, each 7-3/4 miles northeast of Cape Brevoort, one camp on either side of the 82° line, and separated from each other by a distance of only 2-1/4 air line miles or 4-1/2 miles march according to his journal.
These several data, then, are the bases for designating the type locality of M. e. polaris, in the way that I have stated it at the beginning of this account of the subspecies.
The light-colored upper parts and more intensely yellow underparts well differentiate this subspecies from arctica or semplei. Intergradation is suggested by a skin, no. 1462, Copenhagen Zoological Museum, from Axel Heibergs Land, the color of the underparts of which agrees with that of specimens from Greenland. Also the color of the upper parts is decidedly nearer that of animals from Greenland than to that of specimens from Ponds Inlet, Tulican and Gifford River. No other specimens west or south of Greenland suggest intergradation. In Greenland itself, one adult, a female from Turner Sund, East Greenland, has the underparts no more yellowish than in some specimens from Melville Peninsula. This female is darker on the back than any one of the other 10 specimens from Greenland in summer pelage examined at the same time, but even so is not so dark colored as animals from Baffin Island or other islands to the west of Greenland.
The final summation of information about this subspecies would have been more precise if I had been able to have actually in hand, at the time of writing, specimens preserved in the Copenhagen Zoological Museum. The war made it impractical to secure the loan of these as previously planned. Even so, the measurements and notes on color that I obtained from this material, in 1937, in Copenhagen, suffice to prove that the subspecies polaris is well set off in color from the other American subspecies of Mustela erminea.
The best material of this subspecies is in the University Zoological Museum at Copenhagen, Denmark.
Specimens examined.—Total number, 35, arranged by locality from the western end of the north coast of Greenland, eastward and then southward down the east coast. Unless otherwise indicated, specimens are in the Universitetets Zoologisk Museum, Købnhavn, Danmark.
Gap Valley, 7-1/4 mi. NE Cape Brevoort, 82° N, 59° 20´ W, 1 (British Mus.); Dragon Point, 1; Danmarks Havn (Fjeldene ved Baadskjeret, 1; lille Fjeld, 1; Lyservig, 1; harefjeldets, 4; Rypefjeldet, 1; Baadskjeret, 1; Danmarkshavn, 3) 12; Christians Havn, 1 (not found on map); Shannon Island, 4; Germania Havn, 2; Claveringoen, 1; Carls Havn, 1; Myggbukta, 2 (British Mus.); Ymer[s] Island, 2 (Mus. Comp. Zool.); Kap Hoegh, Jamesonsland, 1 (Berlin Zool. Mus.); Scoresby Sund, 3; Turner Sund, 4.
Mustela erminea semplei Sutton and Hamilton
Ermine
Mustela arctica semplei Sutton and Hamilton, Ann. Carnegie Mus., 21:79, February 13, 1932.
Mustela arctica labiata Degerbøl, Rept. 5th Thule Exped., 2 (no. 4):25, 1935, type from Malugsitaq, Melville Peninsula, Canada.
Mustela erminea semplei, Hall, Journ. Mamm., 26:179, July 19, 1945.
Type.—Male, subadult, skull and skin; no. 6470, Carnegie Mus.; Coral Inlet, South Bay, Southampton Island, Canada; October 8, 1929; obtained by George Miksch Sutton, original no. 3M.
The skull has two holes in it: one is immediately above the left canine, and the other (2 × 5.5 mm.) is 3 millimeters to the left of the median line at the juncture of the frontal and parietal bones. From this last mentioned hole a fracture extends back halfway to the lambdoidal crest. The tip of the left upper canine is broken off. Otherwise the skull is complete, and the teeth all are present and entire. The skin is well made and in fresh white winter pelage except for a trace of the old brown summer pelage on the back, on the tail, on the anterior borders of the ears, and in a spot 11 mm. long and 8 mm. wide on the nose.
Range.—Baffin and Southampton islands, Melville Peninsula and west side of Hudsons Bay as far south as Eskimo Point. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. arctica, in that, in males, hind foot less than 44 and basilar length less than 41 and in that females average smaller, their skulls being only about 10 per cent lighter; from M. e. polaris in darker upper parts (Raw Umber rather than Buckthorn Brown) and less-intensely-colored underparts that are Sulphur Yellow, Colonial Buff or Primrose Yellow rather than Buff Yellow, and in lesser size in the same fashion as from arctica; from M. e. richardsonii, of both sexes, in that proximal two-thirds of under side of tail colored same as underparts rather than same as upper parts and by least interorbital breadth amounting to more, instead of less, than distance between glenoid fossa and posterior border of external auditory meatus.
Description.—Size.—Male: Ten adults and subadults, from Southampton Island, yield average and extreme measurements as follows: Total length, 282 (267-318); length of tail, 77 (59-87); length of hind foot, 40 (38-43).
Female: Four subadults from Southampton Island yield average and extreme measurements as follows: Total length, 271 (256-288); length of tail, 71 (69-74); length of hind foot, 35 (33-38).
Color.—As described in M. e. arctica except that least width of color of underparts averaging, in 7 males, 59 (45-81) per cent of greatest width of color of upper parts. Black tip of tail in 19 male topotypes averaging 72 (64-83) mm. which is 91 (75-122) per cent of length of tail-vertebrae.
Skull.—Male (based on 2 adults and 10 subadults from Southampton Island): See measurements and plates 2-4. As described in Mustela erminea richardsonii except that: Weight, 2.0 (in one subadult) grams; basilar length, 37.5 (35.7-39.9); length of tooth-rows more than length of tympanic bulla; breadth of rostrum more than a third of basilar length; interorbital breadth more than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth more than distance between last upper molar and jugular foramen.
Female (based on 1 adult and 4 subadults from Southampton Island): See measurements and plates 9-11. As described in Mustela erminea richardsonii except that: Weight, 1.35 (in one adult) grams; basilar length, 34.2; breadth of rostrum more than 30 per cent of basilar length; interorbital breadth more than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth more or less than (approximately same as) distance between last upper molar and jugular foramen.
In comparison with richardsonii, the skulls of males averaged smaller in every measurement taken except breadth of rostrum and interorbital breadth which are more, and zygomatic breadth and length of inner lobe of M1 which are approximately the same; skull about 20 per cent lighter; in relation to basilar length, preorbital region longer and broader in every part measured. Female averages larger, in every part measured; 23 per cent heavier; in relation to basilar length, every other measurement more. It is noteworthy that the skull of the male is smaller and the skull of the female larger than in richardsonii.
Differences from arctica are: Size less, in each sex; males about 40 per cent and females 10 per cent lighter; in males, skull more rounded in outline as viewed from above because zygomatic arches arise less abruptly from skull; in males tympanic bullae do not project so far ventrally from squamosal floor of braincase; with these exceptions, skull of semplei can be said to be a smaller edition of that of arctica.
From polaris, semplei differs, cranially, in the same way as from arctica.
Remarks.—There is a slight increase in size of ermines toward the north which probably is the result of intergradation between semplei and arctica. Specimens from the northern part of Baffin Island are larger than those from farther south. Specimens from the mainland west of Southampton Island may owe their smaller (than in arctica) size to intergradation with richardsonii almost as much as to intergradation with semplei.
Degerbøl's name Mustela arctica labiata was applied to specimens, which to me are indistinguishable from topotypes of Mustela arctica semplei, which latter name has three years priority. Degerbøl (1935:34) states that Malugsitaq, Melville Peninsula, is the type locality. He did not designate a type specimen. Reference to his account (op. cit.:26) shows that he lists five specimens from the type locality, or more precisely as "Malugsitaq, Lyon Inlet. 5 summer skins. ♂ ♂ June-July 1922. P. F., CN. 2262-2266." On labels attached to these specimens, "Lyon Inlet" is replaced with "Melville Peninsula." On July 28, 1937, Degerbøl and I together examined these specimens in his laboratory. Because no. 2262 is first mentioned I regard it as the type. It is a juvenal male, skull and skin, no. 2262 (20.5 1931.8), Univ. Zool. Mus. Copenhagen, obtained in June or July of 1922 by Peter Freuchen whose original number was / s 2324. The specimen is one of 5 males taken at the same locality by the same collector and they bear identical data as to date. They look to be of the same litter for all are roughly of the same size and each retains milk teeth.
Additional females, with external measurements carefully taken, are much needed from Southampton Island, because the available females are insufficient to show the degree of sexual dimorphism. If the meager data available be accepted, the difference in size between the two sexes is less than in other subspecies. My own feeling is that a better sample of females would show the secondary sexual difference in size to be more than available data indicate.
Specimens examined.—Total number, 183, arranged from north to south by islands, or regions attached to the mainland, and from north to south in each region or island. Unless otherwise indicated, specimens are in the Zoological Museum, University of Copenhagen, Denmark.
Baffin Island. Pond[s] Inlet, 8; (5[77]); Tulukan (sometimes spelled Tulukat), 6; Cape Eglinton, 1[7]; Gifford River, 2; Clyde, 3[86]; head of Cumberland Sound, 1[91]; Pangnirtung, 2[77]; Kingnait Fiord, 1[91]; Kikkulin Island, Cumberland Sound, 1[7]; Blacklead Island, Cumberland Gulf, 1; merely Cumberland Gulf, 1[7]; merely east Baffin Island, 34[7]; Cape Dorset, 2[2]; SW coast of Baffin Island, 1[75].
Melville Peninsula. Iglulik, 3; Pingerqalik, 2; Kingadjuaq, Amitsog, 3; Rae Isthmus, 3; Lyons Inlet, 13(9[2]); M[N?] alugsitaq, Lyon Inlet, 5; Itibdjeriang, 2; Repulse Bay, 27 (22[2], 2[19]); Drichetts Cove, Hurd Channel, 1[2]; Gore Bay, 1; Haviland Bay, 1; Cleveland Harbor, Frozen Strait, 1.
Southampton Island and adjacent islands. Danish Island, 11; Vansittart Island, 4. Southampton Island: Coral Inlet, 19 (1[77], 18[9]); Prairie Point, 1[9]; Munnimunnek Point, South Bay, 5[9]; Native Point, 1[9]; Ranger Rim, 1[9]; Koodloatok (not found on map), 1[77]; merely Southampton Island, 1[77]; Gore Bay, 1[2]; Fox Channel, 2[2].
Mainland to west of Southampton Island. Cape Fullerton, 3 (1[77], 2[2]); Chesterfield Inlet, 4 (1[77], 1[9]); Tavane, 1[77]; N of Wagner Inlet, 1; Eskimo Point, 1[86].
Mustela erminea kadiacensis (Merriam)
Ermine
[Putorius arcticus] subspecies kadiacensis Merriam, N. Amer. Fauna, 11:16, June 30, 1896.
Putorius kadiacensis, Preble, Proc. Biol. Soc. Washington, 12:169, August 10, 1898.
Mustela kadiacensis, Miller, U. S. Nat. Mus. Bull., 79:97, December 31, 1912.
Mustela erminea kadiacensis, Hall, Journ. Mamm., 26:179, July 19, 1945.
Type.—Male, subadult, skull and skin; no. 65290, U. S. Nat. Mus., Biol. Surv. Coll.; Kodiak Island, Alaska; April 25, 1894; obtained by B. J. Bretherton, original no. 304.
The skull lacks the basioccipital, part of the basiphenoid, the occipital region on the right side and the posterior part of the right tympanic bulla. The third, upper, left incisor is missing. Otherwise the teeth all are present and entire.
The white, winter skin is only moderately well stuffed but in a good state of preservation. The spring coat is appearing along the back. This coat is visible at only two places unless the hair be parted when the new brown pelage, which is coming in, can be seen all along the midline of the back.
Range.—Kodiak Island, Alaska. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. arctica in hind foot less than 33 in females and in zygomatic breadth amounting to less, instead of more, than distance between last upper molar and jugular foramen irrespective of sex.
Description.—Size.—Male: One adult and 3 subadults yield average and extreme measurements as follows: Total length, 341 (318-360); length of tail, 93 (86-102); length of hind foot, 47 (44-49).
Female: An adult measures: Total length, 258; length of tail, 70; length of hind foot, 31.
Color.—As described in M. e. arctica, except that least width of color of underparts averaging 54 (40-83) per cent of greatest width of color of upper parts. Black tip of tail in 3 males in summer pelage averaging 80 (70-90) mm. which is 85 (69-96) per cent of length of tail-vertebrae.
Skull.—Male (based on 2 adults): See measurements and plates 2-4. As described in Mustela erminea richardsonii except that: Weight 3.1 grams; basilar length, 42.6 (42.1-43.2); length of tooth-rows more than length of tympanic bulla; breadth of rostrum measured across lacrimal processes averaging more than a third of basilar length; interorbital breadth more than distance between glenoid fossa and posterior border of external auditory meatus.
Female (based on one adult, no. 98042): See measurements and plates 9-11. As described in Mustela erminea richardsonii except that: Weight, 1.2 grams; basilar length, 33.0; length of tooth-rows more than length of tympanic bulla.
Comparison with arctica has been made in the account of that subspecies. Although richardsonii and kadiacensis are described as having the zygomatic breadth less than the distance between the last upper molar and jugular foramen, the zygomatic breadth is considerably more in kadiacensis than in richardsonii; consequently the two dimensions are more nearly equal than in richardsonii. Except for being slightly narrower, the skull of kadiacensis is only a slightly smaller edition of that of arctica.
Remarks.—When naming the weasel from the mainland of Alaska as new, under the name Putorius arcticus, Merriam (1896:16) wrote: "A small form of arcticus occurs on Kadiak Island. . . . It is probably worthy of recognition as subspecies kadiacensis." The informality of this description possibly was in part due to the describer's recognition of the fact that the degree of difference between arcticus and the insular kadiacensis was slight. Specimens collected after Merriam proposed the name for the weasel of Kodiak Island show the animal there to be less different from arctica of the adjacent mainland than he thought; small size is the most pro nounced distinction of kadiacensis and Merriam's male type specimen is smaller than any of the five additional males saved from Kodiak Island since that time. Even so the differences fully warrant subspecific recognition, in my opinion, although kadiacensis is not a strongly differentiated race. More adult females are needed to ascertain the norm of form and size for that sex. If the one female known is typical, the difference from arctica is more pronounced in females than in males. The lesser size of kadiacensis can hardly be credited entirely to the effect of insularity, for animals from the southern part of the mainland, on Kenai Peninsula for example, are smaller than those from central and northern Alaska and provide evidence of intergradation of a sort between kadiacensis and arctica.
Specimens examined.—Total number, 9, all from Kodiak Island, Alaska, and unless otherwise indicated in the U. S. National Museum.
Karluk, 1 (Stanford Univ.); Kodiak, 7; Kodiak Island, 1 (Field Mus. Nat. Hist.).
Mustela erminea richardsonii Bonaparte
Ermine
Mustela richardsonii Bonaparte, Charlesworth's Mag. Nat. Hist., 2:38, 1838.
Putorius cicognanii, Baird, Mamm. N. Amer., p. 161, 1858 (part).
Putorius richardsonii, Baird, Mamm. N. Amer., p. 164, 1858 (part-Halifax, N. S.).
Putorius (Gale) erminea, Coues, Fur-bearing animals, p. 109, 1877 (part).
Putorius richardsoni, Bangs, Proc. Biol. Soc. Washington, 10:16, February 25, 1896.
Putorius cicognani richardsoni, Merriam, N. Amer. Fauna, 11:11, June 30, 1896.
Putorius (Arctogale) cicognanii cicognanii, Bangs, Proc. New England Zoöl. Club, 1:18, February 28, 1899.
Putorius microtis Allen, Bull. Amer. Mus. Nat. Hist., 19:563, October 10, 1903. Type from Shesley, British Columbia.
Putorius arcticus imperii Barrett-Hamilton, Ann. and Mag. Nat. Hist., 13(ser. 7):392, May, 1904. Type from Fort Simpson, Mackenzie, Canada.
Putorius cicognanii richardsoni, Preble, N. Amer. Fauna, 27:231, October 26, 1908.
Mustela microtis, Miller, U. S. Nat. Mus. Bull., 79:96, December 31, 1912.
Mustela cicognanii mortigena Bangs, Bull. Mus. Comp. Zoöl., 54:511, July, 1913. Type from Bay St. George, Newfoundland.
Mustela cicognanii, Sheldon, Journ. Mamm., 13:201, August 9, 1932.
Mustela cicognanii richardsonii, Miller, U. S. Nat. Mus. Bull., 79:95, December 31, 1912; Hall, Univ. California Publ. Zoöl., 40:368, November 5, 1934.
Mustela cicognanii cicognanii, Hall, Canadian Field-Nat., 52:108, October, 1938.
Mustela erminea richardsonii, Hall, Journ. Mamm., 26:77, February 27, 1945; Hall, Journ. Mamm., 26:180, July 19, 1945.
Type.—Male, age unknown, skin; no. 43.3.3.4, British Museum of Natural History; probably from Fort Franklin, Canada; presented to British Museum on or before March 3, 1843; may be the type.
In September, 1937, when I searched in the British Museum for the skull, I found no trace of it nor mention of it in catalogues. The skin is in white, winter pelage, mounted on a pedestal. See under remarks for Mustela e. cicognanii for reasons for and reasons against regarding this specimen as the holotype.
Range.—Hudsonian and Canadian life-zones of the greater part of Canada from the Atlantic to the Pacific. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. arctica, polaris, semplei and haidarum, in both sexes, by proximal two-thirds of under side of tail colored same as upper parts rather than same as underparts, and interorbital breadth less, rather than not less, than distance between glenoid fossa and posterior border of external auditory meatus; from M. e. bangsi, in that, in both sexes, least width of color of underparts averages two-fifths rather than about a third of greatest width of color of upper parts, and in that skulls of males are a fourth heavier, basilar length averaging more than 40; from M. e. cicognanii, in both sexes, in that least width of color of underparts averages two-fifths instead of less than a third of greatest width of color of upper parts, in females by 20 per cent heavier skull (1.1 versus 0.92), in males by skull more, rather than less, than 1.9 grams, and basilar length more, instead of less, than 38; from M. e. invicta, in males, by skull more, instead of less, than 1.9 grams; mastoid breadth more, instead of less, than 19.9 mm.; depth of skull at anterior margin of braincase more, instead of less, than 12.4 mm.; in females, by same measurement of depth more, instead of less, than 10.1, and weight of skull averaging more, instead of less, than one gram; from M. e. fallenda in both sexes upper lips white rather than brown, in males, hind foot more than 41, basilar length more than 38.3, in females hind foot more than 29, basilar length more than 31.4, and breadth of rostrum amounting to less, instead of more, than 30 per cent of basilar length; from M. e. alascensis in males in that black tip of tail more than 43, total length more than 320, tympanic bullae more than 14 and longer than tooth-row rather than less than 14 mm. and sometimes shorter than tooth-row, females not individually distinguishable.
Description.—Size.—Male: Four adults (Fort Franklin, Fort Simpson, Mts. W Fort Nelson, and Govt. Hay Camp, Wood Buffalo Park) yield average and respective measurements as follows: Total length, 331 average (340, 325, 330, 328); length of tail, 93 (102, 91, 93, 87); length of hind foot, 45 (48, 43, 45, 44). Weight of 4 adults from the Belcher Islands is 175 (135-180) grams. Of 10 subadults from Belcher Islands it is 119 (92-137) grams.
Female: Three adults from Great Slave Lake (Willow River, Fairchild Point, and Fort Resolution) yield average and respective measurements as follows: Total length, 252 (237, 238, 282); length of tail, 69 (63, 60, 85); length of hind foot, 32 (31, 32, 34). Corresponding, average measurements for three adults from Glacier Lake are 240, 60, 32 and for 3 adults from the Athabasca Delta, 243, 65, 30. Weight of 8 subadults from the Belcher Islands is 69 (64-78) grams. Weight of adults would be more.
Color.—Winter pelage all white except tip of tail. Summer pelage with upper parts uniform in color and darker (16n) than Raw Umber, and about tones 3 to 4 of Chocolate of Oberthür and Dauthenay, pl. 343. Underparts Sulphur Yellow, Colonial Buff, or Primrose Yellow, often nearly white on chin and insides of forelegs; color of underparts extends narrowly over upper lips, distally on posterior sides of forelegs onto antipalmar faces of toes and sometimes over most of antipalmar surfaces of forefeet, on medial sides of hind legs to a point between knee and ankle but reappears on antiplantar faces of toes and in some individuals is narrowly continuous onto toes. Least width of color of underparts averaging, in a series of 12 males from the Athabasca Lake Region, 40 (25-54) per cent of greatest width of color of upper parts. Black tip of tail averaging 56 (45-63) mm. in 5 adult males from same region and thus 60 (48-70) per cent of length of tail-vertebrae.
From arctica, polaris, semplei and kadiacensis, richardsonii differs in: Color darker; ventral side of tail same color as upper parts; light-colored underparts a fifth narrower; black tip of tail by actual measurement a fifth shorter and averaging less than two-thirds rather than more than four-fifths of length of tail-vertebrae. From cicognanii, richardsonii differs in that the underparts are a fourth wider and in some specimens more brightly colored. The width of the underparts is likewise a fourth more than in bangsi. In invicta the underparts are not so brightly colored as in some specimens of richardsonii. From fallenda, richardsonii differs in that the upper parts often are lighter colored, upper lips white rather than colored like upper parts, and underparts as wide again. In comparison with alascensis, the black tip of the tail averages three-fifths rather than a half of length of tail-vertebrae.
Skull.—Male (based on 6 adults from 3 miles south of Big Island, Great Slave Lake): See measurements and plates 2-4; weight, 2.5 (2.1-2.9) grams; basilar length, 40.9 (39.6-43.7); length of tooth-rows less than length of tympanic bulla; breadth of rostrum measured across lacrimal processes less than a third of basilar length; interorbital breadth less than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth less than distance between last upper molar and jugular foramen.
Female (based on 4 adults: from Willow River, 1; Fort Resolution, 1; Athabasca Delta, 2; and 2 subadults, one from 3 mi. S Big Island and one from 15 mi. above Smith Landing): See measurements and plates 9-11; weight, 1.1 (0.9-1.4) grams; basilar length, 33.1 (31.5-34.2); length of tooth-rows less than length of tympanic bulla; breadth of rostrum less than 30 per cent of basilar length; interorbital breadth less than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth less than distance between last upper molar and jugular foramen.
The skull of the female averages 56 per cent lighter than that of the male.
Comparison of the skull with that of arctica, polaris, semplei, kadiacensis, haidarum, cicognanii, bangsi, invicta, fallenda, and alascensis is made in the accounts of those subspecies.
Remarks.—M. e. richardsonii has the most extensive geographic range of any American race of erminea, is centrally located with respect to the other races, is more abundantly represented by study specimens in zoölogical collections than any other race, and is a sort of average for the species as a whole in most structural features. Therefore richardsonii is used as a standard of comparison and accordingly is more fully described than any one of the other races each of which by reference to richardsonii is described in comparative fashion. This comparative description has the virtue of more clearly indicating differences between subspecies and also makes for brevity.
John Richardson, Bernard R. Ross, and names of their companions, as written on the labels of the older specimens recall to the student's mind early explorations of the north country. Edward A. Preble obtained important specimens at several places and in recent years J. Kenneth Doutt and G. G. Goodwin have made the reviser's work easier by preparing specimens in series from areas not previously well represented.
The nomenclatural history of this subspecies begins with references in the literature that identify the animal as the Old World species, Mustela erminea—an identification which the study here reported upon shows to have been correct in the specific, although not in the subspecific, sense. Richardson, for example, in his "Fauna Boreali-Americana" published in 1829 so identified the animal. In 1838, Bonaparte, basing his description on Richardson's account of 1829, proposed the new name richardsonii. Richardson himself, the following year in the "Zoology of Beechey's Voyage," accepted Bonaparte's name and it has been applied to the animal in the central part of the northern timber-belt of North America ever since, except as authors used the name Mustela erminea in the belief that richardsonii was not distinct from erminea.
The north and south boundaries of the range assigned to richardsonii varied according to the notions of the particular writer who was employing the name. Until Merriam in 1896 named arctica as distinct, animals from the far north were generally included under the name richardsonii along with populations to which the latter name now is applied. Because richardsonii grades gradually into the smaller cicognanii of more southern occurrence the boundary between the two has been set farther north by one writer and farther south by another, depending probably upon what the writer felt was the halfway point in size. This point of course depended upon the samples selected as typical of richardsonii on the north and cicognanii on the south. Because Bangs, in 1896, took as representative of richardsonii the far northern and hence large-sized animals (now separated as M. e. arctica), his halfway point in size between them and the small cicognanii of New England naturally fell farther north than it would have had he used as representative of richardsonii specimens from places south of the range of arctica.
In 1903 J. A. Allen proposed the name Putorius microtis for a specimen from Shesley, northwestern British Columbia, a place approximately 50 miles northwest of Telegraph Creek. Considering the great disparity in size between this one specimen and the other larger specimens of normal size, from the general region, available to Allen at that time, it is not surprising that he thought two full species were represented. In 1943 when G. G. Goodwin called to my attention two males, as small as the type of microtis and taken by him approximately 300 miles east of Shesley, in the valley between the Musqwa and Prophet rivers, I for a second time examined all available specimens and data with the possibility in mind that microtis was a species or subspecies distinct from M. e. richardsonii, but again concluded that only one subspecies was involved because no character except size was found to distinguish the large from the small individuals of a given sex and there are, preserved from northern British Columbia, individuals of intermediate size. Putorius microtis Allen seems to have been based on an individual of M. e. richardsonii near the lower limit of size for that subspecies and microtis is regarded as a synonym.
Barrett-Hamilton in 1904 named the animal at "Fort Simpson, British Columbia" Putorius arcticus imperii. Preble (1908:232) pointed out that Fort Simpson on the Mackenzie undoubtedly was the place intended, and arranged imperii as a synonym of M. e. richardsonii. The type specimen of imperii was stated to have been received from B. M. Ross who is known to have collected specimens, including specimens of this species (now in U. S. Nat. Mus.), at Fort Simpson on the Mackenzie. I know of no Fort Simpson in British Columbia. If, as seems improbable, Port Simpson, British Columbia, was the place that Barrett-Hamilton intended to designate (where so far as I know Ross did not collect), the name imperii still would seem to be a synonym of richardsonii because richardsonii seems to be the race of weasel at Port Simpson. In proposing the name Putorius arcticus imperii, Barrett-Hamilton stressed that the weasel, which he was naming, was a subspecies of P. arcticus, gave characters which applied perfectly to richardsonii but made no reference to richardsonii. Barrett-Hamilton did not refer to richardsonii possibly because he relied on Merriam's classification of 1896 wherein richardsonii is treated as a species distinct from arctica. Merriam, it will be remembered, held that slight degree of morphological difference rather than intergradation was the criterion for subspecies. Although I have no record of having examined the type specimen of imperii I have but little hesitancy in treating it as a synonym, and would have no hesitancy at all in so doing if the type was certainly known to have been obtained at Fort Simpson on the Mackenzie.
The name Mustela cicognanii mortigena Bangs, 1913, proposed for the ermine of Newfoundland, is placed as a synonym of richardsonii only after repeated, detailed comparisons. In advance of study I supposed that the isolation of the ermine, in Newfoundland, had contributed to its differentiation, which, however, the original describer, Bangs, indicated was slight. Bangs was a careful worker and I am confident that the differences he described really existed between his specimens. Material more nearly adequate than he had from the mainland, shows the males, so far as my measurements and comparisons go, to be in nowise different from those in Newfoundland. Females in Newfoundland may have, on the average, slightly longer hind feet than on the opposite mainland but I am not certain that they do and even if there is a slight difference in this regard as suggested by available data, I think it insufficient basis, alone, for according subspecific status to the insular animal.
The name richardsonii was based by Bonaparte on Richardson's description which in turn was drawn from a specimen taken at Fort Franklin, that thus becomes the type locality. It is fortunate that Preble, in 1903, succeeded in taking specimens there because the place is near the belt of intergradation between arctica and richardsonii. Of Preble's two adult males (see Preble, 1908:232) I have examined no. 133847, which is in transitional pelage and therefore gives no clue in so far as coloration is concerned, as to affinities with arctica versus richardsonii. Specimens in the summer pelage are much to be desired from Fort Franklin. Regardless of what their coloration may be, specimen no. 133847, in external measurements and most certainly in cranial features is of the race to the south and not the race that Merriam named arctica. Because all specimens from localities to the south of Fort Franklin likewise differ from arctica of the barren grounds, considerable additional confidence is felt in allocating the name richardsonii to the animal which ranges from Fort Franklin southward rather than to the one, here designated arctica, that occurs to the northward of Fort Franklin.
Although in most structural features richardsonii is a sort of average for the American races of the species, it is the extreme in high degree of sexual dimorphism. The difference in size between the males and females is greater than in any other race except possibly M. e. kadiacensis in which so little is known of the female that the difference between the two sexes cannot be accurately judged. It will aid in understanding the high degree of secondary sexual difference in richardsonii to visualize two kinds of weasels distributed over the northern half of the continent, thinking now of the geographic area in America occupied by the whole species Mustela erminea of which the subspecies richardsonii is only a part. One of the two kinds of weasel is the male ermine and the other the female. The decrease in size of the male, as measured by the weight of the skull, is in the ratio of 7 in the north to 2 in the south. This decrease is gradual whereas the corresponding decrease from 3 to 1 in the female is not gradual; half of the decrease in the female occurs in the short north to south distance comprised in the belt of intergradation, along the northern boundary of richardsonii, between it and arctica. As a result richardsonii is composed of females with medium sized skulls and males with relatively large skulls, the ratio by weight being approximately 5 to 2. The disproportion in races of ermines both to the north and to the south is less. Actually in the north (arctica) the approximate ratio by weight is 2-1/3:1; in richardsonii, 2-1/2:1; in the south (muricus), 1-2/5:1. Indicated in still another way in richardsonii the skull of the female is 56 per cent lighter than that of the male and the skull of the male is 127 per cent heavier than that of the female. Intergradation with races whose ranges border on that of richardsonii is complete. On the northern boundary of the range of richardsonii along the western shore of Hudsons Bay for perhaps a hundred miles north of Eskimo Point, there are intergrades with arctica. As judged by their lesser size, individuals of this population are influenced by the semplei-stock. Otherwise, intergradation on the northern boundary, with arctica, is abrupt whereas intergradation at the south, between richardsonii and cicognanii, is gradual. Intergradation is similarly gradual between richardsonii on the one hand and bangsi and invicta on the other. By speaking of the intergradation as abrupt, it is intended, in this instance, to indicate that in a relatively narrow belt, between the geographic ranges of arctica and richardsonii, ermines intermediate in color-pattern, shape of skull, and size, bridge the gap between the ermine of the tundra (arctica) and that in the forest belt (richardsonii). It may be added that the degree of difference between the two subspecies just mentioned is approximately twice as much as between richardsonii and cicognanii. The intergradation between cicognanii and richardsonii is gradual. By gradual it is meant that the change from one kind to the other is achieved in a wider area where ermines from locality A do not differ appreciably from those taken at, say, locality B, 50 miles farther south, although ermines from A and those from a third locality, C, say, 130 miles south, clearly show differences indicative of geographic variation.
Specimens examined.—Total number, 1035, as follows. Arranged alphabetically by provinces and districts and from north to south in each province or district. Unless otherwise indicated, specimens are in the United States National Museum.
Alberta. 15 mi. above Smith Landing, 2; Fort Smith, 2 (1[77]); Smith Landing, 2; LaButte, Fitzgerald, 1[77]; Egg Lake, 15 mi. NW Ft. Chippewyan, 4 (2[75]); Lobstick Island, near Ft. Chippewyan, 1; Athabasca Delta, 9 mi. above mouth of main branch, 1; Athabasca Delta, Long Creek, 1 mi. W of main branch, 2; Ft. Chippewyan, 1; Peace Point, 1[75]; 18 mi. below Peace Point, 1; Embarass River, 7 (4[75]); Athabasca River, 1[2]; Ft. McMurray, 1; Athabasca River, Middle Rapid, 2; 60 mi. above Grand Rapids, 1; Boiler Rapid, 1; Entrance, 3[2]; St. Albert, 2.
British Columbia. Fort Halket, 1; Shesley, 1[2]; Dorothy Lake, Mts. W of Ft. Nelson, 4000 ft., 3[2]; valley between Musqwa and Prophet rivers, 3800 ft., SW of Ft. Nelson, 2[2]; Sikanni Chief Riv., 1; Telegraph Creek, 7 (6[2]); head of Bad River, 2350 ft., on lake, 1; Six Mile, 5[74]; Tuchodi Lake, 2[2]; Iskoot River, 2[14]; Level Mtn., 1[2]; head of Tatletuey Lake, 12 mi. W Thudade Lake, 2; Robb Lake District, 5[2]; Ft. Grahame, 12 (2[77]); Sustut Mts., on trib. Sustu Riv., 25 mi. SE Thudade Lake, 2; Laurier Pass, 1; Omineca Mts., 1[85]; Point Creek and Clearwater River, 2; Kispiox Valley, 23 mi. N Hazelton, 5[74]; Hazelton, 3[77]; NW arm Tacla Lake, 7; N end Babine Lake, 1; Pt. Simpson, 1; Metlakatla, 1; Stuart Lake, 27; S Fk. Salmon Riv., 1[77]; mouth Salmon Riv., 1[77]; Vanderhoof, 4[77]; Wistaria P. O., near Burns Lake, 1[77]; Kruger Lake, 9[74]; Indianpoint Lake, 23[74]; Quesnel, 1; Ahbau Lake, 3[74]; Isaacs Lake, 6[74]; Beaver Pass, 56[74]; Lightning Creek, 54[74]; LaFontaine, 16[74]; Barkerville, 1[74]; Barkerville District, 34[74]; Swift River, 27[74]; Cunningham Creek, 34[74]; Itcha Mts., 1[31]; Anahim Lake, 1[74]; Chezacut Lake, 8[31]; Kleena Kleene, 18[74]; 158 mi. House (Cariboo on labels), 3[60]; Rivers Inlet, 6 (5[94]; 1[77]); Horse Lake, 4[22]; Kingcome Inlet, 8[77]; Loughborough Inlet, 7[77]; McGillivary Creek, 1; Camel Back, Pemberton Meadows, 1[31]; Arrow Rapids, mainland opposite Stuart Island, 1[77]; Butte Inlet, 9[77]; Green Lake, 1[31]; Mt. Whistler, 1[86]; Alta Lake, 2 (1[31]; 1[21]); Mons, 1[31].
Keewatin. Foot of Baker Lake, 1.
Labrador. Okak, 3[75]; Nain, 22 (11[75]; 11[60]); Hopedale, 24[75]; Kippokak Bay, 7[75]; Ailik, 1; Makkovik, 26[75]; Labrador, 55° N, 3; Hamilton Inlet, 2[75]; NW River Post, interior Labrador, 5[1]; Cartwright, 5; Paradise, 12; Sandwich Bay (Muddy Bay, 6; North River, 6), 12; Battle Harbor, 1[7]; St. Marys River, 3[7]; Black Bay, 16 (15[75]; 1[76]); Lanceau Loup, 17 (1[75]).
Mackenzie. Ft. Franklin, 1[2]; Ft. Rae, 12; Fairchild Point, 6[9]; Fort Simpson, 10 (2[2]); Hot Springs (61°, 125°), 1[2]; Willow River, near Ft. Providence, 1; 35 mi. N Big Island, 7; Big Island, 9; 3 mi. S Big Island, 7; Ft. Resolution, 9; 100 mi. N Ft. Smith, 2; 75 mi. NW Ft. Smith, 1; Ft. Liard, 2; Sucker Creek, 4[77]; Govt. Hay Camp, Wood Buffalo Park, 2[77].
Manitoba. Egg Is., Rabbit Point, 1; Ft. Churchill, 1; Ft. York, W Hudsons Bay 57° N, 1[7]; Oxford House, 11; Gypsumville, 1[86]; Lake St. Martin.
New Brunswick. Restigouche County: Bird Bait, north Camp, 6 mi. NE Nictau Lake, 2[59]; Red Brook, Tobique River, 1[59]. Victoria County: Trousers Lake, 3[2]. Glouchester County: Youghall, 1[77]; Miramichi Road, 15 mi. from Bathurst, 13[77]. York County: Scotch Lake, 2.
Newfoundland. Nicholsville, 3[75]; Bay St. George, 48 (26[75]; 2[7]; 1[9]); Codroy, 9 (7[75]; 2[60]).
Nova Scotia. Victoria County: Cape North, 2[77]. Inverness County: Fizzleton, 3[77]. Richmond County: St. Peters, 1[77]. Pictou County: Glengary, 1[4]. Guysborough County: East Roman Valley, 5[77]. Kings County: Wolfville, 5 (3[74], 2[77]); near Wolfville, 1[77]. Halifax County: Hammond Plains, 1. Annapolis County: Annapolis Royal, 1. Digby County: Digby, 3. No locality more definite than Nova Scotia, 3.
Ontario. Severn River, 1[77]; R. C. Mission, Yellow Creek, near mouth of Albany, 2[86]; Ft. Albany, 4; Charlton Island, 1; Moose Factory, 10 (7[9]; 3[77]); Abitibi, 1[4].
Quebec. Fort Chimo, 10[77]; Ungava Forks, 1; Belcher Islands, Hudsons Bay (Tukarak Island, 29; Eskimo Harbor, 2; Innetalling Island, 1; S tip Gibson Peninsula, 2; Flaherty Island, 1), 35[9]; Cairn Island, Richmond Gulf, 2[9]; Manitounuk Sound, 4[9]; about 15 mi. S Great Whale River, 1[9]; Ft. George, 1[9]; Charlton Island, 1[9]; Waswonaby Post, 1[77]; Mistassinnay Post, 3[77]; Godbout, 36; Mt. Albert, 7 (4[78]; 3[2]); St. Anne River, 1500 ft., 1[77]; Ste. Anne des Monts, 3[2]; "Federal Mine," 1[77]; Berry Mountain Camp, 1[77]; Berry Mountain Brook, 1[2]; Cascapedia River (Middle Camp, 2; Tracadie, 2; Square Forks, 1), 5[2].
Saskatchewan. Poplar Point, Athabasca Lake, 1[75]; Fair Point, Athabasca Lake, 1[75]; Emma Lake, 1[74]; Harper Lake, 2[77]; Livelong, 3[55]; Fairholme, 2[74]; Touchwood Hills, 2[7]; Indian Head, 1[86].
Yukon. Hoole Canyon, 1; Teslin Lake (30 mi. N of, 1; Lake itself, 1; "near" the lake, 1; Mts. "near," 2; Snowden Mts., 2; Teslin Post, 2; Eagle Bay, 1; Morley Bay, 2; Nisutlin River, 1; Nisutlin Flats, 2; Wolf River, 1; Wolf Lake, 5), 21[77].
Mustela erminea cicognanii Bonaparte
Ermine
Mustela cigognanii [sic.] Bonaparte, Charlesworth's Mag. Nat. Hist., 2:37, 1838.
Putorius vulgaris, Emmons, Quadrupeds of Massachusetts, p. 44, 1840.
Mustela pusilla DeKay, Zool. of New York, Pt. 1, Mammalia, p. 34, pl. 14, fig. 1, 1842. Type from New York State.
Putorius pusillus, Audubon and Bachman, Vivip. Quadrupeds of N. Amer., 2:100, pl. 64, 1851 (pl. 1846) and erroneously labeled Mustela fusea, as pointed out on page 102 of text.
Putorius cicognanii, Baird, Mamm. N. Amer., p. 161, 1858.
Putorius richardsoni cicognani, Bangs, Proc. Biol. Soc. Washington, 10; 18, figs. 4, 4a of pls. 1 and 2, and pl. 3, figs. 2, 2a, February 25, 1896 (part).
Putorius cicognani, Merriam, N. Amer. Fauna, 11:10, pl. 2, figs. 3, 3a, 4, 4a and pl. 5, figs. 2, 2a, June 30, 1896.
Mustela cicognanii cicognanii, Miller, U. S. Nat. Mus. Bull., 79:95, December 31, 1912; Bishop, Journ. Mamm., 4:26, February 9, 1923.
Mustela cicognanii, Jackson, Journ. Mamm., 3:15, February 8, 1922.
Mustela erminea cicognanii, Hall, Journ. Mamm., 26:77, February 27, 1945; Hall, Journ. Mamm., 26:180, July 19, 1945.
Type.—No type specimen designated; type locality, eastern United States.
The restriction of the type locality from the general region of northeastern North America, as given by Merriam (1896:10) to the less inclusive area of the eastern United States as earlier given by Bangs (1896:18) is supported by Bonaparte's remarks in connection with the proposal of the name cicognanii. He says (1838:37-38) "During my stay in the United States, I only saw a small species of Mustela, very common throughout the Union . . . ." This animal constituted basis for the name cicognanii which name, he points out, is bestowed in order that the Americans ". . . should have constantly under their eye, this very common little animal, as the perpetual memorial . . ." to the Italian Governmental representative ". . . who, for upwards of fourteen years had served, in diplomatic and commercial concerns, . . . two countries, . . . so different . . . as the Roman and the United States. . . ." Clearly he had in mind principally, if not exclusively, the animal of the United States.
Range.—Transition and higher life-zones of northeastern United States south to Connecticut, central Pennsylvania and extreme northeastern Ohio; in Quebec and Ontario westward from the latitude of central Maine to Lake Nipigon and Lake of the Woods. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. richardsonii of both sexes, in that least width of color of underparts averages less than a third rather than two-fifths of greatest width of color of upper parts, in males skull less, instead of more, than 1.9 grams and basilar length less than 38, in females by 16 per cent lighter skull (0.92 versus 1.1 grams); from M. e. bangsi, in males hind foot less instead of more than 40, linear measurements of skull averaging 11 per cent less (depth of skull at plane of molars 10.0 versus 11.4), in females averaging smaller, hind foot 30 versus 32 and depth of skull at plane of molars 8.6 versus 9.1.
Description.—Size.—Male. Seven adults and subadults from New York and Pennsylvania, yield average and extreme measurements as follows: Total length, 266 (240-295); length of tail, 74 (66-80); length of hind foot, 36 (33-39). Hamilton (1933:294) gives the weight of 31 adults from New York as 81 (66-105) grams.
Female: Twelve adults and subadults from Maine and the area south to central Pennsylvania, yield average and extreme measurements as follows: Total length, 243 (225-260); length of tail, 63 (55-72); length of hind foot, 29.8 (26-32). Hamilton (1933:294) gives the weight of 15 adults from New York as 54 (45-71) grams.
Color.—As described in Mustela erminea richardsonii except that underparts in summer Marguerite Yellow or even more whitish; least width of color of underparts averaging, in adult males from New York and Pennsylvania, 29 (27-32) per cent of greatest width of color of upper parts. Black tip of tail in same series averaging 42 (30-51) mm. which is 57 per cent of length of tail-vertebrae.
Skull.—Male (illustrated by 4 adults in table of cranial measurements, which see): See plates 2-4. As described in Mustela erminea richardsonii except that: Weight, 1.5 (1.2-1.7) grams; basilar length, 35.7 (33.8-37.6).
Female (illustrated by adult and subadults recorded in table of cranial measurements, which see): See plates 9-11. As described in Mustela erminea richardsonii except that: Weight of 2 subadults, 0.92 (0.86-0.98) grams; basilar length, 32.4 (31.4-33.3).
The skull of the male, in linear measurements, is approximately 13 (12-16) per cent smaller and 40 per cent lighter than in M. e. richardsonii. In relation to the basilar length, the skull averages slightly narrower, slightly shallower as measured in the vertical plane touching the posterior borders of the last upper molars, and the preorbital part is slightly longer. In skulls of females of cicognanii, linear measurements average 3 (0-6) per cent less, the weight is 16 per cent less and the teeth are 5 per cent shorter. In relation to the basilar length, measurements of the skull are approximately the same or slightly less in cicognanii.
In comparison with bangsi, the male sex in linear measurements of the skull and teeth averages 11 per cent less than in bangsi from Aitkin, Minn., and 6 per cent less than in bangsi from Elk River, but in relation to the basilar length the preorbital region is larger. The weight is approximately a fourth less. In females the measurements average less, some being the same, and in relation to the basilar length, the bullae are shorter and the skull is shallower. The weight is about the same.
Remarks.—In January, 1838, in Charlesworth's Magazine of Natural History, C. L. Bonaparte proposed for three kinds of American weasels the names Mustela cicognanii, Mustela richardsonii and Mustela longicauda.
In this paper Bonaparte indicates that he previously had written (for his Iconografia della Fauna Italica ...) an account of Mustela cicognanii using this same name. Fasciola XXII of the Iconogr. d. Fauna Italica, presenting his account of Mustela, like the English paper was published in the year 1838. In his article in Charlesworth's Magazine, Bonaparte refers to his book published [used the past tense] in Rome but whether it actually appeared first I am unable to determine and hence am uncertain which of the two constitutes the original description.
Reference to the Italian account suggests as basis for the name M. cicognanii, (1) specimens possibly seen in the United States by Bonaparte, or (2) Godman's published account of the animal.
In the English publication, however, Bonaparte actually says that (1) he saw the small species in the Union [= United States]. Also, he (2) mentions his earlier written Italian account, (3) mentions that "all the [American?] naturalists" used the name M. vulgaris for this animal, (4) incidentally mentions Godman's account, and (5) in naming two other American species cites accounts of them by Richardson. Also, Bonaparte in this English article makes clear that when he wrote [not necessarily published] his Italian paper he did not know of the existence of two of the three American species.
In the register of mammals at the British Museum of Natural History, there appears:
43.3.3.3 Mustela longicauda Bonap N Amer. presented by Dr. J. Richardson
4 Mustela Richardsonii Bonap "
5 " Cicognanii Bonap "
To the right of these entries there appears, in three lines, the notation: "The three specimens examined by Prince Canino on which he established the three species."
Every part of each of the above entries is in the hand writing of J. E. Gray, in charge of the collections from 1824 to 1840 and associated with them as Keeper until 1875. The three specimens are in good condition considering their age. The catalogue or register number shows, among other things, that they were entered in the register on March 3, 1843.
Questions which might occur to anyone are:
(1) Was there a type specimen of Mustela Cicognanii Bonaparte? If so is it no. 43.3.3.5?
(2) If there was no type specimen was there a type locality? If so what is it?
Among other things that may have bearing on these questions, are these: Bonaparte in Charlesworth's Magazine appears to base the two names Mustela Richardsonii and Mustela longicauda on Richardson's published account of Mustela erminea. At any rate immediately following each of the two names, Bonaparte writes "Nob. (M. erminea Rich. F. Bor. Amer.)." Bonaparte's other, first newly proposed name, Mustela Cicognanii, in Charlesworth's Magazine has following it only "Nob. North America," although in a paragraph above he did point out that this was the animal which all naturalists, at the time he was in America, considered as M. vulgaris.
Turning to Richardson's account (Fauna Boreali Americana, ... Quadrupeds, pp. 45-47. 1829) one finds that he recognized two species, M. vulgaris and M. erminea. Of the first he gives measurements "of an old female killed at Carlton House." Of the second species he distinguishes two varieties, the first represented by a specimen, of which he gives measurements, "killed at Fort Franklin, Great Bear Lake" and, the second variety "of a larger size, having a longer tail and longer fore-claws" he indicates the size of by giving measurements of a specimen taken "in the neighborhood of Carlton House."
The last variety is clearly the basis of Bonaparte's M. longicauda. The specimen from which Richardson took his measurements I have been unable to locate [no. 43.3.3.3 in the British Museum, appears to be another specimen, although of the same subspecies and provided by Richardson].
The first variety of Richardson's Mustela erminea, clearly is the basis of Bonaparte's M. Richardsonii. The specimen from which Richardson took his measurements may well be no. 43.3.3.4 now preserved in the British Museum of Natural History, but I could not be certain about this.
Richardson's M. vulgaris is accompanied by measurements of a female which I have ascertained to my full satisfaction is the identical specimen now bearing catalogue number 43.3.3.5 said by Gray to be the specimen on which Bonaparte based his name Mustela cicognanii.
Gray probably saw his guest, Bonaparte, at work on these weasels and Gray's own written indication perhaps should be accepted at its face value. I found only 4 Richardson specimens of North American weasel in the British Museum in 1937 and it is conceivable that Bonaparte, 100 years before, actually had at hand only one specimen each of two kinds and 2 specimens of the third. This I think is not an important consideration, though, for Gray says just which specimens did serve as basis for Bonaparte's names and there is only one specimen for each name according to Gray.
But I wonder if a type specimen can be made in this way? That is to say, after a name is published in a manner which makes it available, and if two or more specimens of the kind of animal involved, were, or may have been, available to the describer, can a person, even the author, himself, make a type specimen by saying that one particular specimen is beyond doubt the specimen on which a given name was established even though no particular specimen was designated in the original description? I incline to the view that a specimen so designated would at most be only a lectotype, unless it were a cotype.
However, if a holotype can be made by action such as Gray took, then (1) is no. 43.3.3.3 the type specimen of Mustela longicauda Bonaparte and, (2) is no. 43.3.3.4 the type specimen of Mustela Richardsonii Bonaparte?
Incidentally, Mustela longicauda Bonaparte whether based on no. 43.3.3.3 or on Richardson's account will continue in its present application. The same is true of Mustela richardsonii. If the basis of Mustela cicognanii Bonaparte [the diagnosis in the Iconografia d. Fauna Italica ... makes it clear that the name applies to the short-tailed species] was a weasel from the eastern United States or a description of a weasel or weasels from there, the name will continue in its present application. If, instead, the name is based on no. 43.3.3.3 (from Carlton House, Saskatchewan) or on Richardson's account of M. vulgaris, the name will apply to a different subspecies (now called richardsonii and richardsonii will fall as a synonym of cicognanii) and the ermine of the eastern United States will take the next available name. Bonaparte probably named (in manuscript at least) cicognanii before he ever saw the specimen in the British Museum. This is indicated by his statement in Charlesworth's Magazine (1838:37) that "I have now [Italics mine] found two [other] American species. . . ." Whereas the names richardsonii and longicauda are based on Richardson, the name cicognanii, even if it dates from the account in Charlesworth's Magazine, appears to have a composite basis composed at the very least of (1) animals seen by Bonaparte in the United States, and (2) those called vulgaris by some other authors. Conceivably the specimen no. 43.3.3.3 in the British Museum, was part of the basis. From the nature of the case it can be argued that there could be no type and that if someone should bring to light a specimen in, say, Philadelphia, bearing the notation "this is the specimen seen in the United States by Bonaparte" it would immediately become as important as the one in London. Any American weasel or weasels (then alive or preserved in a zoölogical collection) that Bonaparte saw in the United States probably were of the eastern United States. Bangs (1896:18-21), for one, previous to the present consideration of the name cicognanii, restricted it to the ermine of the eastern United States. Consequently, the name cicognanii, in the present account is applied to the ermine of the eastern United States. In my opinion there was and is no type. Almost certainly there was no type if the Fauna Italica appeared before the account in Charlesworth's Magazine did.
Specimens examined.—Total number, 172, arranged alphabetically by provinces and states, then (except where indication is given to the contrary) by counties from north to south within each state or province. Unless otherwise indicated, specimens are in the U. S. National Museum.
Connecticut. Windham County: S. Woodstock, Woodstock Lake, 1[2]. Hartford County: Windsor, 1[5]. New London County: Liberty Hill, 3[75].
Maine. Aroostook County: Quimby, 1[75]; Ashland 2[75]. Piscataquis County: tableland on top of Mt. Katahdin, 1; Chimney Pond, 3; T. 5, R. 13, 3[5]; "vicinity of Chesnucook," 1[5]; T. 4, R. 13, 1[5]; Moosehead Lake, 7[75]; Grenville, 10 [75]; Barnard, 3 (1[86]). Penobscot County: South Twin Lake, 1[2]; Lincoln, 11 (7[1], 2[14], 2[50]). Franklin County: Seven Pond Township, 7[75]. Oxford County: Umbago Lake, 1[75]; Upton, 4[86]; Bethel, 1[75]. Hancock County: Bucksport, 17[75]; Naskeag, 1. Lincoln County: Booth Bay, 1[5].
Massachusetts. Middlesex County: Wilmington, 2; Burlington, 6 (1[75]); Worcester County: Cambridge, 5 (1[5], 3[75]); Sterling, 1[5]. Plymouth County: Middleboro, 7 (1[75]).
New Hampshire. Carroll County: Ossipee, 5. Rockingham County: Greenland, 1[76]. Cheshire County: Dublin, 1.
New York. St. Lawrence County: Ogdensburg, 1[74]. Franklin County: Malone, 1[58]. Lewis County: Locust Grove, 1. Warren County: Lake George, 1. Montgomery County: Amsterdam, 1. Albany County: Albany, 1[80]. Rensselaer County: Berlin, 2[2]; Schoharie, 1[2]. Thompkins County: Cascadilla Creek, Ithaca, 1[58]. Allegany County: Ford Brook, Wellsville, 1[58]. Ontario County: Phelps, 1[50]. Cattaraugus County: Cattaraugus, 1[5].
Ontario (localities locally north to south, then west to east). Thunder Bay Dist.: Grand Bay, Lake Nipigon, 5[86]; Macdiarmid, 2[86]; Oscar, 2[14]; 20 mi. SW Fort Williams, 1[76]; Michipicoten Island, 3[102]. Algoma Dist.: Michipicoten, 1; Franz, 1[74]; Pancake Bay, 2[77]. Parry Sound Dist.: French River, Georgia Bay, 1[2]; Seguin Falls, Twp. Montieth, 1[86]. Sudbury Dist.: Casselman, Rathbun Twp., 1[86]. Nipissing Dist.: Smoky Falls, near Kapuskasing, 4[86]; Franks Bay, Lake Nipissing, 1[86]. Haliburton County: Gooderham, 1[60]. Simcoe County: Orillia, 1[2]; no locality more definite than county, 1[60]. Carleton County: Britannia, 5 mi. W Ottawa, 1[77]; Ottawa, 1[77]; Constant Bay, NE? of Ottawa, 1[77]. Wellington County: Mt. Forest, 2[75]; Guelph, 1[31]. Addington County: Buckshot Lake, Abinger Twp., 1[86]. Fontenac County: Clear Lake, Arden, 1[77].
Pennsylvania (by counties from west to east). Crawford County: North Shenango Township, Pymatuning Swamp, 2[9]; Linesville (3 mi. NW, 1; 3-1/2 mi. W, 2; 3 mi. W, 1; 2 mi. SW, 1; 7-1/2 mi. SW, 1) 6[9]. Potter County: Cherry Springs Farm, Abbott Township, 1; 3 mi. S Inez, South Fork Sinnamahoning Creek, 1[9]. Sullivan County: Lopez, 1[74]. Lackawanna County: Scranton, 1[1]. Wayne County: Waymart, 1.
Quebec (west to east). Labelle County: Kamika [= Kiamika] Lake, 2[77]; Lacoste, 2[77]; Trout Lake, probably in this county, 2[77]. Megantic County: Black Lake, 1[77].
Rhode Island. Newport County: Middletown, 2[5].
Vermont. Lamoille County: Mt. Mansfield, 1. Windsor County: Barnard, 1[5].
Mustela erminea bangsi Hall
Ermine
Mustela erminea bangsi Hall, Journ. Mamm., 26:176, July 19, 1945.
[Putorius] cicognani, Mearns, Bull. Amer. Mus. Nat. Hist., 3:235, June 5, 1891.
Putorius richardsoni cicognani, Bangs, Proc. Biol. Soc. Washington, 10:18, February 25, 1896 (part).
Putorius cicognanii, Cory, Mamm. Illinois and Wisconsin, p. 375, 1912.
Mustela cicognanii, Aldous and Manweiler, Journ. Mamm., 23:250, August 13, 1942.
Mustela cicognanii cicognanii, Bailey, N. Amer. Fauna, 49:169, January 8, 1927; Leraas, Journ. Mamm., 23:344, August 13, 1942.
Type.—Male, subadult, skull and skin; no. 11541, D. R. Dickey Coll.; Elk River, Sherburne County, Minnesota; November 1, 1925; obtained by Bernard Bailey, original no. A 606.
The skull is complete and the teeth all are present and entire. The skin is well made and in a good state of preservation.
Range.—Southern Manitoba, northeastern North Dakota, the whole of Minnesota, Wisconsin and Michigan and northern Iowa. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. richardsonii, in that, in both sexes, least width of color of underparts averages about a third, instead of two-fifths, of greatest width of color of upper parts, and in that skulls of males are a fifth or more lighter, basilar length averaging less than 40; from M. e. cicognanii, in that hind foot more than 40 in males, averaging 32 versus 30 in females, and in larger skull, depth of skull at plane of molars being 11.4 versus 10.0 in males and 9.1 versus 8.6 in females.
Description.—Size.—Male: Twelve adult and subadult males from Aitkin, Minnesota, yield average and extreme measurements as follows: Total length, 316 (291-341); length of tail, 87 (70-101); length of hind foot, 43 (40-44). Two adults from Aitkin each weigh 170 grams.
Four adult and subadult females from Elk River and Fort Snelling, Minnesota, yield average and extreme measurements as follows: Total length, 249 (240-260); length of tail, 61 (55-65); length of hind foot, 32 (30-33).
Color.—As described in Mustela erminea richardsonii except that, least width of color of underparts averaging, in males from Minnesota, 32 (19-51) per cent of greatest width of color of upper parts. Black tip of tail in 12 male topotypes in white winter pelage averaging 52 (45-58) mm. which is 60 (53-66) per cent of length of tail-vertebrae.
Skull.—Male (based on adults from Aitkin): See measurements and plates 2-4. As described in Mustela erminea richardsonii except that: Weight of 2 adults from Aitkin, 2.2, 2.3 grams (9 subadults from T. 61 N, R. 26 W, average 1.95 grams); basilar length, 39.7 (38.5-40.7); length of tooth-rows rarely more (usually less) than length of tympanic bulla.
Female (based on adults from Minnesota as listed in table of cranial measurements, which see): See plates 9-11. As described in Mustela erminea richardsonii except that: Weight, of a subadult from T. 61 N, R. 26 W, 0.91 grams; basilar length, 32.8 (31.8-33.6); breadth of rostrum rarely equal to as much as 30 per cent of basilar length.
From richardsonii, topotypes of bangsi differ in that cranial measurements in males are approximately 7 (5-9) per cent less, linear measurements of teeth are 10 (9-11) per cent less and the skull is a fifth lighter. In relation to basilar length the tympanic bullae of bangsi are longer. Skulls of females are individually indistinguishable, those of bangsi averaging approximately 1 per cent less in linear measurements. Comparison with the smaller cicognanii is made in the account of that subspecies.
Remarks.—Before the subspecific name bangsi was proposed, individuals of this subspecies ordinarily were recorded in the literature as Mustela cicognanii. The best single lot of material is in the zoölogical collection of the University of Wisconsin. The late naturalist Albert Lano preserved a large share of the material from Minnesota. The large series from Elk River of that same state was mostly collected by Bernard Bailey although his Aunt, Anna (Bailey) Mills, and her brother the late Vernon Bailey, at an earlier time saved some specimens from Elk River. The name bangsi was proposed in recognition of the superior work done on American weasels by the late Outram Bangs.
From the range of M. e. invicta in the Rocky Mountains, that of bangsi is separated by the Great Plains from a large part of which region the species is unknown. M. e. bangsi differs from invicta in greater degree of sexual dimorphism in size, and in each sex by larger size, narrower light-colored underparts, and deeper braincase as measured at the anterior margin of the basioccipital. In bangsi the braincase is deeper relative to the length of the skull as well as, of course, actually deeper.
Of the two subspecies whose ranges do meet that of bangsi, it more closely resembles richardsonii than cicognanii. From richardsonii, especially from southeastern populations of the same in which the skull is of the same size as in bangsi, the latter differs in longer hind feet. This is an average difference and by one interpretation the animals here referred to bangsi might be lumped with some of the populations from the southeastern part of the range of richardsonii and the whole lot treated as intergrades between richardsonii and cicognanii. Nevertheless, the animals here referred to bangsi are not geographically intermediate between richardsonii and cicognanii and this consideration had much to do with the decision to recognize as a separate subspecies the animals here named bangsi.
Within the range of the subspecies there is some geographic variation; the hind feet of animals from Iowa average slightly shorter than those of animals from Minnesota and Wisconsin but are nowhere nearly so short as in cicognanii at the same latitude in the eastern United States.
It is noteworthy that the few specimens seen from Isle Royal have the long hind feet of bangsi and not the short hind feet of cicognanii which occurs all along the northern mainland.
Because an oft cited record of occurrence even though erroneous, has a way of being repeated in later works, attention is here called to the alleged occurrence of this ermine in northwestern Ohio at New Bremen. Henninger (1921:239) published the original account of the supposed occurrence but as I pointed out in 1937 (p. 304), the specimen concerned proved upon examination to be a female of Mustela frenata noveboracensis. Henninger was misled probably by the short tail; the end of the tail had been lost and healed over before the animal's death. The present study has revealed that M. erminea everywhere east of the Cascade Mountains assumes a white winter coat. Had this been known when Henninger obtained his specimen he probably would not have wrongly identified the animal from New Bremen which was in the brown, winter pelage.
Specimens examined.—Total number, 222, arranged alphabetically by provinces and states and, arranged from north to south, by counties in each state. Unless otherwise indicated, specimens are in the University of Wisconsin Museum of Zoölogy.
Iowa. Dickinson County: W side Lake Okobogie, 1[48]. Winnebago County: Lake Mills, 7[65]. Worth County: Northwood, 1[65]. Clay County: "Dewey's Pasture, near Ruthven," 1[76].
Manitoba. Aweme, 4[47]; Red River Settlement, 1[91].
Michigan. Isle Royal: Tobin Harbor, 1[76]; Bell Isle, 1[76]; Washington Harbor, 3[76]. Ontonagon County: Ontonagon, 2 (1[76], 1[14]); T. 51N, R. 43W, S. 17, Porcupine Mts., 1[76]. Gogebic County: Little Girls Point, 5[76]; Ironwood, 1[76]. Iron County: no locality more definite than county, 1[76]. Luce County: Tahquamenon River Falls, 1[91]. Chippewa County: Sault Ste. Marie, 2[76]. Emmet County: Wilderness State Park, 2[76]. Cheboygan County: Univ. Mich. Biol. Station, 1[76]. Washtenaw County: Ann Arbor, 1[76].
Minnesota. Kittson County: no locality more definite than county, 1[2]. Roseau County: Deer Township, 1[14]; Falun Township, 2[14]. Marshall County?: Moose River, 5[93]; Warren, definitely in Marshall County, 1[93]. Cook County: Grand Marais, 3 (2[76], 1[14]). St. Louis County: 2 mi. E Babbitt, 14[93]; Burntside [= Burnside] Lake, 1[91]. Itasca County: T. 61N, R. 26W, 23. Clay County: Moorhead, 3[9]. Aitkin County: Aitkin, 13 (11[60], 1[7], 1[4]). Otter Tail County: Arthur, 3[60]; Ten Mile Lake, 1[76]; Parkers Prairie, 2[75]. Chisago County: North Branch, 1[60]. Sherburne County: Elk River, 42 (16[91], 5[14], 20[59], 1[74]). Hennepin County: Lake Minnetonka, 1[75]; Minneapolis, 1[91]; Fort Snelling, 5 (4[2], 1[60]).
North Dakota. Pembina County: Walhalla, 1[91]. Nelson County: Stump Lake, 1[91]. Eddy County: Brantford, 2[76].
Wisconsin. Douglas County: T. 44N, R. 13W, 1; Gordon, 1. Bayfield County: Brinks Camp, Washburn, 1[2]; "near Cable," 1. Ashland County: Bear Lake, 2. Iron County: Fisher Lake, 4; Mercer, 5. Vilas County: Mamie Lake, 16[91]; Ox Bow Lake, 1[91]. Oneida County: Tomahawk Lake, 1[60]. Langlade County: T. 34N, R. 11E, 3. Rush County: Ladysmith, 1. Dunn County: Colfax, 2. Door County: Mink River, Ellison Bay, 1[76]. Dodge County: Fox Lake, 1[50]; Beaver Dam, 12[50].
Mustela erminea invicta Hall
Ermine
Plates 2, 3, 4, 9, 10, 11 and 41
Mustela erminea invicta Hall, Journ. Mamm., 26:75, February 27, 1945; Hall, Journ. Mamm., 26:180, July 19, 1945.
Putorius cicognanii, Preble, N. Amer. Fauna, 27:230, October 26, 1908.
Type.—Male, subadult, skull and skin; no. 101122, Mus. Vert. Zoöl.; Benewah, Benewah County, Idaho; October 24, 1926; obtained by William T. Shaw.
The skull has a hole in the right squamosal bone on the floor of the braincase, and lacks the hamular process of the left pterygoid. The postmolar part of the right lower jaw is missing. The teeth all are present and entire. The skin is in white, winter pelage, well made, and in a good state of preservation.
Range.—Central Rocky Mountain region from Jasper Park south over Alberta, southeastern British Columbia, Washington east of the Cascades, and north and central Idaho and northwestern Montana. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. richardsonii, in males, by skull lighter than 1.9 grams, mastoid breadth less than 19.9, depth of skull at anterior margin of basioccipital less than 12.4, in females by corresponding measurement of depth less than 10.1, and weight of skull less than one gram; from M. e. fallenda, in both sexes, by upper lips white (not brown), in males by skull averaging longer (37.0 versus 35.7), in females by breadth of rostrum less, instead of more, than 30 per cent of basilar length; from M. e. streatori, gulosa, and muricus by hind foot more than 36 and basilar length more than 35 in males and by hind foot more than 29.5 and basilar length more than 30.5 in females; further distinguished from streatori by white (not brown) upper lips and from gulosa by black tip of tail more than half length of tail-vertebrae.
Description.—Size.—Male: Ten adults and subadults from central Idaho County yield average and extreme measurements as follows: Total length, 291 (272-328); length of tail, 86 (75-100); length of hind foot, 39.9 (38-44).
Female: Five adults and subadults from the same locality yield average and extreme measurements as follows: Total length, 255 (245-270); length of tail, 71 (68-76); length of hind foot, 32.3 (32-33).
Color.—As described in Mustela erminea richardsonii except that underparts in summer Marguerite Yellow or more whitish; least width of color of underparts averaging, in four females from Idaho and Montana, 38 (33-43) per cent of greatest width of color of upper parts. Black tip of tail in same specimens 38 (31-42) mm. which is 57 (52-65) per cent of length of tail-vertebrae.
Skull.—Male (5 adults from Idaho County): See measurements and plates 2-4. As described in Mustela erminea richardsonii except that: Weight, 1.5 (1.4-1.7) grams; basilar length, 37.0 (35.8-39.8).
Female (illustrated by adult and 4 subadults in table of cranial measurements, which see): See plates 9-11. As described in Mustela erminea richardsonii except that: Weight, 0.72 (0.7-0.9) grams; basilar length, 32.2 (31.6-32.8).
From fallenda, invicta differs in that the skull of the male has a relatively narrower rostrum and relatively shallower braincase. Females show the same differences but the degree of difference is about as great again as in males. The teeth are almost exactly the same size in the two subspecies. The weight is the same in males but in females invicta is 18 per cent heavier.
From streatori, invicta differs in that males average larger in every measurement taken except that the anteroposterior diameter of the inner moiety of M1 is less; 36 per cent heavier; linear measurements of the skull are about 5 per cent larger and those of the teeth, with the one exception noted, about 6 per cent larger; relative to the basilar length the tympanic bullae are longer and the rostrum is relatively narrower. In females, measurements of the skull average 8 per cent more and those of the teeth 7 per cent more except that, as in males, the inner lobe of M1 is actually shorter. Females of invicta are 12 per cent heavier; relative to the basilar length the skull is narrower throughout and the tooth-rows are shorter than in streatori.
From gulosa, invicta differs in that males average larger (about 12 per cent) in every measurement taken, excepting the anteroposterior diameter of M1 which is the same; 50 per cent heavier; relative to the basilar length the length of the tooth-rows and interorbital breadth are less. In females the inner lobe of M1 is smaller but every other measurement taken of the skull and teeth is more, invicta averaging about 8 per cent larger and 22 per cent heavier; relative to the basilar length, the tooth-rows are shorter and the skull is narrower interorbitally, through the rostrum and across the zygomata.
From murica, invicta of corresponding sex differs in being larger in every measurement taken; males average 17 per cent larger in cranial measurements, 13 per cent larger in dental measurements and are 83 per cent heavier; corresponding percentages for females are 11, 9 and 20. Exception must again be made for the anteroposterior diameter of the inner lobe of the last upper molar which is less in females, and only slightly more in males. In males of invicta the tympanic bullae are longer in relation to the basilar length.
From the geographically remote cicognanii, skulls of both males and females of invicta are to me individually indistinguishable. There is, nevertheless, an average difference not apparent to the eye between skulls of males. If the length of the tooth-rows be taken as a standard (100 per cent), the rostrum, of invicta, as measured across the lacrimal processes is broader (89 rather than 84 per cent) but the width across the fourth upper premolars is less, 94 rather than 97 per cent of the length of the tooth-rows.
Since the skull of invicta closely resembles that of cicognanii, it follows that invicta differs from richardsonii and bangsi in about the manner described in the account of cicognanii.
Remarks.—Animals of this subspecies in advance of the present study generally were recorded in the literature under the name Mustela cicognanii. The difficulty in distinguishing individual specimens of invicta on morphological grounds from those of the geographically remote M. e. cicognanii should not be taken to indicate that the populations do not differ appreciably. Actually they differ in several characters although in no one of these is the degree of difference sufficient to allow of using it alone as a certain means of diagnosis. In invicta, as compared with cicognanii, the light-colored underparts are wider in relation to the dark-colored upper parts and the tail is longer by 4 per cent relative to the head and body. Given a population of each of the two subspecies, in which the skull is of the same mass, the hind feet are longer in invicta, there is more sexual dimorphism in size, and the anterior part of the skull differs in some particulars as just described in the comparison of the skull of invicta with other forms. Nevertheless, each of these differences is of an average sort. Therefore, and because overall size is about the same in the two subspecies concerned, one or a few specimens from, say, central Idaho, can be distinguished from animals from western Pennsylvania only with difficulty, if at all. The close resemblance of skulls of invicta and cicognanii may be a function of their living at approximately the same latitudinal position in a climate that has marked seasonal variation.
Intergradation with richardsonii is complete and gradual; in one sense invicta is but little more than a small richardsonii. Intergradation with fallenda is shown by several specimens. These two races differ in large degree in color, and in size and shape of the skull of females. Although the geographic area where intergradation in color occurs is fairly wide, the area in which intergradation in cranial characters in females occurs, appears, from the inadequate material available, to be much narrower. Intergradation occurs freely in Washington with streatori but with muricus so far as known only in the Bitterroot and nearby mountains of northwestern Montana. The Snake River Plains and low country along much of the Columbia River appears to be uninhabited by weasels of the species erminea and hence there is opportunity for intergradation only in the mentioned area of Montana.
Specimens examined.—Total number, 177, as follows. Arranged alphabetically by provinces and states then by localities from north to south in each province and by counties from north to south in each state. Unless otherwise indicated, specimens are in the United States National Museum.
Alberta. Jasper House, 4[77]; Shovel Pass, 2[77]; Jasper Park, 10[77]; head of Smoky River, 9; Henry House, 2 (1[77]); Blindman River, 1[2]; forks of Blindman River and Red Deer River, 2 (1[60], 1[75]); "near Red Deer, Red Deer River," 1[77]; Red Deer River, 2 (1[2], 1[60]); Red Deer, 2[60]; Rosebud, 2[77]; Prairie, 3000 ft., 1; Didsbury, Little Red Deer River, 1; Canadian Nat'l Park, 1[60]; Canmore, 1; Banff, 1[60]; High River, 1[86]; "Waterton Lake Park" in Alberta, 6[77].
British Columbia. Grand Forks of Fraser River, 1; Canoe River, 1[77]; Field, 1; Glacier, 1[58]; E side Beaverfoot Range, 4000-4500 ft., 6 mi. SE Fraser Creek, 8[74]; Wentworth Lake, 1[31]; Revelstoke, 2 (1[77], 1[60]); Spillimacheen[e]en River, 2[2]; Sicamous, 2; Albert River, 7000 ft., 1[2]; Lumby, Creighton Valley, 1[31]; Okanagan, 4 (2[75], 1[94], 1[2]); Kettle River Lake, Gold Range, 4000 ft., 1; Crows Nest Station, 1[74]; Yale District, 3; Fort Hope, 1; Chilliwack Lake, 1[77]; Skagit, 2 (1[77], 1[31]); Skagit Valley, 1[77]; Skagit Summit, 1[77]; Lightning Lakes, 2 mi. N International Boundary, 3; Osoyoos-Bridesville Summit, 2; Westbridge, 1[77]; Rossland, 5[77]; Creston, mouth Goat Creek, 3[77]; Yahk, 4[77].
Idaho. Bonner County: Coolin, 4. Benewah County: Benewah, 1[55]. Idaho County: "Pete Kings, Lochsa River," 1[97]; 2 mi. SSE Selway Falls, 1900 ft., 1[8]; 4 mi. SW Selway Falls, 5800 ft., 3[8]; Newsome Cr., 12 mi. above jct. with S Fk. Clearwater River, 2[74]; Iron Mt., to 14 mi. W thereof, 24[74]; Pilot Cr., 3/4 to 2-1/2 mi. above Newsome Cr., 4[74]; Sawmill Cr., 1-1/4 mi. W Newsome, 1[74]; between Selway River and S. Fk. Clearwater R., 4[74].
Montana. Teton County (of old arrangement of counties): Many Glacier, 4900 ft., 1[74]; Duck Lake, 6 mi. NE St. Marys Lake, 1; St. Marys, Glacier Park, 1[76]; Lower St. Marys Lake, 1[2]. Flathead County: Stanton Lake, 5. County in question: Bitter Root Mts., 1. Ravalli County: Tin Cup District, 1[74]; Bass Creek, 6800 ft., NW of Stevensville, 1; Capitan Peak, 7000 ft., 1; Darby, 2[74]; Girds Creek, 1[74]; Charlos Heights, 2[74].
Washington. Whatcom County: Twin Lakes, Winchester Mts., 3 (1[10]); Chilliwack River, 2600 ft., 2; Cooper Creek, near head, 4500 ft., Hannegan Pass, 1; Cooper Cr., 4300 ft., Hannegan Pass, 1[10]; Beaver Creek (2500 ft., and at McMillan Ranch, 1700 ft.), 2; Barron, Bornite Mine, 5000 ft., 1. Okanogan County: Tungsten Mine, 6800 ft., Bauerman Ridge, 4; Hidden Lakes, 4100 ft., 1; West Fork Pasayten River, 4700 ft., 1. Stevens County: Orin, 1[51]. Pend Oreille County: Ione, 2[51]. Chelan County: Lake Chelan, 1[46].
Mustela erminea alascensis (Merriam)
Ermine
Putorius richardsoni alascensis Merriam, N. Amer. Fauna, 11:12, pl. 2, figs. 2, 2a, June 30, 1896.
Putorius cicognanii alascensis, Miller, U. S. Nat. Mus. Bull., 79:96, December 31, 1912; Swarth, Univ. California Publ. Zoöl., 7:140, January 12, 1911.
Mustela erminea alascensis, Hall, Proc. Biol. Soc. Washington, 57:36, June 28, 1944; Hall, Journ. Mamm., 26:180, July 19, 1945.
Type.—Male, adult, skull and skin; no. 74423, U. S. Nat. Mus., Biol. Surv. Coll.; Juneau, Alaska; August 22, 1895; obtained by Clark P. Streator, original no. 4806.
The skull shows malformation of the frontal sinuses due to parasites and lacks osseous tissue where the parasitic infestation was localized. The left exoccipital condyle and adjacent region is less developed than the right and the posterior part of the skull is bent slightly to the left. Otherwise the skull is unbroken. The teeth all are present and entire. The skin is in the brown summer coat, fairly well made and in a good state of preservation. A few white hairs persist where the proximal line of the black hair of the tip of the tail meets the distal line of the brown hair.
Range.—Mainland of southeastern Alaska from Lynn Canal south to include Mitkof, Zarembo, Wrangel and Revillagigedo islands. See figures 25, 26 on pages 95 and 134.
Characters for ready recognition.—Differs from M. e. arctica and haidarum, in both sexes, by proximal two-thirds of under side of tail colored same as upper parts rather than same as underparts and interorbital breadth less, instead of more, than distance between glenoid fossa and posterior border of external auditory meatus; from M. e. salva, in males, by overall depth of braincase including tympanic bullae less than 89 per cent of orbitonasal length, females not individually distinguishable but averaging shallower through the braincase; from M. e. initis, celenda and seclusa by interorbital breadth less than distance between glenoid fossa and posterior border of external auditory meatus (females of initis, celenda and seclusa unknown); further from initis by total length less than 317 and black tip of tail less than 57 per cent of length of tail-vertebrae; further from celenda by chest white, not mostly covered by brown patch.
Description.—Size.—Male: Eight adults from Windham, Alaska, yield average and extreme measurements as follows: Total length, 298 (288-315); length of tail, 88 (84-94); length of hind foot, 41.3 (37-44).
Female: Two adults from Juneau and Helm Bay measure, respectively, as follows: Total length, 258, 258; length of tail,——, 76; length of hind foot, 32, 34.
Color.—As described in Mustela erminea richardsonii except that least width of color of underparts averaging, in five females, 42 (35-53) per cent of greatest width of color of upper parts. Black tip of tail in same specimens averaging 36 (30-40) mm. which is 49 (48-53) per cent of length of tail-vertebrae.
Skull.—Male (based on 8 adults from Windham): See measurements and plates, 2-4. As described in Mustela erminea richardsonii except that: Weight, 1.8 (1.5-2.6) grams; basilar length, 37.5 (36.5-38.9); length of tooth-rows more or less than (about same as) length of tympanic bulla.
Female (based on 5 adults, from localities listed in the table of cranial measurements): See measurements and plates 9-11. As described in Mustela erminea richardsonii except that: Weight, 0.96 (0.7-1.1) grams; basilar length, 32.7 (31.9-33.2); breadth of rostrum more or less than (about equal to) 30 per cent of basilar length.
From richardsonii, alascensis differs in that the skull of the male averages smaller in every measurement taken and is 28 per cent lighter. Relative to the basilar length, the orbitonasal length is more and the braincase is shallower as measured at the anterior end of the basioccipital. The four adult females seen of alascensis are more variable than those of richardsonii and average smaller in some measurements and larger in others but give no proof of any consistent difference.
From haidarum, alascensis differs in that the rostrum and entire preorbital part of the skull is actually as well as relatively much smaller in both sexes. In males of alascensis the length of the skull, and other cranial measurements of length, is more. In males, the mastoid breadth and zygomatic breadth are about the same as in haidarum, as also is the weight. M1 is larger but m1 and P4 are smaller. In females the anteroposterior extent of the inner moiety of M1 and length of tympanic bulla are about the same in the two subspecies but all other cranial and dental measurements in alascensis are less. It is 29 per cent lighter. The difference in the preorbital region is of about the same degree as in the males.
Comparisons of the skull with those of arctica, salva, initis, celenda, and seclusa are made in the accounts of those subspecies.
Remarks.—The relatively few specimens known of this race seem always to have been referred to in the literature by the name alascensis and the nomenclatural history is therefore simple. The original materials were obtained by the collector Clark P. Streator and the additional series of skeletons, one with skin, from Windham were procured by Stanton Price, a resident there.
The subspecies is well differentiated from both arctica and richardsonii. Although actual intergrades are lacking between alascensis and the two races just mentioned I have no doubt that intergradation occurs with richardsonii and think it probably does also with arctica.
The assignment of the three females from Mitkof Island, Zarembo Island, and Loring on Revillagigedo Island, is tentative because each is so young as not to show diagnostic cranial characters. The two other specimens from Revillagigedo Island (Carroll Inlet), labeled as males, are in white winter pelage. Only one, no. 136358, a subadult, is accompanied by a skull. The small size of each specimen, and its cranial characters which are intermediate between those of males and females of alascensis of the adjacent mainland, indicate the existence of a distinct race of weasel on Revillagigedo Island. On the chance that the one specimen with a skull is a dwarf, or is wrongly sexed as seems improbable, the population is tentatively referred to alascensis.
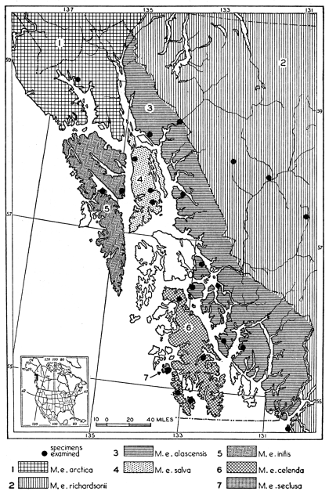
Fig. 26. Map showing known occurrences and probable geographic ranges of the subspecies of Mustela erminea in southeastern Alaska.
Specimens examined.—Total number, 24, arranged by localities from north to south. Unless otherwise indicated, specimens are in the Museum of Vertebrate Zoölogy, University of California.
Alaska. Juneau, 5[91]; Taku River, 1; Windham, 9; Mitkof Island, 1; St. John Harbor, Zarembo Island, 1; Wrangel, 1[91]; Helm Bay, Cleveland Peninsula, 1; Cleveland Peninsula, 2[91]; Revillagigedo Island, Carroll Inlet, 2[91]; Loring, 1[91].
Mustela erminea salva Hall
Ermine
Mustela erminea salva Hall, Proc. Biol. Soc. Washington, 57:35, June 28, 1944; Hall, Journ. Mamm., 26:180, July 19, 1945.
Type.—Male, adult, skull only; no. 74641, Mus. Vert. Zoöl.; Mole Harbor, Admiralty Island, Alaska, December 27, 1936; obtained by A. Hasselborg.
The skull (plates 2-4) shows malformation of the frontal sinuses owing to parasites and lacks osseous tissue where the parasitic infestation was localized. The skull is unbroken. The teeth all are present and entire.
Range.—Admiralty Island, Alaska. See figures 25, 26 on pages 95, 134.
Characters for ready recognition (known only from skulls).—Differs from males of M. e. alascensis in overall depth of braincase which is more than 89 per cent of orbitonasal length; from M. e. initis, in males, in that orbitonasal length and mastoid breadth total less than 35 mm., weight of skull and lower jaws less than 2.1 grams; from M. e. celenda, in males, in that breadth of rostrum measured across lacrimal processes less than a third of basilar length.
Description.—Size.—Male: An adult from Gambier Bay measures: Total length, 320; length of tail, 95; length of hind foot, 45 (41 in dry skin).
Female: A subadult from Hawk Inlet, measures: Total length, 250; length of tail, 70; length of hind foot, 33.
Color.—As described in Mustela erminea richardsonii except that least width of color of underparts in four individuals 40 (38-43) per cent of greatest width of color of upper parts. Black tip of tail, in two individuals for which external measurements are given, amounting to 50 and 40 mm. respectively which is 53 and 57 per cent of length of tail-vertebrae.
Skull.—Male (type and 4 adult topotypes): See measurements and plates 2-4. As described in Mustela erminea richardsonii except that: Weight, 1.7 (1.5-1.9) grams; basilar length, 37.8 (36.4-39.5, extremes are in subadults); length of tooth-rows more or less (usually more) than length of tympanic bulla; interorbital breadth rarely more than distance between glenoid fossa and posterior border of external auditory meatus.
Female (2 ad. and 1 ad.-sad. topotypes): See measurements, and plates 9-11. As described in Mustela erminea richardsonii except that: Weight, 0.9 (0.8-1.0) grams; basilar length, 33.0 (32.0-33.6); length of tooth rows approximately same as length of tympanic bulla; breadth of rostrum approximately 30 per cent of basilar length.
From alascensis, salva differs in that males have the preorbital region slightly wider in relation to the length of the tympanic bulla; also the braincase is smaller, actually as well as in comparison with the preorbital part of the skull. The tympanic bullae do not project so far below the squamosals and the braincase itself is shallower, in adults averaging only 11.5 mm. as against 12.5 mm. The overall depth of the braincase, including the tympanic bullae, when divided into the orbitonasal length gives an average of 93 (90-97) per cent whereas in alascensis the figure is only 85 (78-88) per cent. On this basis alone, everyone of the adult skulls of the two races can be distinguished. The females and subadult males show the same tendency to reduction in depth of braincase but not every individual among them can be surely distinguished. By weight the skull of salva of corresponding sex is only about 6 per cent smaller. Comparisons with initis and celenda are made in the accounts of those subspecies.
Remarks.—Most of the specimens seen were collected by Allen E. Hasselborg, resident on Admiralty Island. On the basis of skulls—few skins, and measurements taken in the flesh, are available—salva more closely resembles alascensis than does any other subspecies so far known from southeastern Alaska. The race on Admiralty Island is only slightly differentiated from alascensis of the adjacent mainland.
Specimens examined.—Total number, 26, all from Admiralty Island, Alaska, arranged in general by localities from north to south, and unless otherwise indicated in the Museum of Vertebrate Zoölogy, University of California.
Alaska. Admiralty Island: Hawk Inlet, 2; Seymour Canal, 4; Mole Harbor, 18 (skulls only); Gambier Bay, 1; no locality more definite than Admiralty Island, 4 (1 in U. S. Nat. Mus.).
Mustela erminea initis Hall
Ermine
Mustela erminea initis Hall, Proc. Biol. Soc. Washington, 57:37, June 28, 1944; Hall, Journ. Mamm., 26:180, July 19, 1945.
Type.—Male, adult, skull and skin; no. 289, Mus. Vert. Zoöl.; Saook Bay, Baranof Island, Alaska; October 9, 1907; obtained by A. Hasselborg, original no. 4.
The top of the skull is fractured on the left side from the anterior nares posteriorly through the postorbital process to the posterior root of the zygomatic arch. On the left lower jaw the canine and three incisors are missing; otherwise the teeth all are present and entire.
The skin is in process of molt, approximately nine-tenths of the incoming white pelage being in place. The skin is well made and in a good state of preservation.
Range.—Chichagof and Baranof islands, Alaska. See figures 25, 26 on pages 95, 134.
Characters for ready recognition (only males known).—Differs from M. e. arctica, in that proximal two-thirds of under side of tail colored same as upper parts rather than same as underparts, zygomatic breadth less than distance between last upper molar and jugular foramen; from M. e. salva in that orbitonasal length and mastoid breadth total more than 35 mm., weight of skull and lower jaws more than 2.1 grams; from M. e. alascensis, by total length more than 317, black tip of tail more than 57 per cent of length of tail-vertebrae, interorbital breadth more than 10.3 and equal to, instead of less than, distance between glenoid fossa and posterior border of external auditory meatus; from M. e. celenda by chest white (not mostly covered by brown patch), breadth of rostrum measured across lacrimal processes less than a third of basilar length; from M. e. seclusa in zygomatic breadth more than distance between last upper molar and jugular foramen.
Description.—Size.—Male: The type and an adult topotype measure, respectively, as follows: Total length, 330, 320; length of tail, 95, 95; length of hind foot, 45, 45.
Female: No external measurements available.
Color.—As described in Mustela erminea richardsonii except that least width of color of underparts averages, in two young female topotypes, 50 (49, 50) per cent of greatest width of color of upper parts. Black tip of tail in three young female topotypes averaging 54 (52-55) mm. which is 67 (63-69) per cent of length of tail-vertebrae.
Skull.—Male (illustrated by type and 1 ad. topotype): See measurements and plates 4-6. As described in Mustela erminea richardsonii except that: Weight, 2.3 and 2.5 grams; basilar length, 39.6, and 40.5; interorbital breadth equal to distance between glenoid fossa and posterior border of external auditory meatus.
Female: No adults available.
From salva, initis differs in that skulls of males average larger in every measurement taken, being 41 per cent heavier. Relative to the basilar length, the interorbital and preorbital parts of the skull are larger; the relatively greater interorbital and mastoid breadths are particularly noticeable. Although the depth of the braincase, including the tympanic bullae, is both relatively as well as actually more than in salva, the depth is relatively less than in alascensis which otherwise differs from initis in about the same way that salva differs from initis. Whereas the interorbital breadth in initis is about equal to the distance between the glenoid fossa and the posterior border of the external auditory meatus, the interorbital breadth is uniformly less than this distance in both salva and alascensis. In comparison with seclusa the teeth are of the same size but all measurements of the skull are larger. The skull of initis is 25 per cent heavier. In relation to the basilar length, the interorbital and preorbital parts of the skull are much less in initis. The preorbital and interorbital regions in initis are relatively smaller in comparison also with arctica. The one measurement of interorbital breadth in initis is greater in relation to the basilar length than in kadiacensis but the rostral region, and all that part of the skull anterior to the braincase, is relatively smaller in initis.
Remarks.—The two adult males, nos. 286 and 289 from Saook Bay, provide convincing evidence of the existence of a distinct race of weasel on Baranof Island. Three other young specimens, almost subadult, from the same place are labeled as males although the basilar lengths of these skulls are only 35.5, 35.9 and 37.3 millimeters as against 39.6 and 40.5 in the two adult males. The difference in size is too great to be age-variation. The fact that 3 are definitely of one category and 2 of the other makes it doubtful that individual variation accounts for the differences. The small size of these 3 specimens and the fact that in each the anterior margin of the tympanic bulla is flush with the squamosal rather than protruded from the braincase, suggests that the three are females. If they are females, the amount of secondary sexual variation is rather less than would be expected by analogy with the amount obtaining in alascensis on the mainland and in salva on Admiralty Island. Another possibility that I can not disprove is that two stocks of weasels persist on Baranof Island, the two larger specimens being descendants of the stock which first became established on the island and the three smaller specimens being descendants of an individual ermine, or of ermines, that were rafted or otherwise transported to the island at a considerably later date. Assuming for the moment that there are two stocks, it must be admitted that each one differs from any stock known from elsewhere. Therefore, each stock would be presumed to have been long resident on the island. But—two stocks as closely related as the two in question would not be expected to persist for long in an area as small as that of Baranof Island because competition would give one the ascendancy. Therefore, the first suggestion, namely that the three smaller animals are really females, seems the more probable. The feasible way to clear up the present uncertainty is, of course, to obtain additional specimens, carefully labeled as to sex. Yet another reason why additional collecting is desirable in this area is to ascertain whether there is subspecific differentiation between the ermines of Baranof and Chichagof islands. The one specimen available from the latter island, although in general like the three smaller animals from Baranof Island, differs in the fuller (less scooped out) medial side of the tympanic bulla and to a slight degree in each of some other features. This specimen from Chichagof Island is labeled as a male also.
Specimens examined.—Total number, 6, arranged by localities from north to south, and in the Museum of Vertebrate Zoölogy, University of California.
Alaska. Chichagof Island, Freshwater Bay, 1. Baranof Island, Saook Bay, 5.
Mustela erminea celenda Hall
Ermine
Mustela erminea celenda Hall, Proc. Biol. Soc. Washington, 57:38, June 28, 1944; Hall, Journ. Mamm., 26:181, July 19, 1945.
Type.—Male, adult, skull and skin; no. 130987, U. S. Nat. Mus., Biol. Surv. Coll.; Kasaan Bay, Prince of Wales Island, Alaska; June 16, 1903; obtained by Cyrus Catt; original no. 4407X.
The skull has a piece 1.5 mm. long broken out of the left zygomatic arch. P2 is absent on both sides. The right I1, and the left I1 and I2 are missing. The skin, in summer pelage, is fairly well made. A scrotal pouch attests to the correctness of the sex recorded on the label. The rostral part of the skull is smaller than in average-sized males of corresponding age.
Range.—Prince of Wales, Dall, and Long islands, Alaska. See figures 25, 26 on pages 95, 134.
Characters for ready recognition (only males known).—Differs from M. e. alascensis and initis in chest mostly covered by brown patch, not white, and breadth of rostrum measured across lacrimal processes more than a third of basilar length, which cranial character serves to distinguish also salva; from M. e. seclusa in zygomatic breadth less than distance between last upper molar and jugular foramen; from M. e. haidarum in chest white (not mostly covered by brown patch), proximal two-thirds of underside of tail colored like upper parts rather than underparts, basilar length more than 38.2 mm.
Description.—Size.—Male: Seven adults and subadults from Prince of Wales Island, yield average and extreme measurements as follows: Total length, 286 (277-304); length of tail, 77 (74-85); length of hind foot, 36 (35.5-40.5).
Female: No specimen available.
Color.—As described in Mustela erminea richardsonii except that upper parts about tone 3 of dark Chocolate Brown of Oberthür and Dauthenay, pl. 342; lower throat and chest covered by a large patch of same color as upper parts; color of underparts extending to toes but in interrupted fashion on both fore- and hind-feet; least width of color of underparts averaging, in four males from Prince of Wales Island, 41 (38-49) per cent of greatest width of color of upper parts. Black tip of tail averaging, in 8 males in winter pelage, 65 (59-78) mm. which is 84 (69-92) per cent of length of tail-vertebrae.
From its geographic neighbors alascensis and initis, celenda differs in darker color of upper parts, presence rather than absence of patch of dark color on lower throat and chest, and longer black tip on tail. From haidarum, celenda differs in darker color of upper parts, presence rather than absence of patch of dark color on lower throat and chest, narrower light-colored under parts, black tip of tail averaging less rather than more than nine-tenths of length of tail-vertebrae and ventral face of tail colored like upper parts rather than like underparts.
Skull.—Male (illustrated by 5 adults): See measurements and plates 5-7. As described in Mustela erminea richardsonii except that: Weight, 2.3 (2.2-2.5) grams; basilar length, 39.5 (38.9-40.7) mm.; length of tooth-rows more than length of tympanic bulla; breadth of rostrum measured across lacrimal processes more than a third of basilar length; interorbital breadth more than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth more or less than (about equal to) distance between last upper molar and jugular foramen.
Female.—Complete skull of adult unavailable.
Differences from richardsonii are indicated in the formal description just given. Additional to differences therein noted, celenda differs from initis in larger interorbital and preorbital parts of the skull although dimensions of other parts of the skull and the teeth are about the same or even less. From salva, celenda differs in larger average size in every measurement taken, except for the inner moiety of M1 which is about the same. The skull of celenda is 35 per cent heavier. In relation to the basilar length the skull of celenda is wider, especially in the interorbital and preorbital regions. In comparison with alascensis the tympanic bullae are of approximately the same length; otherwise essentially the same differences obtain as are noted in comparison with salva and the zygomatic breadth is relatively more in celenda. From seclusa, in which the teeth are of comparable size, celenda differs in that every cranial measurement is more and the skull is 28 per cent heavier. Because the skull of celenda is so much longer, its dimensions in other planes are less in relation to the length than in seclusa. M. e. celenda is larger in every part measured than haidarum, 21 per cent heavier, and in relation to the basilar length the interorbital, and preorbital, parts of the skull are smaller, the braincase is shallower, and the skull is relatively wider across the zygomata and mastoid processes. In comparison with kadiacensis, differences are: 26 per cent lighter, skull shorter; in relation to the basilar length, braincase shallower as measured at the anterior end of the basioccipital, tooth-rows shorter but orbitonasal length more. In comparison with arctica all parts measured of the teeth and skull of celenda are smaller and its skull is 34 per cent lighter. In relation to the basilar length, the interorbital breadth of celenda is only slightly less but its skull is narrower across the rostrum and zygomata, the tooth-rows are shorter, and the braincase is shallower.
Remarks.—The late George Willett in the course of his work in Alaska collected most of the known specimens of this strongly differentiated subspecies. In both coloration and cranial characters the distinguishing features are so well marked that the zoölogist could with reason accord full specific rank to celenda. Nevertheless it obviously is an ermine. Also, races from other islands of southeastern Alaska tend to bridge the gap, as regards cranial features, between celenda and the mainland ermine. The specimen from Dall Island agrees in all respects with topotypes. The specimen from Howkan on Long Island is in white winter pelage and the skull has suffered shrinkage from some chemical solution; the reference of this specimen to celenda is tentative.
Specimens examined.—Total number, 25, as follows: Arranged by localities from north to south. Unless otherwise indicated, in U. S. National Museum.
Alaska. Prince of Wales Island: Craig, 18 (10 in Mus. Vert. Zoöl., and 8 in Los Angeles Mus. Hist. Art and Sci.); Kasaan Bay, 2; no locality more definite than the Island itself, 3; Dall Island, Otter Harbor, 1 (Los Angeles Mus. Hist. Art and Sci.). Long Island, Howkan, 1 (Field Mus. Nat. Hist.).
Mustela erminea seclusa Hall
Ermine
Mustela erminea seclusa Hall, Proc. Biol. Soc. Washington, 57:39, June 28, 1944; Hall, Journ. Mamm., 26:181, July 19, 1945.
Type.—Male, adult, skull alone; no. 31232, Mus. Vert. Zoöl.; Port Santa Cruz, Suemez Island, Alaska; March 24, 1920; obtained by George Willett.
The skull (plates 5-7) is complete and unbroken. Of the upper incisors only right I3 is present. Otherwise the teeth are present and unbroken.
Range.—Known only from the type locality. See figures 25, 26 on pages 95, 134.
Characters for ready recognition (only the male known).—Differs from M. e. celenda in basilar length less than 38.2, from M. e. salva, initis and haidarum in zygomatic breadth more than distance between last upper molar and jugular foramen.
Description.—Size and Color.—No external measurements or skins available.
Skull.—Male: See measurements and plates 5-7. As described in Mustela erminea richardsonii except that: Weight, 1.8 grams; basilar length, 34.3; length of tooth-rows about the same as length of tympanic bulla; breadth of rostrum measured across lacrimal processes more than a third of basilar length; interorbital breadth more than distance between glenoid fossa and posterior margin of external auditory meatus; zygomatic breadth more than distance between last upper molar and jugular foramen.
Female.—Skull not available.
From alascensis and salva, seclusa differs in larger teeth, shorter skull, much larger preorbital and interorbital regions, actually as well as in relation to basilar length. Excepting the teeth, which are of about the same size, the same general differences obtain in comparison with initis which, however, is 29 per cent heavier.
From celenda, seclusa differs in smaller skull in all parts measured, being 22 per cent lighter. The teeth are about the same size. In relation to its length the skull of seclusa is much broader and deeper. From haidarum, seclusa differs in: teeth larger; skull shorter and more convex in dorsal outline along median longitudinal axis; in relation to basilar length, skull broader, deeper and braincase relatively shorter.
Remarks.—The characters shown in the one available skull are far outside the limits of individual variation for other known subspecies. Other specimens are much to be desired to ascertain what the "average" individual is like and to learn the characters of the female.
Specimen examined.—One, the holotype.
Mustela erminea haidarum (Preble)
Ermine
Putorius haidarum Preble, Proc. Biol. Soc. Washington, 12:169, August 10, 1898.
Mustela haidarum, Miller, U. S. Nat. Mus. Bull., 79:97, December 31, 1912.
Mustela erminea haidarum, Hall, Proc. Biol. Soc. Washington, 57:38, June 28, 1944; Hall, Journ. Mamm., 26:181, July 19, 1945.
Type.—Male, adult, skull, skeleton and skin; no. 94430, U. S. Nat. Mus., Biol. Surv. Coll.; Massett, Queen Charlotte Islands, British Columbia; March 17, 1898; obtained by J. H. Keen; original no. 1800x.
The skull is unbroken and complete except for osseous tissue destroyed in the region of each postorbital process; this is the result of infestation of the frontal sinuses by parasites. The skeleton is complete down to the distal ends of the tibiae; the more distal bones are in the skin. The first, right, upper incisor is missing. Otherwise the teeth all are present and entire.
The skin is in the white, winter pelage but the new under fur is visible along the back and on the head although mostly covered with white hair.
Range.—Queen Charlotte Islands. See figure 25, page 95.
Characters for ready recognition.—Differs from M. e. celenda in chest white (not mostly covered by brown patch), proximal two-thirds of under side of tail colored like underparts instead of upper parts, in males basilar length less than 38.2; from M. e. seclusa, in male, in zygomatic breadth less than distance between last upper molar and jugular foramen; from M. e. richardsonii and alascensis, in both sexes, in proximal two-thirds of under side of tail colored like underparts instead of upper parts, interorbital breadth not less than distance from glenoid fossa to posterior margin of external auditory meatus; from M. e. anguinae and fallenda, in both sexes, in light-colored underparts more than half the width of dark-colored upper parts, proximal two-thirds of under surface of tail colored like underparts instead of upper parts, interorbital breadth equal to or more than distance between glenoid fossa and posterior margin of external auditory meatus.
Description.—Size.—Male: Two adults, U.S.N.M., no. 100622, from Cumsheva Inlet, and Amer. Mus. N. H., no. 37411, and the type, measure, respectively, as follows: Total length, 283, 290, 275; length of tail, 70, 75, 60; length of hind foot, 39, 40, 37.
Female: Corresponding measurements of an adult, no. 100624, and a young individual, no. 100623, each from Cumsheva Inlet, are: 252, 250; 63, 61; 31, 32.
Color.—As described in Mustela erminea richardsonii except that underparts not Sulphur Yellow but ranging from near (e) Colonial Buff through Marguerite Yellow to almost pure white; color of underparts extends distally on posterior sides of forelegs and onto toes but in many specimens interrupted at wrist by color of upper parts; color of underparts extends onto proximal three-fourths of under side of tail as length of tail is measured along tail-vertebrae; least width of color of underparts averaging, in 5 males, 79 (66-130) per cent of greatest width of color of upper parts. Black tip of tail in same males averaging 62 (60-70) mm. which is 92 (83-115) per cent of length of tail-vertebrae.
The close correspondence in color-pattern of this weasel with the Arctic races, arctica, polaris, semplei and kadiacensis is noteworthy, and distinguishes it from weasels on the adjacent mainland and adjoining islands to the north and south. The color of the upper parts is darker than in the four Arctic races named.
Skull.—Male (7 adults): See measurements and plates 5-7. As described in Mustela erminea richardsonii except that: Weight, 1.9 (1.7-2.0) grams; basilar length, 36.7 (35.6-37.5); length of tooth-rows more than length of tympanic bullae; breadth of rostrum measured across lacrimal processes more than a third of basilar length; interorbital breadth more than distance between glenoid fossa and posterior margin of external auditory meatus; zygomatic breadth barely less than distance between last upper molar and jugular foramen.
Female (2 adults): See measurements and plates 11-13. As described in Mustela erminea richardsonii except that: Weight, 1.3 and 1.4 grams; basilar length, 34.2; length of tooth-rows more or less than (about equal to) length of tympanic bulla; breadth of rostrum more than 30 per cent of basilar length; interorbital breadth not less than distance between glenoid fossa and posterior margin of external auditory meatus.
From richardsonii, haidarum differs in that skull of the male is actually larger in its anterior part (breadth of rostrum, interorbital breadth and orbitonasal length) but all measurements of other parts average less. In relation to the basilar length, the tympanic bulla is shorter but all other measurements are more. In the skull of the female, which is 23 per cent heavier, the width of the tympanic bulla and anteroposterior extent of the inner lobe of M1 are the same; in all other measurements the female of haidarum is larger, and in relation to the basilar length all measurements are more except the depth of the skull at the anterior margin of the basioccipital and the width of the tympanic bulla, which are less. By actual weight the skull of the male is 25 per cent lighter and the skull of the female 24 per cent heavier than in richardsonii. From fallenda and anguinae, haidarum differs in that measurements of the skulls of both sexes either average more, or are uniformly more, with two exceptions. These are the lesser length and breadth of the tympanic bulla, in comparison with males of fallenda, and the dimensions of M1 which are about the same in all three races concerned. The pre- and interorbital parts are larger in relation to the remainder of the skull. The postorbital breadth is actually a third more than in fallenda. In relation to the basilar length, the tympanic bulla is shorter and the braincase deeper than in males of anguinae. The skull of the male is 27 per cent heavier than that of fallenda and 58 per cent heavier than that of anguinae. The skull of the female is 59 and 50 per cent heavier than those of fallenda and anguinae, respectively. Comparison of the skull with those of alascensis, celenda and seclusa has been made in the accounts of those subspecies.
Remarks.—The available specimens of this ermine were obtained by J. H. Keen in 1898, Wilfred H. Osgood and E. A. Lewis in 1900, W. W. Brown in 1914, J. A. Munro in 1917 and 1918, and Allan Brooks in 1920. M. e. haidarum has more claim to full specific status than any other race of ermine because the diagnostic structural features are numerous and individually of relatively great degree. Indeed, individual variation appears not to bridge the gap between any population of haidarum and other subspecies and strong reasons could be advanced for according haidarum the status of a full species. It differs from the subspecies of erminea on the adjoining mainland and adjoining islands to the north and south and agrees with the Arctic races (arctica, polaris, semplei and kadiacensis) in great extent of the color of the underparts, extension of this color onto the underneath side of the tail, long black tip of the tail and general form of the skull including the relatively heavy preorbital region. The color although darker than in the Arctic subspecies, is lighter than in the insular races immediately to the north and south. In combination, the features mentioned could be taken as indication that haidarum is a relict population from a former glacial period. Assuming that it is a relict population, the color may have become slightly darker since that period but the main response appears to have been a decrease in size for this is a much smaller animal than the Arctic ermines. The size is about what would be expected if one were to judge by the slightly larger ermines on the islands of southeastern Alaska to the north and the smaller ermine on Vancouver Island to the south.
The ermines of the islands of southeastern Alaska, excepting possibly the incompletely known seclusa, have fewer characters of the Arctic races and more characters of the races of the adjoining mainland. Therefore, a possible inference is that the distinctive characters of ermines of the Alaskan islands developed with the aid of isolation from stocks which reached the islands after the glacial period. M. e. haidarum may have found its way to the Queen Charlotte Islands in the glacial period.
Specimens examined.—Total number, 17, as follows. Arranged by locality from north to south. Unless otherwise indicated, specimens are in the U. S. National Museum.
British Columbia. Queen Charlotte Islands. Masset, 7 (4[74], 1[2], 1[59]); Skidegate, 1; Graham Island, 5 (2[94], 1[77], 1[2]); Cumsheva Inlet, 3; no locality more definite than Queen Charlotte Islands, 1[2].
Mustela erminea anguinae Hall
Ermine
Mustela cicognanii anguinae Hall, Univ. California Publ. Zoöl., 38:417, November 8, 1932.
Putorius cicognanii, Baird, Mamm. N. Amer., p. 161, 1858 (part).
Putorius streatori, Swarth, Univ. California Publ. Zoöl., 10:102, February 13, 1912.
Mustela erminea anguinae Hall, Journ. Mamm., 26:79, February 27, 1945; Hall, Journ. Mamm., 26:181, July 19, 1945.
Type.—Male, adult, complete skeleton (no skin); no. 12482, Mus. Vert. Zoöl., French Creek, Vancouver Island, British Columbia; found as a desiccated carcass on May 1, 1910; obtained by Harry S. Swarth.
Range.—Vancouver Island, British Columbia. See figures 25, 27 pages 95, 149.
Characters for ready recognition.—Differs from M. e. haidarum, in both sexes, in light-colored underparts less than half the width of dark-colored upper parts, proximal two-thirds of under surface of tail colored like upper parts instead of underparts, interorbital breadth less than distance between glenoid fossa and posterior margin of external auditory meatus; from M. e. fallenda, in both sexes, anterior margin of tympanic bullae flush with squamosal rather than projecting from floor of braincase, in males by sagittal crest absent, in females by total length more than 238 and tooth-rows about same length as, instead of longer than, tympanic bulla; from M. e. streatori, in male, by sagittal crest absent and hind foot ordinarily more than 33.5, in female by hind foot more than 27.5, basilar length more than 30.2; from M. e. olympica, in males, by greater average size, hind foot ordinarily more than 33.4 and interorbital breadth ordinarily more than 8.5, in females by larger size, total length more than 235, tail more than 65, hind foot more than 27.5, basilar length more than 30.2.
Description.—Size.—Male: Sixteen adults and subadults yield average and extreme measurements as follows: Total length, 272 (261-284) mm.; length of tail, 81 (74-86); length of hind foot, 35.0 (33.5-36).
Female: Five adults and subadults have corresponding measurements as follows: 247 (241-257); 69 (66-73); 30.0 (28.0-32.0).
Color.—As described in Mustela erminea streatori except that: occasionally white in winter; upper parts about tone 2 of Dark Chocolate of Oberthür and Dauthenay; least width of color of underparts averaging, in 7 adult males, 6 (0-15) per cent of greatest width of color of upper parts. Black tip of tail in same series averaging 37 (26-46) mm. which is 46 (32-54) per cent of length of tail-vertebrae.
Skull.—Male (based on 13 adults): See measurements and plates 5-7. As described in Mustela erminea richardsonii except that: Weight, 1.2 (1.0-1.3) grams; basilar length, 34.0 (32.5-35.6); length of tooth-rows more or less (usually less) than length of tympanic bulla.
Female (based on 5 adults): See measurements and plates 11-13. As described in Mustela erminea richardsonii except that: Weight 0.9 (0.77-1.06) grams; basilar length, 31.5 (30.9-31.8) grams; length of tooth-rows more or less than (approximately same as) length of tympanic bulla; breadth of rostrum more than 30 per cent of basilar length.
The sexual dimorphism in the skull is slight, the skull of the male being only a third heavier than that of the female. In fallenda of the adjacent mainland to the east the male is three-fourths heavier than the female. In comparison with fallenda, males are smaller, averaging less in every cranial and dental measurement taken and by weight are a fifth lighter; sagittal crest absent rather than present; tympanic bullae flush with squamosal rather than projecting below floor of braincase; in relation to basilar length, tympanic bullae smaller, braincase deeper and broader, skull wider interorbitally and across zygomata. Females are larger than in fallenda, and with one exception average larger in every cranial and dental measurement taken, being 6 per cent heavier. The one exception mentioned is the lesser actual length of the tympanic bulla in anguinae, in which the length of the tooth-rows is about the same as, rather than less than, the length of the tympanic bulla. The postorbital breadth is greater than in fallenda and the anterior edges of the tympanic bullae are flush with the squamosals rather than projecting below the floor of braincase. In relation to the skull as a whole the preorbital and interorbital parts are larger.
In comparison with streatori, skulls of males are of about the same size, anguinae being only 9 per cent heavier. The length of the tooth-rows is ordinarily less than, rather than about equal to, the length of the tympanic bulla; sagittal crest wanting rather than present since in anguinae the temporal muscles meet usually only at the posterior end of the braincase instead of all along the midline on its top; tympanic bullae narrower and more nearly flush with squamosal (less protruded from braincase). Relative to the basilar length, the zygomatic breadth is more, the tympanic bullae are narrower, and the braincase is deeper at the anterior end of the basioccipital. The female is 41 per cent heavier than streatori, there being no overlap in most cranial and dental measurements. M1, however, is approximately the same size in each subspecies. The tooth-rows and tympanic bulla are of almost equal length whereas in streatori the length of the tooth-rows is less than that of the bulla.
Differences from olympica, in males, are: M1 shorter; all other measurements of teeth and parts of skull averaging larger; skull 20 per cent heavier; tooth-rows averaging shorter than tympanic bulla rather than about the same; relative to basilar length, braincase deeper at anterior end of basioccipital and tooth-rows shorter. The skull of the female is 64 per cent heavier, larger in every measurement taken without overlap; temporal ridges meeting, rather than separated, at lambdoidal crest; length of tooth-rows about equal to, rather than shorter than, tympanic bulla; in relation to basilar length, skull deeper, orbitonasal length more, mastoid and zygomatic breadths more, and tympanic bullae shorter.
Remarks.—References in the literature to this insular race mostly were under the name streatori until 1932 when in the course of the present study the name anguinae was proposed. A few specimens have been taken by nearly every student of small mammals who has collected on Vancouver Island. Arthur Peake and Herbert Laing have probably collected more specimens than any other two zoölogists.
M. c. anguinae is noteworthy for the slight secondary sexual variation in size; the disparity between the two sexes is less than in any other American subspecies of erminea. By linear measurement the body of the female is only 7 per cent shorter than in the male (178 versus 191 mm.). Linear measurements and weights of the skulls of the two sexes are further indicative of this approximation in size. By weight the skull of the female is only a fourth lighter than that of the male, or, stated in another way, the male's skull is only a third heavier (1.2 versus 0.9 grams).
No geographic variation has been detected between lots of specimens from different parts of Vancouver Island. The one specimen available from Salt Spring Island presents no obvious differences from selected individuals from Vancouver Island.
The winter pelage is more often brown than white. Of 17 specimens seen in winter pelage or in transition pelage, only 6 are white. These 6 are from Comox, Stamp River, Hilliers, Jeune Landing and Port Alice. Of the 34 specimens in brown pelage, 7 have the dark color of the upper parts meeting on the abdomen. Six of the 34 have brown color on the pectoral region. In two, this is a separate patch but in the other four the dark color is a continuation of the upper parts and extends in front of each foreleg over part of the pectoral region, but the two extensions, one from either side, do not meet on the underparts. The color of the lips was recorded in 22 individuals: one had both the upper and lower-lips white; 7 had the upper lips brown and the lower lips white; in 14 both the upper and lower-lips were brown.
Specimens examined.—Total number, 40, listed by localities from north to south as follows. Unless otherwise indicated, specimens are in the National Museum of Canada.
British Columbia. Vancouver Island: Cape Scott, 4; Shushartie, 1; Quatsino, 1[74]; Jeune Landing, 1[74]; Port Alice, 5[15]; Marble Creek, Quatsino Sound, 1[22]; Port Hardy, 5; Sayward, 2; Bear Lake, 4; Bear River, 1; Comox, 4(3[85]); Stamp River, Alberni, 1[31]; Errington, 1[74]; French Creek, 1[74]; Hilliers, 1[74]; Craigs Crossing, 1[74]; Nanaimo, 2[22]; Cowichan Lake, 1[22]; Duncan, 2[85]; Salt Spring Island, 1[85].
Mustela erminea fallenda Hall
Ermine
Mustela erminea fallenda Hall, Journ. Mamm., 26:79, February 27, 1945; Hall, Journ. Mamm., 26:181, July 19, 1945.
Putorius streatori Merriam, N. Amer. Fauna, 11:13, June 30, 1896 (part-Sumas).
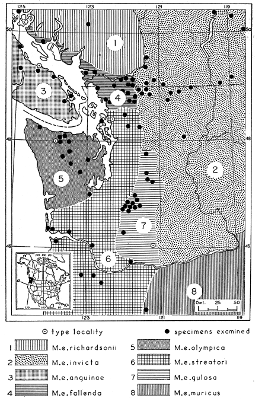
Fig. 27. Map showing known occurrences and probable geographic ranges of the subspecies of Mustela erminea in Washington and parts of British Columbia and Oregon.
Type.—Male, adult, skull and skin; no. 7096, Nat. Mus. Canada; Huntingdon, British Columbia; May 21, 1927; obtained by C. H. Young, original no. 317.
The brown summer skin is well made. The skull (plates 5-7) is complete. Right p2 has the crown broken away; otherwise the teeth all are present and entire.
Range.—On mainland in immediate vicinity of coast from probably opposite Texada Island, British Columbia, south to Lake Whatcom, Washington, and east to Mount Baker Range on International boundary. See figures 25, 27 on pages 95, 149.
Characters for ready recognition.—Differs from M. e. haidarum, in both sexes, in light-colored underparts less than half the width of dark-colored upper parts, proximal two-thirds of under surface of tail colored like upper parts instead of underparts, interorbital breadth less than distance between glenoid fossa and posterior margin of external auditory meatus; from M. e. richardsonii in both sexes, by upper lips brown rather than white, in males hind foot less than 41 and basilar length less than 38.3, in females hind foot less than 29, basilar length less than 31.4 and breadth of rostrum more, instead of less, than 30 per cent of basilar length; from M. e. invicta, in both sexes, by upper lips brown (not white); in males by skull averaging shorter (basilar length 35.7 versus 37.0); in females by breadth of rostrum more, instead of less, than 30 per cent of basilar length; from M. e. anguinae, in both sexes, by anterior margin of tympanic bulla projecting from floor of braincase rather than flush with squamosal (the difference is slight in females), in males by sagittal crest present, in females by total length less than 238 and tooth-rows longer than, instead of about same length as, tympanic bulla; from M. e. streatori, in both sexes, by black tip of tail more than half of length of tail-vertebrae, in males hind foot more than 33.7, tympanic bulla longer than, instead of about same length as, upper tooth-rows; weight of skull more than 1-1/4 grams, in females weight of skull more than 0.7 grams, length of lateral side of P4, 4 mm. or more; from M. e. olympica, in males, length of hind foot more than 33, black tip of tail more than 36.5 mm., weight of skull more than 1.2 grams, basilar length more than 33.5, in females length of hind foot more than 25.5, weight of skull more than 0.66 grams, basilar length more than 28.4; from M. e. gulosa, in both sexes, by anterior margin of tympanic bulla projecting below floor of braincase rather than flush with squamosal (the difference is slight in females), in males hind foot more than 33.5, weight of skull more than 1-1/4 grams, basilar length more than 33.9, in females by total length more than 222, hind foot longer than 26, weight of skull more than 0.7 grams, basilar length more than 29.
Description.—Size.—Male: Seven adult topotypes yield average and extreme measurements as follows: Total length, 278 (249-305); length of tail, 77 (69-81); length of hind foot, 36.5 (34-40). A male topotype of unknown age weighed 113 grams.
Female: Two adult topotypes, with actual measurements in parentheses, average as follows: Total length, 232 (228-236); length of tail, 60 (57-62); length of hind foot, 27 (27-27). An adult from Morovitz Guard Station, Wash., weighed 54 grams.
Color.—Winter pelage rarely white, brown pelage indistinguishable from summer pelage except for slightly more smoky tinge in winter in specimens from some localities; otherwise as described in Mustela erminea streatori except that least width of color of underparts averaging, in seven adult topotypes, 18 (0-37) per cent of greatest width of color of upper parts. Black tip of tail averaging, in same series, 45 (38-52) mm. which is 58 (53-65) per cent of length of tail-vertebrae.
In comparison with richardsonii and invicta, fallenda differs in darker color of upper parts and their extension at the expense of the light-colored underparts which are narrower by a half. In correlation with this restriction in area of the light-colored underparts, the upper lips are brown instead of white. In comparison with anguinae, olympica and streatori, the longer black tip on the tail is the principal difference in color. From gulosa, fallenda differs in slightly darker color of upper parts and in narrow underparts, the width of the same being only about a fifth instead of a third of the width of the dark-colored upper parts.
Skull.—Male (based on 7 adults): See measurements and plates 5-7. As described in Mustela erminea richardsonii except that: Weight, 1.5 (1.3-1.7) grams; basilar length, 35.7 (34.3-38.2).
Female (based on 6 ads.): See measurements and plates 11-13. As described in Mustela erminea richardsonii except that: Weight, 0.85 (0.73-1.0) grams; basilar length, 30.6 (29.4-31.7); breadth of rostrum more than 30 per cent of basilar length.
In comparison with richardsonii, skulls of males differ as follows: averaging smaller in every measurement taken with no overlap in several dimensions; 40 per cent lighter; in relation to basilar length, rostrum (orbitonasal length) longer and skull slightly broader interorbitally. Females average smaller in every cranial and dental measurement taken with no overlap in basilar length, length of tooth-rows and length of tympanic bulla; 22 per cent lighter; breadth of rostrum more, rather than less, than 30 per cent of basilar length; in relation to basilar length, pre- and interorbital parts of skull larger, and mastoid breadth more.
Differences from males of olympica are: size larger with no overlap in most measurements; 50 per cent heavier; tympanic bullae longer than upper tooth-rows rather than of about equal length; in relation to basilar length, rostrum shorter, braincase wider and deeper, zygomata more expanded. Females are larger with no overlap in most measurements; 35 per cent heavier; in relation to basilar length, pre- and interorbital regions narrower, braincase deeper and wider across mastoids.
Differences from streatori, in males, are: skull averaging larger in every cranial and dental measurement taken; 36 per cent heavier; tympanic bulla longer than, instead of about same length as, upper tooth-rows. In females the inner lobe of M1 is shorter anteroposteriorly; otherwise all measurements of fallenda average larger and it is 33 per cent heavier; rostrum and interorbital region broader in relation to remainder of skull.
In comparison with gulosa, skulls of males differ as follows: averaging larger in every measurement taken with no overlap in several dimensions; 50 per cent heavier; tympanic bullae with anterior margins projecting slightly below squamosals rather than flush with same; length of bulla more than, rather than about same as, that of upper tooth-rows. Considering the great difference in size, the relative proportions are remarkably alike. In females, length of inner lobe of M1 about the same; otherwise averaging larger in every measurement taken; 44 per cent lighter; relative to basilar length, tooth-rows longer, skull wider across zygomata and mastoids, rostrum and interorbital regions slightly narrower, skull shallower in plane of last upper molars.
Comparisons with haidarum, invicta and anguinae are made in accounts of those subspecies.
Remarks.—Until the name fallenda was proposed in the course of the present study, most of the specimens of this race were assigned to streatori.
Intergradation with streatori is complete as it is also with invicta and richardsonii, in other words with each of the subspecies whose ranges meet that of fallenda. In color and in size the difference is least between streatori and fallenda. As between fallenda and invicta the size is not greatly different and the intergradation in color is gradual. Between fallenda and richardsonii intergradation is somewhat different and to fully appreciate its nature we should remember that the color of fallenda resembles that of the saturate coastal races, streatori, anguinae and olympica although the black tip of the tail is longer. In this latter feature and in several cranial details, as well as in greater degree of secondary sexual variation in size, fallenda resembles richardsonii. Because the two differ more than do most subspecies of ermine whose ranges meet, some of the intergrades at first inspection appear to be widely different from either parent stock. For example, specimens from Alta Lake, British Columbia, may give this impression because the combination of large size and dark color suggests a kind of ermine different from either fallenda or richardsonii. In no instance, however, has there been found in these intergrades any character other than those occurring in one or the other of the two parent races.
Along the coast in the north part of the geographic range assigned to fallenda, some specimens nearly typical of richardsonii have been taken so near to the place where fairly typical fallenda was obtained that I have doubted whether there is intergradation in the usual fashion in this area; more specimens will have to be obtained from this coastal area to resolve the doubt one way or the other.
The winter pelage is brown in all specimens at most localities. The only white pelage seen was in each of three specimens from Glacier, Whatcom County, Washington. A fourth specimen from there is in brown winter pelage. At any one locality there is much variation in the degree to which the dark color of the upper parts encroaches on the area that in most other races is light-colored. An extreme degree of encroachment is shown by a specimen taken on December 1, 1935, by R. A. Cummings, at Vancouver, British Columbia, in which the light color occurs only in three restricted areas, the chin, the throat and the lower breast; otherwise the coat is brown. There are other specimens, for instance from the type locality, which differ mainly in having an additional white spot in the inguinal region. The opposite extreme, in a specimen also from the type locality, is where the least width of the light-colored underparts on the abdominal region is a third of the circumference of the body. The two extremes are connected by a dozen intermediate stages. Of 64 specimens in which the color of the lips was carefully examined, one, from Vancouver, has both the upper and lower-lips brown; 9 have both the upper and lower-lips white; and 54 have the upper lips brown and the lower lips white.
Specimens examined.—Total number, 72, arranged by localities from north to south. Unless otherwise indicated, specimens are in the National Museum of Canada.
British Columbia. Horseshoe Lake, Stillwater, 2; Vancouver, 1[74]; Point Grey, 1[31]; Port Moody, 5[91]; Chilliwack, 8 (2[75], 4[91], 1[60]); Sumas, 19 (18[75], 1[60]); Thurstons Ranch, 2; Cultus Lake, 2; Mt. Baker Range, 5[75]; Lihumption Park, 1; Huntingdon, 14; Tami Hy Creek, 1.
Washington. Whatcom County: Semiahmoo, 1[91]; New Whatcom, 1[68]; Lake Whatcom, 2[91]; 5 mi. W Glacier, 1[51]; Glacier (3 at 900 ft.), 4[91]; E Side Easton Glacier, Mt. Baker, 1[55]; Morovitz Guard Station, 831 ft., 1[55].
Mustela erminea olympica Hall
Ermine
Mustela erminea olympica Hall, Journ. Mamm., 26:81, February 27, 1945; Hall, Journ. Mamm., 26:181, July 19, 1945.
Mustela rixosa, Svihla and Svihla, Murrelet, 13:24, January, 1932.
Mustela rixosa rixosa, Svihla and Svihla, Murrelet, 14:39, May, 1933.
Type.—Male, adult, skull and skin; no. 90738, U. S. Nat. Mus., Biol. Surv. Coll.; near head of Soleduc River, 4500 ft., Olympic Mountains, Clallam County, Washington; April 28, 1897; obtained by Vernon Bailey, original no. 6213.
The skin is well prepared and in good condition. The skull (plates 5-7) is unbroken and the teeth all are present and entire.
Range.—Olympic Peninsula, Washington, south to Olympia. See figures 25, 27 on pages 95, 149.
Characters for ready recognition.—Differs from M. e. anguinae, in males, by lesser average size, hind foot ordinarily less than 33.4, and interorbital breadth ordinarily less than 8.5, in females by smaller size, total length less than 235, tail less than 65, hind foot less than 27.5, basilar length less than 30.2; from M. e. fallenda, in males, by length of hind foot less than 33, black tip of tail less than 36.5, weight of skull less than 1.2 grams, basilar length less than 33.5, in females length of hind foot less than 25.5, weight of skull less than 0.6 grams, basilar length less than 28.4; from M. e. streatori by smaller size, in males hind foot less than 33.0, basilar length ordinarily less than 32.5, in females by hind foot ordinarily not longer than 24, by breadth of rostrum less than 8.6, depth of braincase at posterior border of upper molars less than 7.6.
Description.—Size.—Male: Twelve individuals of adult proportions yield average and extreme measurements as follows: Total length, 243 (205-269); length of tail, 65 (60-74); length of hind foot, 31 (29-32).
Female: Corresponding measurements of six females are: 196 (188-208), 52 (45-60?), 23.4 (22.7-24.0). An adult weighs 30 grams.
Color.—As described in Mustela erminea streatori except that least width of color of underparts averaging, in 12 males of adult proportions, 5 (0-11) per cent of greatest width of color of upper parts. Black tip of tail averaging, in same series, 26 (20-35) mm., which is 40 (31-58) per cent (average the same as in streatori) of length of tail-vertebrae.
Skull.—Male (based on 5 adults): See measurements and plates 5-7. As described in Mustela erminea richardsonii except that: Weight, 1.0 (0.9-1.1) grams; basilar length, 31.8 (30.6-32.5); length of tooth-rows more or less than (about equal to) length of tympanic bulla.
Female (illustrated by 3 adults): See measurements and plates 12-14. As described in Mustela erminea richardsonii except that: Weight, 0.55 (0.52-0.58) grams; basilar length, 27.1 (26.7-27.5); breadth of rostrum more than 30 per cent of basilar length.
In comparison with streatori, skulls of corresponding sex average smaller in every measurement taken with no overlap in most of those of females. Exception is to be made for the inner lobe of M1 in males where the size is the same. By weight males are smaller by 10 per cent and females by 14 per cent. In relation to other parts of the skull the tympanic bullae are narrower and in females they are shorter as well. Comparison with anguinae and fallenda has been made in the accounts of those subspecies.
Remarks.—The smaller size, especially of females, is the principal feature distinguishing this race from streatori. On the basis of available data the female of olympica is smaller than that of any other race and hence is the smallest adult weasel of the species erminea, in either the Old World or in America.
Intergradation with streatori is indicated by specimens from the southern end of Puget Sound. These specimens are intermediate in size between typical examples of the two races concerned.
The color of the upper parts is uniform and the color pattern varies less than in geographically adjoining races. The white color of the underparts is restricted to a thin line on the abdominal region, but widens out posteriorly in the inguinal region and anteriorly over the pectoral region, throat, chin and lower lips. The upper lips are brown. The brown of the upper parts extends around in front of each foreleg, the two brown areas not quite meeting on the lower throat. The above description applies to each of the 19 specimens examined with regard to these details. Every specimen seen in the winter coat was brown, not white.
Specimens examined.—Total number, 20, arranged by counties from north to south. Unless otherwise indicated, specimens are in the U. S. National Museum.
Washington. Clallam County: Clallam Bay, 2 (1[74], 1[94]); Elwha, 2[10]; Johnsons Ranch, 1[60]; Happy Lake, 1[60]; Boulder Lake, 2[60]; near head of Soleduc River, 4500 ft., 1; 12 mi. S Port Angeles, 1[10]. Jefferson County: Hayes Cr., 2000 ft., Elwha River, 2; head N Fork Quinault River, 4000 ft., 1; Duckabush, 3; N Fork Skokomish River, 1. Mason County: Lake Cushman, 2[76]; 4 mi. S Olympia, 1.
Mustela erminea streatori (Merriam)
Ermine
Putorius streatori, Merriam, N. Amer. Fauna, 11:13, pl. 2, figs. 5, 5a, 6, 6a, June 30, 1896.
Putorius cicognanii, Baird, Mamm. N. Amer., p. 161, 1858 (part unless no. 2395 was a female of M. frenata).
Putorius pusillus, Baird, Mamm. N. Amer., p. 159, 1858 (part).
Putorius (Gale) vulgaris, Coues, Fur-bearing animals, p. 102, 1877 (part).
Mustela streatori streatori, Miller, U. S. Nat. Mus. Bull., 79:96, December 31, 1912; Grinnell, Univ. California Publ. Zoöl., 40:101, September 26, 1933.
Mustela cicognanii streatori, Hall, Murrelet, 12:22, January, 1931; Hall, Univ. California Publ. Zoöl., 38:417, November 8, 1932.
Mustela erminea streatori, Hall, Journ. Mamm., 26:77, February 27, 1945; Hall, Journ. Mamm., 26:181, July 19, 1945.
Mustela rixosa, Beer, Journ. Mamm., 29:296, August 31, 1948.
Type.—Male, adult, skull and skin; no. 76646, U. S. Nat. Mus., Biol. Surv. Coll.; Mount Vernon, Skagit Valley, Skagit County, Washington; February 29, 1896; obtained by D. R. Luckey, original no. 3.
The skull is unbroken and the teeth all are present and entire. The skin, in brown winter pelage, is stuffed and in good condition.
Range.—Western Washington along eastern side of Puget Sound, western Oregon from the Cascades to the coast, and northwestern California south in the humid coastal district nearly to the Golden Gate. See figures 25, 27 on pages 95, 149.
Characters for ready recognition.—Differs from M. e. anguinae, in male, by sagittal crest present and hind foot ordinarily less than 33.5, in female by hind foot less than 27.5, basilar length less than 30.2; from M. e. fallenda, in both sexes, by black tip of tail less than half of length of tail-vertebrae, in males hind foot less than 33.7, tympanic bulla about same length as, instead of longer than, upper tooth-rows; weight of skull less than 1-1/4 grams, in female weight of skull less than 0.7 grams, length of lateral side of P4 less than 4 mm.; from M. e. olympica, by larger size, in males hind foot more than 33.0, basilar length ordinarily more than 32.5, in females by hind foot ordinarily longer than 24, by breadth of rostrum more than 8.6, depth of braincase at posterior border of upper molars more than 7.6; from M. e. gulosa and muricus, in both sexes, by upper lips brown (not white), light color of underparts extending down hind leg no farther than knee, depth of skull at posterior border of upper molars more than 7.7 in females and ordinarily more than 9.6 in males, further from muricus by tail more than 62 in males and more than 49 in females; from M. e. invicta by upper lips white (not brown), in males hind foot more than 36 and basilar length more than 35, in females hind foot more than 29.5 and basilar length more than 30.5.
Description.—Size.—Male: Twelve adults from Blaine and Tillamook, Oregon, yield average and extreme measurements as follows: Total length, 255 (245-275); length of tail, 72 (64-80); length of hind foot, 31.5 (30.0-33.5).
Female: Seven adults from Blaine and Tillamook, Oregon, yield average and extreme measurements as follows: Total length, 214 (193-230); length of tail, 55 (50-63); length of hind foot, 25 (24-27).
Color.—Winter and summer pelages indistinguishable; upper parts uniform and ranging from Raw Umber to slightly darker (16n), and about tones 1 to 3 of Dark Chocolate of Oberthür and Dauthenay, pl. 342; underparts white, in summer rarely with a faint buffy suffusion in pectoral region; color of underparts extends from chin, and often lower lips, posteriorly to inguinal region, distally on posterior sides of forelegs onto antipalmar faces of toes (sometimes interrupted at and above wrist) and on medial sides of hind legs hardly to knee. Least width of color of underparts averaging, in twelve adults from Blaine and Tillamook, 10 (0-47) per cent of greatest width of color of upper parts. Black tip of tail, in same series, averaging 28 (24-33) mm. which is 40 (34-47) per cent of length of tail-vertebrae.
Skull.—Male (based on 12 adults): See measurements and plates 5-7. As described in Mustela erminea richardsonii except that: Weight, 1.1 (1.0-1.2) grams; basilar length, 33.2 (32.5-33.8); length of tooth-rows more or less than (about same as) length of tympanic bulla.
Female (based on 7 adults): See measurements and plates 12-14. As described in Mustela erminea richardsonii except that: Weight, 0.64 (0.60-0.67) grams; basilar length, 28.5 (27.6-29.5); breadth of rostrum more than 30 per cent of basilar length.
Comparison with anguinae, fallenda, olympica, gulosa and muricus is made in accounts of those subspecies.
Remarks.—This weasel is rare in collections and the best material of it was obtained by Alex Walker in Tillamook County, Oregon, where he resides. The almost ideal series of 30 specimens showed the range of secondary sexual, age, and individual variation expectable in the small ermines of the Pacific Coast of the United States and was the means of allowing satisfactory decision on questions of classification in the related subspecies in which individuals are of comparable size.
Intergradation with each of the geographically adjoining subspecies, olympica, fallenda, invicta, gulosa and muricus is shown by specimens examined. With the last mentioned subspecies, intergradation is shown by two specimens from as far south as Siskiyou County, California, assigned to muricus.
The application of the name streatori is difficult because it was based on a specimen from a place where two clines cross. The north-south cline is one of size which decreases to the south. The east-west cline is one of intensity of color, the westernmost (coastal) population being the most intensely colored. The type locality of streatori is at the place where two lines perpendicular to one another, and representing the two clines, cross. This intersection is near the place where the ranges of several subspecies meet. The nomenclatural question is, to which one of 6 subspecies should the name streatori apply. Specimens from barely within the geographic boundaries of four of these subspecies so closely resemble topotypes of streatori that a student with material at his disposal from only the area about Puget Sound naturally would apply the name streatori to all of his specimens, and knowing even of the arrangement adopted in the present account the student will have difficulty in identifying his specimens according to it. Not only will the student find the arrangement difficult, but probably unsatisfactory if he thinks of streatori as being the kind of animal represented by topotypes. I conceive of topotypes of streatori as being nontypical of the subspecies; they are intergrades with fallenda. My aim was initially to work out the geographic ranges of subspecies and only subsequently to apply names, according to which type localities fell within the previously determined geographic ranges. By this procedure no greater weight was given to a holotype and to topotypes than to specimens from any other locality.
Of the 40 specimens seen in winter pelage, only one is white. It is from Darrington in the Cascade Mountains of Washington. The 39 others are brown and I doubt that the white pelage ever occurs in the low coastal territory included within the geographic range of streatori. This subspecies resembles anguinae and olympica in the great extension of area of the dark-colored upper parts at the expense of the area of the light-colored underparts. The usual arrangement is one where the brown of the two sides nearly meets on the midventral line leaving a sizable, inguinal area of light color connected by a thin line to the sizable area of light color on the pectoral region. The light color of the pectoral area ordinarily is continuous with the light-colored area of the throat and chin but the dark color of the upper parts extends around in front of each foreleg. These extensions of dark color meet on the chest in only 2 of the 56 specimens examined in this regard. Across the abdomen the dark color is continuous in 4 of the 56 specimens. The lower lips are brown instead of white in only 3 individuals and in 2 of these the lip of one side is brown and its opposite is white. The variation in color-pattern is less than in anguinae or than in fallenda.
Specimens examined.—Total number, 63, arranged alphabetically by states, then by counties from north to south in each state. Unless otherwise indicated, specimens are in the U. S. National Museum.
California. Humboldt County: 10 mi. NE Carlotta, 1[74]. Mendocino County: Russian Gulch State Park, 1[74]. Sonoma County: Mouth of Gualala River, 1[74].
Oregon. Clatsop County: Astoria, 1. Tillamook County: Tillamook, 16 (14[14], 1[59]); Blaine, 12 (7[14], 2[59], 1[93], 2[76]). Washington County: Beaverton, 1[60]; Forest Grove, 1[36]. Clackamas County: Oregon City, 1[46]. Lincoln County: Newport, 1. Linn County: Sico, 1[46]. Lane County: Vida Fish Hatchery, 2[101]; McKenzie Bridge, 1[101]; Mercer, 1[75]. Klamath County: Deschutes River, 6 mi. E Crescent Lake, 1[101]. Douglas County: Gardiner, 1[60]. Curry County: Port Orford, 1; Gold Beach, 2[60].
Washington. Skagit County: N end Whidby Island opposite Deception Pass, 1; Hamilton, 4; Mt. Vernon, 3. Snohomish County: Oso, 550 ft., 1; Darrington, 600 ft., 1. Pacific County: Wallicut River, 2 mi. E Ilwaco, 1[74]. Wahkiakum County: 4 mi. E. Skamokawa, 3[74]. Cowlitz County: 4 mi. E mouth Kalama River, 2[74]; 6 mi. E mouth Kalama River, 1[74]. Skamania County: 15 mi. N Govt. Springs, 1300 ft., 1.
Mustela erminea gulosa Hall
Ermine
Mustela erminea gulosa Hall, Journ. Mamm., 26:84, February 27, 1945; Hall, Journ. Mamm., 26:181, July 19, 1945.
Putorius streatori Merriam, N. Amer. Fauna, 11:14, June 30, 1896.
Type.—Male, subadult, skull and skin; no. 81998, U. S. Nat. Mus., Biol. Surv. Coll.; Trout Lake, Klickitat County, Washington; February 3, 1897; obtained by P. Schmid, original no. 147.
The skin is in brown winter pelage, and appears to have been made up from a skin split along the midventral line from the anus to the forelegs. It probably was dried by a trapper, is well made, and lacks a patch of hair on the left flank but otherwise is in good condition. The skull lacks the central part of the left zygomatic arch and the posterior two-thirds of the right one. The right m2 is represented only by an abortive stump or the broken root, and i1 and i2 on each side are absent; otherwise, the teeth all are present and entire.
Range.—Cascades of Washington from northeastern King County south to Mount Adams. See figures 25, 27 on pages 95, 149.
Characters for ready recognition.—Differs from M. e. invicta and fallenda, in both sexes, by anterior margin of tympanic bulla flush with squamosal rather than projecting below floor of braincase (difference slight in females), in males hind foot less than 33.5, weight of skull less than 1-1/4 grams, basilar length less than 33.9, in females by total length less than 222, hind foot shorter than 26, weight of skull less than 0.7 grams, basilar length less than 29; from M. e. muricus, in both sexes, by upper parts darker, tone 4 of Chocolate or darker (see description of color), least width of light-colored underparts averaging one-third instead of approximately two-thirds of greatest width of dark-colored upper parts, in males, on the average, tail more than 65, weight of skull more than 0.90 grams, basilar length more than 30.8 mm.; from M. e. streatori, in both sexes, by upper lips white (not brown), light color of underparts extending down hind legs below knee, depth of skull at posterior border of upper molars less than 7.7 in females and ordinarily less than 9.6 in males.
Description.—Size.—Male: One adult and four subadults from Mount Rainier yield average and extreme measurements as follows: Total length, 253 (238-266); length of tail, 75 (70-83); length of hind foot, 31.5 (30-33). Corresponding measurements of 9 subadults from Trout Lake are: 257 (233-282); length of tail, 76 (56-83); length of hind foot, 30.2 (26-33).
Female: Of adults, 2 from Mount Rainier and 2 from Trout Lake measure as follows: Total length, 202, 203, 216, 210; length of tail, 54, 52, 57, 51; length of hind foot, 24, 24, 25, 24. The averages for these females are 208, 54, 24.3.
Color.—As described in Mustela erminea richardsonii except that color sometimes brown in winter (with more smoky tinge than summer coat); upper parts ranging from tone 2 through tones 3 and 4 of Dark Chocolate (pl. 342) into tone 4 of Chocolate (pl. 343) of Oberthür and Dauthenay; underparts (always white in winter) in summer Sulphur Yellow or more whitish; least width of color of underparts averaging, in 5 males from Mount Rainier, 31 (18-45) per cent of greatest width of color of upper parts. Black tip of tail, in same series, averaging 34 (29-40) mm., which is 45 (41-50) per cent of length of tail-vertebrae.
Skull.—Male (based on 2 ad. and 13 sad.): See measurements and plates 5-7. As described in Mustela erminea richardsonii except that: Weight, 1.0 (0.95-1.16) grams; basilar length, 32.3 (30.9-33.4); length of tooth-rows more or less than (about equal to) length of tympanic bulla.
Female (illustrated by 5 adults): See measurements and plates 12-14. As described in Mustela erminea richardsonii except that: Weight, 0.59 (0.53-0.65) grams; basilar length, 28.1 (27.8-28.4); breadth of rostrum ordinarily more than 30 per cent of basilar length.
In comparison with streatori, skulls of males and females average smaller in every cranial measurement taken. Teeth of about same size and males 9 per cent, and females 8 per cent, lighter. In relation to basilar length, skull of female shallower, tympanic bullae slightly shorter and, on the average, zygomata less expanded.
In comparison with muricus, males average larger in every measurement taken; 23 per cent heavier; in relation to other dimensions, braincase shallower at anterior end of basioccipital. Females are of about equal size; in relation to other dimensions, braincase shallower and mastoid and zygomatic breadths less.
Comparisons with invicta and fallenda have been made in the accounts of those subspecies.
Remarks.—This is not a strongly marked race and in most of the characters used for differentiating it from other races it resembles either streatori to the west or muricus to the southeast. Nevertheless, there is a geographic area, the southern Cascades of Washington, throughout which individual characters are combined in essentially the same way and there are a few features, for instance, smaller skull of the female, in which gulosa differs from either of its close relatives. In view of these circumstances and because the animals can not well be included in the subspecies streatori or muricus, gulosa is recognized as distinct. The races gulosa and olympica are what might be termed weakly differentiated subspecies in contrast to the strongly differentiated subspecies streatori and muricus.
Of the 21 specimens in winter pelage, 17 are white and four are brown. The brown winter coat is distinctly paler, with more of a smoky tinge, than the brown summer pelage. The light-colored underparts are narrower than in the subspecies immediately to the east but are wider than in the coastal forms to the west. The dark color of the upper parts extends onto the chest in front of the forelegs, as in the coastal forms, in only one of the 13 specimens in summer pelage and in it on one side only. The black tip of the tail is short as in the coastal forms. One specimen is in transitional pelage. It has acquired approximately half of the white winter pelage and was taken on October 12, 1897, at Keechelus Lake.
Specimens examined.—Total number, 38, arranged by counties from north to south. Unless otherwise indicated, specimens are in the U. S. National Museum.
Washington. King County: 2 mi. E Skykomish, 2[51]. Kittitas County: Keechelus Lake, 3 (1[1]); Martin, 1[1]; Easton, 3. Pierce County: James Lake, 4370 ft., Mt. Rainier, 1; Glacier Basin, 5935 ft., Mt. Rainier, 1; Meslers Ranch, 2000 ft., 1 mi. W Rainier Park, 1. Lewis County: Mt. Rainier Nat'l Park, 5 (1 each from: Paradise Park, 5400 ft.; Reflection Lakes, 4900 ft.; Ohanapecosh [Hot] Springs, 2000 ft.; Tahoma Creek, 1[72]; Bear Prairie); also in Mt. Rainier Nat'l Park, Longmire, 3 (1[72], 1[94]). Skamania County: Mt. St. Helens, 6000 ft., 1. Klickitat County: Trout Lake, 18.
Mustela erminea muricus (Bangs)
Ermine
Plates 7, 8, 12, 13, 14 and 41
Putorius (Arctogale) muricus Bangs, Proc. New England Zoöl. Club, 1:71, July 31, 1899.
Putorius streatori leptus Merriam, Proc. Biol. Soc. Washington, 16:76, May 29, 1903. Type from Silverton, San Juan County, Colorado.
Putorius muricus, Stephens, California Mammals, p. 248, 1906.
Putorius cicognani, Taylor, Univ. California Publ. Zoöl., 7:298, June 24, 1911.
Mustela streatori leptus, Miller, U. S. Nat. Mus. Bull., 79:96, December 31, 1912; Bailey, N. Amer. Fauna, 35:48, September 5, 1913; Dixon, Journ. Mamm., 12:72, February 12, 1931; Whitlow and Hall, Univ. California Publ. Zoöl., 40:246, September 30, 1933.
Mustela muricus, Miller, U. S. Nat. Mus. Bull., 79:96, December 31, 1912; Kellogg, Univ. California Publ. Zoöl., 12:358, January 27, 1916.
Mustela cicognanii lepta, Dice, Journ. Mamm., 1:12, November 28, 1919; Hall, Mamm. Nevada, p. 184, July 1, 1946.
Mustela rixosa, Seton, Journ. Mamm., 14:70, February 14, 1933.
Mustela cicognanii leptus, Miller, Journ. Mamm., 14:368, November 13, 1933; Bailey, N. Amer. Fauna, 55:293, August 29, 1936.
Mustela erminea murica, Hall, Journ. Mamm., 26:84, February 27, 1945; Hall, Journ. Mamm., 26:181, July 19, 1945.
Type.—Male, young, skull and skin; no. 9146, collection of E. A. and O. Bangs in Mus. Comp. Zoöl.; Echo, 7500 ft., El Dorado County, California; July 15, 1897; obtained by W. W. Price and E. M. Nutting.
The skull has a fracture along the sagittal suture and fractures on the left side of the braincase but these have been glued, and no part of the skull is missing except in the region of the right P4 which part has been shot away. On the left side m2 never developed. Excepting this tooth and the right P4, all the teeth are present and entire. The skin is well made but has the soles of the hind feet turned up.
Range.—Near 5300 feet (Denver) to 11000 feet (Santa Fe Baldy); typically boreal but taken in Upper Sonoran Life-zone in winter at Denver; from central and southwestern Montana, southern Idaho, and Blue Mountains of southeastern Washington southward east of the Cascade Divide through the Salmon River Mountains and Sierra Nevada at least into Fresno County of California, in the Great Basin to central Nevada, in the Rocky Mountains into northern New Mexico; eastward to the Black Hills. See figure 25 on page 95.
Characters for ready recognition.—Differs from M. e. invicta by hind foot less than 36 and basilar length less than 35 in males and by hind foot less than 29.5 and basilar length less than 30.5 in females; from M. e. gulosa, in both sexes, by upper parts lighter, tone 2 of Chocolate or lighter (see description of color), least width of light-colored underparts averaging about two-thirds instead of one-third of greatest width of dark-colored upper parts, in males, on the average, tail less than 65, weight of skull less than 0.90 grams, basilar length less than 30.8 grams; from M. e. streatori, in both sexes, by upper lips white (not brown), light color of underparts extending down hind leg below knee, depth of skull at posterior border of upper molars less than 7.7 in females and ordinarily less than 9.6 in males, tail less than 62 in males and less than 49 in females.
Description.—Size.—Male: An adult from Black Butte, California, measures: Total length, 227; length of tail, 55; length of hind foot, 27. Corresponding measurements of another from Wheeler Peak, Nevada, are: 220, 56, 26. Two subadults from Colorado, one from Crested Butte and another from Coventry, measure, respectively, as follows: 238, 227; 66, 60; 30, 30. An adult from Wheeler Peak, Nevada, weighs 57.7 grams and another from 2 mi. W Black Butte, Calif., 54.5 grams.
Female: Two adults from Teton County, Wyoming, measure: Total length, 205, 200; length of tail 52,—; length of hind foot, 23, 23.7. A subadult from 9-1/2 mi. E Pocatello, Idaho, measures: 197, 50, 25. An adult from Wheeler Peak, Nevada, has corresponding measurements of 190, 42, 23, and weighs 33.8 grams.
Color.—As described in Mustela erminea richardsonii except that upper parts tone 2 or lighter of Chocolate of plate 343 of Oberthür and Dauthenay; underparts white, Pale Buff or with faint wash of Sulphur Yellow; least width of color of underparts in male from Black Butte and one from Wheeler Peak, amounting to 65 and 59 per cent of greatest width of color of upper parts. Black tip of tail, respectively, 28 and 33 mm., which amounts to 51 and 59 per cent of length of tail-vertebrae. In two adult females, one from Teton County, Wyoming, and one from Wheeler Peak, Nevada, the least width of the underparts amounts to 55 and 60 per cent of the greatest width of color of upper parts. Black tip of tail, respectively, 23 and 19 mm., which amounts to 44 and 45 per cent of length of tail-vertebrae.
From the other subspecies of small-sized weasels of more northwestern occurrence, namely anguinae, fallenda, olympica, streatori and gulosa, muricus differs in lighter color of upper parts, wider light-colored underparts and relatively longer black tip of tail.
Skull.—Male (illustrated by 5 adults in table of measurements, which see): See plate 7. As described in Mustela erminea richardsonii except that: Weight, 0.78 (Wheeler Peak) and 0.85 (Black Butte) grams; basilar length, 30.6 (29.8-31.2); length of tooth-rows more or less than (approximately equal to) length of tympanic bulla.
Female (illustrated by 6 adults in table of measurements, which see): See plates 12-14. As described in Mustela erminea richardsonii except that: Weight, 0.60 (0.575-0.645); basilar length, 28.0 (27.3-29.4); breadth of rostrum approximately 30 per cent of basilar length.
In comparison with streatori, males average smaller in every measurement taken with no overlap in most dimensions; 25 per cent lighter; anterior margin of tympanic bulla more nearly flush with squamosal, that is to say less protruded from braincase; in relation to other dimensions of skull, braincase shallower anteriorly (at plane of last molars) and deeper posteriorly (at anterior end of basioccipital). Females average smaller in every measurement taken except mastoid and zygomatic breadths which are actually more; 6 per cent lighter; in relation to other parts of skull, preorbital and interorbital parts slightly smaller; in relation to length of skull, braincase shallower. Comparison with invicta and gulosa is made in the accounts of those subspecies.
Remarks.—The smallest males of the entire species are of this subspecies and the females of it are barely larger than those of olympica and gulosa and hence are among the three smallest. The material now available consists only of one or a few specimens from each of several widely separated localities. If as many specimens per unit area were available as there are of the species M. erminea from southern British Columbia, geographic variation warranting the division of muricus into more than one subspecies might be revealed. Evidence pointing in this direction is comprised in the pale color and small size of the pair of adults from Wheeler Peak on the eastern border of Nevada; the suggestion is that there is a distinct pale race of small individuals in the isolated spots of boreal life-zone in the mountains of the desert. The color and size of the specimens from the Toyabe Mountains, and that from the Pine Forest Mountains, both places also in Nevada, nevertheless, lend no support to this suggestion. Comparison of specimens from the Rocky Mountains of Colorado with those from the Sierra Nevada of California gives no basis for recognizing more than one subspecies. Therefore, Putorius streatori leptus Merriam with type locality at Silverton, San Juan County, Colorado, falls as a synonym of the earlier named Putorius (Arctogale) muricus Bangs with type locality at Echo, El Dorado County, California. Furthermore, specimens from northern New Mexico, the southernmost known area of occurrence for the subspecies (and for the species), are as large as specimens from far north in the range of the subspecies, say, in northwestern Wyoming; there is therefore no evidence of progressive decrease in size to the southward as in advance of study I supposed existed in muricus. This erroneous supposition was held because I knew that there was a decrease in size to the southward in the species as a whole and also in each of the subspecies richardsonii and invicta directly to the north of muricus.
Intergradation with invicta is shown by specimens from southwestern Montana. Where the margins of the geographic ranges of invicta and muricus approach one another elsewhere, low-lying territory, zonally unsuited to the existence of the species, occurs along the Snake and Columbia rivers, and precludes any chance of intergradation except around the head of the Snake River Plains. Two specimens, here referred to muricus, from Siskiyou County, California, in both color and cranial characters, are intergrades with streatori and might be referred with almost equal propriety to streatori.
Specimens examined.—Total number, 52, arranged alphabetically by states, then by counties from north to south within each state. Unless otherwise indicated, specimens are in the Museum of Vertebrate Zoölogy, University of California at Berkeley.
California. Siskiyou County: head of Rush Creek, 6400 ft., 1; Castle Lake, 5434 ft., 1. Tehama County: 2 mi. W Black Butte, 6800 ft., 1. Placer County: ridge W of Tahoe Pines, Lake Tahoe, 1; Blackwood Creek, 6250 ft., near Tahoe Pines, 1. El Dorado County: Fallen Leaf Lake, 6500 ft., 1[33]; Echo, 1[75]. Tuolumne County: Ten Lakes, 9200 ft., Yosemite Park, 1. Mariposa County: Vogelsang Lake, 10350 ft., Yosemite Park, 1. Mono County: Mammoth, 1[59].
Colorado. Rio Blanco County: Marvine, 1. Boulder County: Camp Albion, 10600 ft., 1[60]; Boulder, 1[91]. Denver County: Denver, 1[57]. Park County: Jefferson, 1[57]. Gunnison County: near Placita in Gunnison County, 1[26]; Crested Butte, 9000 ft., 3 (1[91], 2[19]). El Paso County: Turkey Creek, SW Colorado Springs, 6000 ft., 1[19]. Chaffee County: Arbourville, 1[91]; Hancock, 1. Montrose County: Coventry, 6800 ft., 1[19]. San Juan County: Silverton, 1[91]; in San Juan County above timberline, 1[87].
Idaho. Bannock County: West Fork of Rapid Creek, 9-1/2 mi. E Pocatello, 1.
Montana. Meagher County: Camas Creek, Big Belt Mts., 4 mi. S Ft. Logan, 1[91]. Beaverhead County: Donovan, 1[91]. County in question: Yellowstone Park, 1[75].
Nevada. Humboldt County: Alder Creek, 6000 ft., Pine Forest Mts., 1. Ormsby County: 1/2 mi. S Marlette Lake, 8150 ft., 1. Nye County: South Twin River, Toyabe Mts., 1[91]. White Pine County: Baker Creek (8500 ft., 8675 ft., 11100 ft.), 3.
New Mexico. Taos County: Twining, 10700 ft., 1[91]. Sandoval County: 9 mi. E Cuba, 9000 ft., 1. Santa Fe County: Saddle S of Santa Fe Baldy, 11000 ft., Santa Fe Range, 1[1].
Oregon. Wasco County: Mill Creek, 20 mi. W Warmsprings, 1[91]. Klamath County: Fort Klamath, 1[91].
South Dakota. Pennington County: 4 mi. SE Hill City, 5300 ft., 2[76]; Pfander's Ranch, 3 mi. SSE Hill City, 5300 ft., 1[76]; Palmer Gulch, 3 mi. SE Hill City, 5300 ft., 1[76]; Spring Creek, 2 mi. W Oreville, 5500 ft., 1[76]. Custer County: 1/2 mi. E Sylvan Lake, 6250 ft., 1[76].
Washington. Columbia County: Butte Creek, 1; Stayawhile Spring, 5150 ft., 1.
Wyoming. Crook County: 5 mi. NW Sundance, 5900 ft., 1[93]. Teton County: Whetstone Creek, 2[76]; 1/4 mi. E Moran, 6700 ft., 1[93]. Sublette County: 1/2 mi. NE Pinedale, 7500 ft., 1[93]. Albany County: 30 mi. N and 10 mi. E Laramie, 6560 ft., 1[93]; 26 mi. N and 4-1/2 mi. E Laramie, 6960 ft., 1[93]. Carbon County: 8 mi. N and 19-1/2 mi. E Savery, 8800 ft., 2[93].
Mustela erminea? angustidens (Brown)
Putorius cicognanii angustidens Brown, Mem. Amer. Mus. Nat. Hist., 9 (pt 4):181, pl. 17, 1908.
Mustela cicognanii angustidens, Hay, Iowa Geol. Surv. Bull., 23:32, 1914; Hay, Carnegie Inst. Washington, Pub. no. 322A:252, October 15, 1924; Hay, ibid., Pub. no. 390 (vol. 2): 528, 1930; Hall, ibid., Pub. no. 473:111, 112, November 20, 1936.
Type.—Female, adult, skull and lower jaws lacking zygomata, right P2 and incisors, no. 12432, Amer. Mus. Nat. Hist.; from Conard Fissure, four miles west of Willcockson, Newton County, Arkansas; obtained sometime in the period 1903 to 1905 inclusive (see plates 8, 14).
Range.—Known only from the Pleistocene deposit in Conard Fissure, at the type locality in northern Arkansas.
Description.—Skull.—Male (based on nos. 12437, 12441 and 12444): See measurements and plates 7 and 8; weight, unknown; basilar length, 38.1 (36.6-39.2); length of tooth-rows more than length of tympanic bulla; breadth of rostrum measured across lacrimal processes less than a third of basilar length; interorbital breadth ordinarily equal to distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth probably averaging approximately the same as distance between last upper molar and jugular foramen.
Female (based on nos. 11766 and 12435): See measurements and plates 8, 12-14; weight, unknown; basilar length, 34.0 (32.5-35.1); length of tooth-rows more than length of tympanic bulla; breadth of rostrum about equal to (more or less than) 30 per cent of basilar length; interorbital breadth less than distance between glenoid fossa and posterior border of external auditory meatus; zygomatic breadth probably less than distance between last upper molar and jugular foramen.
Comparison of the cranial description given above with those of the American races of erminea from the far north will show that many characters are held in common—more than with more southern subspecies of erminea.
Remarks.—The ten specimens studied by the writer fall into two groups of six larger individuals and four smaller. Upon comparing these with each sex of the three species of American Recent weasels, frenata, erminea and rixosa, it is seen that size, and to some degree shape, rule out of consideration both sexes of rixosa and also males of frenata. Thus we are left with females of frenata and males and females of erminea. So far as size is concerned, it can be assumed that the larger specimens are females of frenata and that the smaller are males of erminea. This assumption has in its favor also, the fact that the postglenoidal length of the skull accords with that in Recent specimens. The difference in this regard in Recent animals is that the postglenoidal length of the skull, expressed as a percentage of the total (condylobasal) length of the skull, amounts to:
| in frenata | in erminea |
| ♂ ordinarily less than 46 | ♂ ordinarily more than 46 |
| ♀ less than 47 | ♀ more than 48 |
In the fossils the percentage for the larger skulls is 46; for the smaller skulls it is 48.
It may be that the ten fossil skulls are six female frenata and four male erminea but I think not. In the first place a skull of different shape, seemingly of the frenata stock, is known from the deposit and it is almost certain that two subspecies of the same species would not occur at the same place at the same time. It is possible, of course, that parts of the deposits were laid down at times so far apart that a shift in geographic range of two subspecies had occurred. This one skull, seemingly of the frenata stock, is the type of Putorius gracilis Brown (see p. 404) and was regarded as the only known specimen of gracilis. Regardless of the specific identity of this one specimen named gracilis, the chances of obtaining otherwise from a deposit, like that in Conard Fissure, six females of frenata and four males of erminea without a male frenata or a female of erminea coming to light are so slight as strongly to incline me to the view that the six larger specimens are males of the same species to which the 4 smaller specimens belong. By either this interpretation, or the one initially considered (of female frenata and male erminea), the animals from the fissure are at least subspecifically distinct from any American Recent weasel. Furthermore, by this latter interpretation each sex of this weasel, angustidens, is intermediate between the frenata and erminea stocks in the feature of postglenoidal length which feature, at any place where the two Recent species occur together, serves to distinguish one from the other. In the northernmost subspecies of erminea (arctica for example) the postglenoidal length in some males is no longer than in males of frenata. Considering general size, angustidens agrees better with erminea than with frenata and this circumstance has influenced me to place angustidens as a subspecies of erminea.
Today, erminea is not known to occur nearer Conard Fissure than northern Iowa, more than 400 miles to the northward. In comparison with the race there, bangsi, males of angustidens are of approximately the same size but in the shorter distance between the glenoid fossa and anterior margin of the tympanic bulla, and also in the lesser postglenoidal length of the skull, angustidens resembles the northernmost American subspecies of erminea. Females of angustidens differ more from any living weasel than the males do. The females are much larger than those of bangsi, and among living American races of erminea most closely resemble intergrades between arctica and richardsonii which intergrades are found approximately 1700 miles to the north of Conard Fissure. In females, the preorbital part of the skull in M. e. arctica is broader and in M. e. richardsonii narrower than in angustidens. If it seems strange that females of angustidens resemble one subspecies whereas males, in size, resemble another subspecies almost a thousand miles distant, it should be remembered that the degree of sexual dimorphism varies much from one subspecies to another in the Recent animals. An example is furnished by Mustela erminea fallenda and Mustela erminea invicta.
The assemblage of mammals from Conard Fissure includes several species of boreal predilections which, like Mustela erminea, now occur only much farther north than Arkansas. At one time the edge of the sheet of ice was only about 200 miles north of Arkansas. It may be significant that the cranial characters of the female ermine from the Fissure, and qualitative cranial characters of males from there, are most nearly approximated among Recent weasels by those which live along the southern edge of the frozen tundra.
In view of what has been said, the possibility should be considered that the distinctive cranial features of angustidens may be the result of evolutionary change in time as well as of geographic variation resulting from horizontal placement.
MUSTELA RIXOSA (Bangs)
Least Weasel
(Synonymy under subspecies)
Type.—Putorius rixosus Bangs, Proc. Biol. Soc. Washington, 10:21, February 25, 1896.
Range.—From Norway and Switzerland eastward through Siberia and all the way across North America, but unknown from Iceland, Greenland and the Arctic islands west of Greenland; in North America, from the Arctic Life-zone south to Central British Columbia, Montana and into parts of the Upper Austral Life-zone as in the eastern half of the continent.
The southern extension of range in the Appalachians (to North Carolina) is not duplicated in the Rocky Mountains of western North America probably because the region there suitable for rixosa south of Central British Columbia and Montana is occupied by the almost equally small Mustela erminea muricus and related subspecies which seem to fill the ecological role that rixosa plays where it occurs. The small size of females of M. erminea cicognanii in New England may similarly account for the absence of rixosa there.
Characters for ready recognition.—Differs from both Mustela erminea and Mustela frenata by tail a fourth or less of length of head and body and without a black tip (at most a few black hairs at extreme tip in rixosa), and from M. frenata and from M. erminea in regions where it and rixosa occur together, by basilar length of skull less than 32.5 in males and less than 31.0 in females.
Characters of the species.—Size small: Total length less than 250 in males and 225 in females; tail a fourth or less of length of head and body, and without a black pencil and at most with a few black hairs at extreme tip; caudal vertebrae 11 to 16, normally 15 in M. r. rixosa, and 11 in one M. r. eskimo examined; skull with long braincase and short precranial portion, thus essentially same shape as in M. erminea but the largest males of M. rixosa always with a lesser basilar length that even the smallest females of M. erminea or M. frenata of the same geographic area. In fact no specimens of M. frenata have skulls so small as the largest M. rixosa, and skulls of equal size of M. erminea and M. rixosa, for example, M. erminea muricus of Colorado and M. rixosa eskimo of Alaska, differ in that when the skulls are viewed from directly above those of rixosa have the mastoid processes more prominent, or the braincase is higher in relation to its width or both differences together prevail. Stated in another way, comparison of skulls of equal size of rixosa and erminea shows that in the latter the braincase is more nearly flat and is wider above and in front of the mastoid processes; therefore, the greatest breadth of the braincase equals or exceeds the mastoid breadth, whereas the reverse is ordinarily true of rixosa.
Geographic variation.—In the Old World four subspecies are currently recognized (see Allen, 1933:316) and the same number is here recognized in North America. Length of the tail, length of head and body and hind foot, breadth of the rostral part of the skull in relation to its length, and position on the side of the head of the line of demarcation between the dark color of the upper parts and the white underparts, are the features in which geographic variation has been detected. The general impression is that the amount of geographic variation is much less than in Mustela frenata and only slightly less than in Mustela erminea of the same geographic area.
Nomenclature.—It is exceptional for a species which occurs in both the Old- and New-World to take its specific name from New World material, especially if the name was proposed as recently as 1896; most circumboreal species take their names from descriptions of European specimens. Although the least weasel, Mustela rixosa (Bangs) 1896, seems now to be an exception, it may yet turn out that the first available name was based on European material. Zimmermann (1943) shows that the least weasel actually was named on the basis of European material long before 1896 and concludes that the name Putorius minutus Pomel, 1853, based on a specimen from France, is the first available name.
Because Putorius nowadays is relegated to subgeneric rank under the generic name Mustela, we have for consideration the name-combination Mustela minuta (Pomel). Unfortunately for Zimmermann's conclusion, Mustela minuta Pomel is not available because it is preoccupied by Mustela minuta Gervais [= Palaeogale minuta (Gervais), 1848-1852—see Simpson, 1946: 2, 12], a name applied to another species of small mustelid from the Oligocene or lower Miocene deposits of Europe.
Some other early names thought by Zimmermann (1943:290) to have been based on the dwarf weasel of Europe are judged to be nomina nuda and therefore are to be ignored.
The name Mustela minor Nilsson 1820 was thought by Miller (1912:402) to be a renaming, and hence a synonym, of Mustela nivalis Linnaeus. If that is the case the name does not apply to the dwarf weasel. If the name Mustela minor Nilsson was instead based on the dwarf weasel, the name might still be unavailable, depending on rulings on secondary homonyms, because the name might be preoccupied by [Lutra] minor Erxleben 1777 which is a synonym of [Mustela] lutreola Linnaeus 1766. Two names seemingly available for weasels, and in use for them today, which might replace rixosa as the name of the species, are, first, Mustela boccamela Bechstein, 1801, of Sardinia [= Mustela nivalis boccamela of Miller, 1912, 405] and second, Putorius numidicus Pucheran, 1855, of Morocco and Algeria [= Mustela numidica of Allen, G. M., 1939, 183]. As they stand in the current literature, Mustela numidica is a species distinct from the dwarf weasel and the other name, Mustela nivalis boccamela, is an insular subspecies of the mouse weasel. Zimmermann (1943:292), however, implies that M. numidica may belong to the dwarf weasel group when he says "Ob auch iberica Barr.-Ham. als Unterart zu minuta Pom. zu stellen ist, soll hier nicht untersucht werden, ebensowenig die von Cabrera vermutete Zugehörigkeit der grossen nordafrikanischen M. numidica Puch. zur 'iberica-Gruppe'." The answer to this problem requires a taxonomic, rather than a nomenclatural, decision. Whether either M. numidica or M. boccamela are conspecific with the dwarf weasel I cannot at this time ascertain for want of adequate specimens. Because these two names, M. boccamela, and M. numidica, are assigned to kinds of weasels which are currently regarded as specifically distinct from the dwarf weasel, and because all the other names which certainly have been assigned to Old World populations of the dwarf weasel before 1896, so far as I know, are nomina nuda or are preoccupied, the next available name, Mustela rixosa (Bangs, 1896), is here employed.
Remarks.—This species may have a wider geographic range in northeastern North America than is now known. Strong (1930:7) writes that the Naskapi Indians of the interior country of Labrador between Hamilton Inlet and Ungava Bay "have only one name for weasel, mé-tah-kwut, but they say there are three kinds in their territory, a large, an intermediate, and a very small weasel. The latter suggests the least weasel . . . which has not been recorded from northern Labrador."
In the northern part of the range of the species, the winter pelage is white and the summer pelage is brown. In the southern part of the range, that is in the range of the subspecies allegheniensis, the winter pelage is either brown or white and the time of the molt into winter pelage is irregular; each of eleven individuals from Pennsylvania, Michigan and Ohio, taken in December, January, February and March is mostly white but retains some considerable part of the brown pelage of the previous coat on top of the head and usually also along the midline of the entire dorsum. These eleven animals include individuals of each sex. Of each sex, some are adults and some are subadults. Therefore, the delayed or incomplete fall molt, at present, cannot be correlated with either sex or with any particular age. No wild-taken specimens of M. erminea or of M. frenata of the same region show this delayed or incomplete molt.
Possibly this delay or incompleteness of molt is the result of the same cause that lies behind the birth of some M. rixosa in midwinter. As listed below, several litters of young have been found in midwinter. In fact it appears that in the United States, young may be born in every month of the year although, according to existing information, more litters are produced in spring and in winter than in summer and autumn. Many juveniles and young of allegheniensis examined in study collections clearly were born in spring but about as many seem to have been born in midwinter as at any other time (in the light of present knowledge) and this is in contrast to what we know of the two other species of American weasels since their young, so far as known, are born in spring.
One instance is worthy of detailed comment. An adult female, no. 783 Ohio State Museum, taken on January 31, 1931, at Vinton, Meigs County, Ohio, bears the following notation on the attached label "nest plowed out of ground. Very small young escaped—marked like parent. ♀ was nursing." The enlarged mammae on the dried skin substantiate the statement that the female was nursing young. She has a brown mask continuous from one ear through the eye, across the forehead and through the other eye to the opposite ear. On each side of the body a stripe of brown 5 to 10 mm. wide extends from the upper part of the foreleg back to the thigh and base of the tail, uniting there with its opposite and covering the tail. There are a few spots of brown on the shoulders, and rump and one on the middle of the back. Otherwise the specimen is white. One implication of the statement on the label that the young which escaped were marked like the parent (presumably this female parent) is that this female is a partial albino. I am more inclined, however, to the view that there was an unseasonable activity of the particular glands of internal secretion the hormones of which promote embryonic growth and that these glands, or others controlled by them, were in some way responsible for an abnormal progress of molt, or for a reversal of molt in that one molt began before the previous molt had been completed.
Excepting this one specimen, no. 783 from Vinton, Ohio, all of those in transitional pelage indicate that the direction of the molt pattern is the same as in M. frenata and M. erminea. That is to say, the autumnal molt begins on the midventral line and the molt in spring begins on the mid-dorsal line. Furthermore, the normal progress of each molt appears to follow the same pattern that has been described above for Mustela frenata.
A possible explanation of unseasonal molt in the southeastern area of occurrence of the species Mustela rixosa, and a possible explanation of the abnormal molt of the female from Vinton, Ohio, is that the species has only relatively recently invaded the area, and has had insufficient time to adjust the physiology of its molting mechanism to the longer periods of daylight that obtain later in autumn and earlier in spring than farther north. In the other two species of American weasels, the change in length of periods of light, it will be recalled, is known to indirectly control both molt and some changes in the sexual cycle. Wright (1942B:109) has shown that molt in spring precedes by one or two months the birth of young in M. frenata, that the two phenomena are correlated in a way that is statistically significant, and recognizes that progressively longer periods of daylight may be the causal stimulus. The suggestion made above that M. rixosa does not live in New England or in the Rocky Mountains of the western United States because each of the two areas already is inhabited by weasels of almost equally small size, is in line with the idea that rixosa is a recent immigrant to America, or more precisely that rixosa arrived later than erminea.
Natural History.—Habitat and Numbers.—Soper (1946:136) recounts that near the junction of the Antler and Souris rivers, Manitoba, this species occurs "both in the river valleys and on the upper prairies," and later (1948:55), with reference to the Grand Prairie of the Peace River region of Alberta, writes that the least weasel "inhabits both parklands and mixed wood forest environments."
At most times, wherever found, the least weasel is regarded as rare. Not only mammalogists regard it as rare and as a desirable catch, but Indians likewise value it, probably because of its rarity. For example, Osgood (1901:69-70), who caught a female least weasel at Tyonek, Alaska, writes that: "The natives regard the capture of one of these rare animals as a piece of great good fortune. One old Indian who frequently visited our cabin told us that his brother who had caught one when a small boy had in consequence become a 'big chief'; and he assured me that since I had caught one I must surely be destined to become a man of great wealth and power."
Swenk's (1926:313-330) account of the species in Clay County, Nebraska, shows, however, that the animal was far more abundant in 1916 and 1917 than subsequently and inferentially than it was before 1916. Clearest proof of multiannual fluctuation is provided by P. O. Fryklund's (Swanson and Fryklund, 1935:120-126) receipt of weasels from Roseau County, Minnesota. From 1895 to 1932 he had approximately equal opportunity to receive least weasels each year. Those which came to his attention were distributed by years as follows: 1895-1927, 7 individuals in all; winter of 1927-28, 3 individuals; winter of 1928-29, 59 individuals; 1929-1930, 84 individuals; 1930-1935, 3 individuals. "These records indicate a very definite increase in the abundance of least weasels in the Roseau region [in] the two years from the autumn of 1928 to the spring of 1930. Mr. Fryklund has handled 166 least weasels in his 40 years in Roseau County, and of these, 143 were taken in the two years mentioned."
The maximum home range of the least weasel is two acres and a weasel seldom travels farther than ten rods from its burrow according to Polderboer (1942:146) who, in the period December 20, 1939, to January 2, 1940, studied four least weasels and one long-tailed weasel on a 144 acre farm in Butler County, Iowa.
Behavior
Of the voice, Llewellyn (1942:441) records that his captive specimen taken in Virginia uttered a shrill shriek when seizing prey or when teased. When excessively annoyed the weasel also emitted musk.
The sense of smell is used in hunting as was witnessed by George L. Fordyce; he observed a least weasel following the scent of a Peromyscus and saw the least weasel overtake and kill the mouse (Seton, 1929 (2):637).
At a nest in a clover stack, in Manitoba, Criddle (1947:69), on December 27, 1946, found the least weasel "to have been rather remiss in its sanitary habits as its pile of dung was almost, or quite, touching the nest and only just to the side of its entrance." There were 117 voids.
Enemies
The great-horned owl, barn owl and long-tailed weasel are to be counted as enemies since Nelson (1934:252) found the fur, skull and other fragments of the skeleton of a least weasel in one of 26 pellets of the great-horned owl in Wisconsin; Handley (1949:431) found the skull and other skeletal remains of a least weasel in one of 22 pellets of the barn owl in Virginia; and Polderboer, Kuhn and Hendrickson (1941), in Iowa, found the remains of a least weasel in the den and scats of a Mustela frenata. A domestic cat in Michigan killed a least weasel (Dearborn, 1932B:277).
Food
Mice are killed by the least weasel biting into the back of the head and neck according to Allen (1940:460) who reported upon the growth of five young, from Michigan, that he had in captivity. He further states that a weasel was able to kill a mouse in 30 seconds. One large Microtus introduced into the cage slept with a weasel for several days and ate parts of the mice that the weasel killed but then the weasel killed this mouse! Llewellyn (1942:440-441), in writing of a captive from Virginia, says: "When a live mouse was placed in the cage, the weasel sprang upon it almost instantly. Grasping the mouse by the back of the head, the weasel bit its victim through the skull several times in rapid succession and held on with its sharp teeth. The sound of the teeth piercing the bone was distinctly audible at a distance of several feet. During this interval the weasel hugged the mouse closely with its fore legs and pressed it firmly to its belly through a kicking motion of the hind legs. The hold on the back of the head was not relinquished until the mouse was dead. The killing took only a few seconds. Upon releasing the mouse the weasel usually came to the front of the cage and inspected the observer for an interval of several seconds after which it returned to its prey and began its meal at once. Sometimes the blood would be licked from the wound in the back of the head or perhaps an ear would be chewed a bit and the blood licked off, but never did the weasel 'cut the throat' of its prey and 'suck the blood.'
"The weasel ate the head and brain first, beginning at the back of the head and working forward. Just before reaching the nose the process was reversed and eating then proceeded from the base of the skull toward the tail of the mouse. The tip of the nose, maxilla with teeth, and the tail seemed to be the parts least preferred; they were not eaten when an abundance of food was present. At no time did the weasel place its front feet on the mouse in an attempt to hold it. A second or third mouse was killed immediately upon being placed in the cage even though the first one had not been consumed. The weasel, however, usually returned to the partially eaten mouse and finished it before starting on a new one. Upon completing a meal, especially if the meal had been particularly bloody, the weasel rubbed its chin on the bottom of the cage, scooting along and appearing more snakelike than normal. Whenever I attempted to remove a mouse, or partially eaten one, from the cage, the weasel hung to the mouse tenaciously, and often allowed itself to be lifted up in this manner.
"In the six days that the weasel was kept in captivity it was fed 10 house mice having a total weight of 118 grams. As no food was given on one day, the amount of food eaten is probably slightly below the actual capacity of the animal. Since the weasel weighed only about 32 grams, the average amount of food eaten a day was slightly in excess of one-half the weight of the animal."
Polderboer (1942:146-147) found in three dens, in Iowa, bits of Reithrodontomys (harvest mice) and Peromyscus maniculatus (deer mouse), and in the digestive tract of one least weasel there was a bone fragment and a few hairs of a deer mouse. In the account, given beyond, of a nest, Criddle (1947:69) records the Pennsylvania meadow mouse (Microtus pennsylvanicus drummondi) and the Gapper red-backed vole (Clethrionomys gapperi) as prey at Treesbank, Manitoba. The same author, concerning the same place, earlier (1926:199-200) wrote that in 1922 the meadow mouse, Microtus minor, "went into winter quarters in great numbers and its homes were well stocked with provisions . . . all went well until the middle of February, 1923. Then, within a few days, each was taken possession of by a least weasel (Mustela rixosa) and the inhabitants were quickly destroyed. One dwelling was occupied by one of these weasels for about two weeks during which time I observed that it had dragged several mice over the snow to its temporary home. This residence was examined in April, and in it were discovered six dead Microtus minor, one Evotomys, the head of another, and at least six or eight remnants of small rodents including Microtus drummondi, these last remains being chiefly indicated by the hair-lined nest of the weasel.
"The homes of 27 other vole communities examined at this time were all found to have been entered by weasels, the inhabitants having been killed and partly eaten. Moreover, the weasels had made the homes temporary centers from which they raided other rodent habitations in the vicinity. Thus from being an abundant animal this vole was reduced to insignificance in the course of a few weeks, while all other kinds of mice had suffered severely from the same enemy."
An instance of predation on Peromyscus, revealing some of the methods of capturing prey, is recounted by Seton (1929 (2):636-637) who quotes a letter to him from George L. Fordyce, of Youngstown, Ohio, as follows: "While out in the field this morning (Dec. 26), walking along the bank of a ravine at the edge of our golf course, I saw a Field-mouse run out of the bushes into the rough grass that is just outside of the fair-green of the course. In another instant, what I thought at first to be a white Mouse came out at the same place. The Mouse ran into a wheel track, and disappeared under the grass, coming out about 6 feet from where it went in. The white animal followed through the same course, and when it came out, I saw that it was a small Weasel, very little larger than the Mouse, and that it was following the trail of the Mouse by scent.
"For a time the Mouse ran in circles, and zigzagged about, often . . . within 4 or 5 feet of the Weasel; but the latter seemed so intent on the trail, that it did not notice the Mouse to one side. After a time the latter started toward the open golf course; and when the Weasel reached the point where the trail was straight, it sighted the prey, made a sudden dash forward, and, although 25 feet behind, overtook the Mouse while it was going 3 or 4 feet.
"For a few seconds, they seemed to fight, until the Weasel got the Mouse by the throat, and started for the bushes, dragging the body. When it came to within about three feet of me, I moved a little to see what it would do. It dropped its victim, and ran into the ravine. The Mouse had a drop of bright red blood in the center of its white throat. I waited near by for 15 or 20 minutes, thinking the Weasel might come back, but it did not show up again; even an hour later, the Mouse had not been disturbed."
There are two suggestions, but no proof that I know of, in the literature that the least weasel eats insects. Abbott (1884:27-32—1st ed., 1884) gives considerable information on the food (some insects included) of the "little weasel" which he describes (op. cit.; 27) as having "a little pointed tail of a uniform brown color." Although this suggests Mustela rixosa, Abbott mentions on the next page (page 28) that a specimen of the smaller weasel measured six and a half inches from the tip of the snout to the base of the tail and that the tail itself measured two and a fourth inches to the tip of the last caudal vertebra. These measurements indicate that Mustela erminea was involved. Because of the uncertainty as to the species of Mustela involved, Abbott's interesting data on food, nest and behavior are not recorded in the present work. Seton (1929 (2):636) says that of several least weasels brought to D. Nicholson at Morden, Manitoba, most of them decayed so quickly that they could not be saved as specimens. To Seton this indicated that insects were an important part of the food of the weasels.
In summary: Least weasels are known to eat harvest mice, deer mice, meadow mice and red-backed mice; it is suspected that they eat also insects.
Reproduction
Polderboer (1948:296) has taken six specimens in "northeastern Iowa [in] . . . January and December—all males in winter pelage. None of these males showed signs of sexual activity; in all, the testes were retracted and diminutive in size. . . . A male least weasel in brown pelage was taken November 17, 1945, at Marion, Iowa. This specimen had large testes that had descended into the scrotum. The testes, when removed, were about the size of medium-sized garden peas. Microscopic examination of the testes and the vasa deferentia showed mature sperms to be present. . . ."
On July 1, 1917, in Clay County, Nebraska, a nest with four young was found (Swenk, 1926:321). On July 29, 1939, an adult and five young were plowed out of the ground in Allegan County, Michigan; one of the two young males weighed 40.5 grams two days after capture (Allen, 1940:459-460). On August 12, 1932, ten young with the mother, were found in Roseau County, Minnesota (Swanson and Fryklund, 1935:125). September appears to have been the month of birth of a specimen, no. 8472 in the Carnegie Museum, taken on November 24 in Pittsburgh, Pennsylvania. In October, a young least weasel is recorded from Pennsylvania (Winecoff, 1930:313). Early December was the time of birth of a specimen, approximately 10 weeks old, no. 88077, University of Michigan, taken on February 21 in Allegan County. On December 25, 1927, in Washington County, Pennsylvania, "five full-sized, though young . . . animals were caught under the same pile of corn fodder" (Sutton, 1929:253). The first week of January seems to have been the time of birth of a juvenile, no. 88080, University of Michigan, taken in Livingston County, Michigan, on March 27, 1943, since the specimen is approximately seven weeks old. On January 15, 1929, in Washington County, Pennsylvania, four young with the eyes yet unopened were obtained from a nest (Sutton, 1929:254). On January 25, 1928, young, the eyes of which may not yet have been open, were taken from a den in Washington County, Pennsylvania, by Winecoff (1930:313), who records other young having been taken in the same month as well as in February. On March 10, a female from North Portal, Saskatchewan, gave birth to four young (Dunk, 1946:392). On April 18, 1916, four young, half grown, were taken in Nebraska (Swenk, 1926:317). On April 2, 1929, three young were found in Roseau County, Minnesota, according to Swanson and Fryklund (1935:125) who remark that: "The Pennsylvania and Minnesota records show that least weasels may be born any time from July to early February in the northern states." Now, with all of the above records available, it turns out that November, May and June are the only months in which young least weasels have not been reported. Of course some of the young, for which the ages were not specified, were born in preceding months. Even so, the data now available suggests that, in the United States, young least weasels may be born in every month of the year. The number per litter is 3, 4, 4, 4, 5, 5, and 10, yielding an average of 5.
The rate of growth of the young has not been studied enough to allow of judging if it differs significantly from that of other species of the genus. Allen (1940:459-460), however, tells us that of the three young females and two young males captured on July 29, 1939, in Allegan County, Michigan, one male that was killed on July 31, 1939, weighed 40.5 grams. The male remaining alive increased from 46 grams (August 5) to 62.5 grams on September 20, having eaten 63 mice while in captivity. The females in the period of August 5 to September 4 increased in weight as follows: 41 up to 49 grams; 44 to 50 grams; and 47 to 58 grams, having eaten, by September 26, 60, 64 and 65 mice.
Concerning a nest in which young were found, Sutton (1929:254) writes that on January 15, 1929, near Burgettstown, Washington County, Pennsylvania, an animal was seen to enter a small hole in a creek bank. After the observer dug in a distance of approximately six inches an adult, female least weasel was seen and obtained. Back of the animal, the hole, which turned sharply downward, was full of water. The weasel first seen was a female nursing young. A chamber, to the side of the hole, filled with dead grass, comprised a nest containing four young with the eyes yet unopened. Several nests occupied by adult least weasels or by least weasels that were old enough to shift for themselves have been found. Polderboer (1942:145-147) in the winter of 1939-40, on a 144 acre farm in Butler County, Iowa, found four least weasels living, singly, in burrows dug by moles and pocket gophers. The nests therein made by mice were used by the least weasels. Winecoff (1930:312-313) mentions one den in Pennsylvania that contained the remains of only mice, "and not a hint of a feather." Above, in the account of food of the least weasel, Criddle's (1926:199-200) account of the havoc wrought by least weasels among the meadow mice (Microtus ochrogaster minor) has been given. In this account he mentions the fur-lined nests of the weasels that had appropriated the homes of the Microtus. Criddle's (1947:69) later account of a nest at Treesbank, Manitoba, is as follows: "A Nest of the Least Weasel.—When a least weasel finds its way into a locality that has a large number of mice in it, it selects for its home one of their nests that has been made in a well concealed place. This it immediately starts to improve by lining it with hair plucked from its victims before eating them; and as long as sufficient numbers of mice remain in the district the weasel continues adding their hair to the nest, so that the thickness of its walls give one a good idea of the length of time it has been in use. The nest is not only used for sleeping in, as most of the food is consumed in it. Frozen mice are taken in to be thawed out and the weasel carries those it has recently killed in to prevent them getting frozen, or perhaps to have them warm for its next meal.
"On January 27, 1946, my son Percy called my attention to a nest that he had just uncovered in a clover stack that we were using. This nest had originally been made by a Drummond's vole, Microtus pennsylvanicus drummondii, and taken from it by the least weasel, Mustela rixosa, the tracks of which had been noticed about the stack yard since the first snow in early November.
"The nest had evidently been in use for at least three months and the continual additions made to its walls had been so great that they were nearly an inch thick of hair matted together so closely that it appeared to be felt. The hair alone weighed nearly 22 gm., so that with this for protection the weasel must have been warm and comfortable through the severest winter weather.
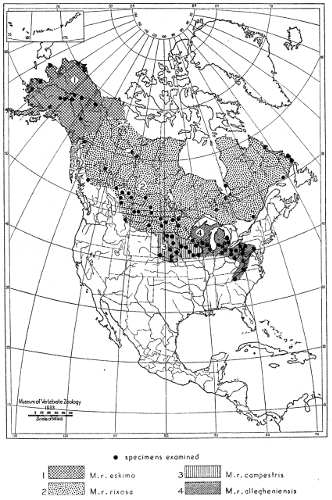
Fig. 28. Map showing occurrences and probable geographic ranges of the subspecies of Mustela rixosa in North America.
"In the nest were two red-backed mice, Clethrionomys gapperi, one of which had the base of its skull eaten out. No hair had been removed from either of them, but a Microtus lying in a side tunnel some feet away had the long hair plucked from its back and sides. In and close about the nest were found forty-three front parts of mice skulls which had evidently been discarded because of the sharp teeth in the maxillaries. Seven full stomachs and eleven hind feet of adult Microtus with parts of leg bones were disclosed in, or under, the weasel's bed and a few small bits of skin with hair attached were scattered among the plucked hair of the nest.
"This weasel seems to have been rather remiss in its sanitary habits as its pile of dung was almost, or quite, touching the nest and only just to the side of its entrance. It was composed of 117 voids all of which contained much hair and broken bone.
"Six other mouse nests found in the same stack, or others adjoining it, had been thinly lined with hair. One of these had two mice in it, a red-backed with its brain eaten out and a Microtus with some hair plucked from its neck. Another nest contained the front part of a skull with teeth and the hind feet and tail of a red-back. Besides the mice found in the nests seven others were discovered tucked away in side tunnels. One of these mice had most of the hair plucked from its back. Whether all these mice and nests belonged to the same weasel or not I am unable to say, but it is usual for them to have several nests in the area surrounding the one that is used as their headquarters or home."
Mustela rixosa eskimo (Stone)
Least Weasel
Putorius rixosus eskimo Stone, Proc. Acad. Nat. Sci. Philadelphia, 1900:44, March 24, 1900.
Putorius (Gale) vulgaris, Coues, Fur-bearing animals, p. 102, 1877 (part).
Putorius rixosus, Bangs, Proc. Biol. Soc. Washington, 10:21, February 25, 1896 (part); Merriam, N. Amer. Fauna, 11:14, June 30, 1896 (part).
Mustela rixosa eskimo, Miller, U. S. Nat. Mus. Bull., 79:96, December 31, 1912; Swenk, Journ. Mamm., 7:327, November 23, 1926; Hall, Univ. California Publ. Zoöl., 30:421, March 19, 1929.
Type.—Female, age in question, no. 848 in Acad. Nat. Sci. Philadelphia; Point Barrow, Alaska; July 25, 1898; obtained by E. A. McIlhenny. Type not seen by me.
Range.—Alaska and Yukon Territory. See figure 28 on page 180.
Characters for ready recognition.—Differs from M. r. pygmaea of eastern Asia in longer tail, averaging 11 rather than 16 per cent of length of head and body, and in study skins reaching only to heel instead of to point between heel and toes; from M. r. rixosa in shorter tail averaging 16 rather than 19 per cent of length of head and body and not extending beyond outstretched hind feet in study skins; white of underparts extending dorsally as a reëntrant angle from upper lip to behind eye, rather than delimited dorsally by a boundary between white and brown color that extends straight across cheeks from upper lip to side of body well below eye and ear; breadth of rostrum measured across lacrimal processes more, instead of less, than 85.5 per cent of orbitonasal length; from M. erminea of same region by basilar length of skull less than 32; tail less than 50 and lacking black pencil.
Description.—Size.—Male: The original describer lists measurements of topotypes as follows: Total length, 204, 230; length of tail, 28, 31; length of hind foot, 20, 22. Allowing 5 per cent for shrinkage, the hind feet of 5 topotypes yield an average measurement of 23 for the hind foot.
Female: Measurements of two topotypes are: Total length, 184, 180; length of tail, 25, 25; length of hind foot, 24, 18. In four other topotypes the hind feet, allowing 5 per cent for shrinkage, yield an average of 21.
Color.—Winter pelage all white, rarely with few white hairs in tip of tail but no black pencil; summer pelage with upper parts about Raw Umber and tone 3 of Chocolate pl. 343 of Oberthür and Dauthenay; underparts white, extending over upper lip, insides of limbs and over all four feet. Line of demarcation between underparts and upper parts extends from upper lip posterodorsally to behind eye down to base of ear, up behind ear for a third or more of its height, and back along side of body. Tail unicolor all around and same color as upper parts. Least width of color of underparts averaging 83 per cent of greatest width of color of upper parts.
Skull.—Based on topotypes; see measurements and plates 14 and 15; weight, 0.82 (0.74-0.93) grams in males, and 0.80 and 0.84 in two females; basilar length, 29.5 (27.6-30.1) in males and 27.8 (27.1-28.8) in females; otherwise as described in M. e. richardsonii.
Remarks.—Among the earliest specimens preserved was one by Edward W. Nelson in the course of his explorations of the Upper Yukon, and one in 1874 by L. T. Turner from St. Michaels, Alaska. Bangs, in 1896 (p. 22) mentioned the occurrence of the species in Alaska, but it was not until 1900 (p. 44) that Stone named the subspecies, and then principally on the basis of specimens obtained two years before by E. A. McIlhenny.
The large size, broad skull, light color and short tail are the distinguishing subspecific characters of the race eskimo, and the three characters first mentioned are distinguishing features also of the subspecies of Mustela erminea, namely arctica, which inhabits the same region. Possibly eskimo also will be found on Banks Island and the other Arctic islands between Alaska and Greenland, as is M. e. arctica; at the present time no specimens of Mustela rixosa are known from these islands although some race of rixosa would be expected to occur there.
Animals from southern Alaska average slightly smaller than those from northern Alaska, and this decrease in size toward the south probably represents intergradation with M. r. rixosa. Further evidence of intergradation is furnished by the short tail of the specimen from 15 miles east of Atlin; in other particulars this specimen agrees with the subspecies rixosa to which it is here referred. Nevertheless, the short tail, and color pattern, namely reëntrant angle of white behind the eye, is to be seen in all Alaskan specimens examined in the brown pelage, even in no. 107591, from Tyonek on Cook Inlet, which Osgood (1901:69) and Swenk (1926:323) thought might not differ from the subspecies M. r. rixosa.
Each of four male topotypes, hardly subadult in age, probably of a single litter, is much larger than any other specimen seen from Point Barrow. The basilar length, for example, is 31.9 as against 29.5, and the weight of the skull (with lower jaws) is as much as 1.5 grams, as against 0.93 in the heaviest of the other males. Initial examination of materials from Point Barrow raised the suspicion that two distinct species were represented—rixosa and a larger one possibly allied to M. nivalis of the Old World. Nevertheless, further study almost completely allayed the suspicion because the only difference discernible is one of size, and it is supposed that additional specimens will bridge the gap in size and show that M. r. eskimo at Point Barrow averages larger than the adult specimens now available indicate. The four large males of subadult age are nos. 42814-42816 and 42818 of the American Museum of Natural History.
Of the fourteen adult and subadult skulls examined, two display lesions resulting from infestation of the frontal sinuses by nematode parasites. None of the young skulls show such infestation.
Specimens examined.—Total number, 42 as follows. Arranged alphabetically by Territory and District and unless otherwise indicated in the United States National Museum.
Alaska. Barrow and Point Barrow, 19 (8[2], 7[74], 2[1], 1[50]); Wainwright, 1[57]; Mts. back of Icy Cape, 1[77]; west of Beechey Point, 1[2]; west edge of Colville River Delta, 1[2]; Koyukuk River, 16 mi. above Beetles, 1; upper Yukon, 1; Fort Yukon, 1; Stephens Village, 1; Wales, 1[57]; McDonald Creek, tributary of Salcha Slough, 1; near head of Toklat River, 1; head of Kantishna River, 1; St. Michael, 4 (2[74]); Tyonek [= Tyonek], 1; Bethel, 3; vic. Bristol Bay, 1.
Yukon. La Pierre's House, 1; Klotassin River, tributary of White River, 1.
Mustela rixosa rixosa (Bangs)
Least Weasel
Putorius rixosus Bangs, Proc. Biol. Soc. Washington, 10:21, pl. 1, fig. 6, pl. 2, fig. 6, pl. 3, fig. 4, February 25, 1896; Merriam, N. Amer. Fauna, 11:14, pl. 2, figs. 7, 7a, June 30, 1896.
Putorius pusillus, Baird, Mamm. N. Amer., p. 159, 1858.
Putorius (Gale) vulgaris, Coues, Fur-bearing animals, p. 102, 1877.
Mustela rixosa, Thomas, Proc. Zoöl. Soc. London, p. 168, March, 1911.
Mustela rixosa rixosa, Miller, U. S. Nat. Mus. Bull., 79:96, December 31, 1912; Swenk, Journ. Mamm., 7:327, November 23, 1926.
Type.—Female, adult, skin and skull; no. 642 Bangs Coll. in Mus. Comp. Zoöl.; Osler, Saskatchewan; July 15, 1893; obtained by W. C. Colt; original no. 79 according to describer.
The skull lacks the basioccipital, basisphenoid, and left zygomatic arch. The "crowns" of the lower canines are missing; otherwise the teeth are present and entire. The skin is fairly well made, with soles of hind feet up, in good condition and in summer pelage.
Range.—From northern British Columbia and Great Slave Lake south on the west side of the Rocky Mountains to Ootsa Lake, British Columbia, and on the east side of the Rocky Mountains, south to central Montana, North Dakota and Minnesota; eastward in Canada, entirely north of St. Lawrence River, to Atlantic Ocean. See figure 28 on page 180.
Characters for ready recognition.—Differs from M. r. eskimo in longer tail averaging 19 rather than 16 per cent of length of head and body and extending beyond outstretched hind feet in study skins, rather than to a point short of tips of toes; boundary between brown upper parts and white underparts extending straight across cheeks from upper lip to side of body well below eye and ear, rather than with reëntrant angle from upper lip carrying white upward to point behind eye, and with breadth of rostrum less, instead of more, than 85.5 per cent of orbitonasal length; from M. r. campestris by smaller size: hind foot less than 25 in males and ordinarily less than 22 in females; in males total length less than 216 and tail averaging less than 34, and in females total length averaging less than 182 and tail averaging less than 29; color said to average darker; from M. r. allegheniensis by three average differences, namely lighter color, longer tympanic bullae and larger size of males; from M. frenata and M. erminea of same region by basilar length of skull less than 32; tail less than 50, and lacking black pencil.
Description.—Size.—Male: Six adults and subadults from Shaunavon, Saskatchewan, yield average and extreme measurements as follows: Total length, 202 (188-208); length of tail, 32.5 (31.5-34.0); length of hind foot, 22.8 (21-24).
Female: One adult and 3 subadults from the same area yield average and extreme measurements as follows: Total length, 172 (162-190.5); length of tail, 27.4 (24-34); length of hind foot, 19.6 (17.5-22).
Color.—Winter pelage all white, rarely brown; as described in M. r. eskimo except that line of demarcation on side of head between upper parts and underparts passes almost straight back without the dorsally directed reëntrant area of white behind the eye and ear; least width of color of underparts averaging 52 per cent of greatest width of color of upper parts.
Skull (Based on those from Shaunavon, Sask.)—See measurements and plates 14 and 15; weight, 0.88 (0.70-0.98) grams in males and 0.55 (0.54-0.56) in females; basilar length, 29.5 (28.4-30.4) in males and 26.1 (24.7-27.0) in females; otherwise as described in M. e. richardsonii.
Remarks.—As early as 1858 (p. 159) Baird recognized an individual of this race from Pembina, Minnesota, as pertaining to a distinct species. Although he used for it the specific name pusillus originally proposed by DeKay for a small weasel from the state of New York, Baird wisely noted that the specimen he described "may be different from the New York species. . . ." After preparing this account, Baird included a second specimen, from Fort Steilacoom, Washington Territory, which he thought might be the same, but the differences that he was careful to point out, in the light of later knowledge, show it to be of the species Mustela erminea. Only a few other naturalists followed Baird in distinguishing the least weasel as a separate species until Bangs in 1896 (p. 21) clearly differentiated it and proposed for it the name Putorius rixosus, which continues in use today and applies to the species.
The accumulation at the National Museum of Canada, through the energy of Dr. R. M. Anderson, of a good series of specimens from Saskatchewan in the general vicinity of the type locality allows for the first time an adequate conception of the amount of secondary sexual variation and individual variation and permits recognition of subspecific characters to differentiate between M. r. rixosa and the subspecies eskimo and campestris. In comparison with the subspecies allegheniensis the basis for segregation is less clear and will remain somewhat in doubt until additional adults of allegheniensis from, say, Pennsylvania, become available with accurate external measurements taken in the flesh and especially with complete skulls.
Intergradation with the subspecies eskimo is suggested by the short tail of the specimen from fifteen miles east of Atlin, British Columbia; in other particulars that specimen, a skin-alone, agrees with the subspecies rixosa. Intergradation with campestris is indicated by increased size of some specimens from North Dakota, and is suggested with allegheniensis by the color of specimens from Wisconsin and Illinois. Three specimens from Winona County, in southeastern Minnesota, unfortunately are skulls-alone without external measurements. Also, two of these skulls are of young animals. The one adult, unsexed, is from Crystal Springs. Selected cranial measurements are: basilar length, 28.5; length of tympanic bulla, 10.9. These measurements accord with those of males of the subspecies rixosa to which the specimens from Winona County, therefore, are here assigned. The possibilities have not been excluded, however, that the adult is an unusually large female of the subspecies campestris or a male of allegheniensis that has tympanic bullae longer than average for that subspecies.
Some hesitation is felt in assigning the specimens, 8 in all, from eastern Canada to the subspecies rixosa. The skin-alone from Eagle River and the skin, with part of the skull, from St. Michael Bay, are in transitional pelage and are of no help in appraising subspecific characters. The one adult specimen which does have a complete skull is from an island south of the Comb Hills. This animal in all respects agrees with selected individuals of M. r. rixosa from Saskatchewan, but each of the five other skins in summer pelage has spots of dark brown color on the breast. Only about one specimen in three of rixosa from Saskatchewan is similarly marked. Furthermore, on some of the specimens from eastern Canada the spots are larger than on any of the animals from farther west. The greater frequency of brown spots on the breast, the larger average size of these spots, and the darker average coloration of the upper parts are suggestive of geographic variation, the existence of which has to be proved by additional and more complete specimens from eastern Canada. For the time being, specimens from there are tentatively assigned to the race rixosa.
Of 56 subadult and adult skulls only 3 (1 North Dakota; 1 Calgary, Alberta; and 1 Island S Comb Hills, Queb.) display lesions resulting from infestation of the frontal sinuses by nematode parasites. None of the young skulls shows such infestation.
Specimens examined.—Total number, 87 as follows. Arranged alphabetically by provinces and states and within each from north to south. Unless otherwise indicated, specimens are in the United States National Museum.
Alberta. Miette River, 1[77]; 5 mi. NW Camrose, 1[77]; Camrose, 2 (1[77], 1[31]); "near Camrose," 2[77]; Forks Blindman and Red Deer rivers, 1[60]; Innisfail, 1[86]; Veteran, 1[93]; Diddsbury [= Didsbury], 1; Calgary, 2 (1[93], 1[2]); Shepard, 1[86].
British Columbia. Clarks Ranch, Halfway River, Peace River Dist., 1[85]; 15 mi. E Atlin, 1[8]; Wistaria, P. O., 3 (2[77], 1[85]); Ootsa Lake, 1[85].
Labrador. Davis Inlet, 1[60]; 30 mi. upriver and 20 mi. toward Groswater Mts., Eagle River, 1; St. Michael Bay, 1.
Mackenzie. Old Fort Reliance, 1[2]; Fort Resolution, 2; Fort Smith, 1.
Manitoba. Gypsumville, 1[86]; Lake St. Martin Reserve, 1[86].
Minnesota. Roseau County: Cedarbend, 2[14]; Grimstad, 1[14]; America, 2 (1[14], 1[74]); Malung, 1[74]; Norland, 1[41]; Falun, 3 (1[14], 1[74], 1[41]); Palmville, 1[41]; Spruce, 1[74]; Stokes, 1[74]. No locality more definite than Marshall County, 1[14]. Clay County: Moorhead, 1[36]. Winona County: "near" Whitman, 1[34]; Altura, 1[98]; Crystal Springs, 1[98].
Montana. Sun River Valley, 1; Wibaux in Wibaux County, 1.
North Dakota. Walsh County: Grafton, 15 (3[60], 1[93], 5[2], 2[14], 1[74], 1[1], 1[76]). McHenry County: 4 and 4-1/2 mi. N Upham, 2. Wells County: 1[36]. Morton County: Mandan, 1[60].
Ontario. Algoma Dist.: Tatnall, near Oba, 1[86]. Moose Factory, 1[75].
Quebec. Island S of Comb Hills, James Bay, 1[9]. Saguenay County: Natashkwan, 1.
Saskatchewan. Osler, 1[75]; "near Regina," 1[77]; Dollard, 2[31]; Shaunavon (and "near" and 1 mi. NE), 9[77]; Klintowel P. O. (about 15 mi. N of Eastend), 1[77]; Eastend and "near" Eastend, 2[77].
Mustela rixosa allegheniensis (Rhoads)
Least Weasel
Putorius allegheniensis Rhoads, Proc. Acad. Nat. Sci. Philadelphia, 1900:751, March 25, 1901.
Putorius rixosus allegheniensis, Cory, Mamm. Illinois and Wisconsin, p. 378, 1912.
Mustela allegheniensis, Miller, U. S. Nat. Mus. Bull., 79:96, December 31, 1912.
Mustela rixosa allegheniensis, Swenk, Journ. Mamm., 7:328, November 23, 1926.
Type.—Probably male adult, skin and skull, no. 6195, Acad. Nat. Sci. Philadelphia; near Beallsville, Washington Co., Pa.; about 1885 or 1886; obtained by Robert Hawkins.
Type not seen by me.
Range.—Wisconsin, northern Illinois, northern Indiana, Michigan, Ohio, Pennsylvania east to Dauphin County and south in the mountains to northwestern North Carolina. See figure 28 on page 180.
Characters for ready recognition.—Distinguished from M. r. rixosa by three average differences, namely, darker color, shorter tympanic bullae, and smaller size of males; from M. r. campestris in smaller size: hind foot less than 25 in males and less than 22 in females; in males total length less than 216 and tail averaging less than 34, color averaging darker; from M. frenata and M. erminea of same region by basilar length less than 31, tail less than 45, and lacking black pencil.
Description.—Size.—Male: An adult or subadult from Fair Oaks, Pa., a subadult from Finleyville, Pa., and an adult from Huttonsville, W. Va., measure, respectively as follows: Total length, 206, 194, 191 (average 197); length of tail, 37, 32, 28 (32); length of hind foot, 23 in each. An adult from Roanoke, Indiana, weighs 40.6 grams.
Female: Two young from Leasuresville, Pa., and Middle Paxton Twp., Pa., measure, respectively, as follows: Total length, 188, 172; length of tail, 33, 30; length of hind foot, 20.5, 21. An adult from Monroeville, Ohio, weighs 40.5 grams and a young individual from Middle Paxton Twp., Pa., 39.3 grams, and a subadult from Swan Creek Exp. Station, Allegan Co., Mich., weighs 49 grams.
Color.—Winter pelage either all white, or brown as in summer; upper parts about Raw Umber, or tone 2 of Carbo Brown of pl. 342 of Oberthür and Dauthenay. Underparts white at least on thoracic region; approximately three-fourths of specimens with brown rictal spot at angle of mouth or with this area covered by brown upper parts which extend down on each side and meet on the underparts in about one specimen out of three; upper lips and hind feet ordinarily brown; toes of forefeet ordinarily white (see under remarks for details of color pattern). Least width of color of underparts in the specimens in which the dark color of the upper parts does not encircle the body averages 60 per cent of greatest width of color of upper parts, or including all specimens the percentage is 42.
Skull (based on specimens from Pa. listed in table of cranial measurements, which see and plates 14 and 15).—Basilar length 29.7 and 28.6 in male and 28.0 in female; weights unavailable; otherwise as described in M. e. richardsonii. The length of the tympanic bullae seems to be actually less, and less in relation to the basilar length, than in other American subspecies of M. rixosa.
Remarks.—Robert Kennicott's mention in 1859 (p. 245) of what seems to be this subspecies is the earliest reference to it that I can identify in the literature. He used the specific name pusillus and it was not until 1900 that Samuel N. Rhoads proposed the name Putorius allegheniensis. Since 1900, several records of occurrence have been published which have made the geographic range of this race better known.
An adequate number of specimens has been gathered only from Ohio and from western Pennsylvania. Many from Ohio are without accurate external measurements taken in the flesh. The majority of the specimens from Pennsylvania owe their preservation to the willingness of local officials, who pay bounties on weasels, to save the skins of Mustela rixosa. These specimens ordinarily comprise the skin with locality but because the feet, external measurements in the flesh, and skulls are unavailable, the material is far from adequate and to give an accurate notion of the usual or average cranial characters of allegheniensis in Pennsylvania, skulls from there are especially desirable.
A smaller percentage of the specimens from Ohio than from Pennsylvania have the brown color of the upper parts meeting on the underparts. Also, more of the specimens from Ohio are lighter colored and this suggests intergradation with the subspecies campestris and rixosa to the westward.
From Pennsylvania 23 animals in brown pelage are available. In 5 there is a rictal spot at the angle of the mouth; in 5 the area is white and in 13 the brown color of the upper parts is continuous over the area in question. Only 2 of 23 have the upper lips white. Eight have the color of the upper parts meeting on the venter thus restricting the white of the underparts to the chin, throat, and pectoral region, and 6 of these have a white area in the inguinal region as well. The toes of the forefeet are white in 3 of 4 animals suitable for examination in this regard and the hind feet are marked with white in 3 of the 8 animals which have the hind feet preserved. Mustela rixosa in Pennsylvania parallels the species Mustela frenata in that in this relatively humid area of the northeastern United States the color of the upper parts is darker and the area of the dark-colored upper parts is increased at the expense of the area of the light-colored underparts. Also Mustela erminea in this same region (range of the subspecies Mustela cicognanii) shows the same tendency to darker color of upper parts and their extension in area at the expense of the area of the light-colored underparts, or was mentioned above.
It is difficult to account for the seeming absence of the species from New England and all that part of Canada and the United States south of the St. Lawrence River and northeastward from Pennsylvania. The size of females of M. erminea cicognanii in that territory is so little more than in rixosa that the latter possibly cannot successfully compete with the erminea stock which may already occupy the ecologic niche to which rixosa is adapted. It will be remembered that in western North America in territory seemingly climatically suitable for rixosa it occurs no farther southward than the line below which M. erminea has become reduced to a size comparable with that of M. rixosa.
Of 41 subadult and adult skulls assigned to this subspecies 24 have obvious lesions in the frontal sinuses evidently resulting from infestation by nematodes. More in detail, none of the specimens from Illinois (3 individuals), Pennsylvania (3 barely subadult), or West Virginia (2) displays lesions. From Wisconsin, Indiana, Virginia and North Carolina there is one specimen each and each specimen displays lesions. From Ohio, 17 of 23 specimens display lesions. From Michigan 3 of 8 specimens display lesions; 2 adults and one subadult have lesions and 5 subadults do not have lesions.
Specimens examined.—Total number, 102 as follows: Arranged alphabetically by states and within each state by counties from north to south. Unless otherwise indicated, specimens are in the United States National Museum.
Indiana. Huntington County: Roanoke, 1. Wells County: Harrison Township, 1[76].
Illinois. Lake County: Deerfield, 3[60]; no locality more definite than county, 1[60]. Cook County: Northfield, 1[60]; La Grange, 1[18].
Michigan. Tuscola County: 8 mi. N Caro, 1[76]. Santilac County: Deckerville, 1[76]. Allegan County: Swan Creek Exp. Station, 1[76]; Swan Creek Farm, 1[76]; T. 2N, R. 14W, 1[76]; Allegan, 1[76]. Livingston County: George Reserve, 1[76]; 1/2 mi. N Unadilla, 1. Oakland County: Rochester, 1[76]. Macomb County: Romeo, 1[76]. Washtenaw County: 5 mi. SW Ann Arbor, 1[76]. Branch County: vic. Coldwater, 1[76].
North Carolina. "near Marshall," 1.
Ohio. Northern part of state, 1[81]. Williams County: Stryker, 1[60]. Lucas County: Monclova, 1[60]. Erie County: Sandusky, 2[76]; marsh near Sandusky, 1[76]; Berlin Heights, 1[76]; no locality more definite than county, 1[2]. Wood County: 10 mi. NE Bowling Green, 1[76]; Bowling Green, 4[76]; 3 mi. E Bowling Green 1[76]; Plain Township, 1[2]; Portage Township, 1[60]. Loraine County: Wellington, 1[81]. Huron County: west of Monroeville, 1[76]. Summit County: Ira, 3[81]. Portage County: Suffield, 1[81]. Hancock County: Vanburen, 1[76]; Findlay, 1[81]; 9 mi. S Findlay, 1[76]; no locality more definite than county, 7 (2[76], 2[81], 3[2]). Mahoning County: Ellsworth, 1. Crawford County: "near Crestline," 1[81]. Delaware County: Sunbury, 1[2]; Lewis Center, 1[81]; no locality more definite than county, 1[81]. Licking County: Johnstown, 1[2]. Fairfield County: Baltimore, 1[81]; Violet Township, 1[81]. Meigs [= Gallia?] County: Vinton, 1[81].
Pennsylvania. Erie County: McKeen Twp. 1. Crawford County: Springboro, 1[1]; Pymatuning Swamp, between Hartstown and Shermansville, Sadsbury Twp., 3[9]. Mercer County: Shenango Twp., 1. Lawrence County: Little Beaver Twp., 1. Butler County: Leasuresville, 1[9]; Clearfield Twp., 1; Valencia, 1[9]. Armstrong County: Ford City, Burrell Twp., 1. Indiana County: Smicksburg, 1; N. Mahoning Twp., 2; White Twp., 1. Allegheny County: South Hills, Pittsburgh, 1[9]; "near Pittsburgh," 1[9]; Fair Oaks, 1[9]. Westmoreland County: Bolivar, 1. Dauphin County: Middle Paxton Twp., 1. Washington County: Finleyville, 1; Rea, 5; Beallsville, 1[1]; Claysville, 1. Green County: Deep Valley, 1; Waynesburg, 1; Jefferson, 1; Cumberland Twp., 1. Fayette County: Acme, 1[9]. Somerset County, 1. Lancaster County, 1.
West Virginia. Randolph County: Huttonsville, 1.
Wisconsin. Sauk County: Sumpter Twp., 1[60]. Dodge County: Beaver Dam, 1[50]. Dane County: Madison, 1; McFarland (= MacFarland), 1.
Mustela rixosa campestris Jackson
Least Weasel
Mustela campestris Jackson, Proc. Biol. Soc. Washington, 26:124, May 21, 1913.
Putorius pusillus, Aughey, Sketches of the physical geography and geology of Nebraska, p. 119, 1880, Omaha.
Mustela rixosa campestris, Swenk, Journ. Mamm., 7:329, Nov. 23, 1926.
Type.—Female, adult, skin and skull; no. 171490, U. S. Nat. Mus., Biol. Surv. Coll.; Beemer, Cuming County, Nebraska; April 18, 1911; obtained by G. Sharp; x catalogue no. 8440.
The skull is unbroken. On the left side, C1 and P2 are missing; the other teeth are present and entire. The skin is excellently made and in a good state of preservation.
Range.—South Dakota, Nebraska and Iowa. See figure 28 on page 180.
Characters for ready recognition.—Differs from M. r. rixosa and M. r. allegheniensis in larger size: Hind foot more than 25 in males and ordinarily more than 22 in females; in males total length more than 216 and tail averaging more than 34; color possibly slightly paler than in M. r. rixosa and averaging paler than in M. r. allegheniensis; from M. frenata and M. erminea of the same region by basilar length less than 32; tail less than 50, and lacking black pencil.
Description.—Size.—Male: Four adults from Nebraska yield average and extreme measurements as follows: Total length, 231 (225-237); length of tail, 36 (32-39); length of hind foot, 29 (28-31).
Female: Six adults from Nebraska yield average and extreme measurements as follows: Total length, 192 (184-225); length of tail, 35 (28-40); length of hind foot, 23 (20.5-26).
Color.—Winter pelage ordinarily white; as described in M. r. eskimo except possibly paler and certainly with line of demarcation on side of head between upper parts and underparts passing almost straight back without the dorsally directed reëntrant angles of white behind the eye and ear; least width of color of underparts in four specimens from Nebraska averaging 80 (49-89) per cent of greatest width of color of upper parts, but in a fifth animal in summer pelage the brown color of the upper parts encircles the body.
Skull.—See measurements in table and plate 15; weight 1.1 grams (male from Brown Co., S. D.); basilar length, 30.7 in male from Clay Co., Neb., and 28.8 in female from same county; otherwise as described in M. e. richardsonii.
Remarks.—In his revisionary treatment of the American races of Mustela rixosa, Myron H. Swenk (1926:313) credits Samuel Aughey with recording this animal, M. r. campestris, from Nebraska, as early as 1880, under the name Putorius pusillus. In 1908, Swenk recorded the animal from the same state under the name rixosus and in 1913 the race campestris was formally named by H. H. T. Jackson.
On the testimony of a friend who had previously obtained several specimens for him, Swenk (1926:321) records the least weasel from Oshkosh, Garden County, Nebraska, which is a marginal record of occurrence to the southwest for M. r. campestris.
At an early stage in the study of American weasels the writer examined the specimens from Nebraska saved by Mr. Myron H. Swenk and recorded measurements of them. However, at the time of writing this account the specimens were not available for examination and the account of coloration is accordingly incomplete.
The large size, particularly the large external measurements, comprises the principal distinguishing character of this subspecies of the least weasel.
Of the four adults examined from Iowa and South Dakota one exhibits lesions such as result from infestation of the frontal sinuses by nematodes.
Specimens examined.—Total number, 21 as follows. Arranged alphabetically by states and by counties, from north to south in each state. Unless otherwise indicated, specimens are in the United States National Museum.
Iowa. Howard County: Chester, 1[12]. Palo Alto County: Emmetsburg, 1[65]. Kassuth County: Algona, 1[65]. Clayton County: National, 1. Storey County: Nevada, 1[65]. Wapello County: Ottumwa, 1[65]. Henry County: Mount Pleasant, 1[66].
Nebraska. Holt County: Page, 1[35]. Madison County: Norfolk 1[35]. Cuming County: Beemer, 1. Hamilton County: Chapman, 1[35]. Clay County: Inland to 1 mi. east thereof, 7[35].
South Dakota. Brown County: shore of Sand Lake, S. 15 T. 126N, R. 62W, 1. Day County: Waubay Migratory Waterfowl Refuge, 1. McCook County: Salem, 1[102].
MUSTELA FRENATA Lichtenstein
Long-tailed Weasel
(Synonymy under subspecies)
Type.—Mustela frenata Lichtenstein, Darstellung neuer oder wenig bekannter Säugethiere, pl. 42 and corresponding text unpaged. 1832.
Range.—From southern Canada southward over all of the United States, México, Central America, Venezuela, and the republics of western South America to southern Perú and extreme northern Bolivia. All the life-zones from Alpine Arctic to Tropical are inhabited. In the extremely desert region of southeastern California and western Arizona the species is scarce or possibly absent although recovery of a skull (see under account of M. f. neomexicana) from near the center of this region at Potholes on the Colorado River, and a reported occurrence in the mountains of Baja California, México, indicate that a few individuals of the species live in favorable habitat even in this desert region.
Characters for ready recognition.—Differs from Mustela erminea, in regions where the two species occur together, by tail more than 44 per cent of length of head and body and by postglenoidal length of skull less than 46 per cent of condylobasal length in males and less than 48 per cent in females (see under characters of the species); from Mustela rixosa by presence of black pencil on tail, caudal vertebrae more than a fourth (2/5-3/4) of length of head and body, basilar length of skull more than 34 mm.; from Mustela africana by absence of thenar pad on forefoot, underparts without longitudinal, median, abdominal stripe of same color as upper parts, upper lips narrowly (rather than broadly) edged with color of underparts, longest facial vibrissae extending to or behind posterior margin of ear; presence of p2; more inflated (see pls. 23 and 30) tympanic bullae.
Characters of the species.—Size large: Total length 300 to 550 mm.; tail two-fifths to seven-tenths of length of head and body, with distinct black pencil at end; caudal vertebrae 19 to 23; skull with long precranial portion; postglenoidal length, expressed as a percentage of the condylobasal length, less than 47 in females and ordinarily less than 46 in males; upper parts brown; light-colored underparts, in summer pelage, tinged with buffy or yellowish and continuous from chin to inguinal region; some subspecies (southwestern United States, México, Central America, and Florida) with white or yellowish facial markings which do not occur in any other American species of the genus Mustela.
Geographic variation.—Forty-two subspecies are recognized, and the species is geographically more variable than any of the other 3 American species. Color, color-pattern especially on the head, relative proportions of the tail, hind feet, body including the head, and shape and size of the skull are the principal features in which geographic variation has been noted. The variation in the skull extends to the basicranial region (shape and size of tympanic bullae and related structures), interorbital region and preorbital region.
Natural History.—Habitat and Numbers.—As has already been remarked, the long-tailed weasel is absent from the extreme desert of the southwestern United States and northwestern México. Possibly the absence of water to drink is the limiting factor. In southern Nevada the finding of weasels only in places that were well watered, even though small rodents suitable as food for weasels were even more abundant in the surrounding desert, supports this possibility that the absence of water to drink is the limiting factor. Also at Berkeley, California, in early December of 1927 in the canyon at the head of Dwight Way and in the autumn and winter of 1928 in Strawberry Canyon on the campus of the University of California, I trapped extensively for this species in different habitats and obtained, in all, four individuals no one of which was farther than 10 feet from water. The lesser cruising range of the individual weasel than of, say, the coyote, probably explains why, in an arid region, for example Pahranagat Valley, Nevada, only the meadow mice and their riparian associates are preyed upon by the long-tailed weasel whereas the coyote preys upon these riparian rodents and also upon the kangaroo rats and other rodents which are so abundant in adjoining habitats that are devoid of water.
In areas where water is available every few hundred yards, no particular habitat seems to be avoided in summer providing there is food for the long-tailed weasel. In winter (January and March) there obviously was a choice of habitat, possibly occasioned by more abundant food or more satisfactory shelter, or both, in Centre County, Pennsylvania, where Glover (1943B) found the population density in the chestnut-oak habitat to be one weasel per 6.5 acres in areas of tree cuttings and slash and one weasel per 13.3 acres in the open forest. In the scrub oak-pitch pine forest type the population was one weasel per 26.4 acres in tree cuttings and slash and one weasel per 38.2 acres in the open forest. No weasel was found in an area of 9.6 acres comprising a wood lot, the edge of the forest, abandoned fence rows and an abandoned orchard. The two types of forest in which he did find weasels, 25 in all, comprised 381.6 acres. Glover's (op. cit.) data is the only precise information known to me on actual numbers of long-tailed weasels in a given area of any considerable size.
Fluctuations which I elsewhere (1946:57) have designated as multiannual fluctuations occur in this species but seemingly not in the degree that they do in Mustela erminea. This difference between the two species is to be expected because M. frenata does not range so far northward toward the polar regions as does M. erminea and populations of most kinds of animals in the polar, at least in the arctic, regions are subject to more extreme and more regular fluctuations than are kinds of animals in temperate or tropical regions. Indication of the means by which decrease in the weasel population is brought about is afforded by Osgood's (1935:156) observations around Rutland, Vermont. In the late winter of 1934, tracks indicated that weasels left their usual haunts and hunted cross lots, vainly trying to find food. Testing of the small mammal population in the spring and summer of 1934 showed that it was at low ebb. In the fall of 1934 mice and shrews were abundant again but weasels seemed to be entirely absent. The decrease in the population of weasels lagged behind the decrease in the population of the herbivorous prey as did the subsequent increase; this, of course, is the normal relation of carnivorous species of mammals and their prey, at least in and above the Transition Life-zone.
The average distance away from the central den which four weasels (sex unspecified) traveled in a single night at Ames, Iowa, was 312 feet; the maximum distance was 642 feet. These data were obtained in the winter of 1939 by Polderboer, Kuhn and Hendrickson (1941:115) who studied the tracks in the snow. In Manitoba, Criddle and Criddle (1925:143) noted that a female which lived in their basement often wandered more than half a mile away in search of food. In Michigan, Quick (1944:75) found the maximum distance traveled in one day (= night?) by a large male to be 3.43 miles although two miles was the average distance traveled by this individual. In 1942, from January 4 to March 4, in Centre County, Pennsylvania, Glover (1943B) studied tracks of 11 males and 10 females, in newly fallen snow, and ascertained that the distance traveled in a single night averaged 704 (60-2535) feet for the male and 346 (20-1420) feet for the female. The weasels in the open timber traveled farther per trip than those in the brushland and dense stands of trees.
Behavior
An adult female (now the holotype of Mustela frenata nevadensis) seen running across a field, and, I think, unaware of my presence, at every bound bent her back up so far that she reminded me of a measuring worm. For part of the time when running, the tail was held off the ground straight out behind, and then, for a while, inclined upward at an angle of about 45°. Another weasel that I saw in the daytime, and that I think was unaware of my presence, was bounding along among the Baccharis bushes on the south-facing slope of Dwight Way Canyon, Berkeley, California. This individual, at each bound, arched the back up so high as to remind me, again, of a measuring worm.
The long-tailed weasel is a land mammal and unlike its close relative, the mink, is seldom seen in the water. That it can swim, however, is attested by the capture of one while it was swimming across the Río Ramos in México (Davis, 1944:381). Also, Green (1936), in May, in Gratiot County, Michigan, saw a weasel, running with a Peromyscus in its mouth. The weasel dropped the mouse, entered the water and swam to a hole among stones.
More instances of climbing, than of swimming, have been reported in the literature for the long-tailed weasel. Seton (1929 (2):625) quotes William M. Graffius of Pennsylvania as having seen a weasel closely pursue a red squirrel nearly to the topmost branch of a large hemlock. When the squirrel loosed its hold and dropped into a stream, the weasel descended to the ground and caught and killed the squirrel when it emerged from the water. Pearce (1937:483), in central New York State, on July 29, 1931, watched a weasel chase a chipmunk up a black cherry tree ten inches in diameter, and noted that the first rush carried the weasel "straight up the trunk for approximately 10 feet, where it hesitated momentarily before continuing. Then, instead of climbing vertically, it made progress by traveling in short ascending spirals around the trunk, scarcely making 3 feet in height for each circuit of the tree. Upon reaching the limb by which the chipmunk escaped, the weasel followed out along this in the same spiral manner. This limb had a diameter of about 4 inches at its base and extended upward at an angle of perhaps 20 degrees above the horizontal . . . it made its way head first almost down to the ground, using the same spiral mode of progress, but at a leisurely pace. . . . While traveling down the side limb it appeared practically to wrap its sinuous body around the limb."
A male long-tailed weasel, from Colorado, which I kept captive was often fed freshly killed mice. These I thrust through one of the small openings in the wire mesh. The weasel quickly learned to seize any part of a mouse thus introduced and his tugging aided in getting the mouse into the cage. Occasionally a mouse too large to be got through the mesh had to be withdrawn. In such an instance, if the weasel had already had hold of the mouse, he would screech frightfully. I have heard no other vocal sounds from a weasel except a kind of purring.
The sense of smell apparently is well developed; at any rate it is keen enough to allow the weasel to follow the trail of an intended victim by the scent left by the latter. Murie's (1935:321-322) account, for example, of a weasel pursuing a snowshoe rabbit gives clear evidence that the weasel relied on scent in following the rabbit.
A captive male weasel obtained at Gainesville, Florida, stamped his hind feet when annoyed (Moore, 1945:259).
A male from Colorado that I kept for months in a cage at Lafayette, California, was several times found in a sleep so deep that he was awakened with difficulty. Seton (1929 (2):629-630) writes: "In my small menagerie, I have had half-a-dozen Weasels of the New York species. Their sleeping dens are arranged so as to be easily and silently opened. Several times I have lifted the lid to find the weasel in a deep sleep—a sleep so profound that I had to poke him vigorously with a stick before he awoke, looked up, and rushed forth with a little puff of wrath, and a little puff of smell."
Feces and urine were ordinarily deposited in one particular place by each of the captive weasels that I have observed. Hamilton (1933:294) records that a large male M. f. noveboracensis, in a week, averaged 10 evacuations every twenty-four hours, that urination immediately precedes defecation, and describes the feces as black or brown, long and narrow and often spiral-shaped owing "to the matted fur of some rodent that had been eaten." Quick (1944:77) writes, concerning four winter dens in Michigan, that "The latrines of weasels were in the entries of used dens and scats could be collected there by the handful." Polderboer, Kuhn and Hendrickson (1941:116) in the spring of 1939 at Ames, Iowa, gathered scats "from latrines found at the entrances of burrows and from latrine chambers found within burrows." Scats were found by them in the linings of some nests.
Courage of a high order might be credited to the long-tailed weasel because individuals have attacked animals much larger than the weasels. Actually, however, in few if any of these instances was the motive for attack known. That a hawk was attacked is suggested by Soper's (1919:45) account of Mustela frenata noveboracensis wherein he repeats a story told to him of a hawk observed in unsteady flight, and obviously in distress, which when it plummeted to earth was with a weasel which escaped from the observer. Charles Tatham, Jr., of Cambridge, Massachusetts, according to Seton (1929 (2): 630, 631) observed one that attacked his dog.
Persons and long-tailed weasels have figured in some rather strange encounters. For example, Oehler (1944:198) recounts that in the autumn of 1940 at Cincinnati, Ohio, an animal, mistakenly thought to be a chipmunk, was seen to dash into a hollow log whereupon pounding on the log brought out the weasel which bit and clung to the hand of one man whose companion was bitten when he attempted to free the man that was bitten first.
Seton (1929 (2): 631) writes that on the night of September 5, 1897, on Roosevelt's old ranch, near Medora, North Dakota, a man turned over his saddle (which was lying on the ground) to dislodge what was thought to be a pack-rat. The animal was a long-tailed weasel which attacked him. It ran up his legs a number of times aiming at his throat before being killed by a dog.
Criddle and Criddle (1925:146) wrote: "August 20, 1919.—A longicauda in the Insectary ran at me this morning apparently with a view to intimidating. It uttered a shrill cry while making the attack, but retreated after advancing within two feet." The same authors (op. cit.: 147) further write that a "Long-tailed Weasel was caught in a trap set for gophers, and, on being released by Miss M. Criddle, at once turned upon its liberator and bit savagely at her boot. It then moved a short distance away to a tub of water, where it drank thirstily, merely glancing at the observer from time to time while doing so, and then ran off out of sight.
"Mr. T. Criddle records a similar experience. After liberating a large weasel from a trap, it immediately rushed at him and persisted in its attack with such ferocity that it was three times picked up and thrown, on each occasion to a greater distance, before it finally abandoned its offensive.
"We have no record of a weasel making an unprovoked attack upon anyone."
Wight (1932: 164) in Michigan, detected a weasel attacking a hen. The weasel fled at Wight's approach but returned and attacked him several times. Finally the weasel went around Wight to reach the hen. In Wight's words "There was no evidence of infuriation, but rather a well directed offense at the one object, regardless of its size, which stood between the weasel and an opportunity to satisfy its desire to kill, which was probably based upon the uncontrollable urge of hunger pangs."
Weasels of each of the three North American species have been successfully kept in captivity. A type of cage satisfactory for keeping the animals in the laboratory is described by Bissonnette and Bailey (1940:761-763). Some of the captives used their teeth to break glass water-containers and to gnaw slivers of wood from the cages. Ingested slivers of wood and bits of broken glass caused the deaths of some of the captives. Weasels kept by me all were of the species Mustela frenata. They thrived on a meat diet but I was always careful to give them, every few days, if not each day, some small rodents entire, thinking that the bits of bone and fur ingested might, in some way unknown to me, keep the digestive tract in better condition than would flesh devoid of hair and bone.
Three young weasels approximately the size of mice, in the Okefinokee Swamp of Georgia, were obtained by a hunter who, according to Harper (1927:303), raised them by feeding "milk for a few days, and then fresh meat." Litters of young born in captivity have been successfully raised by the mothers (Hamilton, 1933) and success in getting the animals to breed in captivity and to rear their young is recorded by Wright (1948A). He has found, however, that the majority of his captive adult males show no interest in mating when placed with females in heat. He, therefore, uses only selected males and when a female in heat is to be bred, he places one of his responsive males with her one day, another of his responsive males with her the second day and thus alternates a couple of males for three or four days. Even so, slightly fewer than half of the females which were thus bred produced young.
A weasel in the white winter coat was used by Audubon and Bachman (1856:177, Quarto edit.) to drive rabbits out of their burrows in the same fashion that ferrets commonly are used. Although these naturalists refer to their animal as an ermine it probably was Mustela frenata noveboracensis, the long-tailed weasel. The animal's teeth (probably canines) were blunted and a long cord tied on its neck. With the aid of this weasel 12 rabbits were caught in one morning and more than 50 in four weeks.
Enemies
Little is recorded concerning enemies of weasels and it may be that other vertebrates are not an important factor in removing the annual increase. Errington (1935:195-198), in Iowa, found four, putrid weasels about dens of red foxes, Vulpes fulvus. No remains of weasels were found in the feces of the foxes and it appears that the foxes do not eat the weasels. The label on an adult female specimen of M. f. spadix from Boone County, Iowa, bears the date May 10, 1938, and the annotation, by T. G. Scott, "fox-killed." Bailey (1931:328) recounts that "Weller saw a coyote carrying one in its mouth" at an elevation of 11,500 feet in the Pecos Mountains of New Mexico. The type specimen, a young female, of M. f. peninsulae from Hudsons, Florida, according to Rhoads (1894:155) ". . . was caught in the woods by a cat." Barber and Cockerell (1898:189) mention one that was killed by a dog in Mesilla Park, New Mexico. Moore (1945:258) records the death of a weasel in Florida. Circumstantial evidence indicated that it was killed by the bite of a water moccasin. In the Biological Surveys Collection of mammals in the United States National Museum, the label with the skull of an adult male weasel, No. 160663, from Banning, California, carries the information that the skull was taken from the stomach of a Crotalus (rattlesnake).
In reporting on a study of owl predation in Delaware County, Pennsylvania, Pearson and Pearson (1947:143) mention that "weasels are found throughout the county but . . . were never eaten by the owls."
The Uinta spermophile at some places and times probably is a prey sought by the long-tailed weasel but Warren (1924:265) records Citellus armatus repeatedly chasing weasels in August, at Camp Roosevelt, Yellowstone National Park, and how the ground squirrels at one time ignored the weasel even when it came within a few inches of a squirrel.
Warren (1932:71), on August 2, 1931, at Grand Mesa, Colorado, obtained a large male weasel with two porcupine quills in it; one was near the mouth and another "in the skull." Osgood (1935:156) writes that near Rutland, Vermont, a male weasel "taken in April, was heavily parasitized and had several short porcupine quills embedded in its neck, head, and shoulders." The remainder of Osgood's account implies that the weasel may have turned to porcupine because the normal food for weasels was scarce at the time. Porcupine quills, then, are a hazard for weasels although it is unlikely that the porcupine is ever to be classed as an enemy of the weasel.
An accident of another sort, which must at the very least have been annoying to the weasel that suffered it, was recorded by Soper (1921:37). The animal had a stick lodged crosswise between the fourth upper premolar teeth.
The recorded actions of several kinds of animals which are too small to be dangerous to the weasel suggest that they recognize that the weasel is a danger to them. Borell and Ellis (1934:21) mention that a weasel in Nevada caused a great disturbance among the chipmunks. Long (1938:250) heard pikas give evidence of terror by a peculiar cry when a weasel was in a rock slide occupied by the pikas. Seton (1929 (2):629) writes "On June 14, 1915, as I prowled around the south side of the lake on my homeland at Greenwich, Conn., my attention was called to a pair of song sparrows and a male towhee that were noisily mobbing a Weasel, twittering around and darting at him, as though they knew full well his evil ways. The weasel paid little heed, but soon dived from sight in a stone wall."
No account has been found of an American weasel or ermine rolling, tumbling and frolicking in a manner that aroused the curiosity of birds to a degree which permitted the weasel to come within leaping distance of the birds. Accounts of such behavior are on record for the English stoat (ermine).
Food and Hunting
Weasels are active both in the daytime and at night. Whether the time of activity varies with the season, with the locality, with the sex or with other conditions, I do not know. Adult, live, free-living, actively moving weasels that I recall having seen all were observed in the daytime: two were in Alameda County, California, two were in White Pine County, Nevada, one was in Scotts Bluff County, Nebraska, and one was in Laramie County, Wyoming. I recall ten adults, from the same three states, and one from Washington State, that got into my traps; two of these certainly got in the traps in the night; one certainly got in the trap in the daytime; the other eight were found in traps which may have caught the weasels either in the night or in the daytime. Soper (1946:136) in speaking of M. f. longicauda north of the International Boundary in Canada remarks that it has the "habit to some extent of hunting at all times of day." Criddle and Criddle (1925:144) in writing of Mustela frenata longicauda in Manitoba record that "The shrill cry of a rabbit [Lepus americanus] in the dark is nearly always due to the weasel's attack. Indeed, we have often watched the latter at work during the twilight hours. First would come the almost noiseless run of the small rabbit with its characteristic dodging and this would be followed by the appearance of the agile foe which, at times, would leap high over obstacles and at others move swiftly beneath them. Then there would follow intermittent cries of the rabbit as the weasel secured a temporary hold of its quarry, for be it noted that this hunter apparently bites anywhere to begin with and it is probable that the blood made to flow acts as an aid to tracking as well as weakening the prey. Several similar close encounters might occur before the rabbit would be finally overcome, but weasels are very persistent when they once get into contact with their victims and it is therefore very seldom that the latter escape. In killing, they either penetrate the brain with their teeth, or dislodge the vertebrae behind the head." These and more than two score other observations which record the time when weasels were seen make it clear that some were active at night and that some were active in the daytime.
As to the routes traveled while the weasels are hunting, Quick (1944:77) says of four individuals that he studied in Washtenaw County, Michigan: "The weasels appeared to prefer hunting certain coverts with noticeable regularity, but rarely cruised the same area on two consecutive nights."
The killing technique of fifteen captive Mustela frenata noveboracensis was studied by Glover (1943A). For the weasels he released 19 mice, 3 brown rats, 6 cottontails and 4 ring-necked pheasants. Most of the mice were killed by a bite on the back of the head, with the body and legs of the weasel hugging the back of the victim. "The weasel shoved the prey in close to the stomach with the hind legs, and the kill was made in a reclining semi-curled-up position." On each of the rats (Rattus) an initial grip was secured at the base of the ear. When the rat rested, a new hold was taken by the weasel. Finally the weasel secured a hold at the base of the skull and near the ear, and a light crushing sound followed. Four of the six cottontails were killed by bites on top of the head and ear; two cottontails succumbed from neck wounds. In three instances, neither of two weasels could be induced to make a determined attack on the cottontails or to kill them. At times the cottontails proved to be able opponents for weasels by striking out with their front feet and by kicking with their strong hind legs. In killing the pheasants the teeth of the upper jaw of the weasel pierced the top of the braincase and the teeth of the lower jaw entered the region of the auditory process. The forelegs hugged the neck of the pheasant, the body of the weasel was extended in a riding position on the back of the bird and no amount of kicking or rolling dislodged the weasel.
Polderboer, Kuhn and Hendrickson (1941) describe a cottontail cached by a weasel as having the muscles of the neck severed from the region behind the right mastoid process and noted "that hemorrhage in the region of the right jugular vein had occurred."
Concerning the methods of killing mammals smaller than cottontails, the accounts by Nichols and Nichols (1935:297-299) and that by Svihla (1931) corroborate Glover's (1943A) account, as do also the accounts of Miller (1931B:164) and Moore (1945:257). The latter says that his captive male, from Gainesville, Florida, customarily bit its rodent prey at the base of the skull and used the feet to manipulate the live prey. Miller (loc. cit.) emphasized that his male weasel (M. f. longicauda) grasped where it could, used its snakelike body to coil over the prey and shifted the grip of its teeth to the nape of the neck or back of the skull. The captives that I have had [one from Salt Lake City, Utah; three from Contra Costa County, California; and the same individual reported upon by Miller (1931:150)] customarily employed the techniques of killing small rodents that were described by Glover and Miller (loc. cit.).
Allen (1938:225-229) experimented with the ability of four different males of M. f. noveboracensis from Michigan to kill adult cottontails. The method used was to place the weasel in a cage of quarter-inch hardware cloth approximately three feet long, two feet wide, and two feet high. The bottom of the box was covered with several inches of straw. One cottontail was offered to each weasel. In two instances the weasel attacked and bit the cottontail, was struck by the hind feet of the cottontail, retired from the attack and died a few hours later as a result of the blows of the cottontail's hind feet. In the other two instances the weasel rendered the cottontail helpless by severing the neck muscles from the skull. Subsequently an incision made by the weasel, in each of the two instances gave access to blood on which the weasel fed until it was full, in one instance by licking "blood as a cat laps milk." One rabbit was subdued in 10 minutes and the other in 15 minutes. Allen (op. cit.) points out that cottontails form a considerable portion of the weasel's food and thinks that they are killed in burrows more easily than they were in the cage.
In writing of the three species of weasels, including Mustela frenata, found at Treesbank and vicinity, Manitoba, Norman Criddle and Stuart Criddle (1925:143, 144), in my opinion, correctly explain the killing of more prey than weasels need. "The fact that weasels frequently kill many more animals than they require for immediate use has been universally interpreted as a lust for killing—a supposition which we believe to be quite erroneous. It is true that weasels often kill more than they need, but the surplus is not necessarily wasted because the animals always store it for future use, in much the same way as do badgers, minks or skunks, and with the same object in view as squirrels have in gathering nuts. We have observed many such stores, but as far as our observations go, the habit of killing in excess occurs much more prominently in the late summer and autumn months than in the spring. Indeed, we have no records of excessive spring slaughter and this indicates that the supposedly blood-thirsty habit of weasels is no more a lust for killing than is the woodsman's foresight in providing his larder with meat for the winter months. It should be noted in this connection that members of the weasel family, when undisturbed, do not leave their victims scattered about, but carefully store them away, and in many instances the bodies are buried with earth or taken under ground to preserve them. We suspect that this instinct for preserving food for future use accounts for most of the excessive killing by carnivorous animals instead of this latter indicating an aimless desire for slaughter which would unnecessarily deplete the food supply of the future. This instinct, however, does not seem to be as definite as that of some rodents, and there is no doubt that much of the stored meat decays before it can be utilized."
Criddle and Criddle (1925:146) note that a weasel in the vicinity of Treesbank was carrying a rat [Rattus] and that "Two small punctures in the throat were the only evidence of the manner in which its death had been brought about."
Considerable information has been recorded concerning the food of Mustela frenata and a little information is on record as to kinds of foods not taken that could have been taken. For example, Ingles (1939:253, 254) on May 14, 1938, near Shasta City, California, noted that nestlings of russet-backed thrushes were ignored by an adult weasel and four young weasels which were feeding instead on meadow mice and a mole. Howard (1935:322, 323) records that a weasel in Michigan which carried bits of meat from beef bones on a porch ignored a red squirrel which drew on the same food supply but which retreated to the end of the porch when the weasel appeared. Quick (1944) records that in the winter of 1940 on a 640 acre area in Washtenaw County, Michigan, four resident weasels did not kill any of the 10 rabbits or several pheasants but subsisted on smaller animals. Glover (1943A) thought that M. frenata kills only a few adult cottontails in the wild. To judge from these observations, M. frenata chooses small mammals as prey in greater measure than it does birds or larger mammals.
Records of prey taken, attacked or pursued by Mustela frenata include the following:
Broad-footed mole (Scapanus latimanus).—One was fed on by an adult M. frenata and four young, on May 14, 1939, "near Shasta City," California (Ingles, 1939:253, 254).
Dusky shrew (Sorex cinereus).—A female weasel, at Majestic, Long Island, N. Y., was shot when carrying a Sorex cinereus that had a small hole in the top of its head (Nichols and Nichols, 1935:297-299).
Big short-tailed shrew (Blarina brevicauda).—One was taken from the stomach of a weasel (Hamilton, 1928:249).
Townsend ground squirrel (Citellus townsendii).—Alcorn saw a weasel five miles west of Fallon, Nevada, carrying a squirrel (Hall, 1946:192).
Richardson ground squirrel (Citellus richardsonii).—The attempted capture of one of these squirrels in Saskatchewan is recorded by Seton (1929 (2):625).
Belding ground squirrel (Citellus beldingi).—Grinnell, Dixon and Linsdale (1937:233) recount that at Tuolumne Meadows, California, a weasel killed a ground squirrel of this species.
Thirteen-lined ground squirrel (Citellus tridecemlineatus).—Errington (1936:406, 407) found a den in Palo Alto County, Iowa, on June 22, 1934, where he collected 32 fecal pellets. Sixteen samples contained thirteen-lined ground squirrels, 9 contained rabbits, 9 contained mice (7 Microtus, 1 Peromyscus and 1 unidentified); red-winged blackbirds and unidentified fringillids were represented as also were ground beetles, grasshoppers and other insects. One red-winged blackbird lay near the entrance of the den.
Franklin ground squirrel (Citellus franklinii).—Sowls (1948:126) records that at Delta, Manitoba, a weasel was observed killing one of these squirrels and that "the weasel had taken the squirrel from its hibernating burrow as evidenced by tracks in the snow." On July 19, 1917, in the vicinity of Treesbank, Manitoba, T. Criddle saw a weasel attacking one of these ground squirrels which was in mortal terror and squeaking continuously. Eventually the squirrel was thrown on its back "and would have been speedily killed but for an interruption" (Criddle and Criddle, 1925:146).
Golden-mantled ground squirrel (Citellus lateralis).—On August 15, 1941, along the Kaweah River in Sequoia National Park, Boyer (1943:99, 100) saw a weasel chasing a Citellus lateralis; three or four times the weasel grasped the back of the neck of the squirrel which each time threw off the weasel until the two, weasel after the squirrel, plunged into the river. The squirrel, bleeding at the base of the skull, was rescued and entered a hole; the weasel got out of the water and under a rotting log. Follett (1937:365) at 2 p.m. in Plumas County, California, saw a weasel have hold of the lower jaw of a golden-mantled ground squirrel near its throat. Alcorn watched a weasel chase a golden-mantled ground squirrel in Nevada (Hall, 1946:192) and Grinnell and Dixon (1919:681) record that on August 4, 1911, near Monache Meadows in eastern Tulare County, California, a weasel pursued, captured and killed a golden-mantled ground squirrel.
Eastern chipmunk (Tamias striatus).—Pearce (1937:483) in central New York State, on July 29, 1931, saw a chipmunk scamper up a tree pursued by a weasel.
Chipmunk (subgenus Neotamias).—Stanford (1931:363) on November 11, 1931, at Fish Lake, Utah, saw a weasel pursuing a chipmunk. On August 5, 1910, "near Independence Lake," Nevada County, California, Louise Kellogg recorded that a weasel seized and ran off with a chipmunk (Grinnell, Dixon and Linsdale, 1937:233). Allen (1938:228) observed that a chipmunk (whether Tamias striatus or T. minimus not specified) was killed in 30 seconds whereas 10 to 15 minutes were required by the caged, male Mustela frenata noveboracensis to kill a cottontail.
Red squirrel (Tamiasciurus).—Seton (1929 (2):625) records the capture of one in Pennsylvania, and Grinnell, Dixon and Linsdale (1937:232), at Cisco, California, saw one closely pursued by a weasel.
Flying squirrel (Glaucomys).—Burroughs (1900:77, 78) records remains of one of these squirrels along with the remains of other animals in a food cache of a Mustela but his account does not make clear whether Mustela frenata or Mustela erminea was the species of weasel involved.
Northern pocket gopher (Thomomys).—In "July, 1939, near Stillwater [Nevada], Alcorn pursued . . . [a] weasel and caused it to drop . . . a pocket gopher [Thomomys bottae] which was about two-thirds grown" (Hall, 1946:192). Grinnell, Dixon and Linsdale (1937:233) write that "at least twice, weasels in the [Yosemite] Valley were seen carrying pocket gophers." Relative to Thomomys talpoides in the vicinity of Treesbank, Manitoba, Criddle and Criddle (1925:146) record that on September 11, 1918, an individual of Mustela frenata longicauda took seven pocket gophers dead. . . . It seized the rodents by the middle of their back and held them high while carrying them away. They were stored in a gopher burrow some two hundred yards distant. On February 17, 1921, "Came across the marks of a weasel carting some object over the snow. An investigation revealed a recently-killed pocket gopher with its captor still in possession." Criddle (1930:279), at Aweme, Manitoba, "frequently observed this weasel [M. f. longicauda] . . . carrying a pocket gopher to its larder, and twice it has been encountered in mid winter with freshly killed gophers in its possession." The evidence already presented that weasels levy heavily on pocket gophers is strengthened by the many references in the literature to weasels having been caught in traps set for pocket gophers in the burrows of those rodents and by the many statements, not quoted here, that living quarters of weasels are in burrows made originally by pocket gophers. For example, the present writer, in an account of the Mammals of Nevada (Hall, 1946:191, 192), has said of the long-tailed weasel, Mustela frenata nevadensis, that "All the three dens that were excavated . . . were originally burrows of pocket gophers. . . . Although we have found weasels in many situations in Nevada, . . . they most often were obtained from the burrows of pocket gophers." Excluding the weasels taken by Alcorn, more specimens of the remaining lot were caught in traps set in the burrows of pocket gophers than by all other means combined. All of the 22 weasels taken by Alcorn [within a radius of 10 miles of Fallon] were obtained in gopher traps.
Mexican pocket gopher (Cratogeomys).—At Chalchicomula, 8000 feet, Puebla, Nelson (1918:470 and letter dated March 9, 1928) saw a weasel fastened to a pocket gopher. Nelson obtained the pocket gopher and found that its neck muscles were torn loose from the skull.
Grasshopper mouse (Onychomys).—Barber and Cockerell (1898:189) found remains of this mouse in the stomach of a weasel at Mesilla Park, New Mexico.
White-footed mice (Peromyscus).—Green (1936) saw a weasel in Gratiot County, Michigan, in May, carrying a Peromyscus. Quick (1944:76), in winter, in Michigan, found one dead, probably killed by a weasel. From Washtenaw County, Michigan, Quick (1944:77) examined 294 scats of free-living weasels and found Peromyscus in 189 scats, Microtus in 83, small birds in 20, red squirrel in 3, and hair of weasels in small quantities (probably from the animals which deposited the scats) in 36. He concludes (op. cit., 78) that the winter food was 65 to 70 per cent Peromyscus, 23 to 33 per cent Microtus, and 2 to 7 per cent small birds.
Wood rats (Neotoma).—A female long-tailed weasel weighing 250 grams was taken one mile north of Kent, Texas, while eating a Neotoma albigula (Davis and Robertson, 1944:263). A wood rat house under observation by Vestal (1937:364) in Contra Costa County, California, was invaded by one weasel which ate two adult wood rats (Neotoma fuscipes) and one young. In the same area he saw a weasel in a wood rat nest some months later (Vestal, 1938:5). Three miles east of Reno, Nevada, on May 13, 1936, W. B. Richardson watched a long-tailed weasel carrying a half-grown round-tailed wood rat (Neotoma lepida) across a rock slide (Hall, 1946, 192). Harper (1927:303) records three wood rats [Neotoma floridana] and two cotton rats [Sigmodon hispidus] found dead in the den of a female weasel and her three young in the Okefinokee Swamp of Georgia. Another female and three young approximately half grown were found in the swamp in a hollow pine log. Contents of the den as described to Harper were nearly a peck of wood rats, whole and in pieces; remains of several kinds of birds including robins and quail, and a piece of joint snake (Ophisaurus ventralis).
Meadow mice (Microtus).—Polderboer, Kuhn and Hendrickson (1941), in 1939, at Ames, Iowa, identified "A total of 118 items . . . in 97 winter scats and 48 in the 38 spring scats." Their combined data are as follows:
| Frequency | Percentage | |
| Meadow mouse | 71 | 42.85 |
| Harvest mouse | 36 | 21.75 |
| Deer mouse | 17 | 10.23 |
| Mearns cottontail | 14 | 8.42 |
| Short-tailed shrew | 9 | 5.42 |
| House mouse | 3 | 1.86 |
| Tree sparrow | 2 | 1.02 |
| Grasshopper | 1 | .60 |
| Shaw pocket gopher | 1 | .60 |
| Least weasel | 9 | 5.40 |
| Unidentified material | 3 | 1.85 |
Polderboer, Kuhn and Hendrickson divide their data into two categories, winter and spring. Items recorded in winter but not in spring are house mouse, tree sparrow, and grasshopper. Items recorded only in spring were pocket gopher and least weasel. The samples of cottontail and least weasel all were from the scats of one large male weasel. Of a total of 14 pheasants, 24 quail and 35 cottontails on the 160 acres involved in the study only two cottontails appear to have been killed by the weasels—really by one weasel of four which lived on the area.
Food items taken from the nests (3) and adjacent caches of food in the dens, were as follows: meadow mouse, 30; short-tailed shrew, 4; pocket gopher, 2; deer mouse, 2; least weasel, 1; tree sparrow, 1. The authors remark that the abundance of several prey species does not cause the weasels to ignore the shrews which are said to be distasteful to carnivores.
Two horned larks, apparently killed by weasels, were found on the 160 acre area studied; the horned larks were not in caches of food, nor were remains of horned larks found in scats.
Dearborn (1932:34, 37) for Michigan, on the basis of contents of (37?) intestinal tracts and "feces collected partly in winter and partly in summer" found that, by frequency of occurrence, mammals comprised 83 per cent of the food, birds 10 per cent and insects 7 per cent. Frequency indices for the genera of mammals in percentages of food items of all kinds were as follows: Microtus, 31 per cent; Peromyscus, 24 per cent; Sylvilagus, 14 per cent; Sorex, 7 per cent; Blarina, 5 per cent; Scalopus, 2 per cent.
Criddle and Criddle (1925:146), for the vicinity of Treesbank, Manitoba, record that on October 3, 1913, a weasel was seen to take a field mouse down a hole. They add (op. cit.:147) that "Once while ploughing, we observed a Long-tailed Weasel carrying a field mouse. . . ." Ingles (1939:253, 254), in June, 1938, near Mt. Shasta City, California, found an adult and four young weasels which fed on several Microtus montanus montanus. Green (1936) in May, in Gratiot County, Michigan, in the vicinity of a nest in which there were four young weasels, found "several" dead Microtus. Hamilton (1933:330) records that in New York State a male weasel, on April 5, 1932, at Ithaca, had eaten a Microtus and that in May, 1927, a female weasel was seen carrying a Microtus in its mouth.
Hamilton's (1933:333) study of the contents of the digestive tracts of bodies of weasels obtained from fur trappers and fur buyers enabled him to publish the following "Frequency Indices of Mammal Genera in Fall and Winter Food of 163 Mustela noveboracensis": Microtus, 33.6 per cent; Sylvilagus, 17.3; mammals undetermined to genus but principally mice, 17.1; Peromyscus, 11.3; Rattus, 9.1; Blarina, 5.9; Sciurus, 2.7; Tamias, 1.0; Condylura, 0.8; Ondatra, 0.8.
Grinnell, Dixon and Linsdale (1937:233, 234) quote W. Fry concerning a weasel which reared six young at Giant Forest, California, in 1919, as follows: "This parent weasel, after the birth of her young, remained at the premises for a period of thirty-seven days; during which time, from actual count, the following numbers of mammal species fell victim to her: mice [genera not specified] 78; gophers 27; moles 2; chipmunks 34; wood rats 3; ground squirrels 4. This is a total of 148 animals for the . . . thirty-seven days . . . not a bird was captured during the period."
Rats (Rattus).—Criddle and Criddle (1925:146), on the farm at Treesbank, Manitoba, record a long-tailed weasel, on July 2, 1918, running away from the farm buildings carrying a rat; July 11, 1919, "Two longicaudas . . . have been seen running off with rats on several occasions."; July 11, 1920, "There are two large weasels about the buildings[;]. . . . Each has been noted with rats and this afternoon one of them was seen running into the woods carrying a rat, followed by two excited swallows." The authors (op. cit.:147) add "In the fall of 1924, Mr. A. Cooper, a prominent poultryman of Treesbank, observed a large weasel carrying a freshly killed rat which it stored below ground and then returned towards the poultry-house, causing no little apprehension to the owner. Within a short time, however, the weasel reappeared with another rat which it hid as before. In this way several rodents were accounted for during the afternoon, and Mr. Cooper assures us that the weasel 'kept up the good work for some days'." Hamilton (1933:330) in New York State in May, 1927, saw a male weasel in possession of a rat.
Big jumping mouse (Zapus major).—In the Warner Mountains of California, on Parker Creek, H. C. Bryant frightened a weasel that dropped a freshly killed jumping mouse (Grinnell, Dixon and Linsdale, 1937:232).
Snowshoe rabbit (Lepus americanus).—Adolph Murie (1935:321-322) writes that: "Four miles north of Funkley, Minnesota, early on the morning of November 13, 1921, . . . watched from the top of a 30-foot spruce a weasel. .. hunting a varying hare. . . . The ground was covered with six inches of fresh snow . . . both animals . . . [had] their [white] winter pelage.
"My attention was first attracted to the hare as it came hopping steadily but unhurriedly from the north. Directly in front of me, about 75 feet from the tree I had climbed, the hare crisscrossed back and forth at various angles over an open area about 20 feet in diameter. After producing a maze of tracks, the hare 'froze' near one edge of the pattern. In a few minutes the weasel appeared, all his faculties focused on the warm trail. Expertly he followed its convolutions, passing at times within a few feet of the watching hare. Not until the weasel had followed every turn of the trail to within three feet of its termination did the hare skip off. It came out to the road almost directly below me, turned at right angles northward and was soon out of sight. At the road the weasel lost the trail, . . . and then ran parallel with it, once more in hot pursuit.
"Ten minutes later the hare emerged from the north as before, came on directly to the tracked-up area, and continuing its stratagem, leisurely hopped about to leave its zigzag trail. Then it sat down quietly to wait. . . . The weasel['s] . . . nose led him through the network with little trouble. He was almost upon the hare before it jumped off and followed the same path [as] . . . before. . . .
"The hare had to show his big heels [a third time] . . . as the weasel approached him. This time the weasel failed to follow. . . . After examining a few brush heaps he vanished into the woods behind me."
Seton (1929 (4):723, 724) writes that in December of 1886 in the sandhills northeast of Carberry, Manitoba, he saw a weasel chasing a snowshoe rabbit which took refuge near his feet under the sleigh and so escaped the weasel. Thurber (1940:356) mentions a month-old varying hare that was rescued from a weasel and of approximately the same size as the weasel.
Criddle and Criddle (1925:146) for the vicinity of Treesbank, Manitoba, record "August 21, 1921.—Heard cries of a small rabbit at dusk to-night, which investigation showed was being attacked by a large weasel. The rabbit was later carried to the weasel's store chamber below ground." They record further (op. cit., 146, 147): "November 8, 1924.—Shot a bush rabbit and left it lying. Two hours later [it] . . . was found to have been dragged beneath a brush pile and partly eaten. Innumerable weasel tracks left no doubt as to the identity of the thief." In describing a weasel that wintered in a nest in a threshing machine, the same authors (op. cit.:143) say that no bird remains were found in the pile of approximately three pounds of droppings adjacent to the nest. In a store chamber some 140 yards away from the nest, two bush rabbits (Lepus americanus) had been dragged to the entrance and numerous smaller rodents were taken below ground. The rabbits were buried beneath the snow and eaten as necessity arose. Narrow selectivity on the part of the weasel in choosing food is almost always shown in instances where the food of weasels has been studied. For example, the weasel which lived in the threshing machine ate rodents and rabbits and not poultry although the weasel had ready access to the poultry building. The weasel which lived in the bag of feathers in the basement of Stuart Criddle's house ignored grouse, approximately 20 in number, in favor of other non-avian food.
Cottontail (Sylvilagus).—Polderboer, Kuhn and Hendrickson (1941) mention that one of 4 weasels which they studied on a 160 acre area at Ames, Iowa, in 1939, had a cache of food in a pocket gopher burrow 10 rods distant from the weasel's den. The cache contained only two cottontails, one partly eaten. Leopold (1937) records seeing a Mustela (probably a long-tailed weasel but possibly an ermine) kill a third-grown cottontail by biting it at the base of the skull. Leopold describes the blood sucking or licking, suggesting that he shared the popular misconception that weasels suck blood. The supposition that weasels suck blood has been refuted by many observers, for example by Svihla (1931). My own observation of captives makes me think that weasels do not suck blood. Seton (1929 (2):626) quotes B. H. Warren as seeing a weasel dragging a freshly killed, still warm, rabbit that contained nine embryos almost ready for birth. A young rabbit was seen being carried by a weasel in Hidalgo County, Texas, in March, 1935 (Mulaik, 1938:104). An instance of a cottontail being chased in June in South Carolina is recorded by Hamilton (1933:330). Addy (1939:372, 373), in Virginia, on August 14, 1939, shot a large weasel which was pursuing a Sylvilagus that was only a foot and a half ahead of the weasel. The rabbit stopped when a shot was fired and permitted itself to be stroked and petted. Tracking showed that the weasel had chased the rabbit for a half mile. On November 20, 1942, at Lake James, Indiana, a weasel was seen by Grosjean (1942:443) attacking a "young rabbit" in the throat of which the weasel had made five large holes from which there was no obvious bleeding. Seton (1929 (4):798) recounts that in 1910 at Base Lake, Michigan, F. C. Hicks saw a cottontail with a weasel hanging to its legs rush to the cottage. When only four feet from Hicks the weasel loosed its hold and the cottontail escaped under the cottage. Burroughs (1939:253) on May 14, 1939, in Saginaw County, Michigan, records that a young cottontail weighing between 200 and 250 grams was carried from the nest and killed. Burroughs was attracted first by the "hissing scream" of the weasel, strode toward the sound, flushed an adult cottontail, and discovered the empty nest from which the weasel had taken the young cottontail.
Brush rabbit (Sylvilagus bachmani).—Vestal (1937:364) in Contra Costa County, California, found a brush rabbit that appeared to have been killed by a weasel.
Reptiles.—Grinnell, Dixon and Linsdale (1937:234) recount that in July, 1889, in Wilson Canyon, near Pasadena, California, a weasel killed a red racer by severing the backbone of the snake. In April, 1935, in Hidalgo County, Texas, a half grown bull snake (Pituophis sayi sayi) was regurgitated by a young weasel. Russell (1930:504, 505) has recorded finding in California a male weasel and a king snake (Lampropeltis getulus boylii) three feet five inches long in mortal combat. The weasel killed the snake but the weasel, incapacitated by the conflict, was easily picked up by hand and was also saved as a specimen.
Wild birds.—In the spring of 1940, in Washtenaw County, Michigan, one bobwhite, of 10 bobwhite living on a 640 acre area, was killed by one of four weasels that lived on the area. No other quail was killed there. The one unfortunate bird was killed in the mouth of an abandoned den where the quail roosted (Quick, 1944:76). A male weasel, subspecies M. f. effera, was seen by Booth (1946:439) attempting to enter the nesting hole of a pair of flickers, Colaptes. One song sparrow (Melospiza melodia), and one slate-colored junco (Junco hyemalis) were recorded by Quick (1944:76) as killed by weasel in Michigan.
Chicken (genus Gallus).—Quick (1944:78) writes that in one year (1938-1939) weasels were reported to have killed 1.03 per cent of all chickens in one township of Washtenaw County, Michigan, and that of the total damage to all kinds of poultry, 59 per cent was done by weasels. Weasels entered 19 per cent of the chicken coops on the study area. Farmers killed 68 per cent of the weasels seen in barn yards. Spring and summer were the seasons in which most of the weasels were observed in barn yards. Internal evidence in Quick's (op. cit.) account leads me to suspect that some losses of poultry were charged to weasels when Rattus was actually responsible.
Criddle and Criddle (1925:146), quote a neighbor in the vicinity of Treesbank, Manitoba, as recording that on October 29, 1917, "A weasel last night made its way into our fowl-house, the door being inadvertently left open. The weasel killed eleven fowl, some of which were dragged into the yard. All the largest fowls were selected, the pullets remaining untouched though they were in the majority. Next night the weasel dug a hole beneath the building and killed a hen and two cocks, returning for another during the day, making a total of fourteen in all." Criddle and Criddle (1925:146) remark that the weasel proved to be a large one, probably an old male. The same authors (op. cit.:147) record that at their farm at Treesbank, Manitoba, on January 31, 1925, "A Long-tailed Weasel killed three hens last night, and rather severely bit a cock about the neck. This, or another weasel, had been around the farm-yard for sometime (The specimen was a large male). . . .
"In the fall of 1924, Mr. A. Cooper, a prominent poultryman of Treesbank, observed a large weasel carrying a freshly killed rat which it stored below ground and then returned towards the poultry-house, causing no little apprehension to the owner. Within a short time, however, the weasel reappeared with another rat which it hid as before. In this way several rodents were accounted for during the afternoon, and Mr. Cooper assures us that the weasel 'kept up the good work for same days'.
"Being a farmer of many years' standing, Mr. Cooper has naturally lost some poultry through the agency of weasels, but while he remarks that 'there are good as well as bad actors among weasels', he has the practical good sense to recognize the value of an efficient ratter even though it be a weasel.
"Our sister, Maida Criddle, writes under date of March 4, 1925:
"'There is another weasel (longicauda) in the fowl-house, a well-behaved one this time. It came and took a piece of meat out of my hand quite nicely, which it carried down a hole and then came and sniffed all over my mitt to see if there was any more. I thought it had been killed when I visited the farm buildings next day as there was a strong smell of musk on the cat and in the fowl house, but the weasel was there as cheeky as ever. It got hold of my skirt twice and tried to pull me down its hole. I think it wanted the cloth for a bed, as it was taking straw and other material down the burrow. The poultry were very frightened at first, but they are getting used to the weasel's presence now'."
In commenting on the economic role of the long-tailed weasel in Manitoba, Criddle and Criddle (1925:145) write as follows: "Supply and demand are prominent factors in governing our weasels' food habits. The two smaller species, as we have already pointed out are so dependent upon mice for a living that they increase or diminish with the fluctuation of these creatures. The Long-tailed Weasel, however, is not so easily checked by the temporary disappearance of any particular kind of game. If mice are scarce it devotes greater attention to gophers or bush rabbits and if these are not in sufficient numbers to satisfy its appetite, the animal raids a poultry house as a last resource. In nine years out of ten, this weasel will find sufficient food about the fields and woods, but on the tenth it may be obliged to temporarily turn to domestic animals. It is at such times that the weasel is seen and its deeds recorded. A thousand mice may have been killed in the meantime, but the destruction of half a dozen hens is alone used as evidence of the weasel's economic standing.
"In the last twenty years we have permitted weasels to frequent the farm buildings at will and the poultry house has been no exception. In that time rats and mice suffered severely from the weasels, while the total number of poultry taken were six. Many times that number, however, have been killed by rats.
"When we review our experiences of the past, we are astonished to find what few poultry have been killed by weasels. Our own losses in forty-two years have not exceeded fifteen birds and even these were usually eatable. There have been reports of losses from time to time from neighbors, but on looking into details we find that there are very few farmers who have experienced more than three separate occasions of weasel depredation and the total loss per farmer in the last thirty years does not, we are sure, exceed ten birds. This is surely a remarkably small payment to weasels in general for the great good done by them in killing rodents.
"We wish to point out, too, that only the exceptional weasel becomes a poultry killer. In most cases apparently it is a fully-grown male that does the killing. There are exceptions, of course, but when we see a large weasel actively engaged in rodent hunting within a few feet of a brood of newly hatched chickens and not even looking at them, we must at least pause to ask if this animal is the enemy that we were taught to believe it to be."
A suggestion that weasels sometimes obtain the prey killed by hawks is offered by Criddle and Criddle (1925:147) who write: "Hawks are not always the aggressors, as is shown by an incident reported by Mr. H. L. Seamans, of Lethbridge, Alberta. Mr. Seamans noted a large buzzard suddenly fly straight upwards from a fence post, and then alight upon another one some distance away. A little while afterward this bird once more arose in the same manner as before, and presently repeated the performance again. An investigation then followed and revealed that a Long-tailed Weasel was following the hawk from post to post.
"We should hardly expect a weasel to attempt to capture a bird of the above type. On the other hand, it is possible that these animals might be able to startle a hawk sufficiently to cause it to drop its prey, which would thus provide food for the weasel."
The following frequency index is compiled from the foregoing data on prey of Mustela frenata.
|
|
More significant than the above compilation, of course, are the results of careful studies of the food of the long-tailed weasel in restricted areas. Examples of such studies are those of Polderboer, Kuhn and Hendrickson (1941) and Hamilton (1933:333).
According to Hamilton's (1933:332) observations on captive weasels, "There seems to be little relative difference in the amount they eat, regardless of their activities.
"In general, more food is taken in summer than in winter. Usually about a third their weight every 24 hours is eaten, but a growing young weasel will consume much more. A young male noveboracensis, weighing 145 grams, consumed an entire chipmunk, fur and bones, weighing 85 grams, in 24 hours. A day later it ate all of a partly grown rat, 105 grams, in the same length of time."
Moore (1945:253) records that a captive male that he obtained at Gainesville, Florida, consumed, on the average, between 63 and 70 grams of flesh and blood per day. The weasel itself weighed approximately 320 grams.
Sanderson (1949:413), concerning seven young weasels from Manitoba, that he raised in captivity, writes: "From the fifth to the seventh week of age, they consumed approximately 22 per cent of their body weight per day; from the eighth to the tenth week (just before reaching mature size) they consumed approximately 24 per cent; but after reaching maturity they consumed only 18 per cent. When given all the food they would take in one day, they ate as much as 40 per cent of their body weight."
Criddle and Criddle (1925:143, 146) say that weasels drinking at a bird trough "held their mouths very close to the water and as far as we could see, lapped the liquid up with rapid movements of the tongue. As a rule, after drinking, they would merely spring to the ground and vanish amid a bunch of scolding birds, but occasionally we have seen an animal slowly drag itself through the water and follow this performance by some rapid gambols, or a quick run, a method of drying which most of us have practiced in our youth." According to Hamilton's (1933:332) observations on captives, "Weasels are great drinkers, and while they take but little at a time, about 25 c.c. is drunk by a large animal during a day. . . ."
Reproduction
Philip L. Wright's several papers (1942A, 1942B, 1947, 1948A, and 1948B) reporting on his detailed studies of Mustela frenata (subspecies oribasus and longicauda) in captivity have yielded a large share of the precise information that we have concerning breeding and reproduction in this species. He has found that a single litter, of up to 9 young is born in the spring, usually in April. At three months of age the females "are full grown." The young males remain sexually immature during the first summer but the young females, as well as the females which are more than a year old, come into heat in the midsummer and are bred by the adult males. After a long period of quiescence lasting for several months, the embryos resulting from these matings become active in early spring and develop to full term in less than 27 days after they become implanted. The adult males are sexually active from April into August, when the testes are at maximal size and are conspicuous in the scrotum. A gradual regression takes place starting in August and extending into September. By October the testes may be fully regressed and the molt to white may start in this month. The white winter weasel, of either sex, is sexually inactive. The testes of the sexually active male in early spring and late summer are seven to eight times the size of the fully regressed testes. Females which had borne and suckled young were first found to be in oestrus 65 to 104 days after birth of the young. Lactation lasts for approximately 5 weeks. In 18 litters the length of the gestation period varied from 220 to 337 days with an average of 279 days. The female in heat has the vulva much swollen and she will remain in this condition for several weeks if not bred. Wright (1948A) describes the actual mating as beginning with a scuffle after which the male grabs the female by the scruff of the neck with his teeth and holds her until she becomes subdued when he clasps her lower abdomen with his front feet and arches his back over her posterior regions. The two animals remain locked in this position usually for two hours and sometimes for longer than three hours. If the animals are left together, copulation may take place again on the same day or upon succeeding days.
Hamilton (1933:316-321) writes of a freshly born M. f. noveboracensis that it ". . . was pink and much wrinkled. The wetness . . . did not entirely obscure a few sparse, rather long, white hairs . . . over its back and head. It had the pronounced and extraordinarily long neck of the adult." At one day of age the average weight of six individuals in the litter was 3.1 grams, which is 3 per cent of the weight of the adult female and 1-1/2 per cent of the weight of an adult male. At two weeks of age "The silky white hair . . . obscures the general flesh color of the skin, evident a week earlier. The hair on the back of the head and neck, also over the shoulders, is slightly longer than that of the back . . ." but there is no crest or mane or pompadour at this or any other age such as characterizes the juvenal ermine. When 21 days old one young male "hurried from the nest chamber and commenced to eat some meat." At three and a half weeks "They are all eating small pieces of meat. . . . The canine teeth have made their appearance in both the upper and lower jaw, but just a hint of the incisors show. Some of the cheek teeth are through, as the meat appears to be thoroughly masticated by the little ones." On the 36th and 37th days the eyes opened. Sanderson (1949:415) found that a litter of seven young of Mustela frenata longicauda, from Manitoba, raised in captivity, "reached the peak of their growth" at approximately ten weeks of age.
Several nests have been found. In Manitoba, Sanderson (1949:412) excavated a burrow at the mouth of which he had trapped the adult female and in which he found eight young approximately five weeks old. The "burrow was about three inches in diameter, with two chambers at a depth of twelve inches. One of these was empty, the other contained the young. The two surface-openings were but two feet apart and the entire burrow was no more than three feet long. . . . The meager nest material consisted entirely of finely chopped grass. There was no mouse hair present, no accumulation of fecal material, and no storehouse containing food."
Charles O. Handley has written me that on January 25, 1929, on the Sinkola Plantation, Thomas County, Georgia, he investigated the living quarters of a family of five weasels, four of which had been shot five days before by a hunter. According to the hunter each of the four which had been killed was approximately two-thirds the size of one which escaped into a hole in the ground. Handley found that the weasels had been using as headquarters a burrow in the trunk of an old uprooted oak as well as a nearby gopher burrow. The burrow in the oak was approximately ten feet long and had been excavated in the rotten wood. In a distance of fifty feet along the gopher tunnel there were several used openings with pathways leading away from each. On February 6, Handley, with the help of a friend, trapped a large male weasel near this place.
Criddle and Criddle (1925:143) describe a female which, one winter, slept in a bag of feathers in a basement of a house occupied by one of the authors; another weasel in winter made its headquarters in a threshing machine. The nest of the latter "was somewhat roughly constructed and consisted of a convenient bunch of straw and chaff under the cylinder."
Harper (1927:303) in the Okefinokee Swamp of Georgia dislodged a weasel from the house of a wood rat and was told of a den found in the swamp "in the trunk of a hollow cypress tree" from which a mother weasel and three young "about the size of mice" were obtained. "The bed contained, I suppose, a bushel or more of rabbit hair, rat hair, and squirrel hair. It looked like it must have been used as a den for several years, although there was no stink that I could detect except the musk from the old Weasel." Another female and three young approximately half grown were found in a hollow pine log.
Between January 6 and April 12, 1940, on 640 acres of land, in Washtenaw County, Michigan, four weasels were studied and each weasel used only one den in this period (Quick, 1944:78). Criddle (1930:279) remarks that M. f. longicauda at Aweme, Manitoba, often makes its temporary headquarters in the burrows of pocket gophers (Thomomys). A female and three young weasels were found by Shaw (1921:167) using a nest of a mountain beaver in the burrow of that animal. Green (1936), in May, in Gratiot County, Michigan, saw a weasel enter a hole under a decayed log and investigated finding four young weasels in a nest mostly of Microtus fur.
In the early part (winter and spring) of 1939, at Ames, Iowa, Polderboer, Kuhn and Hendrickson (1941) studied four weasels living in four separate dens on 160 acres typical of Iowa farmland and excavated three of the dens. One den was in a weed patch in an old mole run. The nest chamber, approximately nine inches in diameter and six inches below the surface of the ground "was filled with grasses packed in a layer-like formation. In the center of this mass was a nest hollow lined with patches of mouse and shrew fur. Beneath this layer of fur and at the sides of the nest were skins, various bones, and skulls of partially eaten mice and shrews . . . scats [were in the nest]. . . . At intervals, layers of clean grass had been laid over the filth of the former bed, thus giving the nest a stratified appearance." A second den, of a large male, was in a field of sweet clover two feet high in the former burrow of a Franklin's ground squirrel. The nest cell, seven inches in diameter and nine inches below the surface of the ground, "was lined with grasses mixed with much rabbit and mouse fur. Some scats, and bones and fur of mice and shrews were matted together in layers at the bottom of the nest." When this den was abandoned the male weasel occupied, for a month, another burrow, 20 rods distant, of a Franklin ground squirrel, in the field of sweet clover. The nest cell measured 11 by nine inches and was 11 inches below the surface of the ground. "Two nest layers were present. The first, composed chiefly of coarse straw and grass, had apparently been occupied at some time by a spotted skunk. . . . On top of the skunk nest was the weasel nest composed of fine grasses, mouse fur, and skeletal remains of mice."
Relation of the Sexes to each other and to the young
Quick (1944:75) writes that on March 28, in Michigan, he found the tracks of a male and those of a smaller animal, supposedly a female, meeting. The two "then led along the fence for about 18 chains and both entered the den of the male. . . . Only the tracks of the smaller weasel left the den on the same date. Observation on April 12 showed that the large male still occupied the den." I am at a loss to explain this behavior since breeding would not be expected to occur in late March and since I suppose that the male and female do not live together except in the breeding season. Consequently, I wonder if the sign was wrongly read.
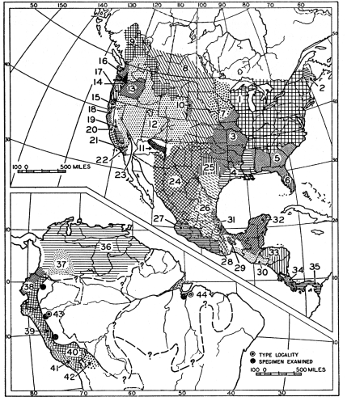
Fig. 29. Map showing the geographic ranges of the subspecies of Mustela frenata and Mustela africana.
| 1. M. f. noveboracensis 2. M. f. occisor 3. M. f. primulina 4. M. f. arthuri 5. M. f. olivacea 6. M. f. peninsulae 7. M. f. spadix 8. M. f. longicauda 9. M. f. oribasus 10. M. f. alleni 11. M. f. arizonensis 12. M. f. nevadensis 13. M. f. effera 14. M. f. washingtoni 15. M. f. saturata 16. M. f. altifrontalis 17. M. f. oregonensis 18. M. f. munda 19. M. f. xanthogenys 20. M. f. nigriauris 21. M. f. latirostra 22. M. f. pulchra |
23. M. f. inyoensis 24. M. f. neomexicana 25. M. f. texensis 26. M. f. frenata 27. M. f. leucoparia 28. M. f. perotae 29. M. f. macrophonius 30. M. f. goldmani 31. M. f. tropicalis 32. M. f. perda 33. M. f. nicaraguae 34. M. f. costaricensis 35. M. f. panamensis 36. M. f. meridana 37. M. f. affinis 38. M. f. aureoventris 39. M. f. helleri 40. M. f. macrura 41. M. f. agilis 42. M. f. boliviensis 43. M. a. africana 44. M. a. stolzmanni |
Hamilton (1933:328), however, writes that M. f. noveboracensis is to "be found in pairs when caring for the young. During mid-May, 1927, I several times saw a male of this species carrying food to a den of young ones." Green (1936), in May in Gratiot County, Michigan, remarks that while he was uncovering and examining a nest of four young weasels, two adults ran about excitedly and one removed a young weasel. In instances where several nearly full-grown young have been obtained from one den it has been my experience (Hall, 1946:191) that the only adult trapped there was the female; no adult male was found or in the one instance when found he was living alone in a den 200 yards away from the den of the female and her young. Data are too few to warrant a definite conclusion about the extent to which the male aids in rearing the young, but I have wondered if he might not do so when the young were less than half grown and then live alone when they were more than half grown.
Mustela frenata noveboracensis (Emmons)
Long-tailed Weasel
Plates 16, 17, 18, 31, 32 and 33
Putorius Noveboracensis Emmons, Quadrupeds of Massachusetts, p. 45, 1840.
Mustela fusca DeKay, Zool. of New York, Pt. 1, Mammalia, p. 34, 1842.
Putorius fuscus Audubon and Bachman, Journ. Acad. Nat. Sci. Philadelphia, 8 (Pt. 2):288, 1842; Audubon and Bachman, Vivip. quadrupeds of N. Amer., 3:234, pl. 148, 1853 (pl. 1848).
Putorius noveboracensis, DeKay, Zool. of New York, Pt. 1, Mammalia, p. 34, 1842; Baird, Mamm. N. Amer., p. 166, 1858; Merriam, N. Amer. Fauna, 11:16, pl. 4, figs. 1, 1a, 2, 2a, pl. 5, figs. 3, 3a, text figs. 4-6, 30, June 30, 1896; Bangs, Proc. Biol. Soc. Washington, 10:13, pl. 1, figs. 2, 2a, pl. 2, figs. 2, 2a, and pl. 3, figs. 3, 3a, February 25, 1896; Cory, Mamm. Illinois and Wisconsin, p. 366, plates, 1912.
Putorius erminea, Audubon and Bachman, Vivip. quadrupeds of N. Amer., 2:56, pl. 59, 1851.
Putorius agilis Audubon and Bachman, Vivip. quadrupeds of N. Amer., 3:184, pl. 140, 1853.
Putorius richardsonii, Baird, Mamm. N. Amer., p. 164, 1858 (part).
Putorius (Gale) erminea, Coues, Fur-bearing animals, p. 109, 1877 (part).
Putorius noveboracensis notius Bangs, Proc. New England Zool. Club, 1:53, June 9, 1899. Type from Weaverville, Buncombe County, North Carolina.
Mustela noveboracensis noveboracensis, Miller, U. S. Nat. Mus. Bull., 79:97, December 12, 1912; Soper, Journ. Mamm., 4:251, November 1, 1923.
Mustela cicognanii, Henninger, Journ. Mamm., 2:239, November 29, 1921; Seton, Lives of game animals, 2:584, 1929 (part, Ohio); Hamilton, Amer. Midland Nat., 14:290, July, 1933 (part, Ohio); Lyon, Amer. Midland Nat., 17:109, January, 1936 (part, Ohio).
Mustela noveboracensis, Jackson, Journ. Mamm., 3:15, February 8, 1922.
Mustela frenata noveboracensis, Hall, Carnegie Instit. Washington Publ., 473:104, November 20, 1936; Hall, Amer. Midland Nat., 18:304, March, 1937.
Type.—Williamstown, Berkshire County, Massachusetts. Type specimen not known to be in existence.
Range.—Altitudinally, sea level to highest parts of mountains of eastern United States; Canadian Life-zone of Ontario and Quebec southward through eastern United States in Canadian, Transition and Upper Austral life-zones to and including upper edge of Lower Austral Life-zone in the Carolinas and northern parts of Georgia, Alabama, and Mississippi; westward from the Atlantic Coast to St. Croix and Mississippi rivers. See figure 29 on page 221.
Characters for ready recognition.—Differs: From M. f. olivacea, in males, by width of tympanic bulla which is less than rather than more than 8.5 mm., and in adult females by total length which is less than rather than more than 345 mm. and by mastoid breadth which is less than rather than more than distance between articular faces of exoccipital condyle and glenoid fossa; from M. f. occisor by a number of average differences including smaller size, relatively shorter tail and relatively narrower skull (see measurements); from M. f. spadix by least width of color of underparts amounting to less than 41 per cent of greatest width of color of upper parts, absence of color of underparts on ankles and feet, adults with hind foot less than 50 in males and 40 in females, orbitonasal length less than 15.5 in males and 13.5 in females, length of tooth-rows less than 18.0 in males and 15.7 in females, mastoid breadth less than 25.5 in males and 22.0 in females; from M. f. primulina in males by interorbital breadth averaging more than 24 per cent of basilar length, orbitonasal length averaging more than 34 per cent of basilar length or 64 per cent of mastoid breadth, tympanic bullae less inflated anteromedially, than posteromedially, and in females by orbitonasal length amounting to more than two-thirds of mastoid breadth, by zygomatic breadth averaging less than 21, and by anterolateral margin of tympanic bullae not projecting below squamosal; from M. f. arthuri in males, by zygomatic breadth more than distance between anterior palatine foramen and anterior margin of tympanic bulla and by convex dorsal outline of skull in longitudinal axis.
Description.—Size.—Male and Female:
| Locality | Number of specimens averaged | Total length | Length of tail | Per cent of body- length | Length of hind foot |
| Massachusetts | 8 ad. ♂ | 415 (390-432) | 146 (127-159) | 54% | 46.0 (41.0-48.0) |
| 4 ad. ♀ | 311 (298-321) | 104 (95-114) | 50% | 33.9 (31.5-37.0) | |
| Liberty Hill, Conn. | 10 ad. ♂ | 411 (379-438) | 141 (124-155) | 52% | 47.1 (43.0-51.5) |
| 6 ad. and sad. ♀ | 318 (303-338) | 105 (80-123) | 49% | 33.0 (31.7-36.0) | |
| Beaver Dam, Wisc. | 10 ad. ♂ | 407 (372-431) | 130 (113-143) | 47% | 46.0 (42.0-50.0) |
| 4 ad. ♀ | 326 (303-338) | 99 (86-108) | 43% | 35.6 (34.6-38.0) | |
| Washtenaw Co., Mich. | 10 ad. ♂ | 371 (350-405) | 130 (115-140) | 54% | 45.0 (40.0-50.0) |
| 10 ad. ♀ | 306 (290-335) | 97 (90-120) | 46% | 34.0 (30.0-40.0) |
The length of the hind foot averages more than the basal length in males whereas the reverse is true in females. The tail, relative to the length of the body, is longer in males than in females. The average differences in external measurements of the two sexes in Massachusetts, are: total length, 104; length of tail, 42; length of hind foot, 12.1. In Michigan, where the males are smaller, corresponding differences are only, 65, 33, and 11. Weight of 19 adult males from New York (Hamilton, 1933:294), 225 (196-267) grams and in 13 adult females, 102 (72-126) grams. Weights of 2 adults from Michigan are: ♂ 258; ♀ 101 grams.
Externals.—Longest facial vibrissae black, brown, or white (often all three colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles as shown in figure 19.
Color.—Upper parts, in summer, Vandyke Brown or darker than tone 4 of Burnt Umber of Oberthür and Dauthenay, pl. 304. Sometimes approaching tone 2 of Warm Sepia of Oberthür and Dauthenay, pl. 305. Underparts, in summer, ranging from white through Napthalene Yellow (Peterboro, N. Y.), Pale Orange Yellow (eastern Mass.), near Primuline Yellow (unusual specimen from Leelanau Co., Mich.) to near (c) Deep Chrome (no. 19053, U. S. Nat. Mus., Roan Mts., N. C.) In winter, all white except tip of tail, or upper parts near (12" 1) Rood's Brown and tone 2 of Raw Umber of Oberthür and Dauthenay, pl. 301, with underparts white or sometimes tinged with yellowish. Tip of tail at all times black. Upper parts of uniform color except for occasional slight darkening of nose. Color of underparts extends distally on posterior sides of forelegs to foot and sometimes over upper sides of toes and on medial sides of hind limbs only to knees. Least width of color of underparts averaging, in a series of twenty-two males, mostly in full winter pelage, from Liberty Hill, Connecticut, 21 (11-40) per cent of greatest width of color of underparts. In eleven females from the same place, corresponding percentages are 20 (14-29). Black tip of tail in same series of males, most of which are in full winter pelage, 70 (60-75) mm. long; thus longer than hind foot and averaging 50 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on ten adults from Massachusetts): See measurements and plates 16-18; weight, 3.6 (3.3-4.4) grams; basilar length, 44.6 (43.3-46.0); zygomatic breadth less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth less than postpalatal length; postorbital breadth more or less than length of upper premolars and greater than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less (usually more) than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate less than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 3 to 6 upper incisors; height of tympanic bulla more or less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum; anterior margin of masseteric fossa behind or directly below posterior fourth of m1.
Female (based on five adults from Mass.): See measurements and plates 31-33; weight, 1.7 (1.2-2.1) grams; basilar length, 36.5 (35.2-38.1); zygomatic breadth less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth more or less than length of upper premolars and more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate more or less (usually less) than greatest length of P4; tympanic bulla as far posterior to foramen ovale as width of 4 to 5-1/2 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer than rostrum.
The skull of the female averages 53 per cent lighter than that of the average male.
Comparisons of the skull with those of M. f. olivacea, M. f. spadix, M. f. primulina, and M. f. arthuri, are made in the accounts of those subspecies. As compared with that of M. f. occisor the skull of adult male noveboracensis, is of smaller average size with relatively (to basilar length of Hensel) lesser mastoid and zygomatic breadths. In addition to the zygomatic arches of noveboracensis being less widely bowed outward they seem to be more rounded posteriorly. Comparisons of subadult females indicate that these differences exist in the females as well as in the adult males.
Remarks.—The earliest of the post-Linnaean references to this weasel mostly were under the specific name erminea in the belief that the American animal was the same as the larger of the two common species of weasel in the Old World. The name noveboracensis, now in use for this subspecies, was applied in 1840 and since that time the males usually have borne that name; the females, because they are smaller, were more frequently confused with some other species. Audubon and Bachman in 1853 even proposed the name agilis for the female in the mistaken belief that it was a species distinct from the male. After 1896, when Bangs correctly classified the weasels of the eastern United States, the males have been correctly identified and the females, except by a few authors, likewise have been correctly named. Because many early American naturalists did their first collecting of mammals in the geographic range of noveboracensis, the person who examines labels of specimens of this subspecies can find data written in the hand of Spencer Fullerton Baird, Theodore Roosevelt, and other naturalists famous for their work as scientists or accomplishments otherwise. The material is more nearly adequate than is that of many other subspecies and the number of specimens is exceeded—and only slightly—by that of the subspecies nevadensis, which like noveboracensis has a relatively large geographic range.
Intergradation with Mustela frenata spadix is indicated by subadult males from western Wisconsin, namely, one from Gordon, three from Colfax and one from Meridean. Linear measurements of the teeth of these specimens are exactly intermediate between those of spadix from Elk River, Minnesota, to the west, and noveboracensis from, say, Beaver Dam, Wisconsin, to the east. The specimens from western Wisconsin show approach to spadix also in that the length of the tooth-rows and breadth of the rostrum are slightly greater than in noveboracensis from farther east, say, Beaver Dam, Wisconsin.
Indeed, animals from as far east as Beaver Dam itself might be thought of as showing some approach to spadix. Although, along the eastern seaboard, the upper lips, with rare exceptions, are the same color as the underparts, farther west, in Michigan and Wisconsin, the lips more often than not are white. Animals from Beaver Dam have slightly shorter black tips on the tails, broader extent of the light color of the underparts and females average slightly larger than typical noveboracensis, say, those from Massachusetts. Each of these differences reflects characters found better developed in the spadix-longicauda stock to the west.
Toward the southern part of its range where noveboracensis meets M. f. olivacea there is a marked increase in yellowness of the underparts. This coloration of the underparts, since it is not so well marked in the northern part of the range of noveboracensis, might be regarded as showing intergradation with olivacea and primulina, each of which has far more intensely colored underparts than does noveboracensis. Excepting this increase of yellow on the underparts, however, there are few if any characters of noveboracensis which undergo marked change as approach to the range of olivacea is made. Indeed, the characters of noveboracensis remain constant to within a relatively short distance of the geographic range of olivacea.
Notwithstanding the state of affairs described above, intergradation seems to take place. Three specimens referred to noveboracensis but which at the same time are regarded as intergrades with olivacea are as follows: No. 28.300, Charleston Museum, from five miles east of York, South Carolina, is an adult female with a badly crushed skull. In external measurements the specimen agrees with noveboracensis. The underparts, as regards color and width, are intermediate. The general proportions of the skull and tympanic bullae agree with those of noveboracensis but the skull is larger than in any female of true noveboracensis and approaches that of olivacea. The same can be said of a young female, no. 80, Ohio State Museum, from Roswell, Georgia.
Another female, no. 171559, U. S. Nat. Mus., from Lookout Mountain, 1500 ft., Fort Payne, Alabama, is barely subadult. The external measurements are nearer those of olivacea. The color and narrowness of the underparts are typical of noveboracensis. The proportions and especially size of the skull show approach to olivacea, though they are nearer to noveboracensis when all features are taken into account. In the northern part of its range individuals of noveboracensis attain larger size than farther south. This tendency reaches its extreme, in males at least, in M. f. occisor of Maine. Specimens of noveboracensis from the Adirondacks of New York average larger (see cranial measurements on page 418) than those from farther south, and thus approach occisor in size as well as in geographic position. Also, occasional individuals which strongly show characters of occisor are found even farther south than the Adirondacks of New York. This is true of no. 96518, U. S. Nat. Mus., ♂ ad., from Lunenburg; Massachusetts. The animal has a large skull of relatively great width much as in occisor, although its external measurements, relative length of tail and long, terminal, black brush place it with noveboracensis rather than with occisor. Of a pair of specimens from Ossipee, New Hampshire, the male, no. 77108, U. S. Nat. Mus., has a long (175 mm.) tail, and short (60 mm.) black pencil as in occisor, although otherwise it is referable to noveboracensis. Still another specimen, a subadult male, no. 4193, Mus. Comp. Zoöl., from Upton, Maine, has a longer (51 mm.) hind foot than noveboracensis although it otherwise agrees with that subspecies. As remarked by Bangs (1899:55), other than fully adult specimens from the range of occisor are "troublesome," and would not be selected as distinct from noveboracensis if placed in a series of that subspecies, say, from New York State. In view of the facts that several specimens from intermediate localities combine the characters of noveboracensis and occisor, that noveboracensis in the northern part of its range averages larger than it does farther south and thus approaches occisor in size, and that occasional large specimens resembling occisor in several, but not all, features sometimes crop up in the northern part of the range of noveboracensis, it appears that noveboracensis and occisor intergrade. Therefore they are treated as two subspecies of the single species, Mustela frenata.
Intergradation with M. f. primulina has been commented on in the discussion of that subspecies. Female, no. 159980, U. S. Nat. Mus., from Golconda, Illinois, has many characters of primulina but two young males from there agree better with noveboracensis.
Examination of 283 adult and subadult skulls for malformation of the frontal sinuses revealed only ten that were not obviously malformed. Two were from New York, one from Massachusetts, one from Pennsylvania, and six from the 52 specimens from Michigan and Wisconsin. In addition, skulls of many young and even juveniles were malformed.
Specimens examined.—Total number, 555, arranged alphabetically by states and provinces and, unless otherwise noted, from north to south by counties in each state. Except as otherwise noted specimens are in the United States National Museum.
Alabama. DeKalb County: Fort Payne, 1.
Connecticut. Litchfield County: Riverton, 1[5]; Gaylordsville, 1. Hartford County: East Hartford, 4 (3[5]); Glastonbury, 2[5]; South Glastonbury, 4[5]. Windham County: Plainfield, 2 (1[14]). Fairfield County: Greenwich, 2[2]. New London County: Liberty Hill, 35 (33[75], 2[7]).
District of Columbia. Washington, 3; near Washington, 1; Eastern Branch, 1; Congress Heights, 1; Benning, 1; no definite locality, 1.
Georgia. Towns County: Young Harris, 1. Cherokee County: Canton, 1. Cobb County: Roswell, 1[81].
Indiana. St. Joseph County: Notre Dame, 2[99]. Porter County: Hebron, 1. Miami County: Denver, 5 (4[75], 1[4]). Wells County: Bluffton, 1. Howard County: Russiaville, 1. Jay County: Salamonia, 1[2]. Boone County, 1[2]. Knox County: Bicknell, 3.
Illinois. Lake County: Camp Logan, 3[60]; Fort Sheridan, 1[60]. Cook County: W Northfield, 2; Flossmoor, 1[60]; no locality more definite than county, 1. Du Page County: Bloomingdale Spg., 1[60]. Carroll County: Savanna, 1[87]. McLean County: Normal, 1[7]. Champaign County: Harwood Township, 1[7]. Pike County?: Milton Spring, 1[60]. Pope County: Golconda, 3.
Kentucky. Woodford County: Midway, 1. Hancock County: Hawesville, 1.
Maine. Oxford County: Upton, 1[75]; Bethel, 1[74].
Maryland. Howard County: Long Corner, 1; Hanover, 1. Montgomery County: Gaithersburg, 1; Garret Park, 1; Chevy Chase, 1; Bethesda, 1. Prince Georges County: Laurel, 18; Plummer Island, 3; Oxon Hill, 1. Talbot County: Easton, 1. Dorchester County: Cambridge, 5[40].
Massachusetts. Middlesex County: Wilmington, 6; Burlington, 6; Lexington, 1[75]; Wayland, 2[75]. Berkshire County: New Marlboro, 1[5]. Worcester County: Lunenburg, 2; Lancaster, 1[75]; Princeton, 2[75]. Norfolk County: So. Weymouth, 1[75]. Plymouth County: Wareham, 5[75].
Michigan. Marquette County: Michigamme, 1. Charlevoix County: Thumb Lake, 1[76]; 1/2 mi. N Thumb Lake, 1[76]. Leelanau County: Leland, 3[76]; Duck Lake, 2 mi. S Leland, 1[76]; Lost Pond, 8-1/2 mi. S Leland, 1[76]. Osceola County: Le Roy, 2[76]. Huron County: Rush Lake, 1[76]. Saginaw County: East Saginaw, 1. Oakland County: Royal Oak, 4[76]; South Lyon, 1[76]. Livingston County: Portage Lake, 1[76]. Washtenaw County: Portage Lake, 6[76]; Waterloo, 2[14]; Lima, 1[76]; Ann Arbor, 11[76]; 3 mi. E Ann Arbor, 1[76]; 2 mi. SE Ann Arbor, 1[76]; 2 mi. S Ann Arbor, 1[76]; 3 mi. S Ann Arbor, 1[76]; Dixboro, 1[76]; Pittsfield, 3 (2[76]); Saline, 1[76]; near Saline, 2[76]; 1 mi. S Saline, 2[76]; York, 2[76]; Manchester, 2[76]. Lenawee County: Morenci, 1[76]. Cass County: Marcellus Township, 1[76]. Berrien County: Harbert, 1[76]; Warren Wood Preserve, 1[76]; Warren Woods, 1[76].
New Hampshire. Grafton County: Franconia, 1[2]. Carroll County: South Chatham, 4 (3[5]); Ossipee, 2; Intervale, 1[5]. Merrimack County: Webster, 2[75].
New Jersey. Morris County: Morristown, 1. Essex County: West Orange, 1[2]. Mercer County: Princeton, 1[1]. Ocean County: Point Pleasant, 1[2]. Camden County: Haddonfield, 1[1]. Cumberland County: Millville, 2[74].
New York. St. Lawrence County: Ogdensburg, 1[74]. Clinton County: Rouses Point, 1[80]. County?: Adirondacks, 12. Essex County: Elizabethtown, 1; Schroon Lake, 1; no locality more definite than county, 1. Lewis? County: Locust Grove, 4; Lyons Falls, 1. Warren County: Lake George, 6; Caldwell, 1. Hamilton County: Beaver Brook, 1/2 mi. above mouth Indian Lake, 1[80]. Oswego County: Scriba, 2[74]; Palermo, 1[74]. Monroe County: Penfield, 3. Madison County: Peterboro, 6 (2[75]). Schoharie County: Schoharie, 1[2]. Rensselaer County: East Shodack, 1[80]. Tompkins County: Taughannock Falls, 2[58]; Ithaca, 4 (3[58]); Glenside, Ithaca, 1[58]; 6 mi. Creek, Ithaca, 1[58]. Green County: Lanesville, 1[2]. Orange County: Poplopen's Pond, 1[2]; Highland Falls, 1[2]. Putnam County, 1[19]. Westchester County: Sing Sing, 4; Armonk, 1[2]; Hastings, 3 (2[2], 1[19]). Nassau County: Flushing Meadows, 1[2]; Flushing, 1[58]; near Flushing, 1[2]; Oyster Bay, 2. Long Island: Cold Spring Harbor, 1; Bridgehampton, 1[2]. County in question: Severance, 3; Lake Grove (Long Island?), 1.
North Carolina (east to west by counties). Wake County: Raleigh, 4 (1[2], 1[75], 2[76]). Mitchell County: Magnetic City, foot of Roan Mountain, 6; Roan Mt., 1; Roan Mt., 6000 ft., 3. Buncombe County?: Valley of Black Mts., 1[2]. Madison County, 2[11].
Ohio. Trumbull County: Warren, 1[93]. Seneca County: Tiffin, 1[81]. Summit County: Ira, 2[81]. Crawford County: Galion, 1[81]. Ashland County: Loudonville, 1[76]. Auglaize County: New Bremen, 3[81]. Franklin County: 3 mi. N Columbus, 1[81]; Minerva Park, Columbus, 5[81]. Fairfield County: Sec. 32, Pleasant Twp., 1[81]; Lancaster, 1[81]. Clinton County: Reesville, 1; 1/2 mi. S and 1/2 mi. W Wilmington, 2[74]. Pike County: Waverly, 1[81].
Ontario. Sudbury District: Metagama, 2[86]. Carleton County: Ottawa, 2[77]. Muskoka County: Lake of Bays, 1; Bracebridge, 1. Haliburton County: Gooderham, 1[60]. Simcoe County: Orillia, 4 (2[2], 2[60].) Prince Edward County: Bloomfield, 1[77]. York County: Toronto, 1[2]. Waterloo County: Branchton, 3[60]; Preston, 2[77]; no locality save county, 1[60]. Welland County: Ridgway, 1[14]. Elgin County: St. Thomas, 1[77]. Essex County: Kingsville, 1[77]; Point Pelee, 1[77].
Pennsylvania (east to west by counties). Crawford County: Pymatuning Swamp, 3-1/2 mi. W Linesville, 1[9]; Meadville, 2[9]. Beaver County: Beaver, 1[9]; Raccoon Creek, 1[9]. Butler County: Mars, 1[9]; Leasuresville, 4[9]. Allegheny County: Allegheny, 1. Warren County: Bear Lake, 2[9]. Westmoreland County: Kingston, 1[9]; Laughlinstown, 2[9]. Somerset County: Confluence, 1[9]; Tub Mill Run, 2 mi. N Springs, 1[9]. Jefferson County: Siegel, 1[9]. Clearfield? County: Penfield, 1[9]. Cambria County: Cresson, 1[9]. Fulton County: Well's Tannery, 1[9]. Clinton County: near Round Island, 2[1]. Cumberland County: Carlisle, 1. Snyder County: 5 mi. S Selinsgrove, 1. Northumberland County: Pottsgrove, 1. Union County: Mifflinburg, 1. Sullivan County: Lopez, 7 (4[1], 3[74]). Chester County: Westtown, 1[1]; Valley Forge, 1[1]; W Bradford Township, 1[1]; no locality more definite than county, 3. Philadelphia County: Holmsburg, 2[1]. Bucks County: 1[1]. Pike County: Milford, 1.
Rhode Island. Providence County: Chepachet, 1. Washington County: Lake Warden, 2.
Quebec. Megantic County: Black Lake 1[77]. County in question: Meach Lake, 1[77].
South Carolina. York County: 5 mi. E York, 1[11]. Laurens County: Laurens, 1[39].
Tennessee. Campbell County: Highcliff, 1. Carter? County: Roan Mts., 1[2]. Hamilton County: Walden Ridge, near Soddy, 3.
Vermont. Windsor County: Hartland, 1[2].
Virginia. Shenandoah County: Toms Brook, 1. Arlington County: Arlington, 1; Ballston, 1; Alexandria, 1. Fairfax County: Falls Church, 3; Mt. Vernon, 2; no locality more definite than county, 1. Prince William County: Occoquan, 1. Essex County: Montague, 1. Prince George County, 1. Norfolk County: Wallaceton, 1. Grayson County: Mt Rogers, 3. County in question: Dismal Swamp, 1; Massanutten Mt., 1.
West Virginia. Hardy County, 1. Pendleton County: radius of 2 mi. Smoke Hole, 1[74]. Greenbriar County: White Sulphur, 2[60].
Wisconsin. Douglas County: Gordon, 1[102]. Vilas County: Mamie Lake, 4. Dunn County: Colfax, 4[102]; Meridean, 1[102]. Door County: state game farm, 17[102]; no locality more definite than county, 1[102]. Dodge County: Rolling Prairie, 1[50]; Beaver Dam, 52[50]. Dane County: Wingra Lake, 1[102]. Waukesha County: Pewaukee, 2[102]. Racine County: Waterford Township, 2[102]. Rock County: Milton, 1[102]; Bowers Lake, 1[102]. Walworth County: Lane's Mill, 8 mi. N Elkhorn, 7 (1[102], 6[54]); Delavan, 7.
Mustela frenata occisor (Bangs)
Long-tailed Weasel
Plates 16, 17, 18, 31, 32 and 33
Putorius occisor Bangs, Proc. New England Zool. Club, 1:54, June 9, 1899.
Mustela occisor, Miller, U. S. Nat. Mus. Bull., 79:98, December 31, 1912.
Mustela frenata occisor, Hall, Carnegie Instit. Washington Publ. 473:104, November 20, 1936.
Type.—Male, adult, skull and skin; no. 9102, coll. of E. A. and O. Bangs in Mus. Comp. Zoöl.; Bucksport, Hancock County, Maine; January 15, 1899; obtained by Alvah G. Door but measured and sexed by O. Bangs.
The skin is well made and in good condition. It is in full, white winter-dress with black-tipped tail. The skull has the posterior half of the left zygomatic arch broken away; otherwise the skull is unbroken and complete. Left I3 and right P3 are missing. The teeth otherwise all are present and entire.
Range.—Maine; possibly north locally to south side of St. Lawrence River in Quebec and possibly occurring in western New Brunswick. Zonal range Canadian and probably Transition. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. noveboracensis by a number of average differences including larger size, relatively longer tail and relatively wider skull (see page 225, and measurements on pages 418, 419).
Description.—Size.—Male: Five adults yield average and extreme measurements as follows: Total length, 443 (430-465); length of tail, 163 (154-175); length of hind foot, 50 (47-54). Tail averages 58 per cent as long as head and body. Length of hind foot averages more than basal length.
Female: Measurements of two subadult female topotypes are as follows: Total length, 346, 318; length of tail, 116, 110; length of hind foot, 39, 35.5.
Tail amounts to 50 per cent and 54 per cent of body-length respectively. Length of hind foot more or less than (about equal to) basal length.
The average differences in external measurements of the two sexes are: Total length, 111; length of tail, 50; length of hind foot, 12.5.
Externals.—As described in Mustela frenata noveboracensis.
Color.—As described in Mustela frenata noveboracensis except that black tail-tip in series of 10 males in full winter pelage 60 (45-80) mm. long; thus averaging 39 per cent of length of tail vertebrae.
Skull and teeth.—Male (based on 3 adults): See measurements and plates 16-18. As described in Mustela frenata noveboracensis except that: Weight, 4.2 (4.1-4.3) grams; basilar length, 45.7 (44.9-46.9); zygomatic breadth more or less than (about equal to) distance between anterior palatine foramen and anterior margin of tympanic bulla; least width of palate rarely less than length of P4; anterior margin of masseteric fossa behind or directly below posterior half of m2.
Female (based on 2 subadults): See measurements and plates 31-33. As described in Mustela frenata noveboracensis except that: Weight, 2.0 (1.9-2.1) grams; basilar length, 37.3, 38.2.
Comparison of the skull with that of M. f. noveboracensis is made in the account of that subspecies.
Remarks.—Excepting a specimen in the Academy of Natural Sciences of Philadelphia, obtained in 1893, and two in the Boston Society of Natural History, obtained in 1925, I have seen no material of this subspecies in addition to that examined by Bangs at the time he prepared the original description in 1899.
Anderson (1945:56, 57) records a specimen, Canadian National Museum Catalogue Number 18426, from Kamouraska County, Quebec, as of this subspecies and thinks that occisor occurs north of Maine "locally to south side of lower St. Lawrence River in Quebec; probably also in western New Brunswick."
So far as the available material of occisor permits one to judge, it is distinguished from noveboracensis by a combination of characters no one of which invariably can be relied upon as diagnostic. Employing adult males, average differences indicate that M. f. occisor is larger in each of the external and cranial measurements; tail relatively longer; black tip of tail relatively shorter; mastoid and zygomatic breadth relatively greater and zygomatic arches more nearly square posteriorly.
Considering the large number of specimens of noveboracensis which are available in comparison with the few of occisor it is not surprising that some noveboracensis should be found which exceed in size those of occisor. This is the case as regards the basilar length of a very old male, no. 96518, U. S. Nat. Mus., from Lunenburg, Massachusetts. Also, the skull is actually broader than any of those of occisor. However, this specimen is much older than any occisor examined. In a female, no. 4260, Mus. Comp. Zoöl., from Liberty Hill, Connecticut, the skull is longer (but narrower) than in either of the two available females of occisor.
The average differences pointed out above which characterize this extreme northern population of noveboracensis-like weasel in comparison with true noveboracensis without much question are geographic variations. Whether or not these variations are of a degree sufficient to warrant nomenclatural recognition is debatable. With equally scanty material from other regions I have not named variations seemingly as great as those shown by occisor. The paucity of material of occisor is a handicap in making a decision in this instance.
Each of the adult and subadult specimens, except the one from Perry, shows malformation resulting from the infestation of the frontal sinuses with parasites.
Specimens examined.—Total number, 18, listed by counties from west to east and unless otherwise indicated in the Museum of Comparative Zoölogy.
Maine. Oxford County: South Andover, 2 (Boston Soc. Nat. Hist.); Umbagog Lake, 1. Franklin County: Seven Pd. Township, 1. Piscataquis County: Grenville, [= Greenville?], 1. Hancock County: Bucksport, 10. Washington County: 3rd Mopang Lake, 1 (Acad. Nat. Sci. Phila.); Perry, 1 (Boston Soc. Nat. Hist.). County in question: Moosehead Lake, 1.
Mustela frenata primulina Jackson
Long-tailed Weasel
Plates 16, 17, 18, 31, 32 and 33
Mustela primulina Jackson, Proc. Biol. Soc. Washington, 26:123, May 21, 1913.
Putorius noveboracensis, Baird, Mamm. N. Amer., p. 166, 1858 (part).
Mustela longicauda longicauda, Dice, Journ. Mamm., 4:108, May 9, 1923.
Mustela longicauda primulina, Linsdale, Journ. Mamm., 9:141, May 9, 1928.
Mustela frenata primulina, Hall, Carnegie Instit. Washington Publ. 473:104, November 20, 1936.
Mustela frenata, Leopold and Hall, Journ. Mamm., 26:143, July 19, 1945.
Type.—Male?, young, skull and skin; no. 168006, U. S. Nat. Mus., Biol. Surv. Coll.; 5 miles northeast of Avilla, Jasper County, Missouri; May 11, 1905; obtained by Hartley H. T. Jackson; original no. 7869X.
The skin lacks the distal part of the tail—the part which bears the black tip. Otherwise the skin is complete and well preserved. The teeth of the permanent dentition all are present and entire. The lower jaws are complete and unbroken. The skull is broken transversely through the interorbital region, transversely through the braincase and longitudinally through the basioccipital. Both zygomatic arches are gone. The type is judged to be a male rather than a female as stated by the original describer, Jackson (1913:123), whose measurements of hind foot, interorbital constriction, maxillary tooth-row, and mandibular tooth-row agree with those of males and are larger than those of any female seen of this subspecies.
Range.—Upper and Lower Austral life-zones west of the Mississippi River in Missouri and Arkansas, the southeastern half of Iowa, eastern half of Kansas and Oklahoma, northern Louisiana and northeastern Texas. Southern and southwestern limits of range undetermined. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. noveboracensis in males by interorbital breadth averaging less than 24 per cent of basilar length, orbitonasal length averaging less than 34 per cent of basilar length or 64 per cent of mastoid breadth, tympanic bullae as much inflated anteromedially as posteromedially, and in females by orbitonasal length amounting to less than two-thirds of mastoid breadth, by zygomatic breadth averaging more than 21 mm., and by anterolateral margin of tympanic bulla projecting below squamosal; from M. f. spadix by least width of color of under parts amounting to less than 40 per cent of greatest width of color of upper parts, by absence of color of underparts on hind leg below knee, and by smaller size (hind foot less than 50 in males and 40 in females; orbitonasal length less than 15.5 in males and 13.5 in females; length of tooth-rows less than 18 in males and 15.7 in females; mastoid breadth less than 25.5 in males and 22 in females); from M. f. longicauda by Brussels Brown rather than near (h) Clay Color of upper parts, least width of underparts less than 40 per cent of greatest width of color of upper parts, absence of color of underparts on hind leg below knee, zygomatic breadth less than 28.8 in males and 24.1 in females; from M. f. neomexicana by Brussels Brown rather than Buckthorn Brown color of upper parts, in absence of white frontal spot and broad white bands on sides of head, in anterolaterally rounded, rather than "square," tympanic bullae and in zygomatic breadth of less than 30 in males and 24 in females; from M. f. frenata and M. f. texensis by absence of white facial markings and smaller size (basilar length of adult males less than 47, tail-length less than 155 in males, and hind foot less than 40 in females); from M. f. arthuri by less evenly spreading zygomatic arches (see pls. 16, 17 and 18), greater inflation of the tympanic bullae anteromedially and more nearly straight (less convex) dorsal outline of skull.
Description.—Size.—Male: Eighteen adults and subadults from Douglas County, Kansas, yield average and extreme measurements as follows: Total length, 397 (371-440); length of tail, 133 (120-147); length of hind foot, 43 (40-47). Tail averages 50 per cent as long as head and body. Length of hind foot averages less than basal length. Corresponding measurements, originally taken in inches and fractions thereof, of 9 adults and subadults from Boone County, Arkansas, are as follows: 413 (384-438); 138 (127-155); 41 (38-44).
Female: Six adults and subadults from Douglas County, Kansas, yield average and extreme measurements as follows: Total length, 339 (317-355); length of tail, 107 (95-115); length of hind foot, 35 (34-37). Tail averages 46 per cent of length of head and body. Length of hind foot less than basal length. Corresponding measurements, originally taken in inches and fractions thereof, of 5 adults and subadults from Boone County, Arkansas, are as follows: 355 (332-397); 113 (101-127); 33 (29-38).
The average differences in external measurements of the two sexes, in Douglas County, Kansas, are: Total length, 58; length of tail, 26; length of hind foot, 8. An adult male from Boone Co., Iowa, weighed 293 grams.
Externals.—Longest facial vibrissae black or dark brown (often both colors in same specimen) and extending beyond ear; carpal vibrissae colored either like underparts or upper parts and extending to apical pad of fifth digit; hairiness of foot-soles as shown in figure 20.
Color.—Upper parts, in summer, Brussels Brown to near (14 n) Brussels Brown, or tones 2 to 4 of Raw Umber of Oberthür and Dauthenay, pl. 301. Chin and rarely upper lips white. Remainder of underparts Picric Yellow to Primuline Yellow. In winter, color essentially the same except for smoke-gray effect in upper parts and more whitish in underparts. Tip of tail at all times black. Upper parts of uniform color except for occasional darkening of nose and mid-dorsal region. Color of underparts extends distally on posterior sides of forelegs onto antipalmar faces of toes, on medial sides of hind legs only to a point between knee and ankle. Least width of color of underparts averaging, in a series of 21 males from Lawrence, Kansas, 23 (9-35) per cent of greatest width of color of upper parts. Black tip of tail in same series, most of which are in full winter pelage, 52 (40-70) mm. long; thus longer than hind foot and averaging 39 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on ten adults from Douglas County, Kansas): See measurements and plates 16-18; weight, 3.7 (3.3-4.2) grams; basilar length, 44.8 (43.8-46.0); zygomatic breadth more or less (less in 80 per cent) than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior end of tympanic bulla (less in 70 per cent); mastoid breadth more or less (less in 80 per cent) than postpalatal length; postorbital breadth less than length of upper premolars and, except in one specimen, more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less (less in 70 per cent) than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate less than outside length of P4 (except in one specimen); anterior margin of tympanic bulla as far posterior to foramen ovale as width of 2-1/2 to 5 upper incisors; height of tympanic bulla more (except in one specimen) than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer (except in one specimen) than rostrum; anterior margin of masseteric fossa behind or just below posterior eighth of m1.
Female (based on 5 adults and subadults from Douglas County, Kansas): See measurements and plates 31-33; weight, 2.2 (2.0-2.4) grams; basilar length, 38.9 (37.6-40.7); zygomatic breadth less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth less than length of upper premolars and except in one specimen, more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate less than greatest length of P4; tympanic bulla as far posterior to foramen ovale as width of 3 to 5 upper incisors; height of tympanic bulla more or less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer than rostrum.
The skull of the female averages 41 per cent lighter than that of the male.
Compared with the skull of M. f. noveboracensis from Massachusetts, that of the male of primulina, in dorsal view, is seen to be shorter anteriorly to the postorbital processes and to have a more marked postorbital constriction. In lateral view the dorsal outline of the skull of primulina is less concave in the postorbital region. In ventral view the skull of primulina is seen to be wider across the mastoid processes and zygomatic arches but the most pronounced difference is in the tympanic bullae. In noveboracensis each bulla is scooped out on the anterior part of the medial face and appears to be narrower anteriorly than posteriorly whereas in primulina the anterior part of the medial face is not scooped out but is moderately inflated and the bulla appears to be of uniform breadth anteriorly and posteriorly. By actual measurement the breadth of the bulla averages 59 per cent of its length in primulina but only 50 per cent in noveboracensis. Other respects in which the skull of the male of primulina differs from that of noveboracensis are as follows: Linear measurements of teeth more; relative to the basilar length, the length of the tooth-rows averages more, whereas the interorbital breadth and orbitonasal length are less.
When skulls of females are compared, each of the differences mentioned above is found to apply, except that the degree of difference is in some parts greater, for example, in the tympanic bullae. In primulina, the bulla is in general like that of the male noveboracensis, whereas in the female noveboracensis it is less inflated, especially anteromedially, shorter, relatively narrower, and in ventral view projects little or none below the squamosal floor of the braincase. The breadth of the bulla averages 51 per cent of its length in primulina but only 47 per cent in noveboracensis. The bullae project below the basioccipital on the average, for a distance of 2.9 millimeters in female primulina and only 2.3 millimeters in female noveboracensis. In primulina the temporal ridges are well developed and fuse to form a low sagittal crest, but in noveboracensis the ridges are absent. Also, in primulina the mastoid processes project farther laterally beyond the braincase. The skull of female noveboracensis is much lighter than that of primulina. Average weights of the two are 1.7 and 2.2 grams. The skulls of females of primulina and noveboracensis differ more than do the skulls of males.
Compared with the skull of spadix, that of the male, and the female, of primulina averages smaller in every part measured. Expressed in percentages of the basilar length, the two depth measurements of the skulls are not significantly different, but, excluding the measurements of the bullae and teeth, the other cranial measurements are less. The main difference in relative proportions is in the tympanic bullae which average only a half millimeter shorter in males of primulina and one and one-tenth millimeters shorter in females. The bullae are, therefore, relative to the basilar length, longer in primulina. The skull of primulina, then, differs from that of spadix mainly in smaller size and relatively longer tympanic bullae, especially in males.
Compared with the skull of M. f. longicauda, that of both sexes of primulina averages smaller in every part measured, except in males where the length of the tympanic bulla, and breadth and length of M1 are the same or slightly larger in primulina. Relative to the basilar length, the length of the tympanic bullae, and in females only, the depth measurements are greater in primulina but all the others, in both sexes, are less. These ratios reflect the relative narrowness of the skull of primulina. Upon direct comparison the narrowness is especially noticeable in the interorbital region, mastoid region, tympanic bullae, and across the zygomata.
Compared with the skull of M. f. neomexicana that of both sexes of primulina averages smaller in every part measured. Excepting the measurements of the teeth, most of the other measurements are constantly larger. Relative to the basilar length, the length of tooth-rows and length of tympanic bulla are more, but excepting the depth measurements, the others are less. Still other differences are, in primulina, less well-developed sagittal crest, anterolateral corner of bulla rounded rather than "square," and in males a transversely convex rather than flat interorbital region.
Compared with M. f. frenata and M. f. texensis, the skulls of males of primulina differ in being smaller in every part measured but relative to the basilar length, have longer tooth-rows, a lesser zygomatic breadth and are less constricted interorbitally.
Compared with the skull of M. f. olivacea, those of both sexes of primulina average smaller in every part measured, have shallower (dorsoventrally) tympanic bullae, a lower sagittal crest and slightly weaker postorbital processes on the frontals. Relative to the basilar length, the several cranial measurements are about the same.
Comparison of the skull with that of M. f. arthuri has been made in the account of that subspecies.
Remarks.—The first specimens of this race known to have been preserved in study collections are one in the United States National Museum, taken at Bridge, Carroll County, Missouri, many years ago by J. Burbage, and less than a dozen specimens preserved before 1900 from eastern Kansas in the University of Kansas Museum of Natural History. In 1913 Hartley H. T. Jackson bestowed a name on this animal on the basis of two specimens taken by him in southwestern Missouri. Later, through the efforts of Charles D. Bunker, and his associates at the University of Kansas, nearly 100 specimens were saved from eastern Kansas, principally from Douglas County. In the course of the present study, Lawrence V. Compton obtained a topotype for the California Museum of Vertebrate Zoölogy, and with the assistance of Mr. B. G. Roberts, a good series of specimens from Boone County, Arkansas, was preserved in the same museum. In the early years of the 20th Century, the late B. H. Bailey at Coe College, Iowa, collected specimens from that state. The specimens from these several sources suffice to give a relatively clear idea of the characters of this subspecies.
Mustela frenata primulina is closely related to M. f. noveboracensis, from which, on the average, it differs in the lighter color of the upper parts of the summer coat, in the more intense coloring of the underparts, and in certain cranial features pointed out above. In the southern part of its range, however, noveboracensis has the underparts only a little less intensely colored with yellow than primulina. Also, the skull of the one topotype from 7-1/2 miles southeast of Carthage, a subadult male in brown, winter pelage, is almost exactly intermediate between that of noveboracensis from Massachusetts and primulina of Douglas County, Kansas, and Boone County, Arkansas. M. f. primulina often has the underparts white in winter, as does this topotype which agrees with the average of noveboracensis in small size of teeth and narrowness across the mastoid processes and zygomatic arches. However, it agrees with primulina in shape and relative size of the rostrum. It is almost exactly intermediate in shape and width of the tympanic bullae.
Three other males, but no females, all in winter pelage, are available from eastern Missouri. Of the two from Silex, Lincoln County, one is nearer noveboracensis and the other nearer primulina on the basis of cranial characters. The third specimen, from four miles south of Lesterville, so far as I can determine by examination of individual cranial characters and tabulation of results, is exactly intermediate. Final decision on the proper allocation of specimens from the parts of Missouri represented can best be made when skulls of females are available. From the fact that the skull of the female referred to noveboracensis from Golconda, Illinois, shows almost as many characters of primulina as of noveboracensis, it is judged that females from as far west as Silex and Lesterville, Missouri, will show even more characters of primulina and so be referable to that form. If this supposition be correct, the present reference of the almost exactly intermediate males, from eastern Missouri, will stand; otherwise, it may not.
Additional intergrades with noveboracensis are available from eastern Iowa. Of five specimens from Hillsboro, Iowa, two males and a female have tympanic bullae like those of primulina but the other two males have bullae like those of noveboracensis. The female is smaller than primulina and in this small size and in general configuration of the skull, viewed dorsally, is more nearly like noveboracensis. As a whole, the population averages almost exactly intermediate. The same is true of 3 males and one female from Muscatine. The subadult male from Keosaqua, to my eye, resembles noveboracensis in the greater length of the skull anteriorly to the postorbital processes, and in the relative narrowness across the mastoidal region, but otherwise is more like that of primulina. Two males and one female from Tipton, although in each instance variously intermediate, are as a whole nearer primulina, No. 2865, Coe College, male adult, from Cedar Rapids, has characters of the three races, spadix, noveboracensis and primulina. In the skull, the width suggests spadix, the narrow mastoid region, noveboracensis, and the tympanic bullae are as in spadix or primulina. One male, no. 12, Coe College, from Dubuque, is as narrow across the mastoid region as is noveboracensis although the bullae are well inflated as in primulina. The skull, without corresponding skin, of a female, no. 140a, Iowa State College, from Green's Island, also resembles noveboracensis in narrowness of the mastoidal region, and in small size of skull, but in larger teeth, broader tympanic bullae, and sagittal crest is referable to primulina. Of two females from Vinton, one adult is typical of primulina but the other, a subadult, is practically indistinguishable from female noveboracensis, from Ann Arbor, Michigan. Three males from Vinton agree well with primulina except that the interorbital region is wider than average and thereby suggests spadix or noveboracensis. An adult female from New Hartford also is typical of primulina except for the broader interorbital region. Three males from Fayette are typical of primulina.
Other specimens from Iowa are intergrades with spadix, or if not with spadix, with the animal of northwestern Iowa which in some ways combines the characters of longicauda and spadix. For example, no. 2665, Coe College, an adult male from Davenport, has the anterior part of the skull (all that is preserved) heavily ridged as in spadix and in addition, the underparts are marked with the shade of reddish displayed by topotypes of spadix and with some yellowish as seen in longicauda. The color pattern, however, is as in primulina. A young male, no. C-51, Iowa State College, from Kelley, Story County, has anteriorly truncate bullae as are more frequently found in the longicauda-spadix stock of northwestern Iowa, than in primulina. In other respects, the animal, in so far as can be judged from the broken skull, agrees with primulina as it certainly does in color, color pattern, and external measurements. An adult male, no. 499a, Iowa State College, from 2 miles east of Ledges St. Park, in Boone County, in short body, size of teeth, and size of skull, in so far as the broken parts can be measured, resembles primulina more closely than it does any other subspecies. The long tail, long hind foot, wide extent of the light-colored underparts, and extension of the color of the underparts onto the hind feet are more as in spadix. Other intergrades with spadix from Iowa are mentioned in the account of spadix.
The specimen from Swartz, Louisiana, suggests intergradation with arthuri in that the anteromedial part of the tympanic bulla is less inflated than in typical primulina.
Intergrades with longicauda are available from Riley and Pratt counties, Kansas. No. 7182, Univ. Kans., subadult male in winter pelage, from near Winkler, has a skull of larger size as in longicauda with which race it seems to agree in large size of body, tail and hind foot, although the collector's measurements are lacking. Color pattern and relative proportions of the skull throughout are as in primulina. The young male, no. 3495, Univ. Kans., from Pratt, Kansas, agrees in external measurements and large size of skull with longicauda, but has the color and color pattern precisely as in primulina. The teeth are smaller as in primulina. Immaturity prevents judging of its relationships on the basis of relative proportions of the skull.
The two specimens, skins only, available from Oklahoma, are provisionally referred to primulina. These are remarkable for the restriction of the color of the underparts and for the intensity of the yellow coloration of the underparts. The specimen from Norman has the color of the underparts entirely absent from the hind legs and not extending posteriorly to the penis. On the chest and lower throat, large spots of color of the upper parts are present and the yellow area of the underparts on the belly is narrower than in any other specimen of primulina examined. The specimen from 8 miles northwest of Stillwater has the color of the underparts only a little less restricted although this color does extend over the inguinal region almost to the knees. The skin of the posterior part of the body of a weasel is available from 10 miles south of Sulphur Springs, Texas. It, likewise, is only provisionally referred to primulina. The coloration is about as in the specimens from Oklahoma but the distribution of the color of the underparts cannot be made out.
The dark color of the upper parts occurs far westward in animals which otherwise display characters of longicauda. Among these intergrades, the larger size of longicauda generally is combined with this dark color. This geographic behavior of the dark color of the upper parts is analogous to the condition described in M. f. spadix. Stated in another way, the dark color of the upper parts is the character, of the eastern animal, last to disappear as one goes westward across the Mississippi Valley toward the range of longicauda which is a subspecies of markedly different size, shape of skull, and coloration.
Only two of 29 specimens from Kansas show infestation of the frontal sinuses. All four of the specimens from Missouri have the frontal sinuses malformed as do 9 of the 14 from Arkansas examined in this respect.
An adult female from Boone County, Iowa, bears the date May 9, 1938, and the annotation by T. G. Scott, "Fox-killed."
Specimens examined.—Total number, 131, arranged alphabetically by states and from north to south by counties in each state. Except as otherwise indicated, specimens are in the University of Kansas, Museum of Natural History.
Arkansas. Boone County: 3 mi. E Bergman, 4[74]; 3 mi. SE Bergman, 1[74]; 3 mi. S Bergman, 1[74]; 3 mi. SW Bergman, 1[74]; 4 mi. SE Bergman, 2[74]; 5 mi. SE Bergman, 1[74]; 4-1/2 mi. SE Bergman, 3[74]; 5 mi. SE Bergman, 1[74]; 5 mi. S Bergman, 2[74]; 5 mi. SW Bergman, 2[74]. Washington County: Fayetteville, 1[96]. Crawford County: 10 mi. S Winslow, 1. Sebastian County: Fort Smith, 1[91].
Iowa. Fayette County: Fayette, 3[12]. Dubuque County: Dubuque, 1[12]; Green's Island, 1[65]. Butler County: New Hartford, 1[12]. Hardin County: Union, 1[65]. Benton County: Vinton, 5[12]. Linn County: Cedar Rapids, 1[12]. Boone County: Worth Township, Sec. 21, 1[65]; 2 mi. E Ledges St. Park, 1[65]. Story County: Kelley, 1[65]. Cedar County: Tipton, 3[12]. Scott County: Davenport, 2[12]. Muscatine County: Muscatine, 4[12]. Henry County: Hillsboro, 5[91]. Van Buren County: Keosaqua, 1[65]; no locality more definite than county, 1[50]. Taylor County, 1.
Kansas. Riley County: near Winkler, 1. Pottawatomie County: Onaga, 1[83]. Atchison County: Doniphan Lake, 1; 5 mi. NE Muscotah, 1; no locality more definite than county, 1. Douglas County: Lawrence, 8; 6 mi. NW Lawrence, 1; 1-1/2 mi. W Lawrence, 1; 6 mi. S Lawrence, 1; 7 to 7-1/2 mi. SW Lawrence, 14; 10 mi. W Lawrence, 1; Clinton, 4; Baldwin, 1; no locality more than county, 29 (2[74]). Woodson County: 1-1/2 mi. S Neosho Falls, 1[59]. Greenwood County: 8 mi. SW Toronto, 2. Pratt County: Pratt, 1.
Louisiana. Quachita Parish: Swartz, 1[71].
Missouri. Carroll County: Bridge Creek, 1[91]. Lincoln County: Silex, 1[74]; 1 mi. E Silex, 1[74]. Reynolds County: 4 mi. S Lesterville, 1[74]. Jasper County: 5 mi. NE Avilla, 1[91]; 7-1/2 mi. SE Carthage, 1[74].
Oklahoma. Payne County: 8 mi. NW Stillwater, 1[82]. Cleveland County: Norman, 1[100].
Texas. Hopkins County: 10 mi. S Sulphur Springs, 1[43].
Mustela frenata arthuri Hall
Long-tailed Weasel
Mustela noveboracensis arthuri Hall, Proc. Biol. Soc. Washington, 40:193, December 2, 1927.
Mustela frenata arthuri Hall, Carnegie Instit. Washington Publ. 473:105, November 20, 1936.
Type.—Male, subadult, skull and skin; no. 37515, Mus. Vert. Zoöl.; Remy, St. James Parish, Louisiana; December 15, 1926; obtained by Stanley C. Arthur.
The skin is stuffed and well preserved. The skull (plates 16-18) is unbroken. The teeth all are present and entire. The presence of a well-developed scrotal pouch shows the specimen to be a male. Contrary to what was stated in the original description the specimen was taken in 1926 and not in 1925.
Range.—Lower Austral Life-zone of southeastern Texas, Louisiana, and into Mississippi. See figure 29 on page 221.
Characters for ready recognition (of males).—Differs from M. f. olivacea in smaller size (adult males with hind foot and basilar length less than 45), depth of skull at anterior margin of basioccipital, ignoring sagittal crest, amounting to more than 63 per cent of mastoid breadth, and greater convexity of dorsal outline of skull in longitudinal axis (see pls. 16-18); from M. f. noveboracensis, in males, by zygomatic breadth not less than distance between anterior palatine foramen and anterior margin of tympanic bulla and by convex dorsal outline of skull in longitudinal axis; from M. f. primulina by evenly spreading zygomatic arches, lesser inflation of tympanic bullae anteromedially than posteromedially, and convex dorsal outline of skull in longitudinal axis; from M. f. texensis and M. f. frenata by absence of white facial markings and postorbital breadth more than distance between posterior borders of P4 and P2.
Description.—Size.—Male: The type, a subadult male, measures (inches and quarter fractions thereof, transposed into millimeters) as follows: Total length, 390; length of tail, 113; length of hind foot, 44. Tail is 41 per cent as long as head and body. Length of hind foot less than basal length.
Typical female unknown.
Externals.—Longest facial vibrissae black, or dark brown (both colors in the type) and extending beyond ear; carpal vibrissae same color as underparts and extending to within 3.5 millimeters of apical pad of fifth digit. Hairiness of foot-soles in type slightly less than shown in figure 20.
Color.—Upper parts in summer tone 4 of Burnt Umber of Oberthür and Dauthenay, pl. 304; underparts as described in M. f. olivacea. In winter, upper parts (based on type) near (1) Brussels Brown or grayer than tone 4 of Burnt Umber of Oberthür and Dauthenay, pl. 304, darker on top of head from nose to a line connecting posterior margins of ears. Chin and posterior third of each upper lip white. Remainder of underparts white with wash of Warm Buff. Tip of tail black. Color of underparts extends distally on posterior sides of forelegs over toes but represented on antipalmar faces of feet by only a few scattered hairs. Color of underparts extends distally on medial sides of hind limbs only to knees. Least width of color of underparts amounting to 15 per cent of greatest width of color of upper parts. Black tip of tail 50 mm. long; thus longer than hind foot and 44 per cent as long as tail-vertebrae.
Skull and teeth.—Male (based on type and 2 subadults): See measurements and plates 16-18. As described in M. f. noveboracensis except that: Weight, 4.0 (3.7-4.3) grams; basilar length, 43.5 (43.3-43.6); zygomatic breadth not less than distance between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth more than length of upper premolars; interorbital breadth more than distance between foramen opticum and anterior margin of tympanic bulla; least width of palate more or less than length of P4; tympanic bulla longer than rostrum.
Female: Typical skull unknown. The skull from 12 miles east of Eagle Lake, Texas, lacks the convexity in the dorsal longitudinal axis and the skull agrees with those of larger individuals of primulina except that the anteromedial faces of the tympanic bullae are less inflated, and the mastoid and zygomatic breadths are greater than in any female seen of primulina. Probably this greater breadth is the result of intergradation with M. f. frenata to the westward.
Compared with the skull of M. f. olivacea that of arthuri differs as follows: Averaging smaller in every part measured; basilar length 5 mm. less; by weight a fourth lighter; relative to basilar length, interorbital breadth greater and zygomatic and especially mastoid breadth less; dorsal outline of skull more convex in longitudinal axis; tympanic bullae narrower and less inflated especially on anteromedial faces. Compared with the skull of noveboracensis that of arthuri has the zygomatic breadth equal to or exceeding the distance from the anterior palatine foramen to the anterior margin of the tympanic bulla, whereas the zygomatic breadth is less than this distance in noveboracensis. Also, in arthuri, the rostrum is relatively shorter, the braincase is more inflated anteriorly, the zygomatic arches are more uniformly spreading, and the dorsal outline of the skull is distinctly convex, both transversely and longitudinally, whereas it is transversely more nearly flat in noveboracensis and longitudinally is concave in the interorbital region.
Compared with M. f. primulina, arthuri has narrower bullae, which are much less inflated on their anteromedial faces, a less marked postorbital constriction, a braincase which is narrower across the mastoid region and broader anteriorly, and a skull, which, in longitudinal axis, has the dorsal outline markedly more convex.
Compared with the skull of M. f. texensis that of arthuri is smaller in every part measured; length one-fifth less; one-half as heavy; postorbital constriction less marked; braincase relatively narrower posteriorly and tympanic bullae less inflated especially anteromedially. Compared with the skull of M. f. frenata that of arthuri is smaller in every part measured; basilar length 6 mm. less; a third lighter; postorbital constriction less marked; relative to the basilar length the rostrum is broader, longer and deeper; the zygomatic expanse and breadth of the braincase across the mastoids is less; the dorsal profile of the skull is more convex in longitudinal axis; zygomata evenly spreading rather than abruptly protruding from skull posteriorly; tympanic bullae less inflated anteromedially.
Remarks.—In 1926, Stanley C. Arthur, then Director of the Division of Wild Life, for the Louisiana State Department of Conservation, obtained specimens of this weasel. Some of them were mounted and the remainder were placed in the collections of the United States National Museum and the Museum of Vertebrate Zoölogy. In 1938 to 1940 George H. Lowery saved specimens from Baton Rouge, which showed the color of the summer pelage and revealed that the size of males was more than was indicated by the original materials. In 1940 and 1941 Rollin H. Baker obtained specimens from eastern Texas which greatly extended the known geographic range.
In addition to the localities represented by specimens examined, Arthur (1928:117) has recorded specimens from Greensburg, St. Helena Parish; Braithwaite, Plaquemines Parish; Geismar, Assumption Parish; Laurel Hill, West Feliciana Parish; French Settlement, Livingston Parish; and Kentwood, Tangipahoa Parish. All these localities lie within the eastern half of southern Louisiana. A skin-only, no. 38902, Mus. Vert. Zoöl., obtained from a fur buyer by Stanley C. Arthur, was taken in Mississippi "south of Jackson." Possibly it is of the subspecies arthuri.
Intergradation with M. f. olivacea is indicated by a specimen from Mobile County, Alabama, commented on in the account of olivacea. Intergradation with primulina is indicated by the shape of the anteromedial part of the bullae of the specimen from Swartz, Louisiana, that is referred to primulina. The lack of specimens from the northern two-thirds of Mississippi and from western Tennessee, prevents any definite statement as to the limits of range of arthuri in those areas. In comparison with animals from the type locality, the slightly larger size of the adult male from Baton Rouge, and the still larger size of the adult male of primulina from Swartz, Louisiana, suggests that the olivacea "influence" may extend farther west in the latitude of northern Louisiana than anywhere else.
None of the skulls examined shows malformation of the frontal sinuses such as results from infestation by parasites in some races. Arthur (1928:115) speaks of the ". . . cut-over swamp land, where the tupelo and cypress have been removed, . . ." as constituting suitable habitat for this animal.
Specimens examined.—Total number, 13, as follows:
Texas. Colorado County: 12 mi. N Eagle Lake, 1[43]; 5 mi. W Eagle Lake, 1[43]; 3 mi. S Garwood, 1[43].
Louisiana. East Baton Rouge Parish: Baton Rouge, 4[71]. Livingston Parish: Springville, 1[74]. Saint James Parish: Convent, 1[91]; Remy, 2 (1[74], 1[45]). Assumption Parish: near Lake Verret, 1[45].
Mississippi. Harrison County: Saucier, 1[71].
Mustela frenata olivacea Howell
Long-tailed Weasel
Plates 16, 17, 18, 31, 32 and 33
Mustela peninsulae olivacea Howell, Proc. Biol. Soc. Washington, 26:139, May 21, 1913.
Mustela frenata olivacea, Hall, Carnegie Instit. Washington Publ. 473:104, November 20, 1936.
Type.—Male, adult, skull and skin; no. 180802, U. S. Nat. Mus., Biol. Surveys Coll.; Autaugaville, Autauga County, Alabama; December 22, 1912; obtained by L. S. Golsan.
The skull (plates 16-18), although cracked at two places in the interorbital region, is in one piece and not warped out of shape. The teeth all are present and entire. The skin is exceptionally well made and in perfect condition except for the extreme tip of the tail which is broken off.
Range.—Lower and Upper Austral life-zones in eastern Mississippi, Alabama, Georgia, South Carolina, and northern Florida. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. peninsulae in finer, softer pelage and shorter (less than 15.8 in ad. ♀) tympanic bullae; from M. f. noveboracensis, in adult males by wider tympanic bulla which is more than rather than less than 8.5, in adult females by total length which is more than rather than less than 345, and by mastoid breadth which is more than rather than less than distance between articular faces of exoccipital condyle and glenoid fossa; from M. f. arthuri in larger size (adult males with hind foot and basilar length each more than 45); depth of skull at anterior margin of basioccipital, ignoring sagittal crest, amounting to less than 63 per cent of mastoid breadth, and lesser convexity of dorsal outline of skull in longitudinal axis (See pls. 16-18).
Description.—Size.—Male and female: External measurements of adults are available as follows:
| Catalogue No. | Sex | Locality | Total length | Length of tail | Length of hind foot | |
| 47165 | ♂ | Box Springs, Talbot Co., Georgia | 454 | 160 | 48 | |
| 47166 | ♂ | Box Springs, Talbot Co., Georgia | 435 | 147 | 47 | |
| 47167 | ♂ | Box Springs, Talbot Co., Georgia | 422 | 145 | 45 | |
| 41023 | ♂ | Thomas Co., Georgia | 443 | 140 | 47 | |
| 41025 | ♂ | Grady Co., Georgia | 395 | 142 | 47 | |
| 223880 | ♂ | Okefinokee Swamp, Georgia | 416 | 145 | 49 | |
| 198 | ♂ | Okefinokee Swamp, Georgia | 425 | 140 | 48 | |
| Average 7 | ♂ | 427 | 146 | 47 | ||
| 49385 | ♀ | Gainesville, Alachua Co., Florida | 396 | 124 | 45 | [not typical] |
| 41024 | ♀ | Thomas Co., Georgia | 380 | 125 | 41 | |
| 51527 | ♀ | Talbot Co., Georgia | 376 | 128 | 43 |
The length of the hind foot averages less than the basal length in both males and females. The tail averages 52 per cent as long as the head and body in males and 51 per cent in females. Average differences in measurements of the two sexes are: Total length, 49; length of tail, 19; length of hind foot, 5. An adult male, no. 41023, and an adult female, no. 41024, each taken in February, 1929, on the Sinkola Plantation, Thomas County, Georgia, weighed 15 ounces (425 grams) and 7 ounces (198 grams) respectively according to Charles O. Handley.
Externals.—As described in Mustela frenata noveboracensis, except that hairiness of foot-soles slightly less than shown in figure 19.
Color.—Upper parts, in summer, near tone 4 of Burnt Umber of Oberthür and Dauthenay, pl. 304. In winter lighter, between tones 3 and 4 of Raw Umber of Oberthür and Dauthenay, pl. 301. Dark spot at each angle of mouth present or absent. Underparts ranging from Massicot Yellow to Cream Buff except on chin and upper lips which are white. Tip of tail black. Upper parts of uniform color. Color of underparts extends distally on posterior sides of forelegs over antipalmar faces of toes and on medial sides of hind limbs to ankles. Least width of color of underparts averaging, in a series of five males from Talbot Co., Georgia, 29 (extremes 24-34) per cent of greatest width of color of upper parts. Black tip of tail in same series, averaging 65 (extremes 60-70) mm. long, thus longer than hind foot and averaging 43 per cent of length of tail-vertebrae.
The spot at the angle of the mouth is absent in one-third of the specimens examined. The upper lips are white in specimens from the southern part of the range of olivacea but in the northern part of the range of the subspecies the upper lips are dark colored as in noveboracensis.
Skull and teeth.—Male (based on 5 adults from Talbot Co., Georgia): See measurements and plates 16-18; weight, 5.3 (5.0-6.4) grams; basilar length, 48.3 (45.8-50.1); zygomatic breadth more or less (usually less) than distance between condylar foramen and M1 and more or less (usually more) than distance between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth more or less than (averaging about equal to) postpalatal length; postorbital breadth less than length of upper premolars and more or less than (about equal to) width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less than (about equal to) distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate less than greatest length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 3 to 5 upper incisors; height of tympanic bulla not less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer than rostrum (one exception); anterior margin of masseteric fossa below posterior half of m2.
Female (based on 2 adults from Thomas Co., Ga., and one from Talbot Co., Ga.): See measurements and plates 31-33; weight, 3.8 (3.5-4.0) grams; basilar length, 43.4 (42.7-44.0); zygomatic breadth less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth less than length of upper premolars and more or less (usually more) than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate less than greatest length of P4; tympanic bulla as far posterior to foramen ovale as width of 3 to 4 (including I3) upper incisors; height of tympanic bulla not less (usually more) than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer than rostrum.
The skull of the female averages 28 per cent lighter than that of the male.
Compared with the skull of M. f. peninsulae, of which only one good skull, and that a female, is available, that of M. f. olivacea averages smaller and has relatively and actually smaller and less inflated bullae. As compared with the skull of M. f. noveboracensis, that of olivacea in the case of males is larger in every part measured and relative to the basilar length is broader across the zygomatic arches and mastoids. However, the rostrum and interorbital region are relatively narrower. The orbitonasal length is relatively less. The tympanic bullae are broader and more inflated. The same differences hold as between females of noveboracensis and olivacea. Indeed, the females of these two races differ more than do the males. Additional, selected differential cranial characters in the females are, in olivacea, as follows: Weight averaging 3.8 grams rather than 1.7 grams; braincase with, rather than without, sagittal crest; anterior border of tympanic bulla separated from foramen ovale by breadth of less than, rather than breadth of more than, 4 upper incisors (including I3); height of tympanic bulla not less than, rather than less than, distance from its anterior margin to foramen ovale; squamosal bone, between anterior margin of tympanic bulla and foramen ovale, ventrally concave rather than ventrally convex. Comparisons of the skulls with those of M. f. arthuri and M. f. primulina are made in the accounts of those subspecies.
Remarks.—Excepting two young specimens from South Carolina in the Charleston Museum, no specimens of this race of large weasel seem to have been preserved until Arthur H. Howell, in the course of his study of the mammals of Alabama, procured specimens on which his name, olivacea, was based. Later, Francis Harper obtained three instructive specimens from Okefinokee Swamp. Really adequate material, for the localities represented, owes its preservation to the alertness of Charles O. Handley, when he resided at Thomasville, Georgia, and to Hallie E. Fuller of Geneva, Talbot Co., Georgia.
The distinctness of M. f. olivacea from M. f. peninsulae is not satisfactorily established due to inadequate material of peninsulae. Differences shown by the specimens seen indicate that, as compared with olivacea, peninsulae is larger, has transversely wider light-colored underparts which possess more yellow, and a larger skull with more inflated tympanic bullae. In each of these characters, olivacea is intermediate between noveboracensis on the north and peninsulae on the south. The question arises, therefore, whether the animals here recognized under the name olivacea really constitute a recognizable subspecies or instead are only representatives of a subspecies which reaches its extreme development in Florida. In the latter event, the name peninsulae would apply to all. Examination of more material from Florida, especially from the southern half of Florida, will be necessary to answer this question.
This large weasel of the southeastern United States is remarkably different from noveboracensis. Indeed, were it not for actual intergrades such as the two from Fort Payne, Alabama, and York, South Carolina, which are described in the account of M. f. noveboracensis, and the six specimens from northwestern Alabama, which are referred to olivacea, the systematist, I believe, would have little or no hesitancy in designating the two as distinct species, especially on the basis of differences to be seen in the skull.
Not only are the two forms structurally more different than usually is the case but between two geographically, adjacent subspecies of the same species of mammal, but the belt where intergradation occurs appears to be narrow. Nevertheless, when material of the two races is laid out in geographic order, and examined in mass, certain features are seen to undergo gradual change as a person's eye travels from specimens from, say, the center of the range of noveboracensis to specimens from southern localities adjoining the territory occupied by olivacea. One of these features subject to gradual change is the color of the underparts. Beginning at the Adirondacks of New York where a large number of the specimens have white underparts, the underparts become more intensely yellowish southward through the range of noveboracensis into that of olivacea. Indeed, this progressive trend seems to continue right on southward through the range of olivacea into that of peninsulae. Turning in the opposite direction we find that the least width of the underparts decreases gradually northward toward the range of noveboracensis. There is, likewise, a decrease to the northward in length of the skull and relative, as well as actual, narrowing of the braincase and tympanic bullae. However, in least width of color of underparts and the mentioned cranial features, the trend stops relatively abruptly at the southern boundary of the geographic range of noveboracensis and does not continue on, northward, into the range of noveboracensis as is the case with the change in intensity of yellowness of the underparts.
Two males, in the United States National Museum, Biological Surveys Collection, from near Leighton, Alabama, no. 178386 from the Tennessee River nine miles north [of Leighton?] and no. 180240 from La Grange Mountain, although clearly referable to olivacea on the basis of cranial characters, show some approach to noveboracensis in lesser size of the skull and agree with noveboracensis in the narrowness of the color of the underparts. Also, these specimens, like others from the northern part of the range of olivacea, for instance, no 31.227, Charleston Museum, from Mayesville, South Carolina, have the color of the underparts extended only part way out on the hind limb toward the foot. In specimens of olivacea from the southern part of its range the color of the underparts is extended onto the hind feet and this trend reaches its extreme in peninsulae, specimens of which have the feet and larger parts of the limbs marked with the light color of the underparts.
An adult female, no. 32.32, Charleston Museum, although typical of olivacea in most respects, is nevertheless an intergrade. The teeth are as small as in some specimens of noveboracensis. The size of the skull is only slightly nearer that of olivacea than it is to that of noveboracensis. The proportions of the skull, however, are distinctly those of olivacea.
Five other specimens, from northwestern Alabama, namely two from eight miles north of Nauvoo, two from Shoal Creek, and one from White Creek, also show intergradation between noveboracensis and olivacea. The remarks concerning color and color pattern of the specimens from Leighton apply equally well to the five from northwestern Alabama. In cranial characters, no. 51658 from Shoal Creek is referable to olivacea, as also is no. 51677 from the same place, providing it is a female rather than a male as sexed by the collector. No. 57146 from White Creek also is referable to olivacea although the skull shows some approach to that of noveboracensis. Of the two males from near Nauvoo, no. 51652 is to me indistinguishable from noveboracensis, but no. 51653 does have some characters of olivacea, although on the whole, the latter, too, seems to be a little nearer noveboracensis than olivacea. However, because the mean of these seven specimens from northwestern Alabama is nearer olivacea than noveboracensis the former name may be applied.
Another specimen from "Souinlonie" Creek, Clark County, Mississippi, has the coloration and rostral configuration of primulina, narrow mastoidal breadth and smaller teeth of noveboracensis and skull of large size with "full" braincase as in olivacea. No. 235364, U. S. Nat. Mus., from the Mobile River at the "L. and N. RR. Crossing," Mobile County, Alabama, although definitely olivacea, shows approach to arthuri in that the dorsal outline of the skull is longitudinally more convex and the tympanic bullae are less inflated than in olivacea and in that the color of the underparts is almost exactly as in the type specimen of arthuri. The young specimen labeled as from "Silver Springs," Florida, has large tympanic bullae (17 mm. long) and several characters that show its relationship to peninsulae as that race is now understood. Because the sex is unknown the identification as olivacea is tentative and is made on the assumption that the specimen is a male. If it is instead a female, the animal is referable to peninsulae.
An adult, female specimen in the Charleston Museum, no. 27.239.1, taken at St. Matthews, South Carolina, on December 8, 1927, contained four embryos which averaged 19 mm. in length and 47.75 centigrams in weight. Another adult female, in the Charleston Museum, no. 32.32, taken on February 21, 1932, at the same place, has prominent mammae, and the collector has noted that two were slightly active.
Sixteen of twenty-nine adults examined show infestation of the frontal sinuses by parasites. However, in none is the malformation of the frontal region so great as frequently occurs in M. f. noveboracensis.
Specimens examined.—Total number, 52, arranged alphabetically by states and from north to south by counties in each state. Except as otherwise indicated specimens are in the University of California Museum of Vertebrate Zoölogy.
Alabama. Lawrence County: White Creek, 1; Little Sand Mt., Shoal Creek, 2. Winston County: 7-1/2 mi. N Nauvoo, 1; 8 mi. N Nauvoo, 1. Lauderdale County: near Leighton, 9 mi. N Tennessee River, 1[91]. Colbert County: Leighton, 1[91]. Autauga County: Autaugaville, 1[91]. Dale County: Midland City, 1[91]. Mobile County: Mobile River, 12 mi. NE Mobile, 1[91].
Florida. Alachua County: Gainesville, 4[61]. Marion County: "Silver Springs," 1.
Georgia. Spalding County, 1. Lamar County, 1. Talbot County: southwest part of county, 1; Box Springs, near Geneva, 3; Upatoie Creek, 1 mi. SW Box Springs, 2; 3 mi. SE Geneva, 1; 4 mi. W Geneva, 1; 5 mi. W Geneva, 1; 2 mi. E Geneva, 1. Chattahoochee County, 2. Grady County: Beachton, 3[91]; locality no more definite than county, 4. Thomas County: Sinkola Plantation, 2; locality no more definite than county, 2. Charlton County: 1/2 mi. E Chesser's Island, Okefinokee Swamp, 1[58]. County in question: Billy's Island, Okefinokee Swamp, 1[91]; Okefinokee Swamp, 1[58].
Mississippi. Clark County: Souinlonie Creek, 1.
South Carolina. Darlington County: Society Hill, 1[91]. Sumter County: Mayesville, 1[11]. Calhoun County: St. Matthews, 2[11]. Georgetown County: Sampit, 1[11]. Charleston County: Rantowles, 1[11]; 8 mi. N Charleston, 1[11]. Beaufort County: Yemassee, 1[2].
Mustela frenata peninsulae (Rhoads)
Long-tailed Weasel
Putorius peninsulae Rhoads, Proc. Acad. Nat. Sci. Philadelphia, 1894:152, June 19, 1894; Bangs, Proc. Biol. Soc. Washington, 10:10, February 25, 1896.
Mustela peninsulae, Miller, U. S. Nat. Mus. Bull., 79:98, December 31, 1912.
Mustela p. peninsulae, Bailey, Bailey Mus. and Library Nat. Hist., 1(no. 5):1, December 1, 1930.
Mustela frenata peninsulae, Hall, Carnegie Instit. Washington Publ. 473:105, September 20, 1936.
Type.—Female, young, part skull and skin; no. 8515, Acad. Nat. Sci. Philadelphia; Hudson's, Pasco County [14 miles north of Tarpon Springs], Florida; before 1895; obtained by W. S. Dickinson.
The skull has been cut vertically in two at the plane of the glenoid fossae. These fossae and all the cranium posterior to them are missing. In addition to the part of the cranium anterior to the glenoid fossae, the lower jaws are preserved complete. The teeth all are present and entire. The prominent sutures on the rostrum and palate show the specimen to be young and its small size leaves but little doubt that the animal was a female. The light facial markings are more extensive than in any of the referred specimens. In the type these light facial markings consist of a median isolated spot immediately in front of the ears, a larger one on the nose, with an interrupted bar on each side extending posteroventrally in front of and anterior to the eye, a wider bar, on each side, extending anterodorsally between the ear and eye and finally an isolated spot at the anterior border of each ear. The skin is stuffed and in fair condition except that the vertebrae remain in the tail.
Range.—Austral and probably Tropical life-zones of Florida south of latitude 29°. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. olivacea in coarser pelage and larger tympanic bullae.
Description.—Size.—Male: No external measurements available. Female: The type a young animal and no. 2379, an adult from Tarpon Springs, measure respectively as follows: Total length, 375, 378; length of tail, 100, 130; length of hind foot, 40, 44.5.
Externals.—As described in Mustela frenata noveboracensis except that hairiness of foot-soles as shown in figure 20.
Color.—Upper parts (in winter) near tone 3 of Burnt Umber of Oberthür and Dauthenay, pl. 304. Dark spot at each angle of mouth present or absent. Tip of tail black. Underparts Reed Yellow except on chin and usually on legs where white. Upper lips white entirely around. Upper parts of uniform color. Color of underparts extends distally on legs over both sides of feet and on front legs over wrists. Proximal part of tail slightly lighter below than above. Least width of color of underparts, in seven specimens, averaging 41 (extremes 31-52) per cent of greatest width of color of upper parts. Black tip of tail, in each of two females, 45 mm. long; thus slightly longer than hind foot and amounting to 36 per cent of length of tail-vertebrae.
The spot at the angle of the mouth is absent in four of the ten specimens and is present on both sides in the other six.
Skull and teeth.—Male (based on an adult from Apopka and the anterior part of an adult from Enterprise): See measurements and plates 16-18. As described in Mustela frenata olivacea except that: Weight, 7.0 grams; basilar length, 49.8.
Female (based on an adult from Tarpon Springs, Florida): See measurements. As described in Mustela frenata olivacea except that: Weight, 4.7 grams; basilar length, 44.2; zygomatic breadth more than distance between anterior palatine foramen and anterior margin of tympanic bulla.
In comparison with M. f. olivacea, the insufficient material of M. f. peninsulae suggests that its skull averages larger and has relatively as well as actually larger and more inflated tympanic bullae.
Remarks.—The first published mention of this weasel seems to have been the original description which appeared in 1894. This description was based on a single specimen sent to Samuel N. Rhoads by W. S. Dickinson, who, in the following year, procured another specimen at Tarpon Springs. So far as known only eight other specimens, as listed under "Specimens examined," have found their way into collections of study specimens.
H. H. Bailey (1930:1) credits the range of this subspecies as extending south "to the shores of Florida Bay and the Gulf of Mexico, where ever high ground occurs."
Evidence of intergradation between M. f. peninsulae and M. f. olivacea is provided by specimens of olivacea from Gainesville, Florida, and the Okefinokee Swamp, Georgia. These specimens, on the average, have the color of the underparts wider, the skull larger, and the tympanic bullae relatively larger than do specimens of olivacea from farther north. In these features, approach to M. f. peninsulae is shown.
Light facial markings occur in this subspecies. They are similar to those possessed by weasels which occur at the same latitude and under corresponding climatic conditions on the Pacific Coast. The type specimen and one from Tarpon Springs have white facial markings. Two of the three specimens from Apopka also show white facial markings, although in reduced amount. One of the four specimens of M. f. olivacea from Gainesville, Florida, has well-developed light (white) facial markings. Also of the four specimens of M. f. olivacea examined from Okefinokee Swamp, Georgia, one has prominent white facial markings. However, in it the pattern is so unusual as to suggest that it is an instance of partial albinism rather than an outcropping of a racial tendency, or a pattern of coloration induced by climatic factors.
None of the eight available skulls show any infestation of the frontal sinuses by parasites.
Specimens examined.—Total number, 10, arranged by counties from west to east.
Florida. Pasco County: Hudson's, 1[1]. Pinellas County: Tarpon Springs, 1[1]. Hernando County, 1[91]. Polk County: Auburndale, 1[91]; no locality more definite than county, 1[91]. Orange County: Apopka, 3[61]. Volusia County: Enterprise, 1[60]. Seminole County: Osceola, 1[2].
Mustela frenata spadix (Bangs)
Long-tailed Weasel
Plates 16, 17, 18, 31, 32 and 33
Putorius longicauda spadix Bangs, Proc. Biol. Soc. Washington, 10:8, February 25, 1896; Merriam, N. Amer. Fauna, 11:21, figs. 10, 11, June 30, 1896; Cory, Mamm. Illinois and Wisconsin, p. 374, 1912.
Mustela longicauda spadix, Miller, U. S. Nat. Mus. Bull., 79:98, December 31, 1912; Bailey, Journ. Mamm., 10:156, May 9, 1929.
Mustela longicauda, Johnson, Journ. Mamm., 11:439, November 11, 1930.
Mustela noveboracensis, Murie, Journ. Mamm., 16:321, November 15, 1935.
Mustela frenata spadix, Hall, Carnegie Instit. Washington Publ. 473:105, November 20, 1936.
Type.—Male, young, skull and skin; no. 3265/1786, Amer. Mus. Nat. Hist.; Fort Snelling, Hennepin County, Minnesota; June 25, 1889; obtained by Edgar A. Mearns; original no. 812.
The skull is complete although there are fractures on the top of the braincase, on the right side of the braincase and at the middle of the right zygomatic arch. The teeth all are present and entire. The skin, although overstuffed, is complete, well preserved, and in summer pelage.
Range.—Upper Austral and Transition life-zones of Minnesota, northern and western Iowa, southeastern North Dakota, eastern part of South Dakota, and northeastern Nebraska. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. noveboracensis and M. f. primulina in that specimens of all ages have least width of color of underparts amounting to more than 41 per cent of greatest width of color of upper parts, and have light color of underparts extended onto hind foot rather than stopped short of ankle; adults with hind feet more than 50 in males and 40 in females; orbitonasal length more than 15.5 in males and 13.5 in females; length of tooth-rows more than 18.0 in males and 15.7 in females; mastoid breadth more than 25.5 in males and 22.0 in females. From M. f. longicauda by color darker than near (h) Clay Color, in males by a flattened occiput in which the depth of the skull, exclusive of the sagittal crest and taken at the anterior border of the basioccipital, amounts to less than 58 per cent of the mastoid breadth.
Description.—Size.—Male: Three adults from Elk River, Minnesota, yield average and extreme measurements as follows: Total length, 458 (444-467); length of tail, 154 (140-165); length of hind foot, 55 (52-59). Tail averages 51 per cent as long as head and body. Length of hind foot averages more than basal length. Corresponding measurements of three subadults from Madison, Minnesota, are as follows: 453 (438-469); 157 (152-165); 50 (47-51). Tail averages 53 per cent as long as head and body.
Female: Three adults from Elk River, Minnesota, yield average and extreme measurements as follows: Total length, 387 (380-391); length of tail, 131 (121-138); length of hind foot, 44 (43-46). Tail averages 51 per cent as long as head and body. Length of hind foot more or less than (approximately equal to) basal length. Corresponding measurements of two adults and one subadult from Madison, Minnesota, are as follows: 385 (379-396); 137 (119-159); 42 (38-44). Tail averages 55 per cent as long as head and body.
The average differences in external measurements of the two sexes from Elk River, are: Total length, 71; length of tail, 23; length of hind foot, 11. At Madison, corresponding differences are 68, 20, and 8. Two adult females from Elk River, Minnesota, weigh 205 and 210 grams.
Externals.—Longest facial vibrissae black, brown, or white (often all three colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles (in summer pelage) as shown in figure 19.
Color.—Winter pelage all white except tip of tail. In southern part of range sometimes assumes a brown winter coat. Summer pelage with upper parts ranging from near (16´) Cinnamon Brown to Vandyke Brown. Chin and upper lips white. Remainder of underparts ranging from near (a) Olive Ocher to Ochraceous Buff and Pale Orange Yellow. Tip of tail at all times black. Upper parts of uniform color except for occasional slight darkening of nose. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and ankles, on medial sides of hind limbs to ankle, over antiplantar faces of toes and distomedial fourth of each tarsus, and over proximal fifth to third of under side of tail. Least width of color of underparts averaging (in 3 specimens from Elk River) 54 (47-59) per cent of greatest width of color of upper parts. Black tip of tail averaging same length as hind foot and 28 per cent of length of tail-vertebrae. Save for the greater width of the light-colored underparts and relatively short black tip of the tail, both features of M. f. longicauda, spadix is variously intermediate, depending on locality, as between noveboracensis and longicauda.
Skull and teeth.—Male (based on 3 adults from Elk River, Minn.): See measurements and plates 16-18. As described in Mustela frenata longicauda except that: Weight, 5.6 (5.0-6.5); basilar length, 49.0 (48.7-49.2); zygomatic breadth sometimes less than distance between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth more or less (about equal to) width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bulla; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4 to 5 upper incisors; height of tympanic bulla more or less than distance from its anterior margin to foramen ovale; length of tympanic bulla less than length of rostrum; anterior margin of masseteric fossa below talonid of m1.
Female (based on 4 adults from Elk River, Minn.): See measurements and plates 31-33. As described in Mustela frenata longicauda except that: Weight, 3.5 (3.3-4.0) grams; basilar length, 42.9 (42.3-43.2); least width of palate more or less than greatest length of P4; tympanic bulla as far posterior to foramen ovale as width of 3 to 5 upper incisors.
The skull of the female averages 33 per cent lighter than that of the male.
Skulls of adult males of spadix from Elk River, Minnesota, as compared with those of longicauda from Alberta, are larger in every part measured. Relative to the basilar length these skulls of spadix are broader across the mastoid region, narrower across the zygomata, deeper through the plane of the postorbital processes, shallower through the braincase and have relatively shorter tympanic bullae. Whereas the tympanic bullae of longicauda are, on the average, approximately as long as the rostrum (orbitonasal length), in spadix the rostrum is longer than the bulla. Viewed posteriorly, the braincase of spadix is seen to be much shallower and wider than that of longicauda. Indeed, the depth of the braincase, measured at the anterior end of the basioccipital, amounts to only 56 per cent of the mastoid breadth in spadix as against 61 per cent in longicauda. The longer, waistlike, postorbital constriction, relatively smaller braincase, and especially the relatively narrower zygomatic expanse in spadix imparts to its skull a more slender appearance than has the skull of longicauda. These differences are not shown by the skulls of females. To be sure, spadix, in most of its cranial measurements, averages slightly larger, has a relatively shallower braincase and is relatively deeper through the postorbital processes, but these differences are so slight that inclusion of one more specimen, of slightly different proportions, in the average might cause the average measurements to read as they do in longicauda.
Compared with noveboracensis, from Massachusetts, adult skulls of spadix, taking sex into account, are larger in every part measured and are relatively as well as actually wider and deeper throughout. Also, in spadix: Sagittal and lambdoidal crests higher, especially in females; anterior margin of tympanic bulla projecting up sharply from squamosal; occiput more flattened in posterior view; tooth-rows relatively and actually longer but orbitonasal length relatively shorter; postorbital processes more robust; zygomatic arches widely bowed outward rather than evenly rounded; canines larger; squamosal less swollen ventrally, especially in females. Between noveboracensis and spadix, the differential cranial characters are greater in number and degree between females than between males. Comparison of the skull with that of M. f. primulina is made in discussion of that subspecies.
Remarks.—Edgar A. Mearns in 1889 and the early nineties took several specimens of this weasel and it was principally on these that Bangs in 1896 (p. 8) based his description. The best material, however, is that from Elk River, Minnesota, collected in later years by Bernard Bailey, and supplemented by one specimen taken in 1885 by Vernon Bailey and another by his sister Anna Bailey in 1891 at the same place.
Mustela frenata spadix has just one structural feature of a "unique" kind which serves to differentiate it from the geographically adjoining subspecies. This feature is large size. The other diagnostic characters ascribed to spadix are of an intermediate sort—intermediate as between two extremes, one found to the westward in longicauda and the other to the eastward in noveboracensis. For example, the dark-colored upper parts are merely darker than in longicauda and merely lighter than in noveboracensis. The color is not "different"; it is only "intermediate." Furthermore, each of the characters ascribed to spadix, including large size itself, undergoes change from one part of its geographic range to another; the characters are not constant over a wide area. Indeed, excepting the large size which remains relatively uniform over the northern two-thirds of the range, no two localities have been found from which the specimens can be said really to agree in characters.
By way of illustration, the coloration of the upper parts may be cited. Near the range of noveboracensis the average coloration of individuals from one locality is only a little lighter than in noveboracensis. Farther westward the average coloration is a little lighter and farther westward yet, toward the range of the extremely light colored longicauda, the average coloration is lighter still. Although all these animals are darker than longicauda and lighter than noveboracensis, those from the three places do not agree among themselves. Because of the lack of more than one character of a "unique" kind and because of the inconstancy, geographically, of other characters, and for that matter, lack of constancy geographically in combination of characters, the writer regards spadix as a barely recognizable subspecies.
Examination of the specimens of spadix shows that the individual variation in a single species is greater in a region of intergradation than it is some distance inside the borders of the geographic range of a well-marked subspecies. This is illustrated by three specimens of M. f. spadix in fresh summer pelage from the single locality, Elk River, Minnesota. In these, the color of the upper parts varies from a little darker than Cinnamon Brown to Vandyke Brown. At any one locality well within the range of longicauda, or noveboracensis, there is nowhere nearly so much variation in color, even in much larger series of specimens.
Study of the specimens here assigned to spadix reveals that some features regarded as of diagnostic value for one or the other of the two races, longicauda and noveboracensis, behave differently. For example, the dark coloration of the upper parts, which is characteristic of noveboracensis, manifests itself far westward within the range of spadix whereas the wider extent of the light-colored underparts, which is characteristic of longicauda, and the Olive Ocher, rather than Pale Orange Yellow, color of these underparts, are seen in varying degree all the way across the range of spadix. Thus, these animals are colored above like noveboracensis and below like longicauda, but not vice versa. In these animals, then, the longicauda type of underparts is dominant, in one sense of the word, over the noveboracensis type of underparts, and the noveboracensis type of upper parts is dominant over the longicauda type of upper parts. Each of these features is subject to actual intergradation and does not always behave as a "unit character," that is to say, one which is either present or absent. However, the noveboracensis type of upper parts is carried much farther west before being diluted than is the noveboracensis type of underparts. Indeed, within the range of noveboracensis itself, the broad extent of the longicauda type of underparts is manifest. This is, of course, near the western margin of the range of noveboracensis.
The large size of males of spadix, as exemplified by specimens from Elk River (see measurements on page 421), seems to be retained across the northern part of the range here assigned to the subspecies. This larger size than is found in longicauda from Alberta, is shown also by some specimens from eastern North Dakota which are assigned to longicauda. However, the average of these Dakotan specimens, all characters considered, is nearer to my concept of longicauda.
Inspection of the cranial measurements of spadix shows also that in addition to its large size it is distinguishable from any one of the geographically adjoining races by its relatively (to basilar length) greater, as well as actually greater, mastoidal breadth. This might be included with size as a unique character distinguishing spadix from longicauda and noveboracensis. However, it is not clear whether or not this greater mastoidal breadth is more than a function of the large size.
Excepting the greater mastoidal breadth and generally larger size of the skull, the cranial features distinguishing males of spadix from longicauda are features in which spadix shows approach to noveboracensis. This is true, in spadix, of the relatively longer (in comparison with longicauda) rostrum, relatively lesser zygomatic breadth, relatively shallower braincase measured at the anterior end of the basioccipital, and relatively deeper skull as measured at the posterior borders of the last upper molars. This same approach to noveboracensis already has been pointed out with respect to color of the upper parts and is evident also in the relative shortness of the tail which averages only 51 per cent of the length of the head and body rather than 55 per cent as in longicauda.
Because the longicauda type of animal previously has been regarded as specifically distinct from the noveboracensis type of animal, comment is offered below on selected specimens, referred to spadix, which are regarded as intergrades with noveboracensis or with other subspecies.
No. 8722, Univ. Wisconsin, adult male, in the white winter coat, from north central Itasca County, Minnesota, obviously has characters of M. f. spadix or longicauda that occur to the west and M. f. noveboracensis of the east. Selected outstanding characters of longicauda are its long tail, anteriorly truncate tympanic bullae and large teeth. Characters indicating its affinities with noveboracensis are smaller size of skull, general narrowness of skull, and relatively low tympanic bullae. The skull is intermediate as regards several individual structural features. For example, although long and narrow and in this feature more nearly approaching noveboracensis, the skull is wider than usual in that subspecies and thus approaches that of longicauda or spadix. The hind foot, in the dried state, measures 47 millimeters. This large hind foot, obviously long tail (the specimen lacks external measurements), and anteriorly truncate bullae constitute basis for here referring the specimen to spadix. However, the seemingly small size of the body and the narrow skull clearly show relationship to noveboracensis.
Specimens, referred to spadix, from northern Iowa, are instructive as showing what happens where the ranges of noveboracensis, primulina, spadix, and perhaps longicauda, meet. No. 47167, Univ. Mich. Mus. Zoöl., a nearly adult female, taken on November 22, 1915, at Island, Clay County, and in process of assuming a brown winter pelage, retains enough of the dark summer pelage to show that the color was slightly lighter than average for spadix. The color pattern, white lips, and extension of light color of the underparts onto the feet, agrees with spadix or longicauda as does also the long tooth-row. The overall length of the skull is intermediate between that of spadix and primulina. The proportions of the anterior part of the skull and of the tympanic bullae resemble those found in primulina. A subadult male skull only, no. 123846, American Museum of Natural History, from Webb, Clay County, shows approach to primulina in the narrowness of the rostrum.
A young male from Ruthven, Iowa, no. 48340, Univ. Michigan, has a large skull approaching in size that of spadix, has the longicauda-spadix type of light-colored underparts and color pattern, and is slightly darker above than true longicauda. Another subadult male in the white winter coat from Palo Alto County, no. 35756, Univ. Michigan, has a large skull, which shows approach to primulina in its narrowness anteriorly and in some other features. Although the tail is of moderate length, the body is large as in spadix or longicauda, and the length of the hind foot suggests spadix or longicauda.
A subadult male, no. 425a, Iowa State College, from Manson, Iowa, in brown winter pelage, agrees with primulina in the restriction of the area of the light color of the underparts and in less expanded zygomatic arches. The teeth are intermediate in size between those of noveboracensis and primulina on the one hand and those of spadix and longicauda on the other. In other respects it agrees with, or is more nearly like, spadix.
An adult female, no. 426a, Iowa State College, from Barnum, in the brown winter coat, agrees with primulina except that the orbitonasal length of the skull is more as in spadix and the presence of some light color on the lower part of the hind legs suggests spadix. The skull only, no. 440a, Iowa State College, labeled merely Webster County, Iowa, is almost a duplicate of no. 426a. A subadult male, no. 427a, Iowa State College, from Moorland, Iowa, only about six miles southeast of Barnum, likewise is indistinguishable from primulina except for having a white winter coat and in being relatively broad in the mastoidal region. Nevertheless, both of these animals are here referred to spadix because the average of specimens from this general area is nearer that of spadix. No. 497a, Iowa State College, an adult female in white winter pelage, from Ames, approaches primulina in the narrow rostrum and smaller teeth but otherwise approaches or even agrees with spadix.
Two adult males, without external measurements, from Pilot Mound, Iowa, have skulls quite like males of longicauda from Alberta. The only approach noted to eastern forms is the restricted color of the underparts on no. 2856, Coe College, which has a brown winter coat. The color of the underparts is not extended so far out on the feet as in longicauda. Also the tympanic bullae of this specimen are a trifle narrower. The other male, no. 2652, is in the white winter coat. The one female from the same place, no. 2660, Coe College, in brown winter pelage, has a skull notably unlike that of longicauda or spadix; the skull is narrower and practically indistinguishable from that of the largest female skull of primulina available from Lawrence, Kansas, save that the tooth-row is much longer. The color pattern also agrees with that of primulina or noveboracensis in that the color of the underparts extends only as far as the knee on the hind legs and is narrow on the belly. Nevertheless, another adult female, no. 120a from Amaqua Township, some 6 miles southwest of Pilot Mound, is in all respects typical of spadix. This is the more remarkable because another comparable specimen from less than 20 miles to the southwest in Worth Township is equally typical of primulina.
Two young females from Chester, Iowa, nos. 2656 and 2874/2873, Coe College, have skulls larger than those of corresponding age of primulina or noveboracensis. The color is as in spadix. The color pattern of the underparts also is as in spadix or longicauda except that the width of the area of light color on the belly is restricted somewhat although not so much as in noveboracensis or primulina. Of four males from the same place, also in the collection of Coe College, no. A2874 is a white skin only and does not provide diagnostic characters. The three other males, each in summer pelage, are marked and colored as are the two females from the same place except that male no. 2861 has the color of the underparts so much attenuated on the hind legs that it barely, uninterruptedly, extends to the feet. No. 2658 is young, or perhaps barely subadult. The skull is large and referable to spadix. The two adults, nos. 2861 and 2657, differ cranially from typical (Elk River, Minn.) spadix only in being slightly narrower across the mastoids and in having the bullae a little narrower. In these departures they show some approach to primulina and to noveboracensis. Another male, subadult, no. 2867, Coe College, from Decorah, which has acquired half of the white winter coat, agrees with the males from Chester except that the preorbital part of the skull is shortened much as in some specimens of primulina.
From Lansing, in extreme northeastern Iowa, a large subadult male, no. 2864, Coe College, of 453 mm. in total length and half through with acquiring the white winter coat, agrees with the males previously described from Chester except in having the palate narrower as in noveboracensis. The adult female available from Lansing, no. 2863/2862, Coe College, in white winter pelage except for the top of the head, although a large skin, has a skull smaller than that of any spadix or longicauda and of about the same size as that of no. 3838, Univ. Kansas Mus. Nat. Hist., of primulina, from Lawrence, Kansas, except that the skull of no. 2863/2862 is much narrower across the mastoids. This specimen, then, shows approach to noveboracensis in narrowness of the mastoidal region, to primulina in other respects and to spadix.
Many of these instructive specimens from Iowa, made available to the present writer by Mr. W. F. Kubichek, were brought together at the Coe College Museum by the late B. H. Bailey. Most of them were obtained from trappers who did not supply the conventional external measurements taken in the flesh. Even though these are lacking, the specimens clearly show that actual intergradation occurs where the ranges of M. f. longicauda, spadix, noveboracensis and primulina meet.
The dark color of the upper parts, restriction of the color of the underparts on the ankles with the result that the color reaches the toes in interrupted fashion, and large skull, of no. 18912 of the Museum of the University of South Dakota, from Roberts County, South Dakota, clearly place this specimen with spadix, rather than with longicauda. Likewise, male, subadult, no. 11376, Univ. South Dakota, from Clay County, South Dakota, is referable to spadix. Although without external measurements, the specimen obviously is large. The patch of summer pelage on its head and neck is darker than the summer pelage of longicauda, and the orbitonasal length is greater than the length of the tympanic bullae; all these features are characters of spadix. The adult male from Fort Sisseton, South Dakota, no. 188407, United States National Museum, figured by Merriam (1896, p. 20, figs. 7-9), is almost exactly intermediate between longicauda and spadix, although here referred to the latter.
Five specimens, nos. 147375, 147432, 147762, 148720 and 148721, U. S. Nat. Mus., including 3 skulls only from Beemer, Cuming County, Nebraska, are intergrades between M. f. longicauda, M. f. primulina and M. f. spadix. One skin is in white winter pelage and the other, a female, is in summer pelage which in coloration and color pattern agrees with that of spadix. External measurements of the male agree with those of longicauda. Measurements of the female agree with those of spadix except that the tail is shorter as in primulina. The skulls are as long as in longicauda but are more slender than in either longicauda or spadix although nearer the latter in this respect. In dorsal aspect, the skulls especially posteriorly to the orbital region, resemble primulina. All points considered, the animals seem best referred to spadix.
Although the degree of development of certain morphological features has been settled upon as indicative of the race spadix, some doubt remains as to where the western boundary of its range should be shown. This results from the fact that color has been taken into account as one diagnostic feature and this feature is lacking in the white winter specimens which, from the following places, are all that are available: Kittson County, Minnesota; Moorhead, Minnesota; Casselton and Valley City in North Dakota; Armour, South Dakota and Clay County, South Dakota. In summary, more specimens in the summer coat will be required to establish definitely the boundary between the ranges of longicauda and spadix.
Surber (1932:49) has referred to additional specimens of this weasel in the University of Minnesota Museum as from Winona, Hennepin and Isanti counties of that state.
At Elk River, Minnesota, B. Bailey (1929:156) found this species to be about half as abundant as Mustela cicognanii and that it is "more often found in the open timber and about the dry ridges and fields." Of seventeen adult or subadult skulls of this race from Minnesota, ten have obvious marks of infestation of the frontal sinuses. In no skull, however, has the infestation resulted in so much malformation, as occurs in noveboracensis.
Specimens examined.—Total number, 76, arranged alphabetically by states and from north to south by counties in each state.
Iowa. Lyon County: Granite, 1[65]. Howard County: Chester, 6[12]. Winneshiek County: Decorah, 1[12]; 8 mi. NE Ossian, 1[76]. Allamakee County: Lansing, 2[12]. Clay County: Island, 1[76]; Webb, 1[2]. Palo Alto County: Ruthven, 1[76]; no locality more definite than county, 1[76]. Calhoun County: Manson, 1[65]. Webster County: Barnum, 1[65]; Moorland, 1[65]; no locality more definite than county, 1[65]. Boone County: Pilot Mound, 3[12]; Amaqua Township, Sec. 19, 1[65]. Story County: Ames, 1[65].
Minnesota. Kittson County, 1[2]. Roseau County: 2-1/2 mi. SW Roseau, Jadis Township, 1[14]. Itasca County: T. 61N, R. 26W, 1[102]. Clay County: Moorhead, 2[9]. Atkin County: Atkin, 1[50]. Otter Tail County: Lake Lizzie, 1[9]; Parkers Prairie, 1[57]. Grant County: 3 mi. NW Barrett, 1[76]. Benton (now Mille Lacs?) County: Princeton, 1[91]. Sherburne County: Elk River, 14 (6[59], 4[14], 3[91], 1[74]). Hennepin County: Fort Snelling, 6 (5[2], 1[91]). Carver? County: Chaska, 1[60]. Lac qui Parle County: Madison, 5 (3[91], 2[1]); no locality more definite than county, 2 (1[68], 1[75]). Yellow Medicine County: Wood Lake, 1[2]. Blue Earth County: Rapidan, 1[64]. County in question: Moore Lake, 1[91].
Nebraska. Cuming County: Beemer, 5[91].
North Dakota. Cass County: Fargo, 1[91]; Casselton, 1[91]. Dickey County: Oakes, 1[91].
South Dakota. Roberts County, 1[102]. Marshall County: Fort Sisseton, 1[91]. Douglas County: Armour, 1[14]. Clay County, 1[102].
Mustela frenata longicauda Bonaparte
Long-tailed Weasel
Plates 16, 17, 18, 31, 32 and 33
Mustela longicauda Bonaparte, Charlesworth's Mag. Nat. Hist., 2:38, 1838.
Putorius longicauda, Baird, Mamm. N. Amer., p. 169, 1858; Coues, Fur-bearing animals, p. 136, 1877; Bangs, Proc. Biol. Soc. Washington, 10:7, figs. 1, 1a of pls. 1, 2 and 3, February 25, 1896; Merriam, N. Amer. Fauna, 11:19, pl. 3, figs. 3, 3a, 4, 4a, pl. 5, figs. 1, 1a, text figs. 7-9, June 30, 1896.
Mustela longicauda longicauda, Bailey, N. Amer. Fauna, 49:166, January 8, 1927.
Mustela frenata longicauda, Hall, Carnegie Instit. Washington Publ. 473:105, November 20, 1936; Hall, Canadian Field-Nat., 52:108, October, 1938.
Mustela frenata, Sowls, Journ. Mamm., 29:126, May 14, 1948.
Type.—Possibly not in existence. No. 43.3.3.3 [from Carlton House, Saskatchewan] in the British Museum of Natural History has been regarded by several zoölogists as the type. It is a subadult female, skull and skin, from North America. See the account of M. erminea cicognanii for reasons for and reasons against regarding this specimen as the holotype.
No. 43.3.3.3 from the collection of Dr. John Richardson is in the white winter coat and now (Sept. 24, 1937) is prepared as a study skin. Evidences of its once having been mounted are: holes in the soles of the hind feet for supporting-wires, large straight wire in the tail, folds in the skin of the now backward-projecting hind feet, and unevenness of the skin on the back resulting from straightening out the specimen. The tip of the tail and some skin from the middle of the belly are missing. Otherwise the skin is intact. The skull is that of an animal in its first year, lacks the zygomatic arch on each side, but otherwise is complete and unbroken. The teeth all are present and entire except that p2 on the right side is missing from its alveolus.
Range.—Transition and Upper Sonoran life-zones of the Great Plains, southward from central Alberta, Saskatchewan and southern Manitoba through eastern Montana, the Dakotas and Nebraska into southeastern Wyoming, northeastern Colorado and western Kansas. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. primulina in near (h) Clay Color rather than Brussels Brown of upper parts, least width of color of underparts more than 40 per cent of greatest width of color of upper parts, color of underparts extended onto hind foot rather than stopped short of ankle, zygomatic breadth more than 28.8 in adult males and more than 24.1 in adult females; from M. f. spadix in lighter color being near (h) Clay Color, in males by deeper occiput in which the depth of the skull, exclusive of the sagittal crest and taken at the anterior border of the basioccipital amounts to more than 59 per cent of the mastoid breadth; from M. f. oribasus in near (h) Clay Color rather than near (14n) Brussels Brown color of the upper parts and in males by deeper occiput in which the depth of the skull, exclusive of the sagittal crest and taken at the anterior border of the basioccipital, amounts to more than 59 per cent of the mastoid breadth; from M. f. alleni in larger size, adult males having a total length of more than 400 millimeters, hind foot more than 45, basilar length more than 43.5, and females having a total length of more than 375 and basilar length not less than 40.0; from M. f. nevadensis in near (h) Clay Color rather than near (14n to 1) Brussels Brown of upper parts, basilar length more than 40 in females and averaging more than 45 in males; from M. f. neomexicana by near (h) Clay Color rather than Buckthorn Brown color of upper parts, absence of white and Argus Brown facial markings, and length of tooth-rows amounting to more than 37 per cent of basilar length.
Description.—Size.—Male: Five adults from Alberta yield average and extreme measurements as follows: Total length, 438 (418-473); length of tail, 158 (140-193); length of hind foot, 50 (46-54). Tail averages 56 per cent as long as head and body. Length of hind foot averaging more than basal length. Corresponding measurements of five adults and subadults from North Dakota are as follows: 465 (445-516); 164 (150-179); 51 (50-54). Tail averages 55 per cent as long as head and body.
Female: Six adults (Alberta, 4; Saskatchewan, 1; Manitoba, 1) yield average and extreme measurements as follows: Total length, 401 (383-425); length of tail, 145 (141-159); length of hind foot, 43 (41-44). Tail averages 57 per cent as long as head and body. Length of hind foot more or less than (approximately equal to) basal length.
The average differences in external measurements of the two sexes are: Total length, 37; length of tail, 13; length of hind foot, 7. General comparisons indicate that the Alberta-taken males may not attain so large a size as those from some other areas. Thus the differences in external measurements might be some greater elsewhere, say, in North Dakota.
Externals.—Longest facial vibrissae black, brown or white (often all three colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles (in summer pelage) only slightly greater than shown in figure 20.
Color.—Winter pelage all white except tip of tail. Summer pelage with upper parts near (h) Clay Color or near tone 3 and 4 of Snuff Brown of Oberthür and Dauthenay, pl. 303. Chin and upper lips white. Remainder of underparts ranging from near (a) Olive Ocher to near (16´) Ochraceous Buff. Upper parts of uniform color except for occasional darkening of head in front of ears. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists, on medial sides of hind limbs to ankles over antiplantar faces of toes and distomedial third of each tarsus, and over proximal fourth to third of under side of tail. Least width of color of underparts averaging, in a series of 10 males from Alberta, 58 (45-60) per cent of greatest width of color of upper parts. Corresponding figures for 10 females from the same place are 57 (50-74). Black tip of tail in same series of males, most of which are in full summer pelage, averaging 43 (35-60) mm. long. Thus, averaging shorter than hind foot and 27 per cent of length of tail-vertebrae.
As compared with M. f. neomexicana, longicauda lacks the white facial markings, black ears, black forehead and nose, but otherwise is similarly colored. As compared with M. f. nevadensis, M. f. oribasus and M. f. spadix, each of color pattern similar to longicauda, selected differences of longicauda are its much lighter color, especially of the upper parts, with less conspicuous darkening on the nose. From M. f. primulina, longicauda differs in lighter color of upper parts, reddish rather than yellowish underparts, and light rather than dark-colored hind feet.
Skull and teeth.—Male (based on 5 adults from Alberta): See measurements and plates 16-18; weight, 4.7 (4.6-4.9) grams; basilar length, 46.0 (44.7-46.8); zygomatic breadth more than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth more than postpalatal length; postorbital breadth less than length of upper premolars and more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth greater than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum more or less (usually less) than length of tympanic bulla; least width of palate less than greatest length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 3 to 4 (including I3) upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum; anterior margin of masseteric fossa below talonid of m1 or anterior half of m2.
Female (based on 5 adults: Alberta, 3; N. D., 1; Sask., 1.): See measurements and plates 31-33; weight, 3.1 (2.8-3.5) grams; basilar length, 42.3 (40.0-43.7); zygomatic breadth more or less (approximately equal to) than distance between condylar foramen and M1 or that between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth less than length of upper premolars and more or less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate not more than greatest length of P4; tympanic bulla as far posterior to foramen ovale as width of 3 to 4 (including I3) upper incisors; height of tympanic bulla not less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-rows and longer or shorter than rostrum.
The skull of the female averages 34 per cent lighter than that of the male.
Comparisons of the skull with those of M. f. primulina, M. f. spadix, M. f. oribasus, M. f. alleni, M. f. nevadensis, and M. f. neomexicana are made in accounts of those subspecies.
Remarks.—Richardson's (1829:47) account on which Bonaparte may be said to have based his name, records measurements in inches and lines which I transpose into millimeters as follows: Total length, 440 mm.; length of head and body, 305; length of tail (vertebrae), 135; length of tail (including fur), 164 mm. Specimen no. 43.3.3.3 in the British Museum, which has by some persons been regarded as the type, yields measurements as follows: Total length, 408 (which allows for 15 mm. loss of the fleshy part of the end of the tail); length of head and body, 272; length of tail (vertebrae), 136 (= 121 + 15); length of tail (including fur), 162 (142 + 20 mm. that appears to have been lost). Richardson's specimen would appear to have been of unusual proportions and to have been larger than no. 43.3.3.3. Some reasons for and reasons against regarding this specimen as the holotype are given in the account of M. erminea cicognanii.
The name longicauda was applied to practically all long-tailed weasels of the western United States at one time but as one after another of the geographic variants in the mountainous regions were designated as separable, the name longicauda came to be restricted to the light-colored, relatively large, animal of the Great Plains.
The intergradation of longicauda with spadix and oribasus has been commented on in the discussions of those subspecies. The larger size and darker color of specimens referred to longicauda from Devils Lake and Grafton, North Dakota, are features indicative of intergradation there with spadix. Two young females from Waterton Lake Park, Alberta, by their darker than average color, suggest intergradation with oribasus, as, for that matter, does the specimen from Waterton Lake [= Chief Mountain Lake, in Montana] itself, which, however, is even darker than the two specimens taken on the Canadian side of the line and hence is referred to oribasus. An adult female, no. 175586, U. S. Nat. Mus., from Moose Pass, Alberta, examined after the above was written, is larger than any other female seen of longicauda and in this respect may show approach to oribasus, which in the northern part of its range is of large size as judged by males from the Bowron Lake region.
One male, no. 8564, Nat. Mus. Canada, from Max Lake, Turtle Mountain, Manitoba, presents puzzling characters. The external measurements of 465, 170, and 57, are in keeping with the great length of the skull which has a basilar length of 48.8. The tooth-rows are 19.3 in length and the mastoid breadth, 25.4. The relative narrowness indicated by the mastoid breadth is maintained throughout the skull. The only other specimens relating to the Turtle Mountains that have been seen are two male, skins without measurements or corresponding skulls, nos. 38902 and 38903, Amer. Mus. Nat. Hist., labeled as from either "Stump Lake or Turtle Mts.," North Dakota. One of these, no. 38902, is much darker than the other. Possibly it is from the Turtle Mountains and the other, lighter-colored one, is from Stump Lake. Study of additional specimens from the Turtle Mountains might show the existence there of a distinct race.
Four specimens, in the collection of Myron Swenk, from Inland, Clay County, Nebraska, are instructive as showing how intergradation occurs between primulina and longicauda. A subadult male, no. 10, is intermediate in external measurements and in color but in each instance is nearer primulina. The same is true of the least width of the color of the underparts. The color of the underparts extends uninterruptedly over the hind legs to the toes as in longicauda, but is absent from the underside of the tail as in primulina. In the skull, the basilar length, breadth of bulla, and size of teeth are nearer longicauda, as are also the ratios to the basilar length of the length of tooth-rows, breadth of the rostrum, length of the tympanic bulla, and depth of the braincase at the anterior margin of the basioccipital. Ratios to the basilar length of the interorbital breadth, mastoid breadth, zygomatic breadth, and depth of the skull at the posterior borders of the upper molars are nearer to those of primulina. The relatively long rostrum, as represented by the orbitonasal length, is nearest to that of spadix. A young, almost subadult, female, no. 7, agrees with primulina in color, color pattern, and length of hind foot. The other external measurements are intermediate, but nearer those of primulina. Size of skull and teeth are as in longicauda. Relative proportions of parts of the skull are not diagnostic in specimens as young as this female. An adult female, skull only, no. 8, agrees with, or approaches nearer to, longicauda in size of skull and teeth and in relative proportion of every part studied. A juvenile, skull only, of questionable sex, no. 9, provides no diagnostic characters. On the basis of color, these specimens from Inland are distinctly nearer primulina. On the basis of cranial characters they are distinctly nearer longicauda. External measurements are intermediate and are a little nearer those of primulina. By placing the most weight on the cranial characters, the animals may be referred to longicauda. The same may be said of 2 skins, one skin with a skull, from Hastings, Nebraska. In each skin the color-pattern is as in primulina; in one the under side of the tail is nevertheless lighter-colored more as in longicauda and the skull, adult male 121651 American Museum of Natural History, approaches nearer to primulina in narrowness but has the large teeth of longicauda.
Intergradation with neomexicana is suggested by one specimen, no. 7936, Univ. Kans., from Thomas County, Kansas, which has well-developed white facial markings.
The specimen, no. 180, Kansas Agric. College, from Glasco, is mounted, of large size, in white winter pelage, and lacks external measurements. On the basis of its obvious large size, and a hind foot measurement of 49 millimeters obtained from the mounted skin, the animal is provisionally referred to longicauda rather than to primulina.
Putorius culbertsoni is a name now credited to Coues (1877:136). Although Coues probably intended only to indicate that Baird wrote this name on the labels of two specimens in the mammal collection of the Smithsonian Institution, Coues gave an "indication" of the application of the name by publishing at the same time the catalogue numbers of specimens whose labels bore the name and thus, in accordance with article 21 of the International Rules of Zoölogical Nomenclature, himself becomes the author of the name. Of the two specimens mentioned by Coues, only the first recorded by him, no. 4320 (with skull no. 37995, U. S. Nat. Mus.), can now be found.
Fortunately, the skull of this specimen labeled (see Lyon and Osgood, 1909:218) as taken at Fort Laramie, Wyoming, is well preserved. Its only defects are a fracture in the left zygomatic arch and the absence of parts of each of the first lower molars. In deciding on the subspecific application of the name Putorius culbertsoni Coues, the skull of the type must be principally relied upon, for there is available only one other specimen, a skin only (no. 12596, U. S. Nat. Mus.), from the same place, and it, like the type, is in white winter pelage and lacks flesh measurements.
The ranges as now known of three subspecies of Mustela frenata approach near to Fort Laramie. These are M. f. longicauda, M. f. alleni, and M. f. nevadensis. The skull of the type of culbertsoni is not typical of any one of the three mentioned races. The small size of its teeth and relative (to basilar length) shallowness of the frontal region of the skull through the postorbital processes of the frontal are as in nevadensis. The zygomatic arches are not so greatly expanded as in some specimens of longicauda and are more like the average for nevadensis or alleni, as also is the relatively (to basilar length) long orbitonasal length. However, each of these characters is subject to variation and alone is not surely diagnostic, especially toward the margin of the range of any one of the subspecies concerned. The same may be said of the relatively great breadth of the skull interorbitally—a feature typically found in longicauda. More important, in my estimation, is the large size of the skull; all parts measured (excepting the teeth, the depth at the posterior border of the last upper molars, the zygomatic breadth, and the depth of the tympanic bullae) equal or approach nearest to the average for males of longicauda of similar age.
The small size of alleni prevents its identification with culbertsoni. The question of application lies between nevadensis and longicauda. If the long-tailed weasel at Fort Laramie is found to be referable to the race earlier named longicauda, no change in current nomenclature will be effected. If, on the other hand, the long-tailed weasel from Fort Laramie is found to be referable to nevadensis this name will have to fall before the earlier proposed name culbertsoni. There is, however, a third possibility, namely, that the long-tailed weasel of the Transition and Upper Sonoran zones of southern Wyoming and northern Colorado, as for example, at Lay, Colorado, may represent a recognizable race characterized by size about as in longicauda, relative proportions of skull about as in nevadensis and coloration intermediate, to which the name culbertsoni may apply. For more detailed discussion of this possibility, see remarks under M. f. nevadensis.
Satisfactory application of the name Putorius culbertsoni Coues requires an adequate series of adult specimens, of both sexes in the summer coat with external measurements taken in the flesh, from the type locality and like material from elsewhere in southern Wyoming. On the evidence furnished by the skull of the type of culbertsoni, that name tentatively is placed in the synonomy of longicauda.
Only 2 of 25 adults examined for malformation of the frontal sinuses by parasites showed evidence of disease.
Specimens examined.—Total number, 138, arranged alphabetically by provinces and states and further by districts or counties from north to south except as otherwise indicated. Unless otherwise indicated specimens are in the collection of the United States National Museum.
Alberta. St. Albert, 1; S. Edmonton, 3; Islay, 4[77]; Battle River, south of Camrose, 1[77]; Daysland, 1[77]; Moose Pass, 1; Blindman River, 2 (1[75], 1[2]); Red Deer, 3 (2[2], 1[60]); Bearberry Creek near Sundre, 1[77]; Canad. Nat. Park, N.W. Territory, 1[60]; Red Deer River, Didsbury, 1; Canmore, 1; Calgary, 11 (6[60], 2[1], 1[86]); Red Deer River, 3[2]; Little Sandhill Creek, Red Deer River, 1[77]; Waterton Lake Park, 2[77]; Sweetgrass Hills, 1[77]; Alberta, 1[14].
Colorado. Yuma County: Wray 4 (1[88], 3[74]).
Kansas. Rawlins County: 7 mi. N, 3 mi. W Beardsley, 1[74]; 6 mi. S and 2 mi. E Atwood, 1[74]; 15 mi. SE Atwood, 1[74]. Thomas County: near Brewster, 2[93]; no locality more definite than county, 2[93]. Trego County, 2 (1[2]). Cloud County: Glasco, 1[67].
Manitoba. Portage la Prairie, 3[75]; Carberry, 2 (1[2], 1[1]); Carman, 1[60]; Max Lake, Turtle Mt., 1[77].
Montana. Glacier County: St. Marys Lake, 1; Blackfoot, 1: Blackfoot Agency, 1. Blaine County: 6 mi. east Chinook, 1[74]. Pondera County: 1/2 mi. SE Conrad, 1[74]. Toole County: Shelby Junction, 1. Hill County: Havre, 1. Fergus County: Moccasin Mts., 5 mi. NW Hilger, 1; 7 mi. NE Hilger, 1. Rosebud County: 3/4 mi. N Ingomar, 1. County in question, Milk River, 2.
Nebraska. Dawes County: Chadron, 2[35]. Cherry County: Kennedy, 1; no locality more definite than county, 1. Brown County: Long Pine, 1[68]. Antelope County: Neligh, 1[35]. Adams County: Hastings, 2[2]. Clay County: Inland, 4[35].
North Dakota (arranged by counties from west to east). Divide County: Crosby, 1. Mountrail County: Lostwood, 1. Little Missouri River, 1. Golden Valley County: Sentinel Butte, 1. Billings County: Medora, 1[60]. McLean County: 3 mi. W Elbowoods, 1. Oliver County: Ft. Clark, 2. Morton County: Mandan, 1. Sioux County: 3 mi. N Cannonball, 1. Logan County: 6 mi. SW Napoleon, 1. Rolette County: Turtle Mts., 1[76]; Fish Lake, 1. Benson County: Ft. Totten, 3[14]; Sully Hill Nat. Park, 1. Ramsey County: Devils Lake, 2. Stump Lake or Turtle Mts., 2[2]. Nelson County: Stump Lake, 1. Grand County: Larimore, 1. Walsh County: Grafton, 11 (4[76], 3[74], 2[2]). Stutsman County: Jamestown, 1. Barnes County: Valley City, 1.
Saskatchewan. Wingard, 5; Osier, 2[75]; Simpson, 1[2]; Touchwood Hills, 4[7]; South arm Last Mountain Lake, 1[77]; Rush Lake (Assiniboia, N.W.T.), 2[75].
South Dakota. Pennington County: Rapid City, 1.
Wyoming. Goshen County: Fort Laramie, 2.
Mustela frenata oribasus (Bangs)
Long-tailed Weasel
Plates 16, 17, 18, 31, 32, 33 and 40
Putorius (Arctogale) longicauda oribasus Bangs, Proc. New England Zoöl. Club, 1:81, December 27, 1899.
Putorius longicauda, Coues, Fur-bearing animals, p. 136, 1877 (part).
Mustela longicauda oribasus, Miller, U. S. Nat. Mus. Bull., 79:98, December 31, 1912.
Mustela longicauda oribasa, Hall, Univ. California Publ. Zoöl., 40:368, November 5, 1934.
Mustela frenata oribasa, Hall, Carnegie Instit. Washington Publ. 437:105, November 20, 1936.
Type.—Female, adult, skull and skin; no. 9058, collection of E. A. and O. Bangs, but now in collection of Mus. Comp. Zoöl.; source of Kettle River, 7500 feet [the summit between middle fork of Kettle River and Cherry Creek at Pinnacles—oral information from the collector, Feb. 12, 1936], British Columbia; September 10, 1898; obtained by Allan Brooks; original no. 1368.
The skull (plate 40) is complete and unbroken. The teeth all are present and entire except right I3 which has the anterior half broken away. The skin is complete, fairly well made, and in summer pelage.
Range.—Canadian and Hudsonian life-zones from near 56°N in the Rocky Mountains of British Columbia and Alberta and Ootsa Lake along the Fraser and Chilcotin rivers south to Alta Lake, in the Caribou and Monashee mountains, probably in the Selkirks and Rockies, and through the Rockies of Montana into extreme northern Wyoming. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. longicauda by near (14 n) Brussels Brown rather than near (h) Clay Color of upper parts and in males by relatively shallower occiput in which the depth of the skull, exclusive of the sagittal crest and taken at the anterior border of the basioccipital, amounts to less than 59 per cent of the mastoid breadth; from M. f. nevadensis by greater average size, see measurements.
Description.—Size.—Male: Two adults from Florence, Montana, measure as follows: Total length, 440, 440; length of tail, 165, 161; length of hind foot, 47, 49. Corresponding measurements of an adult male from Quesnel, British Columbia, are: 443; 168; 55. Tail amounts to 60, 58, and 61 per cent as long as head and body. Length of hind foot averages more than basal length.
Female: The type specimen, the only typical adult or subadult specimen of this sex of which external measurements are available, measures: Total length, 392, length of tail, 150, length of hind foot, 46. Tail is 63 per cent as long as head and body. Length of hind foot amounts to more than basal length.
The differences in external measurements, between the one female and the average of the three males are: Total length, 49; length of tail, 15; length of hind foot, 4.
Externals.—Longest facial vibrissae brown or white (often both colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to or beyond apical pad of fifth digit; hairiness of foot-soles (in summer pelage) slightly less than shown in figure 19.
Color.—Upper parts, in summer, near (14 n) Brussels Brown, more blackish and less reddish than tone 4 of Burnt Umber of Oberthür and Dauthenay, pl. 304; in type near tone 4, pl. 301 of Oberthür and Dauthenay. Underparts, in summer, Buff Yellow or near (20 c) Amber Yellow. In winter, all white except tip of tail which is at all times black. Upper parts of uniform color except for occasional slight darkening of top of head and along mid-dorsal line of back. Color of underparts extends distally on posterior sides of forelegs over feet, on medial sides of hind limbs over antiplantar faces of toes and over proximal two-thirds of ventral side of tail. Least width of color of underparts amounting to 43 per cent of greatest width of color of upper parts, 75 per cent in male from 4 miles northeast of Quesnel, British Columbia, and 52 (33-66) in four males from Montana. Black tip of tail in four males from Montana averaging 50 (44-60) mm. long. Thus averaging approximately as long as hind foot and 33 per cent of length of tail-vertebrae.
Color not different than in many specimens of M. f. nevadensis. Color comparison with M. f. longicauda has been made in the account of that subspecies.
Skull and teeth.—Male (based on 5 adults and 2 subadults from British Columbia and 4 adults from Montana): See measurements and plates 16-18. As described in Mustela frenata longicauda except that: Weight, 5.0 (3.8-6.0) grams; basilar length, 46.7 (43.6-48.8); postorbital breadth in one of nine instances less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 2-1/2 to 5 upper incisors; length of tympanic bulla not less than length of lower molar and premolar tooth-row and shorter than rostrum.
Female (based on the type, specimen): See measurements and plates 31-33, 40. As described in Mustela frenata longicauda except that: Weight, 3.5 grams; basilar length, 41.6 mm.; zygomatic breadth more than distance between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate more than outside length of P4; tympanic bulla as far posterior to foramen ovale as width of 4-1/2 upper incisors; height of tympanic bulla less than distance from anterior margin of tympanic bulla to foramen ovale; length of tympanic bulla less than length of rostrum. If more than one skull were available of the female of oribasus it is believed that the description would agree with that of longicauda in nearly all features.
The skull of the female is 30 per cent lighter than that of the average male.
Comparison with longicauda reveals that, on the average, skulls of males are larger, relative to the basilar length broader across the mastoids, shallower through the braincase as measured at the anterior end of the basioccipital exclusive of the sagittal crest, with longer rostrum. Compared with nevadensis, the skull averages larger in all measurements taken, and has a relatively broader rostrum, relatively greater mastoid breadth and a braincase which is shallower relative to the basilar length. By weight, the skull of nevadensis is a fourth lighter, and in linear measurements 5 to 18 per cent smaller.
Remarks.—Some of the specimens from Montana, which here are referred to oribasus, more than half a century ago were listed by Coues (1877:138) under the name longicauda. It was not until 1899 that this race was given a name by Bangs, who at that time (1899B:81) accurately made out the distinctive color features. Distinctive cranial characters cannot be described with assurance even now because there still are too few specimens.
The type specimen, at one time examined by the present writer, has on the stuffed skin no well-developed mammae, scrotal pouch, or other visible sexual part. Probably the collector's sex mark for female is correct.
As judged by the two skulls of subadult males from the Barkerville region, individuals of this race attain larger size than do those of longicauda. On the basis of larger size than either longicauda or nevadensis, the specimens from the Rocky Mountains of Montana and two from northern Wyoming are referred to this race. The short, wide, flat, tympanic bullae, relatively great mastoidal breadth, and some other features of the specimen from Donovan, Montana, point toward oribasus, whereas nearly as many more cranial features, in this instance mainly differences in size, are indicative of nevadensis to which race the specimen might almost equally well be referred. Another male from Darby, in the Bitterroot Valley of Montana, has a slightly longer hind foot than those from Florence, but a female from Hamilton, agrees more nearly with nevadensis. The average of all the specimens from the Bitterroot Valley is a little nearer oribasus. Four skulls from Buffalo, Wyoming, here referred to nevadensis show approach to oribasus in size of skull. The specimens from Big Snowy Mountains, and the Highwood Mountains of Montana are too young clearly to show size of the adult skull, but are distinctly darker colored than longicauda of the plains country proper. Of two subadult females from Tacy, Montana, the color of the one in summer pelage is distinctly nearer that of oribasus and nevadensis than it is to that of longicauda to which some approach in color might be expected. The reduced size of both of the specimens is further suggestive of nevadensis and it may be that adult specimens from these more eastern mountainous areas in Montana will show that nevadensis is the name proper to apply to animals of this region.
Intergradation with nevadensis is suggested by specimens collected from along the upper reaches of Okanagan Lake, British Columbia, by Major Allan Brooks and Mr. J. A. Munro and by a series of skulls from Ione, Pend Orielle County, Washington, lent me by Mr. Walter Dalquest. At each place, the average of all specimens is nearest to that of nevadensis.
Specimens from near Waterton Lake show several steps in the transition from the light-colored longicauda type of coloration to the darker coloration characterizing oribasus. One taken here, at a time when the body of water referred to seems to have been known as Chief Mountain Lake, is barely dark enough to be placed with oribasus. Two other specimens from across the Canadian Border labeled as "Waterton Lake Park" are slightly lighter colored above, and on this account are placed with longicauda.
The two adult males from Lillooet, British Columbia, are referable to oribasus although neither is quite typical. One has a saturated coloration suggestive of that of altifrontalis and the skull is shorter and broader than in other specimens of oribasus. The female from Lillooet, skin alone, no. 916, Prov. Mus., B. C. is small for oribasus. The female, no. 1539, collection of Kenneth Racey, from Alta Lake, in brown winter pelage, in almost every measurement falls nearly midway between altifrontalis and oribasus but slightly nearer the latter. The skull from Chezacut and 3 animals from Wistaria, British Columbia, probably are females and show a greater average size than specimens from farther to the southeast. For example, the basilar length of the skull, 44.8 (44.3 to 45.1), exceeds that of the type specimen. The animals from Wistaria on Ootsa Lake furnish the northwesternmost station of occurrence of which I have record for this subspecies.
The northernmost records of occurrence, at "Clearwater River, Peace River, B. C," and at Little Prairie, are furnished by a white skin without skull, no. 257450, U. S. Nat. Mus., purchased on August 2, 1932, at the place mentioned by W. H. Sheldon and Richard Borden, and a skull with white winter skin, no. 3585, Provincial Museum, British Columbia, respectively. The characters distinguishing longicauda and oribasus are not shown by white winter skins; the skull shows some features of longicauda, and the reference of these specimens to oribasus rather than longicauda is tentative.
Only the skull from Little Prairie shows evidence of infestation of the frontal sinuses by parasites. In the Barkerville area of British Columbia, Mr. and Mrs. Thomas T. McCabe obtained only 2 skulls of this subspecies from a total of 238 weasel skulls gathered by local trappers. The others were Mustela erminea.
Specimens examined.—Total number, 46, listed by localities from north to south and unless otherwise indicated, in the United States National Museum.
British Columbia. West of Hudson Hope, 1[7]; Clearwater River, tributary to Peace River, 1; Little Prairie, a few miles south of Peace River and about 40 miles west of the main highway between Dawson Creek and Fort St. John, 1[85]; Wistaria, 3[85]; Four Mile Creek, 4 mi. NE Quesnel, 1[21]; Isaacs Lake, 3200 ft., 1[74]; Barkerville region, 1[74]; Clear River, 4800 ft., 1[74]; Chezacut, 1[31]; Lillooet 3 (2[77], 1[85]); Alta Lake, 1[31]; source of Kettle River, 7500 ft., 1[75]; E side Beaverfoot Range, 4000 to 4500 ft. between Fraser Creek and 6 mi. SE of Fraser Creek, 1[74]; Cranbrook, 1[86]; head of Cross River, 10 mi. below Assiniboine Pass, 1[7]; camp east of "Kootanie," 1[7]; camp east of Kootanie River, 1[7].
Alberta. Thoral Creek, 7000 ft., 50 mi. NE Jasper, 1[2].
Montana. Glacier? County: Chief Mt. Lake (= Waterton Lake), 1. Flathead County: Columbia Falls, 1. Chouteau? County: Highwood Mts., 1. Fergus? County: Big Snowy Mts., 1. Wheatland County: Harlowton, 1[74]. Ravalli County: Florence, 2; Hamilton, 1[56]; Darby, 1[56]; Carlos [= Charlos] Heights, 2[74]; Tin Cup District, 2[74]; no locality more definite than county, 2[74]. Beaverhead County: Donovan, 1. Madison County: Sheridan, 1[74]. Gallatin County: Ranch 7-11, Eldridge, 1[60]. Stillwater County: Tacy, 2[76]. County in question: Gallatin Valley, 1; Yellowstone Park, 1[75].
Wyoming. Glen Creek, Mammoth Hot Springs, 1. Park County: Four Bears, 1[2].
Mustela frenata alleni (Merriam)
Long-tailed Weasel
Plates 18, 19, 20, 31, 32 and 33
Putorius alleni Merriam, N. Amer. Fauna, 11:24, June 30, 1896.
Mustela alleni, Miller, U. S. Nat. Mus. Bull., 79:99, December 31, 1912.
Mustela frenata alleni, Hall, Carnegie Instit. Washington Publ. 473:106, November 20, 1936.
Type.—Male, adult, skull and skin; no. 186451, U. S. Nat. Mus. (formerly 4485/5120, collection of Dr. C. Hart Merriam); Custer, South Dakota; obtained by Vernon Bailey; original no. 90.
The skull is complete and unbroken. The upper incisors are missing. All the other teeth are present although the premolars, and especially the canines, are much worn, possibly as the result of the animal's efforts to free itself from a trap. The skin is fairly well made, in a good state of preservation, and entire.
Range.—Canadian, Transition and Upper Sonoran life-zones of the Black Hills of South Dakota and adjacent semi-bad-land territory of Wyoming and Nebraska southward to Mitchell, Scottsbluff County. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. longicauda in smaller size, adult males having a total length of less than 400, hind foot less than 45, basilar length less than 43.5, and in adult females total length less than 375, and basilar length less than 40; from M. f. nevadensis in near Clay Color rather than near (14 n to l) Brussels Brown of upper parts in summer.
Description.—Size.—Male: External measurements of the type specimen are: Total length, 372; length of tail, 137; length of hind foot, 44. Tail is 58 per cent as long as head and body. Length of hind foot more than basal length.
Female: No external measurements for typical adults are available. No. M1 #41 from Mitchell, Scottsbluff Co., Nebraska, an adult female which is an intergrade with the larger M. f. longicauda, measures as follows: Total length, 367; length of tail, 120; length of hind foot, 41.
Externals.—Longest facial vibrissae dark brown or white and extending beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles (in summer pelage) as shown in figure 20.
Color.—Winter pelage unknown; probably white except, of course, tip of tail. Summer pelage as described in Mustela frenata longicauda except that: Least width of color of underparts averaging, in 3 males from Black Hills, 54 (38-62) per cent of greatest width of color of upper parts. Black tip of tail averaging 43 (40-45) mm. long. Thus, averaging approximately same length as hind foot and in type specimen amounting to 33 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on the type and no. 7440 Amer. Mus. Nat. Hist., from Hill City, S. Dak.): See measurements and plates 18-20. As described in Mustela frenata longicauda except that: Weight, 3.1 (3.0-3.2) grams; basilar length, 41.0 (40.9-41.0); mastoid breadth not less than postpalatal length; breadth of rostrum more than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4 to 5 upper incisors; height of tympanic bulla more or less than distance from its anterior margin to foramen ovale.
Female (based on no. 7441, American Mus. Nat. Hist., from Black Hills, S. Dak.): See measurements and plates 31-33. As described in Mustela frenata longicauda except that: Weight, 2.0 grams; basilar length 37.6. The skull of the female is 35 per cent lighter than the average for the two males.
Comparison with M. f. longicauda and M. f. nevadensis reveals that the tympanic bullae average more nearly flat and that the skull is smaller.
Remarks.—Animals of this subspecies were described and named by Merriam in 1896 as a distinct species on the basis of two or possibly three specimens from the Black Hills of South Dakota and the name seems never to have been applied to specimens from other regions. Vernon Bailey obtained only the one specimen, the type, on his trip in 1888, but two more were obtained for the American Museum of Natural History by Walter Granger in 1894.
Mustela frenata alleni combines the light coloration of M. f. longicauda with the small size of M. f. nevadensis. Indeed, the size may average less than that of nevadensis. M. f. alleni seems to reach its extreme of small size in the Black Hills of South Dakota. Specimens from Mitchell, Scottsbluff County, Nebraska, here referred to alleni are of larger size and in this respect are intermediate between the subspecies alleni and longicauda. Of the two specimens available from Chadron, Nebraska, and here referred to as longicauda, the female, M1 #6, is almost exactly intermediate in size between alleni and longicauda, whereas the male, M1 #11, is as large as the average-sized longicauda.
None of the nine skulls (5 adults) shows malformation resulting from the infestation of the frontal sinuses with parasites.
Specimens examined.—Total number, 10, as follows.
Wyoming. Crook County: Sundance, 1[91].
South Dakota. Pennington County: Hill City, 1[2]; 20 mi. N Elk Mt, 1[91]. County in question: Black Hills, 1[2]. Custer County: Custer, 2 (1[91], 1[2]).
Nebraska. Scottsbluff County: Mitchell, 4[35].
Mustela frenata arizonensis (Mearns)
Long-tailed Weasel
Plates 19, 20, 21, 31, 32 and 33
Putorius arizonensis Mearns, Bull. Amer. Mus. Nat. Hist., 3:234, June, 1891; Merriam, N. Amer. Fauna, 11:22, fig. 12, June 30, 1896.
Mustela arizonensis, Miller, U. S. Nat. Mus. Bull., 79:99, December 31, 1912.
Mustela frenata arizonensis, Hall, Carnegie Instit. Washington Publ. 473:106, November 20, 1936.
Type.—Female, adult, skull and skin; no. 2490/1886, Amer. Mus. Nat. Hist.; San Francisco Forest [then (1886?), Yavapai County], Arizona; June 20, 1886; obtained by Edgar A. Mearns.
The skull (plates 31-33) is complete and unbroken save for a small puncture in the right squamosal. The incisors above and below and M2 and P2 on each side are missing. Four canines are preserved separately. Otherwise the teeth are in place. The skin has been taken down from a mount. Some hair has been lost from in front of the ears. Seven mammae are evident and show the animal to have been nursing young. The slightly faded color was mentioned by Mearns in the original description. He says (1891:234): "The memorandum of the colors was made before skinning, the specimen having been subsequently preserved in a solution of alum and salt, which extracted much of the coloring matter."
Range.—Transition to Hudsonian life-zones of Arizona and extreme western New Mexico, along the Colorado River, and south of the Little Colorado River, from San Francisco Mountain region along Mogollon Plateau to extreme western New Mexico. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. neomexicana by near (14 n) Brussels Brown rather than Buckthorn Brown color of upper parts, in absence rather than presence of white frontal spot continuous with color of underparts, in basilar length of less than 44 in males and 39.3 in females; from M. f. nevadensis in that total length averages less than 375 in males and 330 in females, basilar length averaging less than 41 in males and less than 36.7 in females.
Description.—Size.—Male: No. 24679/32071, from Springerville, and no. 248993 from the Kaibab Plateau, measure respectively, as follows: Total length, 363, 367; length of tail, 140, 143; length of hind foot, 41.5, 41.0. Tail is 63, and 64 per cent as long as head and body. These males, the only specimens of that sex of which external measurements are available, probably are grading toward nevadensis and therefore are nontypical.
Female: Three specimens, one young from Little Spring, a subadult from Deadmans Flat and the type specimen, measure respectively as follows: Total length, 323, 296, 302; length of tail, 110, 101, 109; length of hind foot, 38, 33, 36. These average, 307, 107, 36. Tail averages 53 per cent as long as head and body.
Differences in external measurements of the two sexes are: Total length, 56; length of tail, 39; hind foot, 5.5.
Externals.—Longest facial vibrissae black, brown or white (often all three colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot soles (in summer pelage) about as shown in figure 19.
Color.—Winter pelage unknown. Summer pelage with upper parts near (14 n) Brussels Brown or tone 2 of Raw Umber of Oberthür and Dauthenay, Pl. 301, darker on top of head from nose to line connecting posterior margins of ears. Tip of tail always black. Chin and upper lips white. Remainder of underparts Buff Yellow to Straw Yellow and rarely Ochraceous Buff. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists, on medial sides of hind legs to ankles and over antiplantar faces of toes, medial third of tarsus, and over proximal fifth to fourth of ventral side of tail. Least width of color of underparts averaging, in 8 specimens, 44 (29-54) per cent of greatest width of color of upper parts. Black tip of tail, in four females averaging 35 (33-38) mm. long. Thus, averaging shorter than hind foot and 32 per cent of length of tail-vertebrae. Three of the eight specimens before me (no. 242671 from 25 mi. SE Flagstaff, not available at time of this accounting) have the dark spot near the angle of the mouth faintly indicated, whereas the other five lack the spots. The color is as in M. f. nevadensis.
Skull and teeth.—Male (based on 55211, 65231, and 248993; see page 422): See measurements and plates 19-21; weight 2.7 and 3.1 grams; basilar length, 40.4; zygomatic breadth more than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth more than postpalatal length; postorbital breadth less than length of upper premolars and more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate more or less than medial length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 3-1/2 (including I3) upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar-premolar tooth-row and longer or shorter than rostrum; anterior margin of masseteric fossa below talonid of m1.
Female (based on the type specimen): See measurements and plates 31-33; weight, 1.6 grams; basilar length, 35.5; zygomatic breadth less than distance between condylar foramen and M1 and more than distance between anterior palatine foramen and anterior margin of tympanic bulla (nearly equal in each instance); postorbital breadth less than length of upper premolars and greater (7.1-8.4) than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate equal to inside length of P4; tympanic bulla as far posterior to foramen ovale as width of 3 (including I3) upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar-premolar tooth-row and greater than length of rostrum.
The skull of the female averages 41 per cent lighter than that of the male.
Compared with the skull of M. f. nevadensis, that of arizonensis is smaller, less heavily ridged and has more inflated tympanic bullae and a relatively greater mastoid breadth. Comparison with the skull of M. f. neomexicana is made in the account of that subspecies.
Remarks.—In 1891 Mearns (234-235) named this weasel as a full species on the basis of two individuals taken by him in 1886 and 1887. Since that time only a few additional specimens have been preserved. Only four are adults. Although this material does not permit of a definition of the subspecies as precise as could be wished, still, it clearly shows that the animals from the plateau region of Arizona are recognizably different from those farther north in the Sierra Nevada of California and those of the Rocky Mountains and Great Basin region northward to the Canadian border. These more northern animals have gone by Mearns' name, arizonensis, since the date of its proposal until 1939 when the name nevadensis was proposed.
The smaller size, especially of the skull, and the greater inflation of the tympanic bullae are the outstanding characters which distinguish arizonensis from the similarly marked nevadensis. The bullae are relatively much inflated throughout but especially so on the posteromedial parts.
Although the three adult males and two subadult females available of this subspecies are smaller in most parts measured than any of the scores of nevadensis of similar age that have been measured, overlap in size probably will be found as additional specimens of arizonensis become available. A young female, no. 18513, coll. D. R. Dickey, from Little Spring, does have certain cranial measurements as large as are found in the minimum-sized nevadensis from farther north.
Intergradation with the two subspecies whose geographic ranges adjoin that of arizonensis is indicated by specimens at hand. One of these is the adult male from 25 miles southeast of Flagstaff, which shows decided approach to neomexicana, in color and in possessing white facial markings less well developed than in neomexicana. Even better developed white facial markings, with intervening blackish coloration, are displayed by no. 148271, U. S. Nat. Mus., from 8500 feet altitude on Willow Creek, New Mexico. This subadult female shows approach to neomexicana also in larger size of the skull and entire animal. The great inflation of the posterior part of each of its bullae and the dark color of the upper parts are characters of arizonensis. The color of the underparts stops at the ankles leaving the hind feet dark colored, in which respect the specimen is unlike either neomexicana or arizonensis. If additional specimens showing the same characters as this one be found at other nearby localities they probably should be given recognition as a separate subspecies. For the present it seems best to regard the specimen merely as an intergrade. Although it might, with almost equal propriety, be referred to either neomexicana or arizonensis, the specimen is here placed with the latter. The subadult male from Springerville, Arizona, is of larger size than the topotypical male of arizonensis and in this respect shows slight approach to nevadensis. The narrower mastoidal breadth and slightly less inflated tympanic bullae of the male from the Kaibab Plateau may reflect merely individual variation or may represent intergradation in these features with nevadensis.
The statement made by Merriam (1896:22) that, "The type specimen . . . is an immature female and is of unusually small size. A male obtained by him [Mearns] near the same place is of the normal size, as is another male in the Department collection from Springerville, Ariz., collected by E. W. Nelson," needs correction. The female is not immature. The specimen obtained by Mearns near the same place probably refers to Amer. Mus. No. 2489, from Quaking Asp Settlement, which lacks both the skull and external measurements. As stuffed it is of small size for a male. The male from Springerville, as shown by the external and cranial measurements, is not of normal (i. e. average) size, but is smaller than the average for the other populations of similarly colored weasels referred to by Merriam (op. cit.) as arizonensis but here described under the name nevadensis.
None of the skulls shows signs of infestation of the frontal sinuses by parasites.
Specimens examined.—Total number, 17, arranged alphabetically by states and from north to south by counties in each state. Unless otherwise indicated specimens are in the collection of the United States National Museum.
Arizona. Coconino County: VT Park, Kaibab Plateau, 1; Deadman Flat, 6400 ft., 1[74]; Little Spring, 1[59]; Government Prairie, near Parks, 1[74]; Coconino? County: San Francisco Forest (Yavapai Co., in 1886), 1[2]; 25 mi. SE Flagstaff, 1; Quaking Asp Settlement, 1[2]. Apache County: Springerville, 1; North Fork White River, White Mts., 8200 ft., 4[87]; head San Francisco River, Judd Ranch, Alpine, 1[74]; 2 mi. SE Big Lake Knoll, 8700 ft., 24 mi. S Springerville, 1[74]. Greenlee County: S end Blue Range, 9000 ft., Prieto Plateau, 1; Beaver Creek, 7000 ft., 1[74].
New Mexico. Grant County: Mogollon Mts., Willow Creek, 8500 ft., 1.
Mustela frenata nevadensis Hall
Long-tailed Weasel
Plates 19, 20, 21, 33, 34, 35 and 39
Mustela frenata nevadensis Hall, Carnegie Instit. Washington Publ. 473:91, November 20, 1936.
Putorius longicauda, Coues, Fur-bearing animals, p. 136, 1877 (part); Merriam, N. Amer. Fauna, 5:83, July 30, 1891.
Putorius (Gale) brasiliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877 (part).
Putorius arizonensis, Merriam, N. Amer. Fauna, 11:22, figs. 13, 14, June 30, 1896 (part); Stephens, Mammals of California, p. 247, 1906.
Mustela arizonensis, Grinnell and Swarth, Univ. California Publ. Zoöl., 10:376, October 31, 1913; Whitlow and Hall, Univ. California Publ. Zoöl., 40:247, September 30, 1933.
Mustela arizonensis arizonensis, Grinnell, Univ. California Publ. Zoöl., 40:102, September 26, 1933.
Mustela frenata, Boyer, Journ. Mamm., 24:99, February 20, 1943.
Type.—Female, adult, skull and skin; no. 41053, Mus. Vert. Zoöl.; three miles east Baker, White Pine County, Nevada; May 30, 1929; obtained by E. R. Hall and W. C. Russell; original no. 2674, E. R. H.
The skull (plates 33-35) is complete and unbroken. The teeth all are present and entire. The skin is fairly well made. Eight mammae are evident and show the animal to have been nursing young.
Range.—Altitudinally, 700 feet at Wenatchee, Washington, to the highest parts of the mountains of the western United States; Upper Sonoran Life-zone to Arctic Alpine Life-zone; southern British Columbia in the Cascades and territory west to Monashee Mountains, and Nelson, southward in the Cascades of northern Washington, over western Washington, Idaho, Utah, and Nevada to northeastern Arizona and northern New Mexico; westward from the eastern base of the Rocky Mountains in Colorado to the western base of the Sierra Nevada and Cascades of California and to the Cascades of southern Oregon. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. oribasus by smaller average size, see measurements; from M. f. longicauda by near (14 n to l) Brussels Brown rather than near (h) Clay Color of the upper parts, and in males by a shallower occiput in which the depth of the skull, exclusive of the sagittal crest, and taken at the anterior border of the basioccipital, amounts to less than 59 per cent of the mastoid breadth; from M. f. alleni by near (14 n to l) Brussels Brown rather than near (h) Clay Color of upper parts in summer; from M. f. neomexicana by near (14 n to l) Brussels Brown rather than Buckthorn Brown color of upper parts, in absence of white frontal spot continuous with color of underparts, in basilar length of less than 46 in males and 40 in females; from M. f. arizonensis by total length averaging more than 375 in males and 330 in females, basilar length averaging more than 41 in males and 36.7 in females; from M. f. inyoensis by absence of white facial markings; from M. f. pulchra by absence of light facial markings, near (14 n to l) Brussels Brown rather than near (16 j) Buckthorn Brown color of upper parts, and lesser size, hind foot less than 40 in females and basilar length averaging less than 46.0 in males; from M. f. xanthogenys by absence of light facial markings and near (14 n to l) Brussels Brown rather than Buckthorn Brown color of upper parts; from M. f. munda by absence of white facial markings, presence of color of underparts on ventral face of proximal third of tail, and hind foot of less than 50 in males; from M. f. saturata by presence of light color of underparts on tail and ankle and in lesser average breadth across mastoid processes of skull (see measurements); from M. f. oregonensis by absence of nasofrontal white patch, presence of light color of underparts on ventral face of tail, and shorter skull, which, relative to its length in males, is deeper through the braincase; from M. f. washingtoni by presence of light color of underparts on ventral face of tail, by skull which in male relative to basilar length is shorter in the preorbital region and wider across the zygomata and mastoid processes, and in female has longer preorbital region and larger bullae (see measurements); from M. f. altifrontalis by lighter colored upper parts which are tones 1 to 3 of Raw Umber, pl. 301, rather than tone 4 of Brownish Drab, pl. 302, of Oberthür and Dauthenay, by Buff-Yellow to Straw Yellow rather than near (14´ a to 16´ c) Ochraceous-Buff color of underparts, by least width of color of underparts amounting to more than 37 per cent of greatest width of color of upper parts, by presence of color of underparts on ventral side of tail and on hind leg over ankle, and by lesser depth of skull through frontal region; from M. f. effera by larger size, males averaging 12-1/2 per cent larger in external measurements, 8 per cent larger in linear measurements of skull, and 22 per cent heavier in weight of skull, total length averaging 400 rather than 360, basilar length averaging 43.6 rather than 40.5.
Description.—Size.—Male: Twenty-one adults from the southern half of the Sierra Nevada of California yield average and extreme measurements as follows: Total length, 400 (356-428); length of tail, 150 (125-178); length of hind foot, 46.1 (42-50). Tail averages 60 per cent as long as head and body. Length of hind foot averaging more than basal length. Corresponding measurements of twelve adults from extreme southern and southwestern Colorado are as follows: 407 (355-431); 150 (133-170); 46.0 (42-49).
Female: Ten adults from the Sierra Nevada of California yield average and extreme measurements as follows: Total length, 349 (336-362); length of tail, 127 (120-133); length of hind foot, 36.3 (32-39). Tail averages 57 per cent as long as head and body. Length of hind foot less than basal length. Corresponding measurements of ten adults from the Rocky Mountains of central Colorado are as follows: 347 (325-375); 123 (111-141); 40 (32-43).
The average differences in external measurements of the two sexes, in the Sierras of California are: Total length, 51; length of tail, 23; length of hind foot, 9.8. Weight of 7 adult males from California is 267 (226-345) grams. Two adult females from there weigh 148 and 115 grams and 3 from White Pine County, Nevada, 134, 122 and 124, giving an average of 129 grams.
Externals.—Longest facial vibrissae black, brown or white (often all three colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles (in summer pelage) about as shown in figure 19.
Color.—Upper parts, in summer, near (14 n to l) Brussels Brown or tones 1 to 3 of Raw Umber of Oberthür and Dauthenay, pl. 301, darker on top of head from nose to line connecting posterior margins of ears. Chin and upper lips white. Remainder of underparts Buff-Yellow to Straw Yellow and sometimes Ochraceous-Buff especially in young, and in some adults from southern Colorado. In winter, all white, except tip of tail, or upper parts near (j) Snuff Brown or lighter than Brussels Brown with a smoked effect, and underparts white. Tip of tail at all times black. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists, on medial sides of hind legs to ankles, over antiplantar faces of toes, medial third of tarsus and usually over proximal tenth to three-fourths of ventral side of tail. Least width of color of underparts averaging, in a series of twenty males from the southern half of the Sierra Nevada of California, 59 (37-76) per cent of greatest width of color of upper parts. In seven males from southern Colorado corresponding percentages are 55 (37-71). Black tip of tail in series from Sierra Nevada averaging 50 (40-60) mm. long; thus longer than hind foot and averaging 33-1/3 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 25 adults, from Sierra Nevada of California): See measurements and plates 19-21; weight, 3.7 (2.9-4.9) grams; basilar length, 43.6 (40.6-46.1); zygomatic breadth more than distance between condylar foramen and M1 (save in four instances) and more than distance between anterior palatine foramen and anterior margin of tympanic bulla (save in two specimens); mastoid breadth more (80 per cent of specimens) or less (20 per cent) than postpalatal length; postorbital breadth less than length of upper premolars and more or less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate less than medial length of P4 (except in two specimens); anterior margin of tympanic bulla as far posterior to foramen ovale as width of 3 to 5 upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum; anterior margin of masseteric fossa not carried farther forward than point directly below hypoconid of m1.
Female (based on ten adults from Sierra Nevada of California): See measurements and plates 33-35; weight, 2.2 (1.8-2.4) grams; basilar length, 38.2 (36.7-39.5); zygomatic breadth more (except in one specimen) than distance between condylar foramen and M1 and more (save in two specimens) than distance between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth less than length of upper premolars and less than (except in one specimen) width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate more or less than either outside or inside length of P4 but generally less than inside length; tympanic bulla as far posterior to foramen ovale as width of 3 to 5-1/2 upper incisors; height of tympanic bulla more or less (usually more) than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and more or less than length of rostrum.
The skull of the female averages 41 per cent lighter than that of the average male.
Compared with the skull of M. f. longicauda, that of both sexes averages smaller in every measurement taken. Males of nevadensis, on the average, relative to the basilar length, are narrower in the interorbital region and across the zygomata but have the orbitonasal length greater. Stated in another way, the rostrum of longicauda appears to be shorter and broader and the zygomata are more expanded. Females of nevadensis, on the average, relative to the basilar length are narrower across the mastoid processes and zygomata and have the braincase deeper at the anterior margin of the basioccipital. Also in nevadensis the mastoid processes do not project so far laterally beyond the braincase, the lambdoidal crest and postorbital processes are less well developed and except in the interparietal region, the temporal ridges hardly meet and they form a sagittal furrow rather than a low sagittal crest which characterizes adult females of longicauda. Each of these differences separating the females of longicauda from those of nevadensis are of the same nature, although not necessarily of the same degree, as those which appear in longicauda with increasing age. The differences mentioned above are readily appreciable when series of specimens are compared. However, none of the differences is of great degree, and most parts of the skulls of the two subspecies are of similar relative proportions. Even so, there is but little overlap in actual size. Comparisons with the skulls of M. f. oribasus, alleni, neomexicana, arizonensis, inyoensis, pulchra, xanthogenys, munda, saturata, oregonensis, washingtoni, altifrontalis, and effera are made in the accounts of those subspecies.
Remarks.—The populations to which the name nevadensis at present is assigned have gone by the name arizonensis since Mearns proposed this name in 1891. Before that time Coues (1877:141) had included individuals of this race under the name Putorius longicauda.
Among the populations here assigned to M. f. nevadensis, there is some geographic variation but it is of lesser degree than in most other species of mammals which range over the same region. Comparison of 20 adult males from the Rocky Mountains of Colorado with 25 adult males from a place as far distant as the Sierra Nevada of California shows that the two populations closely resemble each other. The specimens from Colorado average a trifle wider across the zygomata, have a longer body and therefore relatively shorter tail, and, except in southern Colorado, a slightly longer hind foot. Comparison of ten adult females from each of the two areas reveals that those from Colorado have a markedly longer hind foot, and a tail somewhat shorter relative to the length of the body. The mentioned differences are the only ones found among the great number of points investigated, except that as remarked by Merriam (1896:23) the Sierran animal has the yellow of the underparts reaching farther up under the chin, the underside of the tail on the average is more suffused with yellowish and the white on the upper lip is more extensive. As regards the last mentioned feature, my check of 34 skins from Colorado reveals that the white extends all the way around the upper lip in every specimen but one, whereas in 69 specimens from the Sierra Nevada the white extends all the way around the upper lip in only 39. However, as further remarked by Merriam (loc. cit.), not only this but the other color features are inconstant in addition to being slight. When the occurrence of the dark spots near the angles of the mouth are tabulated, it is found that in 33 Colorado-taken specimens they are absent in 19, faintly indicated in 13, and well developed in 1. In 62 California-taken specimens they are absent in 37, faintly indicated in 20, and well developed in 5.
In northwestern Colorado, southern Wyoming, and possibly through the Bear River Divide into southeastern Idaho, long-tailed weasels here referred to nevadensis approach longicauda in large size and occasionally in other features, more closely than do specimens of nevadensis from most other places in its range. This tendency is thought to be significant for much of the area in question lies in or below the Transition Life-zone, the same life zones in which farther to the eastward true longicauda occurs.
One specimen that illustrates this approach to longicauda is an adult male, no. 2334, collection of E. R. Warren, from 6160 feet, Lay, Routt [now Moffat] County, Colorado. In large size and, relative to the basilar length, shorter rostrum and shorter tympanic bullae, it agrees with longicauda but the darker color and, relative to the basilar length, narrowness of the rostrum, interorbital region, zygomatic expanse and the shallowness through the region of the postorbital processes place it with nevadensis. Of two other specimens from Steamboat Springs, Routt County, a young male, no. 4010, in the collection of E. R. Warren, has a hind foot (50 mm.) as long as in longicauda; and the other, no. 138195, U. S. Nat. Mus., an adult male, agrees well enough in size and proportions with nevadensis but has the coloration typical of longicauda.
From Wyoming, one subadult female, no. 177553, U. S. Nat. Mus., from Garrett, is intermediate in size and coloration but is nearer to nevadensis in these particulars, as it is in all other points considered except size of the molar teeth which are as large as in longicauda and larger than in any female nevadensis from Colorado or California. Another female, an adult, no. 179304, U. S. Nat. Mus., from Lonetree, Wyoming, agrees with longicauda in size of skull. Indeed, ten of seventeen cranial measurements exceed the maximum for Colorado-taken nevadensis. Where differences exist in relative proportions of the skull as expressed in percentages of the basilar length, the specimen approaches nevadensis in 5 instances and longicauda in only 3. The color is intermediate but much nearer that of nevadensis with which the animal agrees also in external measurements. Ten subadults (5 of each sex) from within 12 miles of Laramie (not Fort Laramie) show greater resemblance to nevadensis but definitely approach longicauda. Average external measurements are: ♂, 408, 155, 44; ♀, 361, 134, 40. The two other specimens examined from this general locality, a young female, no. 2711, Mus. Vert. Zoöl., from Fort Bridger, and a subadult female, no. 188377, U. S. Nat. Mus., from Bridger Pass, show no departures from nevadensis of similar age.
The specimens from scattered localities in the Transition Life-zone of northwestern Colorado and southern Wyoming are larger than nevadensis is elsewhere, and also in certain other features resemble longicauda of the plains to the eastward. Everything considered, the animals in question are much more like nevadensis than longicauda. Study of more specimens, especially from Wyoming, might provide grounds for recognizing as a different subspecies the animals in this large area comprising parts of Colorado and Wyoming from which so few specimens now are available. Possibly the name Putorius culbertsoni Coues would apply. Decision on that point will require adequate material from the type locality, Fort Laramie. See discussion of this name under M. f. longicauda.
In southeastern Idaho males are larger than they are at most other places within the range of nevadensis. An average of 7 adults and subadults from Pegram, Montpelier, Springfield, and the vicinity of Pocatello, reveals, when compared with the average of nevadensis from Colorado and that of longicauda from the Great Plains, that this population from southeastern Idaho is nearest to longicauda in linear measurements of the orbitonasal length, mastoid breadth, length of tympanic bullae, and as expressed in percentage of the basilar length, length of tooth-row, breadth of rostrum, and zygomatic breadth. In all other points of size, relative proportions and color, the animals approach nearer to, or actually agree with, nevadensis.
The specimens commented upon clearly show intergradation between nevadensis and longicauda. Similarly, the specimens from Scottsbluff County, Nebraska, here referred to M. f. alleni, by their larger size suggest intergradation of that subspecies with the larger nevadensis-longicauda stock although the approach is more toward longicauda than nevadensis. Between oribasus and nevadensis, however, there is no lack of material showing intergradation. As set forth in the account of oribasus, specimens from Montana are truly intermediate structurally as well as geographically.
Intergradation with washingtoni is shown by specimens from the northern part of the Cascade Range in Chelan and Okanogan counties, Washington. The adult male, U. S. Nat. Mus., no. 235183, from Bald Mountain, is referable to washingtoni on the basis of cranial characters but all the other adult and subadult specimens examined from Chelan and Okanogan counties are nearer nevadensis on the basis of cranial characters. Indeed, some show no approach to washingtoni in cranial characters. As might be expected on geographic grounds, the specimen from Easton, U. S. Nat. Mus., male subadult, no. 116870, shows approach to washingtoni. This is true of the coloration of the hind limbs, small size of the tympanic bullae, and relatively greater length of the preorbital part of the skull. However, the greater width of the light color of the underparts and relatively great breadth across the mastoid processes and zygomatic arches are points of agreement with nevadensis. Similarly, a series of 7 specimens from the Entait River, 20 miles above its mouth, in tone of color is nearer to washingtoni, as is one of the two skulls of adult males in length of the preorbital region. However, in greater breadth of the skull otherwise, and in the relatively great width of the light color of the underparts, the animals are nearer to nevadensis, to which they are here referred. Some of these characters mentioned above in which departure is shown from typical nevadensis are characters that show approach to altifrontalis. This is especially true of the more intense coloration and restriction of the color of the underparts.
Complete intergradation with effera is shown by specimens from southern Oregon. The change from small effera to the larger nevadensis here is gradual; consequently in northeastern California and southern Oregon the size increases gradually to the northward. Specimens showing complete intergradation with oregonensis and saturata are wanting. However, one specimen from Crescent Lake suggests oregonensis in having near (18) apricot yellow underparts such as occur frequently in oregonensis. Also some specimens from northern California approach saturata in having the color of the underparts reduced in the extent to which it reaches out on the under side of the tail. This fact and the consideration that the two races are less different from one another than are other kinds which definitely are known to intergrade leave no doubt but that material from the intervening localities would show complete intergradation.
Intergradation between nevadensis and munda is indicated by specimens from South Yolla Bolly Mountain, Trinity County, which are commented on at greater length in the account of M. f. munda. M. f. inyoensis is so closely related to nevadensis as to leave no doubt that specimens from suitable localities will show actual intergradation. That intergradation occurs directly with the bridled weasel of the interior valleys of California, M. f. xanthogenys, is shown by specimens from along the west-facing flank of the southern part of the Sierra Nevada. Probably intergradation occurs all along the Sierra Nevada on the western slope but specimens are lacking to show this. Weasels are known to occur in the foothill territory and the lesser attention given to this region by mammal collectors than to the higher parts of the mountains may explain the lack of preserved specimens. Individual specimens, here referred to nevadensis, but, showing varying degrees of approach to xanthogenys are as follows: A female from Hume; a male and a female from 8000 feet elevation, Monache Meadows; a male from 9800 feet elevation on the east fork of the Kaweah River; and 7 specimens, probably one family, from one-half mile south of Mineral King, 7850 feet. Of the specimens from 7850 feet, the adult male has no light facial markings and the head is only slightly darker than the back. The adult female has much restricted, light facial markings and the intervening areas are darker than in the male. The five juveniles trapped in the same burrow as the female, each has more extensive light facial markings than the adult female although the area of this varies from only slightly more than in the female to as much as in typical specimens of xanthogenys. Also, the dark color of the head in these five specimens averages darker than in nevadensis and more as in weasels to the southwestward especially latirostra. One of the five juveniles is lighter colored over all of the upper parts than nevadensis and is suggestive of xanthogenys in this respect. Finally, the adult male has on the underparts small spots of ochraceous orange suggestive of latirostra and some individuals of pulchra. No. 30655/42628, U. S. Nat. Mus., taken on Mount Whitney, also shows white facial markings and some other features of the valley-inhabiting xanthogenys. A suggestion of intergradation with arizonensis is furnished by specimens, referred to that race, from Springerville and the Kaibab Plateau. No specimens happen to be available from the region in which intergradation would be expected between nevadensis and neomexicana. Since neomexicana and arizonensis intergrade it is probable that nevadensis also will be found to intergrade with neomexicana. In summary, nevadensis is judged to intergrade with each of the subspecies of Mustela frenata whose range adjoins that of nevadensis.
This subspecies is remarkably free from injury to the frontal sinuses such as result from the presence of parasites. In 98 adults from Oregon, California, Nevada, and Colorado, no malformation was noted. Only 1 of the 26 specimens from Washington was malformed and it was an intergrade with washingtoni. The single adult from New Mexico was diseased, as were 3 of the 6 from British Columbia, 1 of the 20 from Idaho, and 1 of the 7 from Utah.
Specimens examined.—Total number, 568, arranged alphabetically by provinces and states and from north to south by counties in each state. Unless otherwise indicated specimens are in the collection of the United States National Museum.
Arizona. Apache County: 15 mi. E Luka Chu Kai Navajo School, 8000 ft., 2.
British Columbia. Monte Cr., 20 mi. E Kamloops, 1[21]; Sicamous, 2; Okanagan, 18 (7[2], 6[85], 1[75], 1[86]); Monashee Pass, 1[31]; Swan Lake, near Okanagan Landing, 1[22]; Okanagan Landing, 11 (2[74], 3[31], 3[86], 3[22]); Vernon, 1[74]; Hope-Princeton Summit, 5600 ft., 1[77]; Hope, 1[20]; Similkameen, 1[77]; Osoyoos-Bridesville Summit, 1[77]; Anarchist Mt., Osoyoos, 1[31]; Myer's Creek, 1[77]; Rossland, Mt. Glory, 7000 ft., 1[77]; Cascade, 1[77]; Nelson, 1.
California. Siskiyou County: Hornbrook, 1; Tule Lake Refuge, 5[74]; Upper Mud Creek, 6700 ft., Mt. Shasta, 3; Mt. Shasta, 1. Modoc County: Goose Lake, 1[20]; Joseph Creek, 1[74]; 5280 ft., Parker Creek, near Alturas, 1[74]; Warner Mts., near Alturas, 1[8]; 5 mi. NW Eagle Peak, 7000 ft., 2[74]; Shields Creek, 5000 ft., 1[74]; Jess Valley, 1[8]. Shasta County: Cassel, 1. Lassen County: 3 mi. W Eagle Lake, 5800 ft., 1[74]; 4 mi. S Eagle Lake, 6000 ft., 2[74]; Mill Creek, 5000 ft., S base Mt. Lassen, 1; 6 mi. SW Calneva, 1. Tehama County: Dale's, 600 ft., on Paines Creek, 1[74]. Plumas County: Kelly's, 2 mi. S Willow Lake, 5200 ft., 3[74]; Quincy, 4[68]; Beckwith, Sierra Valley, 1. Butte County: Jonesville, 1[74]. Sierra County: Little Truckee River, 6500 ft., 3 mi. N Independence Lake, 2[42]. Nevada County: Independence Lake, 1[74]. Placer County: Donner, 3; 2 mi. W Soda Springs Station, 6500 ft., 1[74]; Blue Canyon, 5000 ft., 2 (1[74]); 4 mi. S Tahoe City, 1[74]. Eldorado County: 5 mi. S Tallac, 6300 ft., 1; Gilmore Lake, Mt. Tallac, 2[74]; Mt. Tallac, 1[68]; Phillips, 1[59]. Alpine County: 8000 ft., Hope Valley, 1; 8000 ft., Silver Creek, 1. Tuolumne County: Strawberry, 5200 ft., 1[74]; 9300 ft., Ten Lakes, Yosemite Park, 1[74]; Tuolumne Meadows, 8600 ft., Yosemite Park, 1[74]; Tuolumne Meadows (Soda Springs), 1; Tuolumne Meadows, 8500 ft., Yosemite Park, 1[74]; Sequoia, 1. Mariposa County: Chinquapin, 6256 ft., 2[74]; Merced Grove Big Trees, 5400 ft., 1[74]; Wawona, 1; no locality more definite than county, 1. Madera County: Bass Lake, 1[74]. Mono County: Tioga Crest, near Tioga Pass, 4[74]; Warren Creek, 1[74]; Tioga Lake, 1[74]; Ellery Lake, 9600 ft., 1[74]; Mono Lake P. O., Mono Lake, 1[74]; Walker Lake, 8000 ft., 2[74]; Pine City, 1; Mammoth, 13 (12[59], 1[14]); 10300 ft., near Big Prospector Meadow, White Mts., 2[74]. Inyo County: Little Onion Valley, 7500 ft., 1[74]; N Fork Bishop Cr., 10500 ft., 1[74]; S fork Bishop Cr., Andrews Camp, 8000 ft., 1[74]; South Lake, S Fk. Bishop Cr., 9750 ft., 1[74]; Lamarck Cr., 9900 ft., 15 mi. SW Bishop, 1[74]. Fresno County: Hume, 1. Tulare County: Mt. Whitney, 2; Whitney Meadow, 9800 ft., 1[74]; Monache Meadow, 8000 ft., 3[74]; E fork Kaweah River, 9800 ft., 1; 1/2 mi. S Mineral King, 7850 ft., 7[52]; Quaking Aspen Meadow, 7500 ft., 1[52].
Colorado. Moffat County: Lay, 1[19]. Routt County: Steamboat Springs, 2 (1[19]); no locality more definite than county, 1[57]. Jackson County: Higho, North Park, 8400 ft., 1; Buffalo or Illinois Creek, "near Rand," 6[74]. Washington County: 6 mi. NE Hillrose, 1[74]. Larimer County: Estes Park, 2 (1[2], 1[7]); Pinewood, 1; Loveland, 2 (1[57]); no locality more definite than county, 1[7]. Rio Blanco County: Compass Creek, 9000 ft., 1[2]; White River, 6200 ft., 1[21]; Piceance Creek, 6200 ft., 1[2]; Dry Fork, 6200-6600 ft., 4[2]; Meeker, 1; Marvine, 1[74]. Grand County: Crembling [= Kremmling?], 1[50]; Middle Park, 1[57]. Boulder County: Foot Mt. Meeker, 8700 ft., 1[2]; Silver Lake Mine, 1[60]; Boulder, 1[60]; Dixie Lake, 2 (1[2], 1[57]); Caribou, 1[2]; no locality more definite than county, 1. Clear Creek County?: Grays Peak, 1[93]. Jefferson County: 7000 ft., Mt. Parks, 1[57]; 6 mi. W Denver, 1[57]. Adams County: Barr, 1[2]; near East Lake, 2[57]. Denver County: Denver, 2 (1[2], 1[74]). Arapahoe County: Littleton, 1[19]. Summit County: Breckenridge, 1[57]. Eagle County: Eagle, 9500 ft., 1[102]. Park County: Jefferson, 4 (1[2]); 12800 ft., Mt. Bross, 1[57]. Mesa County: Tunnel, 1. Montrose County: near Crawford, Clear Fork of Smiths Fork, 1[19]; Coventry, 3 (1[19]); Naturita, 1; Paradox, 1[94]; West Paradox Valley, 1[57]. Pitkin County: Placita, 2[26]. Gunnison County: Marble, 1[26], Crested Butte, 2[19]; Deckers Ranch, Crested Butte, 2[19]; Sapinero, 7245 ft., 1[19]. Chaffee County: Buena Vista, 1[76]; Hancock, 1[16]; Salida, 5[19]. Teller County: Glencore, Pikes Peak, 1[76]. El Paso County: Monument, 1[76]; Seven Lakes, 1[19]; Lake Moraine, 10250 ft., 1[19]; Colorado Springs, 6000 ft., 1[19]; 5 mi. E Sand Creek, Colorado Springs, 1[19]; no locality more definite than county, 1[50]. Saguache County: Villa Grove, 1[19]; Pierce Place, Cochetopa Nat. Forest, 1; Houselog Creek, Cochetopa Nat. Forest, 1; P. Tevebaugh's Ranch, near Cochetopa Pass, 1; P. Tevebaugh's Ranch, 9 mi. S Cochetopa Pass, 1. Rio Grande County: between Monte Vista and Del Norte, 1[88]. Archuleta County: Upper Navajo River, 2[57]; Navajo River, 5 (4[57], 1[2]); Chromo, 2[57]. Conejos County: Osier, 3[57]. Montezuma County: Ure Peak, 1[57]. County in question: Del Norte Peak, 1[76]; no locality more definite than state, 4[75].
Idaho. Latah County: Cedar Mt., 4000 ft., 12 mi. NE Moscow, 1[55]; Moscow and 1/2 mi. W, 2[97]. Idaho County: Lochsa River (= Locksaw Fork), 1; between Selway Riv., and S Fork Clearwater Riv., 8[74]; Selway Divide, 8[74]; Pilot Creek, 2[74]; Newsome Cr., 1[74]. Lemhi County: Salmon River Mts., (now Lemhi Mts.), 8000 ft., 5; Leadore, 3. Adams County: summit Smith Mt., 7500 ft., 1[41]. Washington County: Midvale, 2. Custer County: Pahsimeroi Mts., 1; Double Springs, 16 mi. NE Dickey, 1[74]; Mackay?, 1; Stanley Lake, 1. Payette County: 2 mi. S Payette, 1[74]. Fremont County: 17 mi. E, 4 mi. N Ashton, 6275 ft., 2[74]. Teton County: 3 mi. S Victor, 1[74]. Jefferson County: 20 mi. W Camas, 1. Blaine County: Sawtooth City, 1; Ketchum, 5 (3[50], 2[75]). Canyon County: Nampa 3. Clark County: Dry Creek, Targhee Nat. Forest, 1[2]; Birch Creek, 2. County in question: North fork of Teton River, 1. Bingham County: Shelley, 1; Alridge, 2; Springfield, 1. Lincoln County: Shoshone, 1. Minidoka County: 1/4 mi. E Heyburn Bridge, 1[74]. Power County: 4 mi. NW American Falls, 1[74]. Bannock County: 3 mi. N Schutt's Mine, Ross Creek, 1[74]; 3 mi. N Pocatello, 1[74]; near (within 10 miles of) Pocatello, 1[74]; 3 mi. S Pocatello, 1[74]; 1 mi. E Portneuf, 1[74]; 2 mi. up Mink Creek, 2 (1[74], 1[41]); Inkom, 2; Swan Lake, 1. Owyhee County: 5 mi. SE Riddle, 1; Three Creek, 2. Cassia County: Elba, 1[52]. Bear Lake County: Geneva, 6171 ft., 1[74]; Montpelier, 1; Paris, 6000 ft., 1[6]; Pegram, 2.
Nevada. Humboldt County: Alder Creek, 7000 ft., Pine Forest Mts., 1[74]; head of Big Creek, 8000 ft., Pine Forest Mts., 1[74]; Cottonwood Range, 1; Calico Mt., Little Owyhee R., 1; Mahogany, Little Owyhee R., 2; Sulphur, 1. Pershing County: Lovelocks, 1. Elko County: Mountain City, 3; Three Lakes, Ruby Mts., 1[41]. Washoe County: Pyramid Lake, 1; 3 mi. E Reno, 1[74]; Incline Creek, 7100 ft., 1[74]; 2-1/2 mi. S Incline, 6250 ft., 1[74]; E side Marlette Lake, 8000 ft., 1[74]; Marlette Lake, 8000 ft., 1[74]. Ormsby County: 1/2 mi. S Marlette Lake, 8150 ft., 1[74]. Churchill County: 4 mi. W Fallon, 1[74]; 3 mi. W Fallon, 1[74]; 2 mi. W Fallon, 1[74]; Fallon, 3970 ft., 1[74]. 5 mi. S Fallon, 4000 ft., 1[74]; 8 mi. S and 3 mi. E Fallon, 1[74]. Douglas County: Mt. Siegel, 1[60]. Mineral County: Lapon Cañon, 8900 ft., Mt. Grant, 1. Nye County: Arc Dome, 1; 10700 ft., 1/2 mi. SW Jefferson Peak, Toquima Range, 1[74]. White Pine County: 3 mi. E Baker, 1[74]; Baker Creek, 6600 ft., 4[74]; Baker Creek, 8400 to 8450 ft., 4[74]; Gleason Creek, 7500 ft., 1[74]. Esmeralda County: Arlemont, 4850 ft., Fish Lake Valley, 1[74]. Lincoln County: 3 mi. S Crystal Spring, 3900 ft., Pahranagat Valley, 1[27].
New Mexico. Taos County: 2 mi. N Twining, 10500 ft., 1; Taos, 2. Santa Fe County: 11600 ft., Pecos Baldy, 1. San Miguel County: 8000 ft., above Willis, Pecos River, Forest Reserve, 2[75]; Ribera, 1.
Oregon (by counties from west to east). Jackson County: Rustler Peak, Crater Nat. Forest, 1[46]; Siskiyou (probably south of), 2. Klamath County: 20 mi. W Crescent, 1[101]; Anna Creek, Mt. Mazama, 2; S Boundary Crater Lake Nat. Park, 1[74]; Fort Klamath, 15; Upper Klamath Lake, 2[4]; Klamath Falls, 1[75]. Lake County: Dog Lake Ranger Station, 30 mi. SW Lakeview, 1. Harney County: Camp Harney, 2[75]; Burns, 2 (1[101]); 20 mi. S Burns, 1[46]; Narrows, 1[59]; Voltage, 1; Shirk P. O., 2; Keiger Gorge, Steen Mts., 4. Malheur County: Riverside, 1; 2 mi. NW Riverside, 2; Barren Valley, Cord, 1; Cedar Mts., 2; Cow Creek Lake, 1; Jordan Valley, 1. County in question: Sageview, 1.
Utah. Cache County: Logan, 1[74]. Rich County: 8000 ft., near Laketown, 1. Boxelder County: Willard, 1[101]. Salt Lake County: Salt Lake City, 1[74]; Barclay, 6500 ft., Wasatch Mts., 1; Mill Creek, 1[101]. Utah County: Provo Bench, 2[6]; Aspen Grove, Mt. Timpanogos, 1[6]; Payson, 1[6]. Juab County: between Santaquin and Starr, 1[103]. Uinta County: Dry Fork Canyon, 20 mi. NW Vernal, 1[9]. Carbon County: Sunnyside, 1[44]; Range Creek, 1[44]. Millard County: Deseret, 1[74]. Sevier? County: Fish Lake Plateau, 1. Grand County: Warner Ranger Station, La Sal Mts., 1[6]. Beaver County: Britts Meadows, 11000 ft., Beaver Range, 1[2]; Britts Meadows, Beaver Range, 1; Puffer Lake, 1[44]. Garfield County: Boulder, 2[6]. Washington County: Pine Valley, 1[44]; St. George, 1. San Juan County: Geyser Pass, La Sal Mts., 2[6]. County in question: Salt Lake, 2; Wasatch Mts., 1; La Sal Mts., 11000 ft., 1.
Washington. Okanogan County: Bald Mt., 6800 ft., 1; Bauerman Ridge, 6800 ft., Tungsten Mine, 1; Hart Pass, Methow River Trail, 1[46]; Conconully, 2 (1[51], 1[49]); 5 mi. NW Loomis, 1; Molson, 3800 ft., 1; Tunk Mt., 3500 ft., 1. Whatcom County: Barron, 5000 ft., 1. Stevens County: Colville, 1; Orin, 11[51]. Pend Oreille County: Ione, 6[51]. Chelan County: Chelan Mts., 1[2]; Lake Chelan, 1[46]; Manson, 1; Entiat River, 1680 ft., 20 mi. from mouth, 7; Dryden, 2[49]; Wenatchee, 1. Kittitas County: Easton, 2 (1[51]); Ellensburg, 1[51]; 4 mi. E Ellensburg, 1[51]. Grant County: Neppel, 1[51]. Lincoln County: Sprague, 1. Spokane County: Spokane, 1[94]; Cheney 2[89]. Whitman County: Pullman, 11 (6[55], 1[68], 1[10]); 6 mi. S Pullman, 1. Garfield County: Snake River, 1. Yakima County: Yakima, 1[74]; 1 mi. W Moxee, 1[74].
Wyoming. NW Wyoming, 1[75]. Yellowstone National Park: Lamar River, 1; Yellowstone Lake, 1. Park County: Greybull River, 1[80]. Teton County: Crystal Creek, 2; Jackson, 1; Whetstone Creek, 2[76]. Johnson County: Buffalo, 4 (2[93]). Fremont County: Continental Divide, 20 mi. NW Dubois, 1[75]. Sublette County: Bronx, 1[75]. Carbon County: Medicine Bow Mts., 1[75]; 15 mi. SE Parco, 1[74]. Albany County: Garrett, 1; 12 mi. W Laramie, 1[74]; 7 mi. W Laramie, 2[74]; 5 mi. W Laramie, 4[74]; "near" Laramie, 1[74]; 3 mi. SW Laramie, 1[74]; 12 mi. S Laramie, 1[74]. Uinta County: Fort Bridger, 6800 ft., 1[74]; Lonetree, 1; Bridger Pass, 1. County in question: Laramie River, 2. No locality more definite than state, 1.
Mustela frenata effera Hall
Long-tailed Weasel
Mustela frenata effera Hall, Carnegie Instit. Washington Publ. 473:93, November 20, 1936.
Mustela arizonensis, Dice, Journ. Mamm., 1:12, November 28, 1919.
Type.—Male, adult, skull and skin; no. 33637, Amer. Mus. Nat. Hist.; Ironside, 4000 ft., Malheur County, Oregon; September 8, 1912; obtained by H. E. Anthony; original no. 267.
The skull (plates 19-21) is complete and unbroken. The teeth all are present and entire. The skin, in summer pelage, is well made.
Range.—Upper Sonoran to Arctic Alpine life-zones of northern two-thirds of Oregon east of the Cascades, and southeastern Washington, south of the Snake River. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. nevadensis in small size, males averaging 12-1/2 per cent smaller in external measurements, 8 per cent smaller in linear measurements of skull, and 22 per cent in weight of skull, total length averaging 360 rather than 400, condylobasal length averaging 40.5 rather than 43.6; from M. f. oregonensis in absence of frontonasal white patch, presence of light color of underparts on ventral face of tail and smaller skull with basilar length averaging less than 41.7 in males; from M. f. washingtoni in presence of light color of underparts on ventral face of tail, in male skull by linear measurements averaging 7 (5-12) per cent shorter and relative to basilar length shorter in preorbital region and broader across mastoid processes and zygomatic arches.
Description.—Size.—Male: Eight (6 adult and 2 subadult) males from northeastern Oregon yield average and extreme measurements as follows: Total length, 360 (340-378); length of tail, 129 (122-136); length of hind foot, 42 (40-44). Tail averages 56 (52-59) per cent as long as head and body. Length of hind foot more or less than (about same as) basal length.
Female: No. 212423 from Vale, and no. 566 V. B. Scheffer, from 15 mi. E Ukiah, measure, respectively: Total length, 312, 306; length of tail, 113, 114; length of hind foot, 35, 35. Tail averages 57 per cent as long as head and body.
Differences in external measurements between the one adult female and the average of the males are: Total length, 51; length of tail, 16; length of hind foot, 7.
Externals.—Longest facial vibrissae black, brown or white (often all three colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles (in summer pelage) about as shown in stage 4 of figure 19.
Color.—Upper parts, in summer, near (14 n to l) Brussels Brown or tones 1 to 3 of Raw Umber of Oberthür and Dauthenay, pl. 301, darker on top of head from nose to, or slightly behind, line connecting posterior margins of ears. Chin and usually all of upper lips white. Remainder of underparts Buff-Yellow to Straw Yellow. In winter all white except tip of tail or upper parts near (j) Snuff Brown or lighter than Brussels Brown with a smoked effect, with underparts white. Tip of tail at all times black. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of toes and wrists, on medial sides of hind legs to ankles over antiplantar faces of toes, distomedial third of tarsus and usually over proximal fourth to three-fourths of ventral side of tail. Least width of color of underparts averaging, in 15 males, 53 (36-69) per cent of greatest width of color of upper parts. Black tip of tail averaging 47 (38-67) mm. long. Thus averaging longer than hind foot, and 36 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 6 adults from northeastern Oregon): See measurements and plates 19-21. As described in Mustela frenata nevadensis except that: Weight, 2.9 (2.5-3.4) grams; basilar length, 40.5 (39.3-41.8).
Female (based on no. 212423, adult from Vale): In so far as parts of the broken skull permit a person to judge, the skull is as described in M. f. nevadensis except that: Smaller; lighter; postorbital breadth more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite.
As compared with the skull of M. f. nevadensis that of effera seems, on the average, to have the preorbital part relatively smaller. Otherwise, the skull is a miniature of the skull of nevadensis, averaging about eight per cent smaller in linear measurements and weighs twenty-two per cent less. Comparisons of the skull with those of M. f. washingtoni and M. f. oregonensis are made in accounts of those subspecies.
Remarks.—This geographic race has long borne the name of Mustela arizonensis (Mearns). Small size differentiates effera from nevadensis and specimens have been allocated to one or the other subspecies on the basis of size, or average size when several individuals are available from one locality. Complete intergradation with each adjoining subspecies is indicated by numerous specimens, more of which are assigned to these adjoining subspecies than to effera itself.
The minimum of size in M. f. effera is found in the Blue Mountain region of northeastern Oregon. Specimens from the area intervening between these mountains and the Cascades average larger but are nearer the mean of typical effera than they are to the means of washingtoni, oregonensis or nevadensis.
Two males, nos. 204883, adult, and 204884, young, from Sisters, Oregon, near the eastern base of the Cascades, show approach structurally to M. f. washingtoni as it is represented at the nearby locality, Permilia Lake, at the west base of Mount Jefferson. Everything considered, however, the two specimens from Sisters are nearer to effera. A male from Condon, Oregon, shows approach to the Cascade race in slightly increased size.
No perfect skulls of adult females are available from the part of northwestern Oregon in which effera reaches its typical state of development as judged by the small size of the skull of the adult male. Skulls of adult females are available, however, from more nearly marginal localities. These, though smaller than in nevadensis, show relatively less difference in size when compared with nevadensis than do skulls of males. Even so the females at these marginal localities are smaller than those of nevadensis of comparable age and adequate material of adult female effera from the region where the males attain their extreme of small size probably will show about the same relative difference in size between nevadensis and effera as is known to exist between the adult males of these two subspecies. The small size of a subadult female, no 74631, U. S. Nat. Mus., from Asotin, Washington, constitutes partial basis for this opinion.
Of 14 adults examined none showed malformation of the frontal sinuses due to infestation by parasites.
Specimens examined.—Total number, 53, arranged within each state by counties from north to south. Unless otherwise indicated specimens are in the collection of the United States National Museum.
Oregon. Wasco County: 4 mi. S The Dalles, 1[74]; Wapinita, 1; Antelope, 2; 7 mi. E Antelope, 5. Gilliam County: Condon, 1[46]. Morrow County: 10 mi. S Hardman, 1. Umatilla County: Umatilla, 2; 15 mi. E Ukiah, 4000 ft., 1[49]. Union County: Elgin, 1; 20 mi. E Lehman, 1[46]. Wallowa County: Horse Creek, 15 mi. N Paradise, 1; Enterprise, 1[46]; Wallowa Lake, 1[46]; Wallowa Mts., 8300 ft., 1. Baker County: Haines, 1[49]; Anthony, 3[2]; Bourne, 2. Grant County: Long Creek, 1[46]; Canyon Creek, 1[46]; Strawberry Mts., 2; Silvies, 1[14]. Crook County: Prineville, 4. Deschutes County: Sisters, 2; Bend, 1. Lake County: 3 mi. W Stauffer, 1; Fort Rock, 1[46]. Harney County: 25 mi. NW Burns, 1. Malheur County: 4000 ft., Ironside, 2[2]; 1-1/2 mi. S Vale, 2.
Washington. Walla Walla County: Prescott, 4 (2[76], 1[60], 1[74]); Ft. Walla Walla, 2 (1[75]); Wallula, 1[76]. Asotin County: Asotin, 1.
Mustela frenata washingtoni (Merriam)
Long-tailed Weasel
Plates 19, 20, 21, 34, 35 and 36
Putorius washingtoni Merriam, N. Amer. Fauna, 11:18, pl. 4, figs. 3, 3a, 4, 4a, June 30, 1896.
Mustela washingtoni, Miller, U. S. Nat. Mus. Bull., 79:98, December 31, 1912.
Mustela frenata washingtoni, Hall, Carnegie Instit. Washington Publ. 473:106, November 20, 1936.
Type.—Male, adult, skin and skull; no. 76322, U. S. Nat. Mus., Biol. Surv. Coll.; Trout Lake, Mt. Adams, Klickitat (?) County, Washington; December 15, 1895; obtained by D. N. Kaegi; original no. 2.
The skull is unbroken. The left incisors above are missing. Otherwise the teeth are present and entire. The skin is well made, in brown winter pelage, lacks collector's measurements, has no bones in the feet, but by large size is judged to be a male.
Range.—Altitudinally from near 2000 feet at Trout Lake up to the highest parts of the Cascade Range from Mount Jefferson, Oregon, north to Mount Rainier, Washington; Upper Sonoran Life-zone to Arctic Alpine Life-zone. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. altifrontalis in lighter color of upper parts and underparts, latter ranging from Buff-Yellow to Naples Yellow rather than near (14 a to 16 c) Ochraceous-Buff, in shallower skull in both sexes (see measurements), in males, a longer preorbital region, narrower skull with shorter bullae, and in females, a smaller skull with interorbital breadth averaging less than 24 per cent of basilar length; from M. f. nevadensis in absence of light color of underparts on ventral face of tail, in skulls of males, by longer preorbital region and narrower skull across mastoid processes and zygomatic arches, in skulls of females, by shorter preorbital region, and smaller bullae (see measurements); from M. f. effera in absence of light color of underparts on ventral face of tail, in skulls of males, by linear measurements averaging 7 (5-12) per cent larger, and relative to basilar length, longer in the preorbital region and narrower across mastoid processes and zygomatic arches; from M. f. oregonensis in absence of frontonasal white patch, longer skull in males, which in percentage of basilar length has, on the average, orbitonasal length amounting to more than 35, mastoid breadth less than 55, and zygomatic breadth less than 63, and in females, smaller skull with least width of palate less than length of P4, upper tooth-rows less than 38-1/2 per cent of basilar length, bullae smaller, averaging less than 13.4 in length.
Description.—Size.—Male: Fifteen subadult topotypes yield average and extreme measurements as follows: Total length, 400 (357-437); length of tail, 149 (122-171); length of hind foot, 47.6 (42-59). Tail averages 59 per cent as long as head and body. Length of hind foot averaging more than basal length. Corresponding measurements of one adult and 3 young from Mount Rainier are: 415 (405-423); 155 (145-164); 51 (50-53).
Female: Five adult topotypes yield average and extreme measurements as follows: Total length, 349 (330-393); length of tail, 124 (114-133); length of hind foot, 38 (36-39). Tail averages 55 per cent as long as head and body. Length of hind foot averaging about same as basal length. Corresponding measurements of two adults and 6 young from Mount Rainier are: 338 (320-360); 121 (115-132); 36 (34-40).
The average differences in external measurements of the two sexes, from Mount Adams, are: Total length, 51; length of tail, 25; length of hind foot, 9.6. Corresponding differences between the specimens from Mount Rainier are: 77; 34; 15.
Externals.—Longest facial vibrissae black or brown (often both colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to or beyond apical pad of fifth digit; hairiness of foot-soles slightly less than shown in figure 19.
Color.—Upper parts in summer near (14 n) Argus Brown or tone 4 of Burnt Umber of Oberthür and Dauthenay, pl. 304; one topotype Buckthorn Brown or tone 3 to 4 of Snuff Brown of Oberthür and Dauthenay, pl. 303. Dark spot at each angle of mouth present or absent, and when present, often fused with color of upper parts, which rarely covers lower lips. Chin, and usually lower lips, white. Remainder of underparts Buff-Yellow to Naples Yellow. In winter, all white except tip of tail which is at all times black, or upper parts near (14) Brussels Brown to near (j) Snuff Brown with smoked effect and underparts white, rarely with trace of yellowish. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and usually all of wrists, on medial sides of hind legs anywhere from knee to tips of toes. Least width of color of underparts averaging in ten topotypes, 24 (10-37) per cent of greatest width of color of upper parts. Black tip of tail in same series averaging 55 (45-60) mm. long, thus longer than hind foot and averaging 37 per cent of length of tail-vertebrae.
The color of the underparts is not so narrow in the specimens from Mount Rainier and it is believed that the slender bodies used in stuffing the topotypes has accentuated in them the appearance of narrowness of the light-colored underparts.
Skull and teeth.—Male (based on 22 adult topotypes): See measurements and plates 19-21; weight, 3.5 (2.8-4.7) grams; basilar length, 43.7 (40.0-47.7); zygomatic breadth more or less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth more or less than postpalatal length; postorbital breadth less than length of upper premolars and greater than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bullae; breadth of rostrum less (except in no. 82180) than length of tympanic bulla; least width of palate more (except in no. 81954) than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 2 to 5 upper incisors; height of tympanic bulla more or less than distance from its anterior margin to foramen ovale; length of tympanic bulla more (except in two instances) than length of lower molar and premolar tooth-row and shorter (except in two instances) than rostrum; anterior margin of masseteric fossa below m2.
Female (based on 11 ad. topotypes): See measurements and plates 34-36; weight, 2.0 (1.8-2.2) grams; basilar length, 37.6 (37.0-38.9); zygomatic breadth less (except in no. 70945) than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth less than length of upper premolars and more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate less (except in one specimen) than greatest length of P4; tympanic bulla as far posterior to foramen ovale as width of 3-1/2 to 5-1/2 upper incisors; height of tympanic bulla more or less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum.
Compared with M. f. nevadensis, the skull of the male of washingtoni averages more slender, as shown by the mastoid and zygomatic breadths and has the preorbital part longer, on the average, as shown by the greater ratio (to the basilar length) of the length of the tooth-rows and orbitonasal length. Also, on the average, the postorbital constriction is longer than in nevadensis and the tympanic bullae are smaller. In females, the skull is lighter, the tooth-rows are shorter, the tympanic bullae are smaller, and the preorbital part of the skull is shorter and narrower as shown by the orbitonasal length and interorbital breadth. Except that the tympanic bullae are actually, although not relatively, smaller in males of effera, it differs from washingtoni in the same way as does nevadensis as regards relative proportions, but, of course, the actual difference in size is greater since effera is smaller than nevadensis. Comparison of the skull with that of oregonensis is made in the account of that subspecies.
Remarks.—M. f. washingtoni was described and named in 1896 by Merriam as a distinct species. Subsequently, specimens which here are regarded as intergrades between altifrontalis and nevadensis, were classified as washingtoni.
The external measurements given for the specimens from Mount Adams are those recorded on the labels in inches and fractions thereof. Instead of total length there sometimes is written "tip to tip." In the series of 19 winter-taken topotypes the hairs project beyond the end of the caudal vertebrae for an average distance of 28 (19-40) millimeters. If the hairs on the end of the tail were included in the measurements, 28 millimeters should be subtracted from the averages. Probably the measurements should stand as given, since an adult male topotype, no. 226758, U. S. Nat. Mus., taken subsequently by Walter P. Taylor measures 405; 152; 51.
Mustela frenata washingtoni is not a strongly marked geographic race. In many features it is intermediate between M. f. altifrontalis and M. f. nevadensis. This is especially true of coloration. In the series from Mount Adams and that from Mount Rainier, some individuals have the light color of the underparts extended down the hind legs over the feet and over the proximal face of the ventral third of the tail as in nevadensis, whereas others from the same place have the light color of the underparts absent from the tail and extending no farther down the hind limbs than the knees. The light color of the underparts in the series of topotypes is so restricted that the transverse extent at the narrowest place amounts to only 24 (10-37) per cent of the greatest width of the color of the upper parts. This narrowness of the color of the underparts has been likened by Merriam (1896:18) to the condition in Mustela frenata noveboracensis. So it is, but it is similar to the condition found also in the geographically adjoining M. f. altifrontalis.
Of the 37 skulls of subadults and a few adults, 11 had the frontal sinuses malformed as a result of infestation by parasites.
Specimens examined.—Total number, 56, arranged within each state by localities from north to south. Unless otherwise indicated specimens are in the collection of the United States National Museum.
Oregon. Mt. Jefferson, Permilia Lake, 1.
Washington. Pierce County: 5500 ft., Spray Park, Mt. Rainier, 1; Spray Park, 1[74]; 5935 ft., Glacier Basin, Mt. Rainier, 5 (1[10]); 5051 to 5100 ft., Owyhigh Lakes, Mt. Rainier, 7 (1[10]), Tahoma Creek, 1[72]; Nisqually entrance, 1[72]; Longmire, 1[72]; Mt. Rainier Nat'l Park, 2[72]. Klickitat County: Trout Lake, S Base Mt. Adams, 35; 3500 ft., Gotchen Creek, Mt. Adams, 1.
Mustela frenata saturata (Merriam)
Long-tailed Weasel
Putorius saturatus Merriam, N. Amer. Fauna, 11:21, June 30, 1896.
Mustela saturata, Miller, U. S. Nat. Mus. Bull., 79:98, December 31, 1912.
Mustela arizonensis saturata, Grinnell, Univ. California Publ. Zoöl., 40:102, September 26, 1933.
Mustela frenata saturata, Hall, Carnegie Instit. Washington Publ. 473:106, November 20, 1936.
Type.—Male, adult, skull and skin; no. 65930, U. S. Nat. Mus., Biol. Surv. Coll.; Siskiyou, Jackson County, Oregon; June 6, 1894; obtained by C. P. Streator; original no. 3905.
The skull (plates 19-21, 30) lacks the middle part of each zygomatic arch. The teeth all are present although much worn, probably from gnawing at the trap which captured the animal. The skin, in fresh summer pelage, is fairly well made.
Range.—Transition and Boreal life-zones of Siskiyou and Trinity mountains in southern Oregon and northwestern California. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. nevadensis in lacking light color of underparts on tail and ankle and in greater average breadth across mastoid processes of skull (see measurements); from M. f. oregonensis in lacking white nasofrontal spot, in having color of underparts interrupted at ankle; from M. f. munda in lacking white nasofrontal spot, in smaller and relatively deeper skull of males and smaller skull of the female.
Description.—Size.—Male: Four adult males (the type, 1 from Mt. Ashland and 2 from Jackson Lake) yield average and extreme measurements as follows: Total length, 414 (402-437); length of tail, 150 (136-160); length of hind foot, 46 (43-50). Tail averages 57 (49-62) per cent as long as head and body. Length of hind foot more or less than basal length.
Female: One young from the summit of the Trinity Mountains east of Hoopa and one nontypical adult from 5500 feet elevation on South Fork Mountain, Humboldt County, measure respectively as follows: Total length, 330, 325; length of tail, 115, 123; length of hind foot, 37, 37. Tail is 53 and 61 per cent as long as head and body. Length of hind foot less than basal length.
Average differences in external measurements between the two sexes, indicated by the unsatisfactory material available, are: Total length, 86; length of tail, 31; length of hind foot, 9.
Externals.—Longest facial vibrissae black or dark brown and extending beyond ear; carpal vibrissae same color as underparts and extending as far as apical pad of fifth digit; hairiness of foot-soles, in summer pelage, as shown in figure 19.
Color.—Upper parts, in summer, Brussels Brown to near (n) Brussels Brown or lighter than tone 3 of Raw Umber of Oberthür and Dauthenay, pl. 301, usually darkest on nose and forehead. Chin white. Remainder of underparts Buff-Yellow to Warm Buff. Tip of tail black. Winter pelage unknown. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and sometimes wrists, on medial sides of hind legs only to ankles, but toes sometimes with isolated white markings. Least width of color of underparts in the type and 2 adults from Jackson Lake averaging 35 (30-40) per cent of greatest width of color of upper parts. Black tip of tail averaging 54 (53-55) mm. long; thus longer than hind foot and averaging 37 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 4 adults: Type, Mt. Ashland, 1; Jackson Lake, 2): See measurements and plates 19-21, 30. As described in Mustela frenata nevadensis except that: Weight, 3.8 (3.5-4.3) grams; basilar length, 44.4 (42.6-45.8); zygomatic breadth more or less than distance between condylar foramen and M1 or than distance between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth more than postpalatal length; least width of palate less than medial length of P4 (except in one specimen).
Female (based on one adult possibly not typical, from 5500 ft., South Fork Mt.): See measurements. As described in Mustela frenata nevadensis except that: Weight, 2.2 grams; basilar length, 38.1; zygomatic breadth less than distance between condylar foramen and M1 and less than distance between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite.
The skull of the male of saturata, relative to the basilar length, is broader across the mastoids and narrower across the rostrum and interorbital region than that of nevadensis. Skull not known certainly to differ from that of oregonensis. Compared with the skull of munda, that of the male of saturata is smaller in every part measured except depth of tympanic bullae which averages 3.6 millimeters, rather than 3.5 as in munda. Also, the skull of saturata has a less-marked postorbital constriction, is less heavily ridged, less angular, does not have the impressions of the temporal muscles carried so far forward on the frontal bones and is relatively much narrower across the zygomatic arches.
Remarks.—In 1896, Merriam named M. f. saturata as a distinct species on the basis of one specimen, taken by Clark P. Streator at Siskiyou, Oregon, and a second specimen taken the year previously by Allan C. Brooks at Chilliwack, British Columbia. On the basis of these two specimens, Merriam (1896:22) ascribed to the race a range ". . . on the Cascade and Siskiyou mountains of Oregon and Washington, reaching a short distance into British Columbia." Since that time, this name, saturata, has been employed for the dark-colored weasels, of the coastal region of Oregon, Washington, and extreme southwestern British Columbia, which here are arranged under the name M. f. altifrontalis. M. f. saturata proves to be restricted to the humid mountainous region inland from the coast in northern California and in the Siskiyou Mountains of southern Oregon. Its range is separated by that of M. f. oregonensis from the range of the darker-colored, deeper-skulled, M. f. altifrontalis of the humid costal region proper.
On May 5, 1933, Mr. Clark P. Streator, informed the writer that he remembered taking the type specimen of Mustela frenata saturata (Merriam) in the town of Siskiyou, Oregon. The exact place, he said, was reached, at the time of his work there, by going one or two blocks east of the depot, then through a garden into the thick woods where there were springs and numerous burrows of the rodent, Aplodontia. Two other weasels labeled as taken at Siskiyou, on September 28 and 29, 1893, by Mr. Streator, are much lighter colored than the type of saturata and have the color of the underparts extended distally on the hind legs to the tips of the toes and in other features of coloration are more like nevadensis, the subspecies to which they are referred, than saturata. Probably these did not come from exactly the same place that the type specimen of saturata did. Although Mr. Streator does not remember the taking of these particular specimens in 1893, he does remember that on this visit to Siskiyou, he walked southward through the railroad tunnel and collected on the opposite side of the ridge from Siskiyou. Here on more southern exposures, the country was markedly different than in the thick forest at Siskiyou. Probably these two specimens taken in 1893, and referred to nevadensis, came from a little way south of Siskiyou and from a different habitat and life-zone than the type specimen of M. f. saturata.
Of the 6 specimens examined, only one, the type, shows malformation of the frontal sinuses such as result from infestation by parasites.
Specimens examined.—Total number, 6, as follows:
California. Siskiyou County: Jackson Lake, 5900 ft., 2, Mus. Vert. Zoöl. Humboldt County: South Fork Mt., 5500 ft., 1, Mus. Vert. Zoöl. County in question, Trinity Mts., summit east of Hoopa, 5800 ft., 1, U. S. Nat. Mus.
Oregon. Jackson County: Mt. Ashland, 1, Univ. Oreg.; Siskiyou, 1, U. S. Nat. Mus.
Mustela frenata altifrontalis Hall
Long-tailed Weasel
Plates 1, 19, 20, 21, 34, 35 and 36
Mustela frenata altifrontalis Hall, Carnegie Instit. Washington Publ. 473:94, November 20, 1936.
Putorius (Gale) brasiliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877 (part).
Putorius saturatus Merriam, N. Amer. Fauna, 11:21, June 30, 1896 (part).
Mustela saturata, Miller, U. S. Nat. Mus. Bull., 79:98, December 31, 1912.
Type.—Male, adult, skull and skin; no. 42093, Mus. Vert. Zoöl.; Tillamook, Tillamook County, Oregon; July 10, 1928; obtained by Alex Walker; original no. 717.
The skull is complete and unbroken. P3 on the left side is missing; otherwise the teeth all are present and entire. The skin is well made and the enlarged scrotal pouch shows the collector's sexing of the specimen to have been correct.
Range.—Altitudinally from sea level up to at least 4800 feet (Mount Baker) in the Transition Life-zone of the humid, coastal region of Oregon, Washington and extreme southwestern British Columbia. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. nevadensis in tone 4 of Brownish Drab, pl. 302, rather than tones 1-3, of Raw Umber, pl. 301, of Oberthür and Dauthenay of upper parts, in near (14 a´ to 16 c´) Ochraceous-Buff rather than Buff-Yellow to Straw Yellow of underparts, in that least width of color of underparts amounts to less than 37 per cent of greatest width of color of upper parts, in absence of color of underparts on ventral side of tail and on hind leg distal to knee, and in greater depth of skull through frontal region; from M. f. washingtoni in darker color of upper parts and underparts, latter near (14 a´ to 16 c´) Ochraceous-Buff rather than ranging from Buff-Yellow to Naples Yellow, in deeper skull in both sexes (see measurements), in males a shorter preorbital region, broader skull with longer bullae and in females a larger skull with interorbital breadth averaging more than 24 per cent of basilar length; from M. f. oregonensis in frontonasal white patch absent, color above darker (tone 4 of Brownish Drab, pl. 302, rather than tone 2 to 3 of Raw Umber, pl. 301 of Oberthür and Dauthenay), light-colored underparts narrower and not extended distally beyond knee, in females tooth-row shorter, amounting to less than 38 per cent of basilar length.
Description.—Size.—Male: Eight adult topotypes yield average and extreme measurements as follows: Total length, 426 (392-445); length of tail, 160 (148-170); length of hind foot, 47 (42-53). Tail averages 60 per cent as long as head and body. Length of hind foot averages more than basal length.
Female: Five adults from Tillamook and Blaine, Oregon, yield average and extreme measurements as follows: Total length, 347 (320-370); length of tail, 125 (114-131); length of hind foot, 38 (35-44). Tail averages 56 per cent as long as head and body. Length of hind foot less than basal length.
The average differences in the external measurements are: Total length, 79; length of tail, 35; length of hind foot, 9.
Externals.—Longest facial vibrissae black, brown or white (often all three colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to or beyond apical pad of fifth digit; hairiness of foot-soles (in summer pledge) slightly less than shown in figure 19.
Color.—Upper parts, in summer, near (n) Argus Brown or tone 4 of Brownish Drab of Oberthür and Dauthenay, pl. 302. Dark spot at each angle of mouth well developed; often fused with color of upper parts which sometimes covers lower lips. Chin white. Remainder of underparts near (14 a´ to 16 c´) Ochraceous-Buff. In winter, upper parts near (14) Argus Brown with smoked effect and Warm Buff to Naples Yellow below. Tip of tail at all times black. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and usually all of wrists, on medial side of hind legs typically only to knee but sometimes to ankle. Tips of toes of hind feet almost always marked with color of underparts. Least width of color of underparts averaging in a series of 14 males from Blaine, Oregon, 23 (14-36) per cent of greatest width of color of upper parts. Black tip of tail in 8 adult males from Blaine, Oregon, averaging 59 (47-70) mm. long; thus longer than hind foot and averaging 37 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 9 adults from Blaine, Tillamook Co., Oregon): See measurements and plates 19-21; weight, 4.4 (3.3-5.3) grams; basilar length, 45.6 (42.4-47.7); zygomatic breadth more or less (usually more) than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth more or less (usually more) than postpalatal length; postorbital breadth less (except in some instances of malformations of frontal sinuses which result from infestation by parasites) than length of upper premolars and more or less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate more or less than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 3 to 4 (including I3) upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and more or less than orbitonasal length; anterior margin of masseteric fossa directly below m2.
Female (based on 4 adults): See measurements and plates 34-36; weight, 2.2 (2.2-2.3) grams; basilar length, 38.1 (37.8-39.7); zygomatic breadth more or less (less in three of four specimens) than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; relation of postorbital breadth to other measurements in doubt because of malformation of frontal sinuses by parasites; least width of palate not less than greatest length of P4; tympanic bulla as far posterior to foramen ovale as width of 3-1/2 to 5-1/2 upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum.
Compared with the skull of M. f. washingtoni that of each sex of altifrontalis averages slightly larger in every measurement taken, except measurements of teeth which are approximately the same, and is relatively deeper through the frontal region and through the braincase as measured at the anterior margin of the basioccipital. Skulls of females of altifrontalis have a relatively broader interorbital region. Skulls of males of altifrontalis further differ in having relatively, as well as actually, longer tympanic bullae, relatively lesser orbitonasal length and a greater relative breadth across the mastoids and across zygomata. Compared with M. f. nevadensis, the skull of the male of altifrontalis averages slightly larger and heavier although the skulls of females are of approximately the same size and weight. Relative to the basilar length, the skulls of both sexes are deeper through the braincase and narrower across the mastoids; the rostrum is broader, especially in males; the tooth-rows are shorter and the interorbital breadth less, especially in females. Comparison with the skull of oregonensis is made in the account of that subspecies.
Remarks.—Until the present study was begun, animals of this race have gone under the name Mustela saturata (Merriam). The United States National Museum has a juvenile taken, in 1858, by Wayne at Astoria, O. T.; the Samuel N. Rhoads collection contained one specimen taken in 1891, at Tacoma, Washington; one in the Bangs' collection was taken at Chilliwack, British Columbia, in 1895, and the Field Museum has one taken on the Olympic Peninsula in 1898. The best material is that collected by Alex Walker, at Tillamook, Oregon.
Intergradation with nevadensis is indicated by several specimens. The coloration of the one adult female, no. 90, Chas. R. Conner Mus., from Swamp Creek, Washington, has the color of the underparts extended down the hind legs over the feet, and over the proximal third of the ventral face of the tail as in nevadensis although the other two specimens from the same place have the color pattern of altifrontalis. Of the four specimens from British Columbia referred to this subspecies, only the specimen from Chilliwack is typical as regards color pattern. The one from Cultus Lake has the color pattern of nevadensis and might be referred to that race almost as well as to altifrontalis. The two specimens from Lihumption Park are intermediate between the two races in tone of color. Neither has the color of the underparts extended onto the tail or continuously over the hind feet as in nevadensis but each does have the color of the underparts less restricted and of lighter hue than in altifrontalis. Only one of the specimens, no. 7848 Canad. Nat. Mus., from Lihumption Park is adult and it has a skull which agrees with that of altifrontalis rather than nevadensis.
After writing the above, a good representation of the weasel population along the eastern side of Puget Sound was made available by friends in that area. Study of the weasels from there shows that their color is intermediate between that of altifrontalis and nevadensis. On the whole, they (specimens from Bellingham, for example) resemble one subspecies about as much as the other. In cranial characters some specimens, in certain features, approach nevadensis but most specimens agree with altifrontalis and all are more nearly like altifrontalis to which race all are referred.
The color of these animals is to me indistinguishable from that of washingtoni. The color of washingtoni is merely intermediate between that of nevadensis and altifrontalis. Nevertheless, the race washingtoni has cranial characters (long narrow skull) which set it off from both altifrontalis and nevadensis. This shape of skull is not found in the specimens from along the eastern side of Puget Sound; these animals have skulls like that of altifrontalis and when departures from this occur they are in the direction of nevadensis and not washingtoni.
The above, then, explains why specimens which are colored like those of washingtoni are not referred to that race but instead to the race altifrontalis.
Of 23 adult skulls examined, 19 have the frontal sinuses malformed as the result of infestation by parasites.
Specimens examined.—Total number, 80, arranged within states by counties from north to south. Unless otherwise indicated specimens are in the United States National Museum.
British Columbia. Chilliwack, 1[74], Lihumption Park, 4750 ft., 2[77]; Cultus Lake, 1[77].
Oregon. Clatsop County: Old Fort Clatsop, 1[74]; Astoria, 1. Tillamook County: Tillamook, 12 (7[14], 2[74], 2[2], 1[46]); Netarts, 1[46]; Blaine, 16 (13[14], 1[93], 1[76], 1[59]). Lane County: Reed, 1; Mercer, 1[46]. Curry County: Langlois, 1[46].
Washington. Whatcom County: Nooksack River, 2000 ft., 14 mi. E Glacier, 1; Swamp Creek, 2050 ft., Nooksack River, 3[10]; Lookout, 4800 ft., Mt. Baker, 2[10]; Bellingham, 8[25]; 5 mi. S Bellingham, 1[49]. Skagit County: Rockport, 300 ft., 1. King County: Bothell, 2[94]; N Seattle 1[51]; Seattle, 1[49]; Tye, 1[51], 2 mi. E Skykomish, 1[51]; 7 mi. E Kent, 1[76]; Auburn, 3[94]. Pierce County: Tacoma, 1[1]. Clallam County: Sequim, 1[49]; Soleduc Riv., near [sic.] Sappho, 1[49]; Happy Lake, 1[60]; mouth of Boulder Creek, Elwha River, 560 ft., Olympic Mts., 1; Hume's Ranch, 1000 ft., Elwha River, 1; Bogachiel Riv., 1[49]. Mason County: Lake Cushman, 2; 4 mi. N Shelton, 1[51]. Thurston County: Olympia, 2[49]; Tenino, 1[51]. Pacific County: 2-1/2 mi. SE Chinook, 3[74].
Mustela frenata oregonensis (Merriam)
Long-tailed Weasel
Plates 19, 20, 21, 30, 34, 35 and 36
Putorius xanthogenys oregonensis Merriam, N. Amer. Fauna, 11:25, June 30, 1896; Bangs, Proc. New England Zoöl. Club, 1:57, June 9, 1899.
Mustela xanthogenys oregonensis, Miller, U. S. Nat. Mus. Bull., 79:99, December 31, 1912.
Mustela xanthogenys munda, Grinnell, Univ. California Publ. Zoöl., 40:102, September 26, 1933 (part).
Mustela frenata oregonensis, Hall, Carnegie Instit. Washington Publ. 473:107, November 20, 1936.
Type.—Male, subadult, skull and skin; no. 32019/43828, U. S. Nat. Mus., Biol. Surv. Coll.; Grants Pass, Rogue River Valley, Josephine County, Oregon; December 19, 1891; obtained by C. P. Streator; original no. 1404.
The skull (plates 19-21, 30) is complete and unbroken. P3 on the left side is missing. Otherwise the teeth all are present although worn probably as a result of gnawing at the trap which captured the specimen. The skin, in brown, winter pelage, is fairly well made.
Although the label on the skin and the label in the skull vial each give the sex of the specimen as female, and although Merriam (1896:25) regarded the specimen as a female, the present writer regards the specimen as a male.
It is as large as other undoubted males and larger than any known female of this subspecies. The labels with the skull and skin give the locality as "Rogue River Valley, Oregon." The listing here of the more restricted locality, Grants Pass, is made on the basis of Merriam's (1896:25) original description of the subspecies.
Range.—Transition and Canadian life-zones along coast of northern California and southern Oregon from Humboldt County, California, north through Curry County, Oregon, thence inland, west of the Cascades, north to the Columbia River. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. altifrontalis in presence of frontonasal white patch, lighter color above (tone 2 to 3 of Raw Umber, pl. 301, rather than tone 4 of Brownish Drab, pl. 302, Oberthür and Dauthenay), wider extent of light color of underparts which is extended distally beyond knee, and in females, longer tooth-row which amounts to more than 38 per cent of basilar length; from M. f. munda in shorter hind foot of males which is less than 50, and in both sexes, smaller, less rugose skull (see measurements and plates); from M. f. saturata in presence of frontonasal white patch, in having color of underparts extended uninterruptedly over ankle onto foot; from M. f. nevadensis in presence of frontonasal white patch, lack of light color of underparts on ventral face of tail, and longer skull, which relative to its length in males, is shallower through braincase; from M. f. effera in presence of frontonasal white patch, lack of light color of underparts on ventral face of tail, and larger skull with basilar length averaging more than 41.7 in males; from M. f. washingtoni in presence of frontonasal white patch, shorter skull in males, which in percentage of basilar length has, on the average, orbitonasal length amounting to less than 35, mastoid breadth more than 55, and zygomatic breadth more than 63; and in females larger skull with least width of palate more than length of P4, upper tooth-rows more than 38-1/2 per cent of basilar length, bullae larger and averaging more than 13.4 long.
Description.—Size.—Male: Five males (3 adults and 2 subadults from Eureka, Ferndale, and Carlotta, California) yield average and extreme measurements as follows: Total length, 392 (347-430); length of tail, 138 (110-160); length of hind foot, 46 (43-50). Tail averages 54 (46-61) per cent as long as head and body. Length of hind foot more or less than basal length. The type specimen, and an adult from Goldbeach measure, respectively, as follows: Total length, 412, 386; length of tail, 155, 137; length of hind foot, 44, 46.
Female: Three adults (2 from Fortuna and 1 from Carlotta, California) yield average and extreme measurements as follows: Total length, 367 (360-374); length of tail, 130 (123-134); length of hind foot, 40 (39-40). Tail averages 55 (52-57) per cent as long as head and body. Length of hind foot less than basal length. A subadult from Goldbeach, an adult from 13 mi. SW Grants Pass, and an adult from Medford, measure, respectively, as follows: Total length, 316, 344, 294; length of tail, 114, 120, 122; length of hind foot, 36, 40, 38.
The average differences in external measurements of the two sexes in the vicinity of Carlotta, are: Total length, 25; length of tail, 8; length of hind foot, 6. Corresponding differences, at Goldbeach, are: 70, 23, 10. Probably the females at Fortuna reflect the large size of munda more than do the males at Carlotta and the differences between the measurements of the two sexes probably, therefore, are actually more than are indicated by the figures above.
Externals.—Longest facial vibrissae black, brown or white (often all three colors in same specimen) and extending beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles, in summer pelage, as shown in figure 20.
Color.—Upper parts, in summer, near (16 l) Brussels Brown or tone 2 of Raw Umber of Oberthür and Dauthenay, pl. 301, to slightly darker than tone 3 of same plate. Darker on nose and top of head, usually with frontonasal white patch but lacking white bar in front of each ear, except in the type and 2 specimens from Salem. Chin, lower lips, angle of mouth, and usually posterior seventh of upper lip white. Remainder of underparts Pale Orange-Yellow. In winter usually lighter above with underparts Warm Buff to Straw Yellow. Tip of tail at all times black. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists, on medial side of hind leg, typically over ankle in extremely narrow line which widens out over distal phalanges of antiplantar faces of toes but sometimes interrupted at ankle. Least width of color of underparts averaging, in twenty available specimens, 39 (27-54) per cent of greatest width of color of upper parts. Black tip of tail in five adults averaging 50 (43-60) mm. long; thus averaging longer than hind foot and 33 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 4 adults and subadults from Eureka, Requa, Goldbeach, and Grant Pass): See measurements and plates 19-21, 30. As described in Mustela frenata nevadensis except that: Weight, 3.5 (3.5-4.1) grams; basilar length, 42.9 (41.8-44.0); least width of palate more or less than medial length of P4.
Female (based on 2 adults, one from Carlotta and one from 13 mi. SW Grants Pass): See measurements and plates 34-36. As described in Mustela frenata nevadensis except that: Weight, 2.4 (2.2-2.6) grams; basilar length, 37.7 and 39.5; zygomatic breadth less than distance between condylar foramen and M1 and less than distance between anterior palatine foramen and anterior margin of tympanic bulla. See under "Remarks" for additional data on variation in size of skulls of females.
The skulls of the female averages 31 per cent lighter than that of the average male.
Because there is much geographic variation between specimens here referred to oregonensis, the person who is guided by the present account should keep in mind that results, here reported, of comparisons of the skull with those of other races, were obtained by employing specimens of oregonensis from Carlotta and Eureka, California. These specimens from California are judged to have more of the characters of the subspecies munda than do specimens of oregonensis from more northern localities.
Compared with that of M. f. washingtoni the skull of the male is shorter, especially in the preorbital region and is relatively broader across the mastoidal processes and zygomatic arches. The skull of the female is longer in the preorbital region, has a less cylindrical braincase and differs less from the male skull than is the case in M. f. washingtoni. Compared with M. f. effera, the skull of the male is smaller in every part measured and relative to the basilar length is broader across the mastoids and has relatively shorter tympanic bullae. From M. f. nevadensis the skull of the male differs in the same way except that size is about the same. The skull of the female oregonensis is more heavily ridged and is relatively broader across the mastoids than that of effera. From M. f. saturata, oregonensis is not surely known to differ in cranial characters. From M. f. munda, oregonensis differs in having the skull of both sexes smaller, and on the average, in all parts measured, has a less marked postorbital constriction, relatively narrower interorbital region and relatively more expanded zygomata. From M. f. altifrontalis, males of oregonensis differ on the average, in having larger teeth, and relative to the basilar length, a greater mastoid breadth and a shallower braincase as measured at the anterior margin of the basioccipital. Females of oregonensis differ in larger average size of skull, except for breadth of rostrum and interorbital breadth which, therefore, are relatively less in oregonensis, as also is the relative depth of the skull measured at the posterior borders of the upper molars and at the anterior margin of the basioccipital. However, skulls of females of oregonensis have relatively longer tooth-rows and are relatively broader across the zygomata and mastoidal processes.
Remarks.—In 1896, Merriam named oregonensis as a subspecies of the California bridled weasel on the basis of a single specimen taken by Clark P. Streator. Three additional specimens were acquired in later years, by workers of Dr. Merriam's bureau, from near the type locality and specimens from farther north in Oregon have been accumulated at the University of Oregon. The most satisfactory material is that saved from Humboldt County by the late H. E. Wilder, which, when brought together, is adequate to give some idea of the range of variation that can be expected in a given population.
Of two specimens from Goldbeach, one shows approach to altifrontalis in that the color of the underparts stops at the ankle, and in one, the angle of the mouth is dark colored. Specimens from Eugene and vicinity lack the white facial markings, and in this feature approach the adjoining washingtoni-effera-nevadensis stock. A specimen from 6 miles south of Medford shows approach to saturata in the interruption, on the ankle and lower tibial region, of the color of the underparts. One adult female, no. 1413, Univ. Oregon, from the Rogue River Valley, 13 miles southwest of Grants Pass, stands out prominently, among the other specimens from extreme southern Oregon and northwestern California, by reason of the near (18) Apricot Yellow color of the underparts, but this same color occurs in specimens from the more northerly localities of Buchanan, Eugene, Vida Fish Hatchery, and McKenzie Bridge, as well as in no. 2178, Univ. Oregon, from Cresent Lake. The last mentioned specimen is here referred to nevadensis.
Two females referred to oregonensis from southern Oregon differ so greatly in size of skull that they challenge one's imagination in any attempt to provide an explanation for so wide a range of variation in one subspecies. One of these, no. 244520, U. S. Nat. Mus., is an adult female from Medford. The other, no. 224034, U. S. Nat. Mus., is a subadult female (though labeled male) from 43 miles northeast of Grants Pass. The skull of the adult from Medford has a basilar length of 41.5, upper tooth-rows, 16.1 in length, and a weight of 2.75 grams, whereas corresponding figures for the subadult are only 33.8, 12.9, and 1.4. Two other adult females are intermediate in size: No. 1413, Univ. Oregon, from 13 miles southwest of Grants Pass, Oregon, approaches the specimen from Medford in size, and the second specimen, no. 34325, Mus. Vert. Zoöl., from Carlotta, California, is smaller.
Not only is there a difference in length between the skulls of the two extremes of the females but this difference extends to all other dimensions of their skulls, and is most pronounced in the preorbital region. The differences in breadth of the braincase and other parts of the skull are relatively less than the differences in length. Differences of the same nature, although of lesser degree than found in the females, are to be seen in two males. The skull of an adult no. 51590, Mus. Vert. Zoöl., from 6 miles south of Medford, has a basilar length of 46.4, upper tooth-rows, 17.6 mm. long, and a weight of 4.0 grams, whereas corresponding figures for the subadult type specimen from Grants Pass, are only 43.0, 16.2, and 3.3.
The wide range of variation in size of skull of both sexes, together with the considerable variation in color pattern of the specimens here referred to oregonensis raises the suspicion that we are using the name in a composite sense; nevertheless, to recognize more than one subspecies with the material now available would be unwise.
A subadult female, of abnormal color, no. 47149, Mus. Vert. Zoöl., taken by Mr. H. E. Wilder at Carlotta, California, on December 20, 1930, in a region where weasels do not turn white in winter, is white, except for the black tip of the tail, but has a suffusion of orange. This specimen, discussed at greater length on page 43, is instructive in that it suggests that there are separate determiners for the brown and red elements of the pelage. It is interesting also as suggesting how natural selection may tend to eliminate from the population a conspicuous color-variation of this kind. At any rate, Mr. Wilder (Ms.) states: "This specimen was picked up in a field, where it evidently had been dropped by a hawk or an owl." The braincase of the skull is crushed in three places as though by a raptor's beak. None of the several other weasels, all normally colored, saved by Mr. Wilder from this general locality gives evidence of having fallen a victim to a raptor.
Only 2 skulls of the 12 adults and subadults examined show malformation of the frontal sinuses such as results from the presence of parasites.
Specimens examined.—Total number, 29, arranged within states from north to south by counties. Unless otherwise indicated specimens are in the collection of the United States National Museum.
California. Del Norte County: Requa, 1[8]. Humholdt County: Eureka, 2 (1[74], 1[75]); Ferndale, 1[74]; Fortuna, 2[63]; Carlotta, 6 (3[74], 3[59]); 12 mi. E Bridgeville, 1[59]; 2 mi. W Bridgeville, 1[59].
Oregon. Washington County: Forest Grove, 1. Marion County: Salem, 2. Benton County: Buchanan, 1. Lane County: McKenzie Bridge, 1[101]; Vida Fish Hatchery, 1[101]; Eugene, 1[101]. Douglas County: Anchor, 1. Curry County: Gold Beach, 2[60]. Josephine County: Rogue River Valley (Grants Pass), 1; 13 mi. SW Grants Pass, 1[101]. Jackson County: Medford, 2; 6 mi. S Medford, 1[74].
Mustela frenata munda (Bangs)
Long-tailed Weasel
Plates 1, 19, 20, 21, 22, 23, 30, 34, 35, 36 and 40
Putorius xanthogenys mundus Bangs, Proc. New England Zoöl. Club, 1:56, June 9, 1899; Stephens, California mammals, p. 247, 1906.
Mustela frenata, Audubon and Bachman, Journ. Acad. Nat. Sci. Philadelphia, 8 (Pt. 2):291, 1842 (North California about 40° latitude).
Mustela xanthogenys munda, Miller, U. S. Nat. Mus. Bull., 79:99, December 31, 1912.
Mustela frenata munda, Hall, Carnegie Instit. Washington Publ. 473:107, November 20, 1936.
Type.—Male, adult, skull, os penis and skin; no. 5459, collection of E. A. and O. Bangs, but now in collection of Mus. Comp. Zoöl.; Point Reyes, Marin County, California; June 19, 1896; obtained by C. A. Allen; original no. 931. (See comments under "Remarks," below, on places in California to which the name Point Reyes has been applied.)
The skull (pls. 19-21, 30) is complete and unbroken. I1 on each side and right I2 are broken away; p2 and p3 on each side have been aborted and the only alveoli remaining are two for the right p3. Otherwise all teeth are present and entire. The skin is fairly well made and in good condition.
Cranially, the type is a "runt"; its small size and the circumstance that the tympanic bulla is longer than the lower molar and premolar tooth-row and longer than the rostrum are features which differentiate the type from any other specimen seen of this race.
Range.—Sea level to at least 6,000 feet (South Yolla Bolly Mountain, Trinity County, California); Upper Sonoran and Transition life-zones of the coast and Coast Range of northwestern California from the Golden Gate northward into southern Humboldt and Trinity counties. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. oregonensis in longer hind foot of males which is more than 50 mm., and in both sexes, larger, more prominently ridged skull (see measurements and plates); from M. f. saturata by presence of nasofrontal white spot, larger and relatively shallower skull of males and larger skull of female; from M. f. nevadensis by presence of well-developed, white, facial markings; absence of color of underparts on ventral face of proximal third of tail; and hind foot of males more than 50; from M. f. xanthogenys by near (l) Sudan Brown to near (l) Antique Brown rather than Buckthorn Brown colors of upper parts and greater size, and in adult male basilar length more than 45 and hind foot more than 47; from M. f. nigriauris by having inside of ears same color as back rather than much darker than back.
Description.—Size.—Male: Three adults and two young from Point Arena and Gualala, Mendocino County, yield average and extreme measurements as follows: Total length, 447 (434-470); length of tail, 167 (150-185); length of hind foot, 53 (50-60). Corresponding measurements of three adults from 5 and 6 miles west of Inverness, Marin County, are: 430 (420-440), 154 (141-160), 48 (48-49). Corresponding measurements of four individuals (3 adults and 1 young of large size) from South Yolla Bolly Mountain, Trinity County, are: 383 (374-400), 134 (130-138); 44 (43-44). The tail averages 60 per cent as long as the head and body in the series from Point Arena, 56 per cent in the series from Point Reyes, and 53 per cent in the series from South Yolla Bolly Mountain. In every specimen except two, length of hind foot less than basal length. The two exceptions are no. 19720, M.V.Z., male adult from Point Arena in which the hind foot is recorded as 60 (probably an error in measurement), and no. 19721, M.V.Z., from the same place, in which the skull has not yet attained its full growth.
Female: One adult from Point Arena measures as follows: Total length, 383; length of tail, 134; length of hind foot, 43. Corresponding measurements of an adult from seven miles north of Laytonville, Mendocino County, are: 336, 121, 33 (= 36 on dried skin). Corresponding measurements of an adult from South Yolla Bolly Mountain, Trinity County, are, 326, 113, 37. In these three specimens, the tail is, in the order given, 54, 56, and 53 per cent as long as the head and body. Length of hind foot more than basal length.
Differences in external measurements of the two sexes as indicated by the five males and one female from Point Arena, are: Total length, 64; length of tail, 33; length of hind foot, 10. Weights of 2 adult males are 265 and 221 grams and of one adult female 155 grams.
Externals.—As described in Mustela frenata nigriauris.
Color.—Spot between eyes, narrow band or spot confluent with color of underparts on each side of head anterior to each ear, chin, lower lips, and rarely posterior third or less of each upper lip white; dark spot posterior to each angle of mouth uniformly present and of large size; tip of tail black; remainder of upper parts near (14 l) Sudan Brown and tone 4 of Raw Umber of Oberthür and Dauthenay, pl. 301; occasionally, slightly darker brown on forehead, nose, and about eyes. Underparts near (a to c) Ochraceous-Buff and sometimes Orange-Buff. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists, on medial sides of hind limbs over antiplantar faces of toes. Least width of color of underparts averaging, in a series of 5 males from Mendocino County, 57 (46-67) per cent of greatest width of color of upper parts; 38 (35-40) in 3 males from Point Reyes, Marin County. Black tip of tail in Mendocino County series averaging 53 (46-60) mm., which is same length as hind foot and 32 per cent of length of tail. In Point Reyes males, black tip of tail averages 44 (34-52) mm., which is less than length of hind foot and 45 per cent as long as tail-vertebrae.
Several specimens of the smaller, inland variant (see under "Remarks") are near (l) Antique Brown rather than near (14 l) Sudan Brown above and hence do not differ in this respect from nigriauris.
Skull and teeth.—Male (based on 3 adults from Mendocino County): See measurements and plates 19-23, 30. As described in Mustela frenata nigriauris except that: Weight, 6.0 (5.4-6.3) grams; basilar length, 47.6 (46.5-48.2); length of tympanic bulla more than length of lower molar and premolar tooth-row.
Female (based on no. 19723, M.V.Z., from Point Arena): See measurements and plates 34-36, 40. As described in M. f. nigriauris except that: Weight, 3.0 grams; basilar length, 42.3.
The skull of the female is 50 per cent lighter than that of the average male.
Compared with the skull of the male of nevadensis that of munda averages larger in every part measured and specimens from Point Arena are nearly as heavy again, have relatively more expanded zygomata and mastoid processes but are relatively narrower anteriorly as shown by the breadth of the rostrum, interorbital breadth and postorbital breadth. Also the braincase is less inflated anteriorly, the tympanic bullae are lower and the skull is more angular. Females show the same differences although in different degree. Compared with the skull of the male of M. f. nigriauris, that of munda from Point Arena averages larger in every part measured except for the length of the upper tooth-rows. Relative to the basilar length, the skull of munda averages broader across the mastoids and across the zygomata, is deeper through the braincase at the anterior end of the basioccipital, and has a greater development of the lambdoidal crest.
Remarks.—The skin and part skull, no. 536/1849, U. S. Nat. Mus., taken by Lieutenant W. P. Trowbridge at San Pablo Bay, is the first specimen known to have been saved of this subspecies. Since 1899 when O. Bangs diagnosed munda as of small size, the weasel of the humid costal belt north of San Francisco Bay has been regarded as smaller than bridled weasels from farther south in the State. Actually, however, the weasel of the humid costal belt shares with M. f. pulchra the distinction of being one of the two largest weasels in California.
M. f. munda may be a composite subspecies, for the variation in facial markings, in coloration otherwise, in external measurements and in size and shape of skull is great. At one time in the course of the present study, manuscript accounts of two subspecies were prepared for the animals now all called munda and there is still much justification for recognizing two subspecies, one, along the coast proper, the larger, darker-colored animal with reduced white facial markings and large, wide, heavily ridged skull from Point Arena, and 6 miles south of Laytonville, Mendocino County, along with the specimens from 5 and 6 miles west of Inverness, Marin County, and the other, an inland race, which is a smaller, lighter-colored animal with more extensive white facial markings and a smaller, narrower, skull, known by specimens from Point Reyes [station?], Nicasio, 15 mi. north of San Rafael, Freestone, Vallejo, and Mount Sanhedrin. The differences between these two lots of specimens are of great degree. However, a female from Fort Bragg proves to be no larger than three females labeled as from Point Reyes. Also, a male from 2 miles south and one mile east of Stewarts Point on the coast has a skull no larger than the animal from Vallejo, whereas the skin alone of an adult female from 3 miles south of Stewarts Point is large and agrees with the specimens from Point Arena. Consequently, no logical ranges can be worked out for the two variants with the material now available.
Finally, the type specimen of munda is a "runt," smaller than any other male seen. This specimen, purchased by E. A. and O. Bangs from C. A. Allen, who collected and sold specimens widely, was labeled as from Point Reyes. So far as this place-name is concerned, it might refer to: (1) The point of land by that name which projects out into the Pacific Ocean, (2) an abandoned ranch house bearing that name at the head of Drakes Bay, 6 miles north and 3-3/4 miles east of the actual point, or (3) the railway station by the same name at the head of Tomales Bay, 12 miles east and 4-3/4 miles north of the actual point. Allen, himself, lived near San Geronimo (then Nicasio) about nine miles southeast of the Point Reyes railway station. All these places are in Marin County, but differ markedly as regards climate and flora. The first two are treeless, windswept and have much fog, whereas Point Reyes Station is more often sunny, and is situated in a shallow valley, inland, where the open grass-covered west-facing slopes meet the east-facing wooded ones. From which one of these three places the type specimen came, I do not know. The same may be said of the three female specimens labeled Point Reyes; two of these are in the United States National Museum and one in the Field Museum.
The specimens in the Museum of Vertebrate Zoölogy from 5 and 6 miles west of Inverness and those from near the same place in the collection of John Cushing come from within a couple of miles or less of the Point Reyes represented by the abandoned ranch house. These specimens, as remarked above, agree with those from Point Arena in large size, reduced facial markings and wide skull. These are points of difference from the smaller variant suspected of being a recognizable subspecies. It is the smaller variant which the type specimen approaches in size, and with which it agrees in relatively well-developed white facial markings. This suggests that the type specimen came from Point Reyes Station rather than from either of the two other places bearing the name "Point Reyes," from one of which, as just stated, the variant of large size is known. The three females labeled "Point Reyes" also have well-developed white facial markings and are of lesser size than the female of similar age from Point Arena, Mendocino County. The presumption is that these three females also came from Point Reyes Station.
The smaller, inland variant seems to agree in size, cranial characters, and coloration with M. f. nigriauris to the southward of San Francisco Bay, but lacks the black on the head which characterizes nigriauris. The larger variant, on which the description here used for munda is based, comprises animals which differ from nigriauris in larger size, darker color, reduced white facial markings, and larger, relatively wider skull. Both of the variants mentioned above are sharply distinct from nigriauris on the basis of coloration of the inside of the ear which is blackish in nigriauris like the dark facial markings, and in munda is colored like the back. M. f. munda lacks the dark facial markings; an occasional specimen has at most, a trace of the markings but this does not extend back so far as the ears. This difference, blackish versus non-blackish face, persists eastward of San Francisco Bay to at least as far as the Carquinez Straits, where a specimen of munda is available from 4 miles north of Vallejo and one of nigriauris from Glen Frazer Station on the south shore opposite Vallejo.
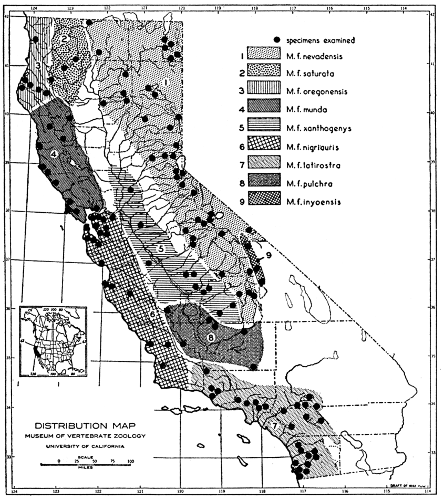
Fig. 30. Map showing the geographic distribution of subspecies of Mustela frenata in California
Intergradation with M. f. nevadensis and possibly with M. f. saturata is indicated by specimens from South Yolla Bolly Mountain, Trinity County. In them the external measurements and measurements of the skull are intermediate. Also the white frontal spot is much reduced in size. The white bars in front of the ears are absent in three specimens, and weakly developed in the other two. The relative proportions of the skulls as a whole are nearer those of nevadensis or saturata than munda. The skull of one of the three adult males and the skull of the adult female suggests M. f. oregonensis in certain features; for example, the dorsal outline of the skull in longitudinal axis is slightly convex as it is in oregonensis.
None of the specimens shows malformation of the frontal sinuses such as results from infestation by parasites.
Specimens examined.—Total number, 37, arranged by counties from north to south. Unless otherwise indicated specimens are in the Museum of Vertebrate Zoölogy.
California. Trinity County: S. Yolla Bolly Mt., 3[91]; 1/2 mi. S S. Yolla Bolly Mt., 1. Tehama County: 2 mi. S S. Yolla Bolly Mt., 1. Mendocino County: 6 mi. N Laytonville, 1; Mt. Sanhedrin, 1[87]; Ft. Bragg, 1; Gualala, 1; Point Arena, 5. Sonoma County: 2 mi. S and 1 mi. E Stewarts Point, 1; 3 mi. S Stewarts Point P. O., 1; Freestone, 1. Napa County: 6 mi. SSW, Napa, 1; 4 mi. N Vallejo, 1. County in question: San Pablo Bay, 1[91]. Marin County: 6 mi. W Inverness, 2; 5 mi. W Inverness, 2(1[28]); Point Reyes, 4 (2[91] 1[60], 1[75]); Nicasio, 2 (1[60], 1[75]); Kehoes Ranch, Pierce Point, 1[28]; Drakes Bay, 1[28]; Tomales Point, about 1/2 mi. SW White Gulch, 1; Point Reyes School, 3-3/4 mi. W Inverness, 1; 15 mi. (by road) N San Rafael, 1[52]; Hurley Ranch, 2 mi. W Tomales, 1. No locality more definite than California, 1[7].
Mustela frenata xanthogenys Gray
Long-tailed Weasel
Plates 21, 22, 23, 28, 30, 34, 35 and 36
Mustela xanthogenys Gray, Ann. and Mag. Nat. Hist., 11:118, 1843.
Putorius (Gale) brasiliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877 (part).
Putorius xanthogenys, Merriam, N. Amer. Fauna, 11:25, June 30, 1896; Bangs, Proc. New England Zoöl. Club, 1:56, June 9, 1899.
Mustela xanthogenys xanthogenys, Miller, U. S. Nat. Mus. Bull., 79:99, December 31, 1912.
Mustela frenata xanthogenys, Hall, Carnegie Instit. Washington Publ. 473:107, November 20, 1936.
Type.—Male, adult, skull and skin; skull no. 197a-43.6.4.55, skin no. 234a-42.11.21.4, British Museum (Nat. Hist.); from the bank of Sacramento River below mouth of Feather River, or from north shore of San Francisco Bay, California; taken in "1837 or 1838"; presented by Captain Edward Belcher.
The skull (plate 28) lacks the occiput, the right mandible posterior to m1, and the right pterygoid; the right zygomatic arch is fractured. The teeth are not greatly worn. The skin was originally mounted for exhibition (R. I. Pocock in Litt.) but in 1937 when I saw the skin, it was prepared as a conventional study skin. The skin is in fairly good condition; some hair is missing on the hind quarters and the skin of the tail is torn at one place.
Range.—Altitudinally, less than 600 feet (Fair Oaks); Lower Sonoran and Upper Sonoran life-zones of all but southern end of the San Joaquin Valley, and probably Sacramento Valley, California. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. nevadensis by presence of light facial markings and Buckthorn Brown rather than near (14n to l) Brussels Brown color of upper parts; from M. f. munda by Buckthorn Brown rather than near (l) Sudan Brown, or near (l) Antique Brown color of upper parts and lesser size, in adult males basilar length less than 45 and hind foot less than 47; from M. f. nigriauris by lighter color in same way as from munda and also by having inside of ears same color as back rather than much darker than back; from M. f. pulchra in hind foot of males less than 46 and narrower skull, in males having breadth of rostrum less than 13.9 and mastoid breadth less than 26.0, see comparison of skulls in the account of pulchra.
Description.—Size.—Male: Three adults, from Fresno, Selma and Los Banos, measure, respectively as follows: Total length, 425, 417, 450; length of tail, 152, 154, 180; length of hind foot,—, 43, 44. Tail averages 61 per cent as long as head and body. Length of hind foot less than basal length.
Female: Adults from Selma, Los Banos, and 4 mi. SW Turlock, measure respectively as follows: Total length, 357, 365, 395; length of tail, 133, 132, 145; length of hind foot, 40, 38, 41. Tail averages 58 per cent as long as head and body. Length of hind foot less than basal length.
The average differences in external measurements between the two sexes, as represented by these six specimens, are: Total length, 65; length of tail, 25; length of hind foot, 3.5. One adult male weighs 274 grams and 2 adult females 182 and 214 grams.
Externals.—As described in Mustela frenata nigriauris.
Color.—Spot between eyes, band confluent with color of underparts on each side of head extending anterodorsally anterior to each ear, and posterior half to third of each upper lip white, or whitish tinged with some shade of yellowish; chin and lower lip white; dark spot posterior to each angle of mouth of varying size but uniformly present; tip of tail black; remainder of upper parts Buckthorn Brown of Ridgway or a trifle browner than tone 4 of Brown Pink of Oberthür and Dauthenay, pl. 297. Upper parts of uniform color except for slight darkening of head-markings anterior to ears. Underparts Ochraceous-Buff to Warm Buff. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists, on medial sides of hind limbs over antiplantar faces of toes and sometimes tarsal region. Least width of color of underparts averaging, in 9 specimens from Fresno, Selma and Los Banos, 54 (32-74) per cent of greatest width of color of upper parts. Black tip of tail in three males (one subadult and 3 adults) averages 55 (50-60) mm. long. Thus longer than hind foot and averaging 34 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 2 adults from Fresno and one from Selma): See measurements and plates 21-23, 30. As described in M. f. nigriauris except that: Weight 3.8 grams; basilar length, 43.7 (43.4-43.9); least width of palate more or less than lateral length of P4; length of tympanic bulla more than length of lower molar and premolar tooth-rows.
Female (no. 2626 W. E. Snyder, from Selma): See measurements and plates 34-36. As described in M. f. nigriauris except that: Weight, 2.5 grams; basilar length, 39.4.
The skull of the female is 34 per cent lighter than the average for the three males.
Compared with skulls of nevadensis from the Sierra Nevada, those of the two adult males from Fresno differ as follows: M1 wider (transversely); tympanic bullae narrower; preorbital part of skull smaller. Comparison with pulchra is made in the account of that subspecies. Compared with skulls of adult males of nigriauris, from Santa Clara County, the two skulls from Fresno are generally smaller and in basilar length, length of tooth-rows and measurements of the teeth fall below the minimum for nigriauris. Relative proportions of the skulls are approximately the same. Comparison with munda reveals essentially the same differences as does comparison with nigriauris except that the difference in size is greater.
Remarks.—The name Mustela xanthogenys Gray was long applied to all the weasels of the interior valleys of California and of the coast of that state south of San Francisco Bay. Gray, when he named the species and when referring to it in later accounts, never defined the locality whence the specimen came more definitely than "California." In 1896, Merriam (1896:25) gave the type locality as "Southern California, probably vicinity of San Diego" and later writers have not contradicted him. The type specimen was obtained in the course of the voyage of the British ship Sulphur, under command of Sir Edward Belcher. Examination of Belcher's (1843, vol. 1, p. 129) narrative of the voyage indicates the following places in California at which the specimen of weasel, described by Gray, could have been obtained: Fort Ross, Bodega, vicinity of San Francisco Bay and up Sacramento River to the mouth of the Feather River, Monterey, Santa Barbara, Buenaventura, San Pedro, San Juan, and San Diego.
Reginald I. Pocock has kindly compared the type specimen in the British Museum with several specimens sent for that purpose. In the first place, comparison of skulls shows that the type specimen is a member of one of the races north of San Diego. In the second place, comparison of skins shows that the inside of the ears are not blackish but similar in color to the back. In fact, Pocock writes under date of February 12, 1929, regarding the type specimen, that "It is practically uniformly colored from the snout to the base of the tail, there being scarcely a trace of the darkening of the head, or muzzle, observable in your specimens [those sent for comparison]." This character of coloration of the ear excludes all the weasels of the Coast region of California from San Francisco Bay southward, namely, M. f. latirostra and M. f. nigriauris. My own examination of this type specimen at a date later than that on which Pocock compared it satisfies me as to the accuracy of his statement above.
Accordingly, the name xanthogenys would seem to apply to one of the two subspecies here called munda and xanthogenys. Perusal of Belcher's narrative of the voyage (loc. cit.) shows that little, if any, opportunity was afforded to obtain vertebrate specimens at Fort Ross or Bodega, both localities within the range of the subspecies here called munda. Furthermore, the type specimen is smaller than individuals of munda from 5 to 6 miles west of Inverness and from Point Arena with which the animals from Fort Ross and Bodega would be expected to agree in size. Weasels from along the north shore of San Francisco Bay are smaller than those on the coast north of the bay. Possibly the type specimen of xanthogenys came from the north side of San Francisco Bay but probably it came from the bank of the Sacramento River and almost certainly not farther up stream from San Francisco Bay than the junction of the Sacramento and Feather rivers. The statement of Belcher (1843, vol. 1, p. 129), regarding the trip up the Sacramento River as far as Point Victoria, lat. 38°46´47" north, and return to San Francisco Bay, that "Cuyote or jackal—fox, racoon, land otter, weasel, and squirrel were obtained" lends strong probability to the idea that this type specimen was taken along the Sacramento River, possibly in the vicinity of the existing city of Sacramento. Unfortunately no specimens are available from the Sacramento Valley. If some were available, a comparison of them and specimens of munda from along the north side of San Francisco Bay and Carquinez Straits with the type specimen of xanthogenys should determine the correct application of the name. For the present it seems best to retain the name munda and apply the name xanthogenys to the weasels inhabiting the northern part of the San Joaquin Valley and presumably the southern part of the Sacramento Valley.
Efforts to obtain specimens of weasels from the Sacramento Valley have been in vain. A juvenal specimen taken five miles south of Fair Oaks, Sacramento County, by Mr. John Fitzgerald, Jr., in December, 1927, was examined at his home and found to agree in coloration with specimens from farther south. Geographically, this specimen probably is more nearly a topotype than any other examined.
Most of the specimens examined are immature and adequate adult cranial material has not been seen. Two adults, one of each sex, from Los Banos have skulls of large size which agree with those of nigriauris. The same is true of one adult and one young female from 4 miles southwest of Turlock, which, unlike the animals from Los Banos, show a darkening of the head extending in reduced degree even to the inside of the ears, as in nigriauris. The slightly darker than average (for xanthogenys) color on the back may indicate intergradation with nevadensis. Intergradation with M. f. nevadensis is shown by specimens, from the southern part of the Sierra Nevada, mentioned in the account of nevadensis.
None of the skulls shows malformation of the frontal sinuses such as result from infestation by parasites.
Specimens examined.—Total number 30, arranged by counties from north to south.
California. Sacramento County: Bank of Sacramento River, 1[7]; 5 mi. S Fair Oaks, 1[29]. San Joaquin County: 4 mi. W Stockton, 1[74]. Merced County: Tegner School, 4 mi. SW Turlock, 2; Los Banos, 4 (2[74], 1[91] 1[87]). Fresno County: Mendota, 1[74]; Biola, 1[30]; Clovis, 1[55]; Fresno, 5 (1[74], 1[91], 2[55], 1[1]); 5 mi. W Fresno, 1[14]; Selma, 3 (2[50], 1[104]); 4 mi. NW Sanger, 1[55]; 5 mi. S Selma, 1[62]. Tulare County: Monson, 1[74]; 1-1/2 mi. N Goshen, 1[74]; Milo, 1[91]; 2 mi. N Tipton, 1[74]; Poplar, 2[53]. No locality more definite than California, 1[4].
Mustela frenata nigriauris Hall
Long-tailed Weasel
Plates 22, 23, 24, 34, 35, 36 and 41
Mustela frenata nigriauris Hall, Carnegie Instit. Washington Publ. 473:95, November 20, 1936.
Putorius xanthogenys, Baird, Mamm. N. Amer., 1858, p. 176 (part).
Mustela xanthogenys Gray, Ann. and Mag. Nat. Hist., 14(ser. 4):375, 1874 (part?).
Putorius (Gale) brasiliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877 (part).
Putorius xanthogenys xanthogenys, Grinnel, Proc. California Acad. Sciences, fourth series, 3:292, August 28, 1913.
Mustela xanthogenys xanthogenys, Miller, U. S. Nat. Mus. Bull., 79:99, December 31, 1912; Grinnell, Univ. California Publ. Zoöl., 40:102, September 26, 1933.
Type.—Male, adult, skeleton and skin; no. 32820, Mus. Vert. Zoöl.; Half Moon Bay, San Mateo County, California; received at Museum of Vertebrate Zoölogy, May 4, 1922, through A. L. Hagedoorn, after having been in captivity a few days where death occurred owing to injuries received in trap; original no. 1590.
The skull has each of the zygomatic arches and the anterior end of the nasals broken through. The only part missing is the central two millimeters of the left zygomatic arch. The teeth all are present and entire. The skeleton appears to be complete except for the bones of the feet, which are preserved within the skin. The skin is well made and in good condition.
Range.—Altitudinally, sea level to more than 4000 feet; Sonoran and Transition life-zones of Coast Range and coast of California from San Francisco Bay south to Point Conception, Santa Barbara County, California. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. munda, xanthogenys, and pulchra by having inside of ears darker than back rather than same color as back, and from xanthogenys and pulchra in near (l) Antique Brown color of upper parts rather than Buckthorn Brown or near (16 j) Buckthorn Brown to near (h) Yellow Ocher respectively; from M. f. latirostra by postorbital breadth, of adult males and females, less, rather than more, than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite.
Description.—Size.—Male: Five adults from Palo Alto, Santa Clara County, yield average and extreme measurements as follows: Total length, 447 (412-465); length of tail, 167 (147-175); length of hind foot, 46 (45-47). Corresponding measurements of four adults from San Francisco are: 412 (394-435); 153 (145-160); 43.5 (41-46). Corresponding measurements of five adults and subadults from Berkeley, Alameda County, are: 419 (390-448); 148 (135-160); 44 (42-47). Tail averages 59 per cent as long as head and body in series from Palo Alto and in one from San Francisco. The average of 55 for the Berkeley series probably reflects a lesser average age. Length of hind foot less than basal length. The type specimen measures, 415, 150, 43. It is smaller than the mean.
Female: A subadult from Palo Alto measures: Total length, 368; length of tail, 126; length of hind foot, 39. An adult and two subadults from Berkeley measure, respectively, as follows: Total length, 347, 365, 340; length of tail, 134, 123, 125; length of hind foot, 37, 38.4, 36.5. In these four females the tail averages 55 per cent as long as head and body. Length of hind foot less than basal length.
The average differences in external measurements of the two sexes, as represented by specimens from Berkeley, Alameda County, are: Total length, 68; length of tail, 21; length of hind foot, 7. Eight adult males weigh 249 (217-335) grams and one adult female 123 grams.
Externals.—Longest facial vibrissae brownish like dark color of head and extending beyond ear; carpal vibrissae mostly color of underparts and extending to apical pad of fifth digit; hairiness of foot-soles slightly more than shown in figure 20.
Color.—Spot between eyes, band, confluent with color of underparts, on each side of head extending anterodorsally anterior to ear, and posterior third of each upper lip tinged with color of underparts or, less often, pure white; chin and lower lips white; remainder of sides and top of head posteriorly to, or a little behind, a line connecting posterior margins of ears, blackish; inside of pinna of ear, and sometimes outside of pinna, blackish; dark spot posterior to each angle of mouth present on each side in three-fourths of specimens; tip of tail black; remainder of upper parts near (l) Antique Brown, and with more yellow than tone 3 of Raw Umber of Oberthür and Dauthenay, pl. 301. Often with more blackish and red in winter. Underparts near (a to c) Ochraceous-Buff or Ochraceous-Salmon. Ochraceous-Salmon in some juveniles. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists, and on medial sides of hind limbs over antiplantar faces of toes. Least width of color of underparts averaging, in 17 adult males (Berkeley, 5; San Francisco, 5; Palo Alto, 7), 55 (40-73) per cent of greatest width of color of upper parts. Black tip of tail in same series of males averaging 51 (35-60) mm., thus averaging longer than hind foot and 33 per cent of length of tail (Palo Alto and San Francisco, 31 per cent; Berkeley, 35 per cent). In 8 adult females, least color of underparts amounts to 55 (47-62) per cent of greatest width of color of upper parts. Black tip of tail averages 41.5 (28-50) mm., thus averaging longer than hind foot and 32 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on six adults from Stanford Univ. and vicinity): See measurements and plates 22-24; weight (four adults), 5.4 (5.0-5.9) grams; basilar length, 47 (46.1-48.1); zygomatic breadth more than distance between condylar foramen and M1, or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth more than postpalatal length; postorbital breadth less than length of upper premolars (less than distance between posterior borders of P4 and P2) and less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth not greater than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate less than lateral length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 3 or 4 (including I3) upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more or less than (about equal to) length of lower molar and premolar tooth-row and longer or shorter (usually shorter) than rostrum; anterior margin of masseteric fossa below anterior half of m2.
Female (based on three adults, Hayward, Palo Alto, and Morro): See measurements and plates 34-36; weight (no. 43574, from Morro) 2.7 grams; basilar length, 41.2 (40.2-42.2); zygomatic breadth more or less than distance between condylar foramen and M1 and more or less than distance between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth less than length of upper premolars and less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate less than lateral length of P4; tympanic bulla as far posterior to foramen ovale as width of 3 (including I3) upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum.
The skull of the female averages 50 per cent lighter than that of the average male.
Comparisons of the skull of the male with those of M. f. latirostra, pulchra, xanthogenys, and munda are made in the accounts of those subspecies.
Remarks.—Like M. f. latirostra, nigriauris long bore the name xanthogenys. The fairly adequate lot of specimens is divided between the collections of several institutions. The most satisfactory material in any one collection is in the Stanford University Natural History Museum where local specimens have been accumulated over a period of many years.
No actual intergrade between nigriauris and xanthogenys has been seen, although the specimens from Los Banos, referred to xanthogenys, have large skulls as in nigriauris. Intergradation with latirostra is shown by specimens, referred to latirostra, from the Los Angeles area. Also the one adult male from 5 miles southeast of Santa Margarita, San Luis Obispo County, is of small size and in this respect approaches latirostra. The range of nigriauris is separated from that of munda by San Francisco Bay, Carquinez Straits, and I suppose by the lower part of the San Joaquin River. On the basis of color of the inside of the pinna of the ear, the two subspecies are uniformly distinct. Intergradation is assumed to occur through the subspecies xanthogenys.
None of the 26 adult and subadult specimens examined for evidences of infestation of the frontal sinuses by parasites shows malformation of the sinuses.
Specimens examined.—Total number, 103, arranged by counties from north to south. Unless otherwise indicated specimens are in the Museum of Vertebrate Zoölogy.
California. Contra Costa County: Glen Frazer Station, 1; 2 mi. W Pinole, 1[13]; 1 mi. E Pinole, 1; Richmond, 1[13]; Lafayette, 1; 7 mi. E Clayton, 1; Moraga Valley, 1; Pinehurst, Redwood Canyon, 1; Concord, 1. Alameda County: Berkeley, 11; Oakland, 1; Piedmont, 1; Haywards, 2; near Haywards, 2; 10 mi. E Haywards, 1[91]; Redwood Canyon, 1; Calaveras Dam, 1. San Francisco County: San Francisco, 11 (5[8], 2[91], 1[60], 1[7]); Ocean View, 1[68]; Visitation Valley, 1. San Mateo County: Moss Beach, 1; Half Moon Bay, 1; Redwood City, 1[87]; Menlo Park, 9 (5[87], 2[68]); no locality more definite than county, 1[8]. Santa Clara County: 1/4 mi. N Milpitas, 1; 1/4 mi. S Milpitas, 1; Stanford University, 6 (4[68], 2[91]); Palo Alto, 11 (6[41], 2[60], 1[75], 1[87]). Santa Cruz County: 3 mi. E Santa Cruz, 1; 2-1/2 mi. E Santa Cruz, 1; Santa Cruz, 6 (2[91], 1[68], 1[4]). Monterey County: 1 mi. E mouth Salinas River, 10 ft., 1[37]; Pacific Grove, 1[8]; Monterey, 2 (1[7]); Carmel, 1[8]; Carmel Valley, 1[68]; Point Lobos, 1; Gonzales, 1. San Luis Obispo County: 5 mi. SE Santa Margarita, 1; Morro, 1[91]; 3-1/2 mi. S Oceano, 6. Santa Barbara County: Santa Maria, 1[87]; 5 mi. N Las Cruces, 1; 7 mi. W Gaviota, 1; Gaviota, 1.
Mustela frenata latirostra Hall
Long-tailed Weasel
Plates 1, 22, 23, 24, 34, 35 and 36
Mustela frenata latirostra Hall, Carnegie Instit. Washington Publ. 473:96, November 20, 1936.
Putorius xanthogenys, Baird, Mamm. N. Amer., p. 176, 1858 (part); Stephens, California mammals, p. 246, 1906; Merriam, N. Amer. Fauna, 11:25, June 30, 1896 (part).
Putorius (Gale) brasilianus frenatus, Coues, Fur-bearing animals, p. 142 (part).
Mustela xanthogenys xanthogenys, Miller, U. S. Nat. Mus. Bull., 79:99, December 31, 1912; Grinnell, Univ. California Publ. Zoöl., 40:102, September 26, 1933.
Mustela arizonensis, Grinnell and Swarth, Univ. California Publ. Zoöl. 10, 376, October 31, 1913.
Type.—Male, adult, skull and skin; no. 3257, Mus. Vert. Zoöl.; San Diego, San Diego County, California; May 20, 1907; obtained by Frank X. Holzner.
Right M1 is missing and the part of the jaw bearing this tooth is broken away. With this exception the skull is complete and unbroken and the teeth are all present and entire. The skin is fairly well made and in good condition except that it is slightly soiled.
Range.—Altitudinally sea level to 8000 feet (Tahquitz Valley, San Jacinto Mountains); Sonoran and Transition life-zones of coast and mountains west of Mohave and Imperial deserts of southern California from Point Conception and Cuyama Valley southward at least to Mexican boundary. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. nigriauris by having postorbital breadth of adult males and females, more, rather than less, than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; from M. f. pulchra by having tympanic bulla longer than rostrum (orbitonasal length) and by near (l) Antique Brown rather than near (16 j) Buckthorn Brown to near (h) Yellow Ocher color of upper parts.
Description.—Size.—Male: Six adults and subadults from San Diego yield average and extreme measurements as follows: Total length, 439 (428-449); length of tail, 153 (142-160); length of hind foot, 45 (40-47). Corresponding measurements for a series of eight adult males from the vicinity of Los Angeles are: 416 (394-428); 158 (151-166); 44 (40-47). In the series from San Diego the tail averages 54 per cent as long as head and body. In the series from Los Angeles the average is 61 per cent. Length of hind foot in each series, less than basal length. The type specimen measures, 435, 142, 42.
Female: No. 5070, adult, from San Diego, measures 367, 141, 38. Nos. 22 and 6748 from Santa Ysabel, measure: 359, 380; 130, 140; 39, 35. No. 7194 from Jamacha measures, 358, 125, 35. Three adult females from Los Angeles yield the following: Total length, 373, 345, 368; length of tail, 150, 122, 134; length of hind foot,—, 41, 41. In no. 5070 the tail is 62 per cent as long as the head and body and in the three from Los Angeles it averages 60 (55-67) per cent. Length of hind foot, in each case, less than basal length.
The average differences in external measurements of the two sexes as shown by the six males from San Diego and the four females from San Diego County are: Total length, 73; length of tail, 19; length of hind foot, 8. Corresponding differences shown by the eight males and three females from Los Angeles are: 54, 23, 3.
Externals.—Longest facial vibrissae brownish, like dark color of head and extending beyond ear; carpal vibrissae mostly color of underparts and extending to apical pad of fifth digit; hairiness of foot-soles slightly more than shown in figure 20.
Color.—Spot between eyes, band confluent with color of underparts on each side of head extending anterodorsally anterior to ear, and posterior third of each upper lip tinged with color of underparts or, less often, white; chin and lower lips white; remainder of sides and top of head posteriorly to near line connecting posterior margins of ears, blackish; inside of pinna of ear, and sometimes outside of pinna, blackish; dark spot posterior to each angle of mouth present on each side in three-fourths of specimens; tip of tail black; remainder of upper parts near (l) Antique Brown, and with more yellow than tone 3 of Raw Umber of Oberthür and Dauthenay, pl. 301. Underparts Ochraceous-Buff to Warm Buff and in some specimens from Los Angeles and Ventura counties Ochraceous-Orange, especially in young and juveniles. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists and on medial sides of hind limbs over antiplantar faces of toes. Least width of color of underparts averaging, in 15 adult and subadult males from San Diego County, 54 (35-75) per cent of greatest width of color of upper parts. Black tip of tail in same series of males averaging 54.5 (46-60) mm. long. Thus averaging longer than hind foot and 35 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 6 adults from San Diego County). See measurements and plates 22-24. As described in M. f. nigriauris except that: Weight (4 specimens), 3.9 (3.8-4.0) grams; basilar length 43.8 (41.9-47.0); postorbital breadth more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth not less than distance between foramen opticum and anterior margin of tympanic bulla; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 2 to 2-1/2 (including I3) upper incisors; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer than rostrum; anterior margin of masseteric fossa below m2.
Female (based on 4 adults from San Diego County): See measurements. As described in M. f. nigriauris except that: Weight, 2.6 (2.2-2.8) grams; basilar length, 40.0 and 40.1; postorbital breadth more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; length of tympanic bulla more than length of rostrum.
The skull of the female averages 34 per cent lighter than that of the average male.
The skull of the male of latirostra, compared with that of nigriauris, is by weight, more than one-fourth lighter, has a lesser basilar length, a lesser mastoid breadth, a lesser zygomatic breadth and a narrower M1. In these features no overlap has been observed between adults from the general vicinities of the type localities of the two forms. In adult males of latirostra the postorbital breadth, with one exception, is more than the combined length of P4 and P3 whereas the reverse is true in adult males of nigriauris. Both males and females of latirostra have a generally smaller skull with relatively broader interorbital and postorbital parts and the tympanic bullae are relatively larger, rounder and more inflated.
Compared with the skull of the male of pulchra that of latirostra is, by weight, more than one-fourth lighter, has a lesser basilar and orbitonasal length, lesser zygomatic and mastoid breadth and a more nearly flat braincase. In these features no overlap has been observed between adults from the general vicinities of the type localities of the two subspecies. Also, in latirostra the tympanic bulla is longer than the rostrum whereas the opposite is true in pulchra. The skull of latirostra is generally smaller and relatively, on the average, has the preorbital part of the skull deeper and broader with longer tooth-rows, although with shorter rostrum, while the zygomatic and mastoid breadths are less. Study of skulls of subadult females of pulchra indicate that females of latirostra and pulchra differ in the same fashion as do males.
Remarks.—This subspecies long has gone by the name M. xanthogenys and the type locality was generally supposed to be in the vicinity of San Diego. This supposition seems to have originated with Merriam's (1896:25) statement that the type locality was "Southern California, probably vicinity of San Diego." Nevertheless, as set forth in the account of M. f. xanthogenys the type specimen concerned now is thought to have come from much farther north.
Although 76 Recent specimens are available from southern California, additional adults are needed to understand the geographic variation there. M. f. latirostra may be a composite—made up of more than one geographic race. Specimens from San Diego County differ so much in relative length of the tail that at one stage in the present study it was thought that a difference in this respect existed between the coastal animals and those from farther inland. Material received later did not wholly substantiate this view and because of the uniformly small size of all of the skulls from that county, the animals were later regarded as of the same subspecies. Eventually, even this supposed common feature proved to be inconstant for an adult male from Jamacha, no. 7098, of the San Diego Society of Natural History, and another adult male from San Marcos, no. 8869, collection of Ralph Ellis, were later examined and found to have skulls as large as those of average-sized, adult males of nigriauris.
Despite these puzzling local variations, it is established that the long-tailed weasels of southern California are smaller than those from farther north. Also, the southern animal averages smaller in weight and size of skull, and the skull is differently proportioned. Specimens in series from Los Angeles County definitely are intermediate in size and shape of skull between latirostra from San Diego County and nigriauris from, say, Santa Clara County, but definitely more closely resemble latirostra from San Diego County than they do nigriauris. A skull of a young animal, not here identified to subspecies, from Potholes, in the Colorado River Valley, 10 miles northeast of Bard, Imperial County, California, may have closest relationship to M. f. latirostra. Additional comment on this specimen is offered in the account of M. f. neomexicana.
From the asphalt pits of Rancho La Brea, in Los Angeles County, a total of 57 skulls have been examined, more than half of which are reasonably complete. I have been unable to learn whether these came from pits regarded by students of the deposit as wholly Recent, from pits regarded as of Pleistocene age, or from both. Suffice to say that only two specimens were found which could be distinguished from skulls of the subspecies of weasel living in that area today.
These two specimens, lent to me by Professor Chester Stock, were with other skulls received from the Los Angeles Museum of History, Art and Science and bore identifying numbers as follows: 16/20-27, the anterior part of the skull of an adult, and 16, the skull posterior to the cribiform plate of a subadult or possibly young individual. The latter has a mastoid breadth of 28.0 millimeters, a tympanic bulla 16.1 long and other measurements in proportion. It is larger than any specimen of weasel, of any subspecies, seen from California and in the subgenus Mustela seems to be exceeded in size only by certain individuals of M. f. texensis. M. f. neomexicana attains relatively large size and comparisons were made with individuals of that subspecies. However, the young specimen from Rancho La Brea differs from neomexicana in that the tympanic bullae rise less steeply on the medial sides and the inferior lip of the external auditory meatus is less developed laterally. Age considered, the sagittal crest is less developed and the mastoid processes project more abruptly from the skull. The anterior part of the skull of the adult, no. 16/20-27 is larger than any specimen seen of M. f. latirostra or adjoining subspecies, and among California-taken specimens is equaled in size only by the largest males of M. f. munda from the northwest coastal district in Mendocino County. This adult from Rancho La Brea differs from neomexicana, sex and age taken into account, in greater postorbital breadth, lesser rostral width in comparison with the interorbital breadth, and in having the temporal ridges at the anterior end of the sagittal crest spread out into a Y-shaped, rather than a T-shaped, pattern. All these differences from neomexicana are features of agreement with the California bridled weasels of the subspecies latirostra, nigriauris, and munda. The same is true of the characters which set apart the young specimen from neomexicana. In summary: of 57 weasel skulls examined from the asphalt pits at Rancho La Brea, Los Angeles County, all but two are indistinguishable from the skulls of the Recent weasel living in that region today. These two skulls agree in qualitative characters with animals of the California coastal subspecies now living from Los Angeles northward to Humboldt County, but are larger. For the time being these two may be thought of as giants of the same type of animal inhabiting the Los Angeles region today.
Only one of 41 adult and subadult skulls examined for malformation of the frontal sinuses shows infestation by parasites.
Specimens examined.—Total number, 142, listed by counties from north to south. Unless otherwise indicated specimens are in the Museum of Vertebrate Zoölogy.
California. Santa Barbara County: Rincon Point, 1. Ventura County: Cuyama Valley, 2200 ft., 1[91]; Nordhoff, 3[59]; Santa Paula, 1[59]; Ventura, 7. Los Angeles County: near Owensmouth, 1[24]; Cahuenga, 1[91]; Llano, 10 mi. E Littlerock, 1; Flint Ridge, Pasadena, 1[59]; Pasadena, 3; Lankershim, 1[24]; 1 mi. S Lankershim, 1[24]; Duarte, 1[59]; Covina, 1[59]; Claremont, 1[91]; El Monte, 4 (2[75], 1[24]); Montebello, 1; Alhambra, 6 (5[2], 1[91]); El Nogal, 2[8]; Gardena, 1[26]; Palos Verdes Estate, 3; Rancho La Brea asphalt deposits, 57[70] and [92]. San Bernardino County: San Bernardino Valley, 1[75]; San Bernardino, 4 (2[20], 1[91]); Redlands, 2 (1[38]); Bluff Lake, 2 (1[59], 1[33]). Riverside County: West Riverside, 1; Arlington, 800 ft., 1[17]; 3-1/2 mi. E and 1/2 mi. N Beaumont, 2600 ft., 1; Banning, 1[91]; Cabazon, 1[91]; San Jacinto Plain, 1[20]; Tahquitz Valley, 8000 ft., 1; Elsinore, 1[1]. San Diego County: Twin Oaks, 1[91]; San Marcos, 2 (1[87], 1[41]); Escondido, 1; Witch Creek, 1[91]; Ballena, 1[20]; Santa Ysabel, 3 (2[20], 1[87]); Julián, 1; La Jolla, 1; Lakeside, 1[91]; El Cajon, 1[91]; El Vido (not found on map), 1[91]; San Diego, 9 (1[91], 1[20], 1[87], 1[32]); Jamacha, 2[87]; Chula Vista, 1[20].
Mustela frenata pulchra Hall
Long-tailed Weasel
Mustela frenata pulchra Hall, Carnegie Instit. Washington Publ. 473:98, November 20, 1936.
Type.—Male, adult, skeleton and skin; no. 16668, Mus. Vert. Zoöl.; Buttonwillow, Kern County, California; April 30, 1912; obtained by J. Grinnell; original no. 1953.
The skull (plates 22-24) is complete and unbroken (a fracture in the right jugal has healed). All teeth are present and entire. The skeleton lacks the os penis, left fibula, shaft of left tibia and the distal three or four caudal vertebrae. Some of the bones of the feet distal to the radius and tibia are with the skeleton, and the remainder probably are in the skin. The skin is fairly well made and in good condition, except for the left hind leg which was torn when the animal was captured. A well-developed scrotal pouch shows the specimen to have been a male.
Range.—Altitudinally around 300 feet in San Joaquin Valley to 2500 feet at Isabella; Upper Sonoran and Lower Sonoran life-zones of southern end of San Joaquin Valley and in mountains at southern end of Valley, California. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. nevadensis in presence of light facial markings, and from M. f. nevadensis and M. f. inyoensis in near (16 j) Buckthorn Brown to near (h) Yellow Ocher rather than near (14 n to l) Brussels Brown color of upper parts, and greater size with hind foot more than 40 in females and basilar length averaging more than 46.0 in males; from M. f. latirostra in having rostrum (orbitonasal length) longer than tympanic bulla and from M. f. latirostra and M. f. nigriauris by color of upper parts as stated above rather than near (l) Antique Brown, and by having inside of ears same color as back rather than much darker than back; from M. f. xanthogenys in hind foot of males more than 46 and broader skull which in males has breadth of rostrum more than 13.9 and mastoid breadth more than 26.0.
Description.—Size.—Male: The type specimen and five other adults yield average and extreme measurements as follows: Total length, 454 (428-477); length of tail, 178 (153-184); length of hind foot, 50 (47-55). Tail averages 65 per cent as long as head and body. Length of hind foot approximately equal to basal length. The type specimen measures, 460, 184, 49.
Female: Three subadult topotypes yield average and extreme measurements as follows: Total length, 399 (383-411); length of tail, 154 (140-161); length of hind foot, 42 (41-42). Tail averages 63 per cent as long as head and body. Length of hind foot less than basal length.
The average differences in external measurements of the two sexes are: Total length, 55; length of tail, 24; length of hind foot, 8.
Externals.—As described in Mustela frenata nigriauris.
Color.—Spot between eyes, band confluent with color of underparts, on each side of head extending anterodorsally anterior to each ear, posterior third of each upper lip, lower lips and chin white or more often darker than Ochraceous-Buff and therefore same color as belly; dark spot posterior to each angle of mouth present but small; tip of tail black; remainder of upper parts near (16 j) Buckthorn Brown to near (h) Yellow Ocher and from tone 2 to 4 of Brown Pink of Oberthür and Dauthenay, pl. 297, but with a trifle more reddish brown. Upper parts of uniform color except for occasional slight darkening of nose, forehead, and areas around eyes. Underparts darker (a) than Ochraceous-Buff. Color of underparts extends distally on posterior sides of forelegs over toes, onto antipalmar faces of feet and wrists, on medial sides of hind limbs over antiplantar faces of toes, tarsal region and sometimes in diluted fashion on proximal third of underside of tail. Least width of color of underparts averaging, in 6 male topotypes, 55 (43-81) per cent of greatest width of color of upper parts. Black tip of tail in same series of males averaging 58 (53-63) mm. long; thus averaging longer than hind foot and 33 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 6 ads., type and 5 topotypes): See measurements and plates 22-24. As described in M. f. nigriauris except that: Weight (6 ads.), 5.3 (4.5-6.1) grams; basilar length, 47.6 (46.0-48.6); (one skull, no. 335, with postorbital breadth more than distance between posterior borders of P4 and P2); interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bulla; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 2 to 3-1/2 (including I3) upper incisors; length of tympanic bulla more than length of lower molar and premolar tooth-row and shorter than rostrum.
Female: Adult skull of typical female not seen.
As compared with the skull of the type specimen of inyoensis, skulls of adult males of pulchra are larger throughout, relatively broader, especially in the preorbital part of the skull, have more inflated tympanic bullae, and are less convex in dorsal outline. Comparison of the skull with that of latirostra has been made in discussion of that subspecies. Comparison of skulls of adult males of nigriauris and pulchra shows that those of pulchra average larger in every measurement taken except those of m1, M1, P4, and depth of skull at posterior borders of upper molars. The basilar length is only slightly more and it follows that, relative to this length, other measurements of the skull are relatively, as well as actually, larger. In no one measurement is there an entire lack of overlap, but the skulls of adult males, and probably adult females, may be distinguished from those of nigriauris by the combination of the following mentioned, average differences: Tympanic bullae larger in each of three dimensions; preorbital and interorbital parts of skull broader and notably heavier; interorbital breadth greater; zygomatic arches more expanded laterally; mastoid processes more prominent. As compared with xanthogenys, differences of similar nature, but of greater degree, distinguish pulchra. As compared with those of nevadensis (represented by specimens from Mono Co., Calif.), skulls of adult males of pulchra average larger in every measurement taken and no overlap exists in basilar length, orbitonasal length, mastoid breadth, zygomatic breadth, length of tympanic bulla, or depth of skull at either the anterior margin of the basioccipital or at the posterior margins of the upper molars. Relatively, the preorbital portion is about the same size in the two forms.
Remarks.—The best material of this big weasel was obtained in 1910 and 1911 by John Wimmer and forwarded to the California Academy of Sciences through John R. Rowley, although in 1905, one specimen had been obtained by A. S. Bunnell for the collections of the United States Bureau of Biological Survey, another by J. Grinnell for the Museum of Vertebrate Zoölogy in 1912, and in 1933, another by L. M. Huey, for the San Diego Society of Natural History.
The males from the type locality are relatively uniform in size and shape of skull. The one exception is no. 137935, U. S. Nat. Mus., slightly younger than the others. Its skull is relatively more slender than any of the others and does not display several of the differential characters. The male, no. 127566, U. S. Nat. Mus., from Alila (= Earlimart) is intermediate in cranial features between pulchra and xanthogenys as known from specimens taken in the vicinity of Fresno. The skull of the female, no. 127565, from the same place, is too young to provide diagnostic characters. Since the skull of an adult female of topotypical pulchra is unknown, doubt attaches to the identification of the adult, female specimen, no. 115895, U. S. Nat. Mus., from Delano. It has a relatively broad skull in comparison with the adult female of xanthogenys from Los Banos. The adult female, no. 9998, San Diego Soc. Nat. Hist., from 2 mi. SW Simmler, shows approach to nigriauris in slightly reduced size. The skin alone from Coalinga, a male, taken on April 10, 1935, measures 462, 179, 47. The adult female, with crushed skull, from 4 miles east of Coalinga, measures 350, 129, 40. In size, these specimens agree better with pulchra than with xanthogenys. The skin alone from 3 miles south of Coalinga is unsexed and without external measurements. Skulls of adults from Coalinga are needed to permit of more positive identification of the subspecies found there. The female from 4 miles east of Coalinga, taken on February 21, 1936, is in process of molt on the underparts, and the longer hair which is near (20´) Naples Yellow contrasts strongly with the incoming shorter hair which is near (10 c) Salmon-Orange. The skin alone, no. 16270, Mus. Vert. Zoöl., from Isabella, was made up from a decayed animal and is of but little use. It is referred to pulchra purely because of geographic nearness of Isabella to the type locality of pulchra. The most that can be told from the specimen is that it is a relatively light-colored, bridled weasel. The fact that the color is slightly darker than in pulchra may or may not indicate intergradation with nevadensis. No. 54103/41042, U. S. Nat. Mus., consisting of crushed bits of skull and the skin of the head, is from Willow Spring, Kern County. This marginal locality is really in the Mojave Desert rather than in the San Joaquin Valley. The light color of the skin of the head suggests pulchra, but it is realized that a complete specimen might show the animal there to be unlike pulchra.
None of the skulls shows evidence of having had the frontal sinuses infested by parasites.
Specimens examined.—Total number, 18, listed by counties from north to south. Unless otherwise indicated, specimens are in the Museum of Vertebrate Zoölogy.
California. Fresno County: Coalinga, 1[23]; 4 mi. E Coalinga, 1; 3 mi. S Coalinga, 1[8]. Tulare County: Alila (= Earlimart), 2[91]. Kern County: Delano, 1[91]; Buttonwillow, 9 (6[8], 2[91]); Isabella, 1; Willow Spring, 1[91] San Luis Obispo County: 2 mi. SW Simmler, 1[87].
Mustela frenata inyoensis Hall
Long-tailed Weasel
Mustela frenata inyoensis Hall, Carnegie Instit. Washington Publ. 473:99, November 20, 1936.
Putorius xanthogenys, Merriam, N. Amer. Fauna, 11:25, June 30, 1896 (part).
Mustela xanthogenys xanthogenys, Miller, U. S. Nat. Mus. Bull., 79:99, December 31, 1912.
Type.—Male, adult, skull (with skeleton) and skin; no. 25907, Mus. Vert. Zoöl.; Carl Walter's Ranch, 2 mi. N Independence, Inyo County, California; June 26, 1917; obtained by A. C. Shelton; original no. 3143.
The skull (plates 22-24) is complete and unbroken. All teeth are present and entire. The skin is well made and in good condition.
Range.—From 3700 feet (Lone Pine) to at least 4000 feet (Alvord); Lower Sonoran Life-zone of the floor of Owens Valley in Inyo County, California. See figures 29 and 30 on pages 221 and 314.
Characters for ready recognition.—Differs from M. f. nevadensis in presence of white facial markings; from M. f. pulchra in near (l) Brussels Brown rather than near (16 j) Buckthorn Brown to near (h) Yellow Ocher color of upper parts and basilar length of less than 45 in males; from M. f. latirostra in brownish rather than blackish color of inside of ear and orbitonasal length of more than 15.
Description.—Size.—Male: Two adults, the type specimen and no. 25392/32805, measure, respectively, as follows: Total length, 423 and 390; length of tail, 170 and 145; length of hind foot, 42 and 44. Tail is 67 and 59 per cent as long as head and body. Length of hind foot less than basal length.
Female: No. 12400, Field Mus. Nat. Hist., which is young, has the following measurements: Total length, 390; length of tail, 150; length of hind foot, 39. Tail is 63 per cent as long as head and body. Length of hind foot less than basal length.
The differences in external measurements between the two sexes, as represented by the male type specimen and by the young female, are: Total length, 33, length of tail, 20; length of hind foot, 3.
Externals.—Longest facial vibrissae black or dark brown and reaching beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles (in summer pelage) slightly less than shown in figure 19.
Color.—Large spot between eyes, band confluent with color of underparts, on each side of head extending anterodorsally anterior to each ear, upper throat, chin, lower lips and in some specimens part or all of upper lips white; patch between eyes and bars in front of ears tinged with some shade of yellowish in one specimen; dark spot posterior to each angle of mouth present in four of five specimens; tip of tail black; remainder of upper parts, in summer, near (l) Brussels Brown or tones 1 to 2 of Raw Umber of Oberthür and Dauthenay, pl. 301; slightly darker brown on forehead, nose and about eyes. In winter near (j) Snuff Brown or lighter than Brussels Brown with a smoked effect. Underparts Buff-Yellow, winter and summer. Color of underparts extends distally on posterior sides of forelegs over toes onto antipalmar faces of feet and wrists and on medial sides of hind legs over antiplantar faces of toes. Least width of color of underparts averaging, in 5 available specimens 34 (24-42) per cent of greatest width of color of upper parts. Black tip of tail, in two adult males, averaging 53 (45 and 60) mm. Thus longer than hind foot and averaging 34 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on the type): See measurements and plates 22-24. As described in M. f. nigriauris except that: Weight, 4.4 grams; basilar length, 44.7; postorbital breadth not less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; length of tympanic bulla less than length of lower molar and premolar tooth-row.
Female: Adult unknown.
Compared with the skull of the male of nevadensis, no single difference not covered by individual variation in nevadensis has been detected. Selected differences of inyoensis in comparison with latirostra are larger size, less inflated tympanic bullae and relative narrowness of the postorbital, interorbital and preorbital parts of the skull. Comparison of the skull with that of M. f. pulchra is made in the account of that subspecies.
Remarks.—Although two specimens of this subspecies were taken during the Death Valley Survey conducted by Dr. C. Hart Merriam, only three additional individuals are known to have been saved as study specimens since that time.
M. f. inyoensis as now known may be thought of as closely similar to M. f. nevadensis except for the presence of well-developed white facial markings like those found in the weasels of the San Joaquin Valley and coastal region of California south of San Francisco Bay. The nonwhite areas of the head are almost the same color as the back and not distinctly blackish as in M. f. latirostra and M. f. nigriauris. The one specimen in the winter coat, no. 25392/32805, U. S. Nat. Mus., from Lone Pine, is brown rather than white. The brown has the pale smoke-tinge common in the winter pelage of subspecies whose members are either brown or white in winter. The range of this subspecies is thought to include the floor and lower elevations of Owens Valley although it may occur in limited numbers southwestward along the base of the Sierra Nevada and through the mountains in places of low elevation like Walker Pass its range may meet that of pulchra.
The type specimen was taken in an alfalfa field by ranch hands, who, according to A. C. Shelton (MS), stated that the species was common at the type locality. None of the five specimens shows infestation of the frontal sinuses by parasites.
Specimens examined.—Total number, 5, listed by localities from north to south.
California. Inyo County: Alvord, 4000 ft., 1 (U. S. Nat. Mus.); 2 mi. N Independence, 1 (Mus. Vert. Zoöl.); Lone Pine, 3 (2 in Field Mus. Nat. Hist. and 1 in U. S. Nat. Mus.).
Mustela frenata neomexicana (Barber and Cockerell)
Long-tailed Weasel
Plates 1, 22, 23, 24, 34, 35 and 36
Putorius frenatus neomexicanus Barber and Cockerell, Proc. Acad. Nat. Sci. Philadelphia, 1898:188; Lantz, Trans. Kansas Acad. Sci., 19:178, 1905.
Mustela frenata neomexicana, Miller, U. S. Nat. Mus. Bull., 79:100, December 31, 1912; Bailey, Animal Life of Carlsbad Cavern, p. 97, 1928; Hall, Carnegie Instit. Washington Publ. 473:108, November 20, 1936.
Mustela frenatus neomexicanus, Bailey, N. Amer. Fauna, 35:19, September 5, 1913.
Type.—Male, adult, skull and skin; no. 10475, Mus. Comp. Zoöl.; Armstrongs Lake, Mesilla Park, Dona Ana County, New Mexico; February 1, 1898; obtained by A. C. Tryson; original no. 58 of C. M. Barber.
The skull is imperfectly cleaned but unbroken. The right upper incisors, right P2 and left p3 are broken away. The skin is indifferently stuffed but in a good state of preservation except that the distal part of the tail is missing. The animal's coat is ragged, and this imperfect appearance is heightened by injury to the posterior part of the body, probably at the time of capture.
Range.—From 3800 feet (type locality) to 9000 feet (Cloudcroft, N. Mex.); Upper Sonoran and Lower Sonoran life-zones of northern México, southeastern Arizona, New Mexico and western Texas, panhandle of Oklahoma, southeastern Colorado and southwestern Kansas. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. frenata and M. f. texensis by Buckthorn Brown rather than Brussels Brown color of upper parts, mastoid breadth of adult males ordinarily more, rather than less, than postpalatal length; from M. f. leucoparia by Buckthorn Brown rather than Argus Brown color of upper parts, distance from anterior margin of tympanic bulla to foramen ovale less, rather than more, than four-fifths height of tympanic bulla; from M. f. arizonensis and M. f. nevadensis by Buckthorn Brown, rather than near (14 n) Brussels Brown or, in winter, white color of upper parts, in presence of white frontal spot continuous with color of underparts, in basilar length of more than 46 mm. in males and 40 mm. in females; from M. f. longicauda by Buckthorn Brown rather than near (h) Clay Color of upper parts, by presence of white facial markings on Argus Brown head, and by length of tooth-rows amounting to less than 37 per cent of basilar length; from M. f. primulina by Buckthorn Brown rather than Brussels Brown color of upper parts, in presence of white frontal spot and broad white bands on side of head, in anteriorly truncate rather than anterolaterally rounded bullae and zygomatic breadth of more than 30 in males and 24 in females.
Description.—Size.—Male: The type specimen (see Barber and Cockerell, 1898:188) measured: Total length, 500; length of tail, 205; length of hind foot, 50. Tail 70 per cent as long as head and body. Length of hind foot less than basal length.
Female: No. 21779 from Tombstone, Arizona, measured: Total length, 419; length of tail, 165; length of hind foot from dried skin, 41 (probably 43 in flesh). Tail 65 per cent as long as head and body. Length of hind foot less than basal length.
The average differences in external measurements of the two sexes, as known from these two individuals, are: Total length, 81; length of tail, 40; length of hind foot, 7.
Compared with M. f. frenata, the size, proportions of parts and difference in size of the two sexes, appears to be about the same.
Externals.—Longest facial vibrissae colored like upper parts [in the type specimen some of the "long bristles of the upper lip" are white as pointed out by Barber and Cockerell (op. cit.: 188)] and extending beyond ear; carpal vibrissae colored like underparts and extending to apical pad of fifth digit; hairiness of foot-soles as shown in figure 20.
Color.—Broad white bands on sides of head, extending anterodorsally anterior to each ear, confluent with white spot between eyes and with color of underparts; posterior half or all of each upper lip edged with white; usually few white hairs on top of head between ears; remainder of top of head near Argus Brown of Ridgway and Chocolate, tone 4, of Oberthür and Dauthenay; dark spot posterior to each angle of mouth usually absent; tip of tail black; remainder of upper parts varying, in different specimens, from Buckthorn Brown to Dresden Brown of Ridgway, and Brown Pink (tones 3 to 4, pl. 297, of Oberthür and Dauthenay); underparts Antimony Yellow or near (c) Warm Buff of Ridgway, and Brown Pink (tone 1, pl. 297, of Oberthür and Dauthenay); color of underparts extends distally on legs over forefeet and hind feet. Least width of color of underparts averaging 46 (41-55) per cent of greatest width of color of upper parts; black tip of tail 35 to 45 mm. long in females; 43 to 68 mm. long in males and averaging 21 (20-36) per cent as long as tail-vertebrae.
No specimen of this subspecies in the white winter coat has been seen. Animals taken in midwinter are available from Mesilla Park, Willcox, and 10 miles east of Roswell.
From M. f. frenata, neomexicana differs in: upper parts and underparts much lighter colored; white facial markings more extensive; color of underparts more extended onto feet. From M. f. leucoparia, neomexicana differs as follows: above and below, much lighter colored, but white facial markings less extensive and color of underparts less extended onto feet and legs.
Skull and teeth.—Male (based on adults: the type; no. 131582 from Berino, New Mexico; and no. 1485 from Seward Co., Kansas): See measurements and plates 22-24. As described in Mustela frenata frenata except that: Weight, 6.2 (4.9 and 7.5); basilar length, 49.3 (48 and 50.5); mastoid breadth more than postpalatal length; least width of palate less than length of P4; anterior margin of masseteric fossa directly below m2 or heel of ml.
Female (based on three adults): See measurements and plates 34-36. As described in Mustela frenata frenata except that: Weight, 3.1 (2.6-3.5) grams; basilar length, 42.7 (40.8-45.5); zygomatic breadth less than distance between condylar foramen and M1 and more or less than distance between anterior palatine foramen and anterior margin of tympanic bulla.
The skull of the female averages 50 per cent lighter than that of the male.
As compared with the skull of the male of M. f. frenata, that of neomexicana is decidedly more angular and ridged. The postorbital constriction is narrower, the mastoid breadth greater (it is less than the postpalatal length in some subadult males), the sagittal crest much higher with impressions of the temporal and masseter muscles carried farther forward on the frontals, rostrum shorter and tympanic bullae wider and more inflated. Similar, though less marked, differences exist between the females. As compared with M. f. leucoparia and perotae, the same differences as noted above between frenata and neomexicana exist. In addition the tympanic bullae are so far removed from the foramen ovale that the distance from the anterior end of each bulla to the foramen ovale, instead of being less than the height of tympanic bullae, is in leucoparia more than four-fifths this height and in perotae more than the entire height. Also, in perotae, the squamosal, anterior to each tympanic bulla, is ventrally convex rather than ventrally concave as in neomexicana. Compared with M. f. longicauda, neomexicana is relatively narrower in the interorbital region, has relatively shorter tooth-rows, a V-shaped rather than a U-shaped interpterygoid space and in males has the interorbital region flat rather than convex and the sagittal crest is higher. The same differences are to be noted in comparison with nevadensis but here the difference in relative length of tooth-row is less. The same differences exist also in comparison with M. f. arizonensis except that its interorbital breadth, relative to the rest of the skull, is about the same. Difference in size is especially marked here; even females of neomexicana average larger than males of arizonensis.
Remarks.—When Barber and Cockerell named this subspecies in 1898, they had three specimens. Only two others are known to have been taken before this time. These are a skeleton, without corresponding skin, taken at Lozier, Texas, in 1890 by Wm. Lloyd, and no. 21779/36482, U. S. Nat. Mus., taken on April 6, 1893, by R. D. Lusk at Tombstone, Arizona. On the back of a label recently attached to the last mentioned specimen the name C. K. Worthen appears and probably signifies that the specimen was purchased from this dealer in vertebrate specimens.
M. f. neomexicana has a large geographic range. The old male from Liberal, Seward County, Kansas, extends the known range far to the northeast. Geographically, this occurrence is logical for the southwestern desertlike conditions extend to this part of Kansas. Probably the subspecies occurs in southeastern Colorado and in the panhandle of Oklahoma where conditions are similar. Bailey (1905:198) lists neomexicana as a member of the mammalian fauna of Texas. As stated by him (loc. cit.:198) this inclusion is based on geographic grounds and not on actual specimens. Strecker (1926:13) also includes neomexicana in his list of Texas mammals but writes me, under date of January 9, 1928, that "I included Mustela frenata neomexicana as a Texas mammal on the strength of its being mentioned by Bailey. . . ." On better ground, Bailey (1928:97) lists the subspecies as occurring in southeastern New Mexico at Carlsbad Cavern. However, Bailey (loc. cit.) knew of the existence of weasels just below El Paso and at Langtry, Texas. An unsexed skeleton, no. 167891, in the United States National Museum, from Lozier, Texas, is not certainly identifiable to subspecies. If, as I think, the animal is a female, its skull is intermediate between that of frenata and neomexicana although when all features are considered it is seen to be nearest the latter. The large size (basilar length of 46.5 mm.) may reflect some relationship to texensis. The field notes of the collector furnished me by Dr. H. H. T. Jackson (MS), describe the color as brownish yellow above and sulphur below. The admission of this subspecies to the list of mammals of Texas is made certain by the female (no. 1572, Texas Cooperative Research Collection) taken on July 28, 1940, 1-1/2 mi. NW Kent, Texas, by C. E. Scull.
The skull alone from Durango (City of), extends the known range far to the south. This skull is typical of neomexicana. Skins from the same place would be especially interesting as showing the approach, if any, in color, of neomexicana to M. f. leucoparia.
Mr. D. D. Stone of Casa Grande, Arizona, writes, under date of February 2, 1927, that a weasel was seen by an acquaintance of his in a field near Chandler, Maricopa County, Arizona. Probably this was neomexicana. If so, its range extends much farther west than collected specimens show.
Actual intergradation with M. f. frenata is not shown by the material at hand. The two females from Albuquerque, although typically neomexicana as regards color, have smaller, less prominently ridged skulls than females of neomexicana of the same age from farther south and approach M. f. nevadensis.
Probably the geographic ranges of M. f. neomexicana and M. f. latirostra do not meet; the only evidence of the existence of weasels in all of the large area, comprising western Arizona and the deserts of eastern California, which intervenes between the ranges of the two subspecies is the skull of a young individual, no. 68842, Mus. Vert. Zoöl., from 10 miles northeast of Bard, Imperial County, California. There, on December 29, 1932, Jack C. vonBloeker, Jr., retrieved the weathered skull with some of the vertebrae attached, from the top of a wood rat's nest beneath a mesquite tree near the west bank of the Colorado River.
The idea that the carcass may have been washed down the river from far upstream gains no support from a comparison of the specimen itself for the tympanic bullae are larger than in nevadensis and the skull is larger than the largest males seen of arizonensis, the two upriver races. On the basis of size the skull could be either a male of latirostra or a female of neomexicana. These two subspecies, like arizonensis and the skull in question, have much inflated bullae. However, the immaturity of the specimen conceals any other diagnostic cranial features, and prevents referring it certainly to either neomexicana or latirostra. In any event the specimen provides no evidence of intergradation between the two forms last mentioned. Speculating on its identity, I should say that it might be either an intergrade between arizonensis and nevadensis, from southern Utah or northwestern Arizona, or a member of an unnamed race resident in the lower part of the valley of the Colorado River.
Whereas M. f. panamensis and M. f. aureoventris are the darkest-colored weasels and occur in regions of heavy rainfall, M. f. neomexicana is the lightest-colored American weasel and occurs in an extremely arid region where the rainfall and humidity are slight.
According to Barber and Cockerell (1898:189) "The type specimen was shot in the grass on the shore of Armstrong's Lake. . . ." Bailey (1928:97) found the tracks of one of these animals "in the great pit at the west entrance to" Carlsbad Cavern and supposes they "hunt the cave walls for mice and other small game." Data on the label attached to no. 230973 states that the specimen was taken, two miles west of Willcox, Arizona, in a prairie dog town.
Only two of the 23 skulls show evidence of infestation of the frontal sinuses by parasites.
Specimens examined.—Total number, 28, arranged alphabetically by states and from north to south by counties in each state. Unless otherwise indicated specimens are in the United States National Museum.
Arizona. Graham County: Safford, 1. Cochise County: 2 mi. W Willcox, 1; Willcox, 1; 8000 ft., Chiricahua Mts., 1; 6000 ft., Pinery Canyon, Chiricahua Mts., 1[33]; Tombstone, 1; Sulphur Spring Valley, 1[74].
Durango. "Durango City," 1.
Kansas. Seward County: Liberal, 1[93].
New Mexico. Bernalillo County: 3 mi. NW Albuquerque, 2. Lincoln County: 7800 ft., South Fork Eagle Creek, White Mts., 1. Chaves County: Pecos River, 10 mi. E Roswell, 8[74]; Dexter, 1[74]. Otero County: Cloudcroft, 9000 ft., 1[90]. Dona Ana County: Mesilla Park, 2 (1[75], 1[7]); Berino, 2.
Texas. Culberson County: 1-1/2 mi. NW Kent, 1[90]. Terrel County: Lozier, 1.
Mustela frenata texensis Hall
Long-tailed Weasel
Mustela frenata texensis Hall, Carnegie Instit. Washington Publ. 473:99, November 20, 1936.
Mustela frenata, Strecker, The Baylor Bull., 27:14, September, 1924.
Mustela frenata frenata, Strecker, The Baylor Bull., 27:12, August, 1926 (part).
Type.—Male, adult, skull with skin of head, neck and tail; no. 14821, Amer. Mus. Nat. Hist.; Kerr County, Texas; September 17, 1897; obtained by H. P. Attwater.
The skull (plates 22-24) and dentition are complete and unbroken. The preserved parts of the skin are not stuffed.
Range.—Lower Sonoran and possibly Upper Sonoran life-zones of central Texas. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela frenata arthuri in possessing white facial markings and postorbital breadth less than distance between posterior borders of P4 and P2; from M. f. frenata in larger size of body and skull, the basilar length of which in adult males is more than 52.5; from M. f. neomexicana in Brussels Brown rather than Buckthorn Brown color of upper parts and basilar length of skull more than 52.5.
Description.—Size.—Male: Measurements taken from the dried skins of a young male, no. 15476, Mus. Comp. Zoöl., from Kerr County, Texas, and a subadult male, no. 2017, Baylor Univ. Mus., from 5 mi. N Waco, Texas, are, respectively, as follows: Total length, 600 and more than 510; length of tail, 200 and 225; length of hind foot, 52 and 52.
Female: Skins unknown.
Externals.—As described in Mustela frenata frenata.
Color.—As described in Mustela frenata frenata.
Skull and teeth.—Adult male: See measurements and plates 22-24. As described in Mustela frenata frenata except that: Weight, 8 grams; basilar length 54; least width of palate less than length of P4; anterior margin of masseteric fossa anterior to middle of m2.
Female: Skull unknown.
Remarks.—The type specimen, taken by the veteran collector of Texan mammals, H. P. Attwater, appears to have been the first one of these animals to find its way into the collection of any museum or naturalist. The second specimen from Kerr County was secured by, or through, the well-known commercial collector, F. B. Armstrong. Two trade skins, from Kerr County, taken on December 10, 1938, are in the Texas Cooperative Research Collection, as is also the skeleton of a young animal from Fredericksburg. The two other specimens from McLennan County (both males contrary to the statement of Strecker, 1924:14), owe their preservation to the alertness of John K. Strecker, Curator of the Baylor University Museum, who has given a complete account of their history.
The range of this subspecies is thought to include much of central Texas.
The preserved parts of the skin of the type specimen show it to have been generally large. The part of the tail preserved measures 226 millimeters and the skin of the head and neck is correspondingly large. The skin alone, no. 427, from near Waco, Texas, has, as now stuffed, a body 365 millimeters long. Individuals of this race attain larger size than those of any other American member of the subgenus Mustela with the possible exception of Mustela frenata macrophonius from Veracruz, México. In addition to large size, texensis and macrophonius are analogous in that each has a small geographic range at the northern end of an extensive range of its similarly colored southern relative from which it differs mainly in size. Each of the two groups, goldmani and macrophonius on the one hand and perotae, frenata and texensis on the other, has relatively uniform color, color pattern and body proportions over a large region but at its northern extremity develops a "giant" population, M. f. macrophonius and M. f. texensis, respectively. The skull of the type specimen of M. f. texensis is the largest one seen of any American weasel. The type specimen of M. f. macrophonius has a basilar length that is greater by one-tenth of a millimeter but in every other measurement taken the skull of M. f. texensis is the larger. Its weight, 8 grams, also shows it to be larger.
The broad, white bands in front of the ears are confluent with the white patch between the eyes on both sides in two specimens and on one side only in one other specimen. A white patch between the ears is present in four specimens. The dark spot at each angle of the mouth is absent on both sides in four specimens and on one side only in one other specimen. Thus out of a possible twelve cases, the broad bands in front of the ears are confluent with the spot between the eyes in five cases. Four of the six specimens have a white spot between the ears. The dark spot at each angle of the mouth is present three out of a possible twelve times.
The skull of no. 2017, from five miles north of Waco, is smaller than either of the two skulls seen from Kerr County and in this respect approaches M. f. frenata. There is no actual evidence of intergradation with any other subspecies but intergradation probably does take place with M. f. neomexicana and possibly with M. f. arthuri and M. f. primulina.
Strecker (1924:14) remarks that of the two specimens obtained near Waco, one was taken in a trap baited for mink and the other was shot in a hen house. None of the four skulls had the frontal sinuses infested with parasites.
Specimens examined.—Total number, 7, arranged by counties from north to south.
Texas. McLennan County: 5 mi. N Waco, 1[3]; Erath, 1[3]. Gillespie County: Fredericksburg, 1[90]. Kerr County: 4[75]; 1[2]; and 2[90] trade skins.
Mustela frenata frenata Lichtenstein
Long-tailed Weasel
Plates 1, 22, 23, 24, 36, 37, 38 and 40
Mustela frenata Lichtenstein, Darstellung neuer oder wenig bekannter Säugethier, 1832, pl. 42, and corresponding text, unpaged; Seton, Lives of game animals, 2:576, 1929.
Mustela brasiliensis Sevastianoff, Mem. de l'Acad. Imp. Sci. St. Petersburg, 4:356-363, tab. 4, 1813, name on plate only, the description being in the text (not of Gmelin, 1788); Gray, Proc. Zoöl. Soc. London, 1865:114.
Putorius frenatus, Baird Mamms. N. Amer., p. 173, 1858; Merriam, N. Amer. Fauna, 11:26, pl. 3, figs. 1, 1a, 1b, June 30, 1896; Bailey, N. Amer. Fauna, 25:198, October 24, 1905.
Putorius (Gale) brasiliensis aequatorialis Coues, Fur-bearing animals, p. 142, 1877, part? ("merely as a substitute for Gray's [supposedly] preoccupied name" that is, aureoventris).
Putorius (Gale) brasiliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877 (part).
Putorius mexicanus Coues, Fur-bearing animals, p. 142, 1877, [nomen nudum, cited by Coues in synonymy as "Putorius mexicanus, Berlandier, MSS. ic. ined. 4 (Tamaulipas and Matamoros)"].
Putorius brasiliensis frenata, Allen, Bull. Amer. Mus. Nat. Hist., 3:219, April 17, 1891.
Putorius brasiliensis frenatus, Allen, Bull. Amer. Mus. Nat. Hist., 6:197, May 31, 1894; Bangs, Proc. Biol. Soc. Washington, 10:9, February 25, 1896; Allen, Bull. Amer. Mus. Nat. Hist., 8:74, April 22, 1896.
Mustela frenata frenata, Strecker, The Baylor Bull., 27:12, August, 1926; Hall, Carnegie Instit. Washington Publ. 473:108, November 20, 1936.
Type.—Female, adult, skull and skin; no. 991, Berlin Zool. Mus., México City, México; June, 1829; obtained by F. Deppe.
The specimen once mounted, now is remade into a study skin and lacks the distal part of the tail. The skull (plates 36-38, 40) lacks the basicranial region.
Range.—Altitudinally, sea level (Brownsville, Texas) to 7600 feet (Tlalpam, México); from southern Texas as far south as México City; Lower Sonoran to at least Transition life-zone. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. perotae in nonextension of blackish over anterior fourth of neck, least width of color of underparts more than 37 per cent of greatest width of color of upper parts; height of tympanic bulla more than distance from its anterior margin to foramen ovale; from M. f. leucoparia by restricted white facial markings that cover less than half surface of head in front of ears, by nonextension of black of head onto anterior half of neck and by wider (more than 7.8) tympanic bullae; from M. f. neomexicana by Brussels Brown rather than Buckthorn Brown color of upper parts and mastoid breadth less than postpalatal length; from M. f. texensis by smaller size of body and skull (basilar length in adult males less than 52.5); from M. f. arthuri by white facial markings and postorbital breadth less than distance between posterior borders of P4 and P2; from M. f. tropicalis by nonextension of blackish over anterior fourth of neck, least width of underparts more than 37 per cent of greatest width of upper parts, postorbital breadth of adult males less than distance between posterior borders of P4 and P2.
Description.—Size.—Male: Fifteen adults and subadults from Brownsville, Texas, yield average and extreme measurements as follows: Total length, 485 (430-556); length of tail, 202 (165-250); length of hind foot, 48 (40-55). Averages believed to be reliable but extremes probably are not. Tail averages 71 per cent as long as head and body. Length of hind foot less than basal length. Corresponding measurements of an adult male (topotype, no. 50826) from Tlalpam, México, are: 505, 203, 53. Another adult male, from Miquihuana, Tamaulipas, México, measures: 520, 215, 52.
Female: Six adults, subadults and young from Brownsville, Texas, yield average and extreme measurements as follows: Total length, 420 (362-456); length of tail, 173 (126-200); length of hind foot, 41 (40-46). Tail averages 70 per cent as long as head and body. Length of hind foot more (with possible exception of no. 36362/48732 U. S. Nat. Mus.) than basal length.
The average differences in external measurements of the two sexes are: Total length, 65; length of tail, 29; length of hind foot, 7.
Externals.—Longest facial vibrissae black and reaching beyond ear; carpal vibrissae same color as underparts and extending to apical pad of fifth digit; hairiness of foot-soles as shown in figure 20.
Color.—Spot between eyes, broad band, confluent with color of underparts, on each side of head extending anterodorsally anterior to each ear, and posterior two-thirds to one-half of each upper lip, white; remainder of sides and top of head, posteriorly to line connecting posterior margins of ears, blackish; dark spot posterior to angle of mouth present on both sides in about half the specimens; tip of tail black; remainder of upper parts Brussels Brown; chin white; remainder of underparts near (16´a) Ochraceous-Buff (same color in juveniles and young), which color extends distally on posterior sides of forelegs over forefeet and on medial sides of hind legs to feet and sometimes onto upper sides of toes. Least width of color of underparts averaging, in a series of seventeen males from Brownsville, Texas, 47 (extremes 38-53) per cent of greatest width of color of upper parts. Black tip of tail, in same series, averaging 49 (extremes 40-55) mm. long, thus about equal to length of hind foot and averaging 24 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on ten adults from Brownsville): See measurements and plates 22-24; weight (three adults, one topotype and two from Brownsville, Texas), 6.2 (5.3-7.2) grams; basilar length, 49.8 (48.2-51.3); zygomatic breadth more than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoidal breadth less than postpalatal length; postorbital breadth less than length of upper premolars (less than distance between posterior borders of P4 and P2) and not greater (usually less) than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth not greater than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate more or less than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 3 or 4 (including I3) upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter (usually longer) than rostrum; anterior margin of masseteric fossa just behind m2.
Female (based on two adults from Brownsville, Texas): See measurements and plates 36-38, 40; weight, 3.4 (3.3-3.5) grams; basilar length (six, adult to young) 43.3 (41.3-47.3); zygomatic breadth more or less than distance between condylar foramen and M1 and more than distance between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth less than length of upper premolars and more or less than (about equal to) width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate less than outside length of P4; tympanic bulla as far posterior to foramen ovale as width of 2 to 3-1/2 (including I3) upper incisors; height of tympanic bulla more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum.
The skull of the female averages 45 per cent lighter than that of the average male.
Comparison of the skull with those of M. f. arthuri, tropicalis, perotae, leucoparia and neomexicana has been made in accounts of those subspecies. As compared with M. f. texensis (known only from males), the only difference detected is smaller size.
Remarks.—As Merriam (1896:27) has said: "In 1813 a Russian naturalist, Sevastianoff, gave the name 'Mustela brasiliensis' to a weasel brought to St. Petersburg by Capt. A. J. Krusenstern on his return from a voyage around the world. The animal was said to have come from Brazil, but no definite locality was given." This name was long applied by many European naturalists to American weasels which had white facial markings, and several American naturalists adopted the name. However, Lichtenstein in 1832 applied the name Mustela frenata to the weasels of the vicinity of México City and that name was used for bridled weasels from México and the southwestern United States by most subsequent German writers and by several Americans. In 1896 Merriam (1896:27) showed that Sevastianoff's Mustela brasiliensis, 1813, although probably the same as Mustela frenata, was preoccupied by Gmelin's Mustela brasiliensis, 1788, applied to an otter and that Lichtenstein's name must be used as the next available one. Since that time, 1896, frenata has been the name applied to the large bridled-weasels of Texas and the high table land of México south to México City. It may be added that in 1937 search by the writer among the specimens and records at the Russian Academy of Sciences, in Leningrad, failed to reveal any trace of the type specimen of Sevastianoff's Mustela brasiliensis.
The geographic range of this subspecies is relatively large and, as might therefore be expected, specimens show geographic variation. The specimens from Tlalpam, which Merriam (op. cit.:27) regards as topotypes, differ in certain respects from specimens from Texas. The skull of the adult male "topotype," no. 50826, differs from any other adult male seen in that the basilar length, the length of the upper tooth-rows, the orbitonasal length, the ratio of the same to the basilar length, the mastoidal breadth, the zygomatic breadth, the depth of the skull at the posterior margins of the upper molars, and the length and breadth of M1, are greater. The height of the tympanic bullae is less than the average height for these structures in more northern specimens. The specimens from Tlalpam have also larger external measurements than the average of more northern specimens. All of these features show an approach to the subspecies of more southern distribution. On the other hand, the blackish of the head is not more intense or more extended posteriorly onto the neck than in specimens from Brownsville, Texas. The skin, with skull crushed, no. 767, in the Paris Museum, from 3200 meters elevation near Toluca, does have the black color of the head extended 30 millimeters posteriorly to the ears. In this feature, and also in the extensively white face on which the white bar in front of each ear connects with the frontal spot, as well as with the color of the underparts, the specimen resembles leucoparia. Better material from the western part of the state of México may show the range of leucoparia to extend eastward almost or quite to Toluca.
An adult male, taken on July 15 at Miquihuana, Tamaulipas, is unique in several respects. The top of its head is black, rather than blackish, and this color extends posteriorly on the top and sides of the neck almost halfway to the shoulders. All of the upper parts are much more darkly colored than in other specimens of this race. The least width of the color of the underparts is 63 per cent of the greatest width of the color of the upper parts; thus the color of the underparts is considerably more extensive than in any other specimen seen. The underparts are more intensely colored than in the average specimen. The mastoidal breadth is greater than in any other adult male and amounts to more than the postpalatal length. On available maps the elevation of Miquihuana is given as 1892 meters (about 6200 feet). Thus the dark colors can hardly be ascribed to more tropical conditions than those under which animals from Brownsville, Texas, live. Brownsville is only a few feet above sea level and only 235 miles farther north. The difference noted, therefore, seems to be of geographic significance. However, there is from Alvarez, San Luis Potosí, approximately 115 miles south of Miquihuana, a young (nearly subadult) female, no. 21968, which is as light colored as specimens from Brownsville, Texas, or Tlalpam, México. The only distinctive feature of this specimen is the much greater extent of its white facial markings; they are more extensive even than in the specimen from Miquihuana.
Finally, the series from Brownsville, Texas, indicates that the animal there is smaller than frenata from the vicinity of México (city). The skull is similarly proportioned except that relative to the basilar length the orbitonasal length is more. Several other measurements of the skull of the adult male from Tlalpam, as pointed out above, are actually, although not relatively, greater than in any specimen from Brownsville. The similarities between specimens from the two localities, Tlalpam and Brownsville, are striking; since the two localities lie at opposite, extreme ends of the range more geographic variation would be expected. All that is known of the characters of populations from intermediate localities is that the one specimen from Alvarez shows no peculiarities whereas the one from Miquihuana suggests the existence there of a geographic variant.
None of the specimens seen shows actual intergradation with M. f. neomexicana or with M. f. arthuri but it is supposed that frenata intergrades with each of these subspecies. The difference between frenata and arthuri is greater than between frenata and neomexicana. Bailey (1905:198) records tracks of a weasel seen just below El Paso which he supposed had been made by a weasel of the neomexicana type. He also cited the taking of a weasel at Langtry which suggested to him (op. cit.) ". . . a continuous range from the country of frenatus up the Rio Grande to the type locality of neomexicanus at Mesilla Valley," New Mexico. Other records of occurrence in Texas cited by Bailey, in addition to those provided by specimens examined by the writer, are San Diego, Beeville, and Port Lavaca. The Port Lavaca record is the easternmost one assigned to the subspecies frenata; possibly specimens from there would be referable to arthuri.
The series of thirty-four specimens from Brownsville, Texas, permits measuring the amount of individual and age variation in several features. For instance, the material is sufficient to show that external measurements of subadults and those that fall in the upper part of the category designated as "young" may be included with the measurements of adults, because the mentioned measurements are not appreciably greater in adults. The series of skulls, although not providing more than six of any one age, shows the range of variation in size and proportion of certain parts and enables the student the better to evaluate cranial characters of nearby races known from only a few specimens. For example, not one of the twenty skulls of males from Brownsville and immediate vicinity is as large as either of the two specimens of texensis from Kerr County.
The white facial markings vary much in size and shape. In the series of thirty-four skins from Brownsville the broad white bands in front of the ears are confluent with the white patch between the eyes on both sides in three specimens and on one side only in six other specimens. These bands are confluent with the color of the underparts in all but two specimens. In one specimen the connection is lacking on both sides and in the other on one side only. A white patch between the ears is present in two specimens. The dark spot at each angle of the mouth is absent on both sides in eleven specimens and absent on one side only in ten others.
In six other specimens from parts of Texas north of Brownsville, the broad white bands in front of the ears are confluent with the white patch between the eyes on both sides in one specimen. A white spot between the ears is present in one specimen. The dark spot at each angle of the mouth is absent on both sides in six specimens and on one side only in three other specimens.
In eleven specimens from México, the broad white bands in front of the ears are confluent with the white spot between the eyes on both sides in two specimens and on one side only in one other specimen. The white spot between the ears is present in one specimen. The dark spot at each angle of the mouth is absent on both sides, in six specimens, and on one side only in one other specimen.
Thus, in 51 specimens the broad bands (one in front of each ear) are confluent with the white patch between the eyes in nineteen out of 100 instances, and not with the color of the underparts in three instances. A white spot between the ears is present in four specimens. The dark spot at each angle of the mouth is present 47 out of a possible 98 times.
Four juvenal specimens from Brownsville, Texas, with their dates of capture and probable age, are as follows: no. 58574, ♀, three weeks old, taken on February 15; no. 17318/24239, ♂, four weeks old, taken on March 16; no. 45899, ♀, forty days old, taken on May 21; no. 21778/36481, ♂, thirty days old, taken on October 20. In the order given, the dates of birth of these four juveniles would be approximately as follows: January 25, February 15, April 1, and September 20. The dates of birth of other specimens less than three months old as judged by the stage of development of the skull, and reckoning backward from the dates of capture, are as follows: April 1, April 30, May 25, October 12, and December 21. Thus, young appear to be brought forth at Brownsville, Texas, in the fall, winter and spring, that is to say from the latter part of September until the latter part of May.
Mustela frenata frenata is either free of the parasites that infest the frontal sinuses of most weasels, or withstands their presence remarkably well, for only one skull shows a definite pathological condition of the frontal sinuses.
Allen (1896:74) quotes H. P. Attwater, with respect to this species in Bexar County, Texas, as follows: "Not common, but occasionally met within the chaparral and cactus lands, where Wood Rats, Rabbits and Quail abound. They were frequently met with around San Antonio during the great 'Tramp Rat' [= Sigmodon hispidus texianus, see Bailey (1905:116)] invasion of 1889-90."
Specimens examined.—Total number, 63, arranged by counties, and in México by states, from north to south. Unless otherwise indicated specimens are in the collection of the United States National Museum.
Texas. Bexar County: San Antonio, 2 (1[2]). Goliad County: Charco, 1. Nueces County: Corpus Christi, 1[2]. San Diego County (not found), 1. Hidalgo County: La Hacienda, 1. Duval County: San Diego, 2[7]. County in question: Lower Rio Grande, 1. Cameron County: Brownsville, 34 (3[2], 4[1], 3[93], 2[75], 1[59], 1[60], 1[4]); no locality more definite than county, 2.
Nuevo León. Río Ramis, 20 mi. NW Montemorelos, 1[90].
Tamaulipas. Matamoros, 6; Miquihuana (now in Nuevo León), 1[75].
San Luis Potosí. Alvarez, 1[75].
México: Region montagneuse des environs de Toluca, Nevada Toluca, 3200 M., 1[84]
Distrito Federal. City of México, 2 (1[4]); Tlalpam, 2. No locality more definite than México, 4 (1[4], 3[7]).
Mustela frenata leucoparia (Merriam)
Long-tailed Weasel
Plates 1, 24, 25, 26, 29, 30, 36, 37 and 38
Putorius frenatus leucoparia Merriam, N. Amer. Fauna, 11:28, June 30, 1896.
Putorius brasiliensis frenatus, Allen, Bull. Amer. Mus. Nat. Hist., 2:165, October 21, 1889.
Putorius frenatus frenatus, Allen, Bull. Amer. Mus. Nat. Hist., 22:259, July 25, 1906.
Mustela frenata leucoparia, Miller, U. S. Nat. Mus. Bull., 79:100, December 31, 1912; Hall, Carnegie Instit. Washington Publ. 473:108, November 20, 1936.
Type.—Male, adult, skull and skin; no. 34914/47179, U. S. Nat. Mus., Biol. Surv. Coll.; Pátzcuaro, Michoacán, México; July 27, 1892; obtained by E. W. Nelson; original no. 2960.
The skull (plates 29 and 30) lacks most of the braincase; a fragment, consisting of the supraoccipital and the coalesced frontals and parietals remains. The rostrum, left zygomatic arch, palate, left pterygoid, left glenoid fossa and right postorbital process are intact. The teeth all are present and entire. The lower jaw lacks the right coronoid process and the lateral part of the articular condyle. The skin is well made and in good condition. It differs from an adult male topotype (36855, U. S. Nat. Mus.) and other referred specimens in having: the black of the head extended farther posteriorly on the neck, the maximum amount of white on the head, and a white stripe 50 mm. long extending down the middle of the nape from a point between the ears more than half way to the shoulders.
Range.—Sonoran and Transition life-zones of mountains west of México (city) in Michoacán and Nayarit. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. goldmani in least width of color of underparts more than 47 per cent of greatest width of color of upper parts, hind feet colored like underparts rather than like upper parts; postorbital constriction less than, rather than more than, combined length of upper premolars; from M. f. macrophonius by same details of coloration as from goldmani and by ventrally concave rather than ventrally convex pretympanic part of squamosal; from M. f. perotae by least width of color of underparts more than 40 per cent of greatest width of color up upper parts; height of tympanic bulla more than three-fifths distance from its anterior margin to foramen ovale; from M. f. frenata by white facial markings that cover half of surface of head in front of ears, by extension of black of head onto neck halfway to shoulders and by narrower (less than 7.8) tympanic bullae; from M. f. neomexicana by Argus Brown rather than Buckthorn Brown color of upper parts and distance from anterior margin of tympanic bulla to foramen ovale more, rather than less, than four-fifths of height of tympanic bulla.
Description.—Size.—Male: Two adults and one young from Los Reyes and Pátzcuaro, Michoacán, yield average and extreme measurements as follows: Total length, 514 (510-523); length of tail, 206 (196-215); length of hind foot, 55 (52-58). Tail averages 67 per cent as long as head and body. Length of hind foot more than basal length.
Female: One adult from Artenkiki, Jalisco, and one subadult from Pátzcuaro, Michoacán, measure, respectively, as follows: Total length, 412, 400; length of tail, 159, 159; length of hind foot, 41, 42. Tail averages 64 per cent as long as head and body. Length of hind foot equal to or greater than basal length.
The average differences in external measurements of the two sexes are: Total length, 108; length of tail, 47; length of hind foot, 13.
Mustela frenata leucoparia has a greater total length and length of tail than either M. f. frenata or goldmani. The hind foot is longer than that of frenata and approximately the same as in goldmani. Relative to the body length, the tail averages longer than that of goldmani and shorter than that of frenata.
Externals.—As described in Mustela frenata frenata.
Color.—Broad white bands on sides of head, extending anterodorsally anterior to each ear, confluent with white spot between eyes and with color of underparts; posterior third of each upper lip white; remainder of sides and top of head, and neck posteriorly to point halfway to shoulders from ears, black; no dark spots at angles of mouth; tip of tail black; remainder of upper parts Argus Brown; chin white and sometimes also chest, neck and medial sides of hind legs; remainder of underparts near (16´) Ochraceous-Buff (near (a) Ochraceous-Buff in juvenal female), which color extends distally over all of each foreleg (except its lateral face proximally from about middle of forearm) and on medial side of hind leg and over most of upper side of each foot. Least width of color of underparts averaging, in eight specimens, 54 (extremes 44-61) per cent of greatest width of color of upper parts; black tip of tail averaging, in four males, 52 (extremes 38-78) mm. long, thus averaging 25 per cent of length of tail-vertebrae.
As compared with M. f. frenata and goldmani: white facial markings more extensive; color of underparts less restricted and more extended on legs; black tip of tail relatively of about same extent as in frenata and thus much less than in goldmani; black color of head extending farther posteriorly than in frenata but not so far as in goldmani.
Skull and teeth.—Male (adult): See measurements and plates 24-26, 29, 30. As described in Mustela frenata frenata except that: Weight (no. 128972) 6.3 grams; basilar length, 51.2; interorbital breadth less than distance between foramen opticum and anterior margin of tympanic bulla; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4 or 5 upper incisors; height of tympanic bulla more or less than (about equal to) distance from its anterior margin to foramen ovale; anterior margin of masseteric fossa anywhere from slightly anterior, to slightly posterior, to m2.
Female (based on no. 26153): See measurements and plates 37-39. As described in Mustela frenata frenata except that: Weight, 3.6 grams; basilar length, 44.5; zygomatic breadth less than distance between condylar foramen and M1, or than between anterior palatine foramen and anterior margin of tympanic bulla; tympanic bulla as far posterior to foramen ovale as width of 4 or 5 upper incisors; height of tympanic bulla not more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row or than length of rostrum.
The skull of the female is 43 per cent lighter than that of the male.
Comparison of the skull with those of M. f. perotae, goldmani and neomexicana has been made in the accounts of those subspecies. As compared with that of frenata the main difference is the less inflated tympanic bulla, the height of which is approximately equal to, rather than decidedly more than, distance from its anterior margin to foramen ovale.
Remarks.—The first specimen known to have been preserved is the alcoholic in the British Museum of Natural History, taken in September, 1891, on the Río Santiago in Jalisco, by D. A. C. Buller. The other known specimens of this white-faced weasel are divided between the American Museum and the United States National Museum. The two referred specimens from Jalisco were the last of several helpful ones collected in México and Central America by J. H. Batty, and these two were taken less than three months before Batty's tragic death in Chiapas (see Allen, J. A., 1906:191). The five specimens from Michoacán were taken by Nelson or Nelson and Goldman together. Merriam had only three of these when he named the subspecies and remarked (1896:29) that "This form is the poorest subspecies described in the present paper." Although the form is not strongly marked, the two additional specimens from Michoacán and better comparative material than Merriam had confirm several of the differential characters ascribed to it by him and indicate the existence of still other characters.
M. f. leucoparia occurs in the Sonoran and Transition life-zones. No. 27258 from Los Masos, and no. 26153 from Artenkiki (see specimens examined for other spellings) approach true frenata in coloration. Each of these specimens has a few white hairs between the ears and the white patch between the eyes is confluent on one side only with the lateral white bands on the side of the head. No. 27258 from Los Masos has a dark spot at each angle of the mouth. The 7 other specimens are relatively uniform in coloration. Each has the white spot between the eyes confluent on both sides with the extensive white areas on each side of the face. None has a dark spot at either angle of the mouth. Of these 7 specimens, the type specimen and three others have white hairs forming a median line between the ears and a fifth specimen has a white spot behind each ear.
M. f. leucoparia is most like M. f. frenata. Unlike frenata, leucoparia has tympanic bullae that are less inflated, narrower and less projected, at their anterior margins, from the cranium. In these characters leucoparia is intermediate between M. f. frenata and M. f. goldmani. The latter subspecies has the least inflated, narrowest and least projecting tympanic bullae of the three. The black color of the head extends, on the average, farther posteriorly than in M. f. frenata but not so far as in M. f. goldmani. The general color, too, is intermediate between that of M. f. frenata and that of the much darker M. f. goldmani. The white facial markings are more extensive than in either M. f. frenata or M. f. goldmani. This applies to both the white area between the eyes and the one on each side of the head between the ear and eye. M. f. neomexicana, whose range possibly meets that of M. f. leucoparia, also has more extensive white facial markings than M. f. frenata but less extensive markings than M. f. leucoparia.
On the basis of skulls alone, specimens of frenata from Tlalpam and those of leucoparia from Los Reyes can hardly be distinguished. This fact, and the circumstance that the specimens from the northern part of the range of leucoparia closely resemble frenata in color, constitute sufficient evidence for regarding the two as only subspecifically distinct. The female, no. 26153 from Artenkiki, as mentioned above, approaches true frenata in coloration. On this account it is not to be regarded as typical and it was because no other skulls of adult females were available that this one was used for comparison with females of allied races.
M. f. leucoparia is, then, a subspecies of the large, temperate-zone group and is unique in possessing the maximum extent of white facial markings.
None of the seven skulls shows signs of having had the frontal sinuses infested with parasites.
Specimens examined.—Total number, 8, all from México. Localities are listed by states from north to south. Specimens from Michoacán are in the United States National Museum; one from Río Santiago is in the British Museum of Natural History; all others are in the American Museum of Natural History.
Nayarit. Tepic, 1.
Jalisco. Río Santiago, 1; Los Masos, 1; "Artenkiki" (J. A. Allen, 1906, p. 238, writes "Artenkikil" and, on p. 259, "Artenkiki."), 1.
Michoacán. Zamora, 1; Los Reyes, 1; Pátzcuaro, 3.
Mustela frenata perotae Hall
Long-tailed Weasel
Mustela frenata perotae Hall, Carnegie Instit. Washington Publ. 473:100, November 20, 1936.
Putorius frenatus, Merriam, N. Amer. Fauna, 11: pl. 3, fig. 2, June 30, 1896.
Type.—Female, adult, skull and skin; no. 54278, U. S. Nat. Mus., Biol. Surv. Coll.; 12,500 feet, Cofre de Perote, Veracruz, México; May 26, 1893; obtained by E. W. Nelson; original no. 4864.
The skull (plates, 37-39) lacks the right zygomatic arch. Left p2 is missing. The skin is fairly well made and in good condition except that the extreme tip of the tail has been broken off and there are two holes in the right hind leg. The underparts show the beginning of a spring molt.
Range.—From 7500 (?) feet (Perote) to 13,500 feet (Popocatépetl), Upper Sonoran, Transition and Boreal life-zones of mountains along Puebla-México boundary, eastward to western central Veracruz and south into Oaxaca. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. frenata, its nearest relative, in extension from head of blackish onto anterior fourth of neck; restriction of color of underparts (least width of same less than 37 per cent of greatest width of color of upper parts), height of tympanic bulla less than distance from its anterior margin to foramen ovale; from M. f. macrophonius and M. f. goldmani in presence of, rather than absence of, color of underparts on hind feet; upper parts (black) Brussels Brown rather than Argus Brown or darker; from M. f. tropicalis in larger size (adult female with total length more than 400, basilar length more than 40, weight of skull more than 3 grams); postorbital breadth less than combined length of upper premolars; m1 more than 5.4 long; from M. f. leucoparia in white facial markings so restricted that spot between eyes is not confluent with white stripe in front of ear, or, if so, narrowly (less than 4 wide) confluent; color of upper parts extending onto antipalmar face of forefoot, least width of color of underparts not more than 40 per cent of greatest width of color of upper parts; height of tympanic bulla not more than three-fifths distance from its anterior margin to foramen ovale.
Description.—Size.—Male: A nontypical specimen from Cerro San Felipe, Oaxaca, measures: Total length, 500; length of tail, 205; length of hind foot, 52.
Female: The type specimen, measures: Total length, 418; length of tail, 160; length of hind foot, 45.
In this male the tail is 70, and in the female, 62 per cent as long as the head and body. In each the hind foot is longer than the basal length.
The differences in external measurements between these two specimens, representing the two sexes, are: Total length, 82; length of tail, 45; length of hind foot, 7.
Externals.—As described in Mustela frenata frenata.
Color (based on type specimen).—Color and color pattern as described in Mustela frenata frenata except that: blackish of sides and top of head extends one-fourth of way back to shoulders from ears; throat and breast as well as chin white; remainder of underparts near (16´ c) Ochraceous-Buff; least width of color of underparts equals 36 per cent of greatest width of color of upper parts; black tip of tail equal to 28 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on a referred specimen from Cerro San Felipe which certainly is nontypical): See measurements. As described in Mustela frenata frenata except that: Weight, 4.9 grams; basilar length, 49.2; postorbital breadth more than distance between posterior borders of P4 and P2; tympanic bulla as far posterior to foramen ovale as width of 5 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; zygomatic breadth less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla.
Female (based on type specimen, an adult): See measurements and plates 37-39. As described in Mustela frenata frenata except that: Weight 3.4 grams; basilar length, 43.5; zygomatic breadth less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; tympanic bulla as far posterior to foramen ovale as width of 5 or 6 upper incisors; height of tympanic bulla one-half to three-fifths distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer than rostrum.
The skull of the female is 33 per cent lighter than that of the nontypical (and smaller than average) male from Cerro San Felipe.
Comparison of the skull with that of M. f. tropicalis is made in the account of that subspecies. Compared with the skull of M. f. macrophonius, that of the female of perotae is more flattened, has the longitudinal dorsal outline distinctly concave rather than flat just behind the postorbital processes, and much wider tympanic bullae. Accordingly, the basioccipital is slightly narrower in perotae. The more marked postorbital constriction of the type specimen of perotae possibly is due to its relatively greater age. As compared with the skull of M. f. leucoparia, that of the female of perotae has less inflated tympanic bullae, the height of each being half as great as distance from its anterior margin to foramen ovale, whereas, in leucoparia (as represented by no. 26153) the two distances are equal. As compared with that of M. f. frenata, the skull of the female of perotae differs mainly in the lesser inflation of the tympanic bullae and their relative position. The height of each bulla is in perotae only half as much as, but in frenata more than, the distance from its anterior margin to foramen ovale. The anterior margin of the bulla is much less projected from the floor of the braincase in perotae. The squamosal anterior to each bulla is convex ventrally in perotae but flat or concave ventrally in frenata.
Remarks.—The type specimen and a juvenal female from the town of Perote were taken in the spring of 1893 by E. W. Nelson. Of these two, the type specimen was mentioned and figured by Merriam (1898:30, fig. 16 [= fig. 15], pl. 3, fig. 2) as Putorius frenatus. The referred nontypical specimen from Cerro San Felipe, Oaxaca, was referred by Merriam (op. cit.:29) to Putorius frenatus goldmani with the comment that it was intermediate ". . . both in coloration and cranial characters, between typical frenatus and goldmani;. . . ." No other published references to this subspecies, or specimens of it, have been seen. In 1941 and 1942, W. B. Davis and associates took four specimens along the boundary between the states of Puebla and México.
Although the specimen from Cerro San Felipe, Oaxaca, is referred to Mustela frenata perotae, to the description of which it answers best, that specimen, on account of its structural characters and geographic position relative to adjacent races, is in reality an intergrade between several of the adjacent races. Some of its intermediate characters are pointed out in the discussion of M. f. goldmani. In the specimens from 45 and 55 kilometers ESE of México (city) the black color of the top of the head does not extend so far behind the ears as in the holotype of M. f. perotae and in this feature the two specimens show intergradation between the two subspecies, perotae and frenata.
The type specimen taken on May 26, is acquiring new hair on the belly and lower sides which appears to be the result of a normal molt.
As would be expected from its geographic position, M. f. perotae resembles M. f. frenata of northern México and the high mountain forms of southern México more than it does the lowland tropical forms. This is true as regards size of entire animal, proportions of its parts, and size, general angularity and major proportions of its skull. The marked postorbital constriction, convex supralacrymal face of rostrum, width of tympanic bullae and angularity of the braincase place it nearest M. f. frenata as does also the color and color pattern. The ventrally convex squamosal anterior to each tympanic bulla and the slight degree of projection from the cranium of the anterior margin of each tympanic bulla are intermediate in degree between the condition in M. f. macrophonius and that in M. f. frenata. Thus M. f. perotae combines several characters of M. f. frenata on the one hand with some of M. f. macrophonius on the other and in some features, for instance in the size, shape and degree of inflation of the tympanic bullae, presents intermediate stages of development.
On the eastern plain below the high mountain, Cofre de Perote, there ranges the similarly colored, smaller, tropical weasel, Mustela frenata tropicalis. Between M. f. perotae and M. f. tropicalis there is marked differentiation in the skulls with much less differentiation in coloration. The differences in typical skulls of the two subspecies are so pronounced that one would, at first glance, hardly believe it possible for direct intergradation to occur between them on the sides of this mountain. Merriam (1896:30) thought that it did not. The two skulls figured by him (op. cit.:31) are a topotype of M. f. tropicalis from Jico and the one which now is the type specimen of M. f. perotae. They show the great difference in size and proportions and are females of comparable ages, not of different ages as I suspected before examining the skulls. However, despite this marked difference in the skulls, there is some, although not conclusive, evidence of intergradation furnished by a young female from Xuchil, Veracruz. This specimen is described in connection with M. f. tropicalis (see page 366).
None of the seven skulls shows marked deformity of the interorbital region, but two of the three adults appear to have had these parts infested with nematodes.
Specimens examined.—Total number, 7, all from México, listed by localities from north to south. Specimens from Veracruz and Oaxaca in the United States National Museum; remainder in Texas Cooperative Research Collection.
México: Monte Río Frío, 45 Km. ESE México City, 1; 55 Km. ESE México City, 1; N slope Mt. Popocatépetl, 13,555 ft., 1.
Puebla. Río Otlati, 8700 ft., 1.
Veracruz. Cofre de Perote, 12,500 ft., 1; Perote, 1.
Oaxaca. Cerro San Felipe, 10,000 ft., 1.
Mustela frenata goldmani (Merriam)
Long-tailed Weasel
Putorius frenatus goldmani Merriam, N. Amer. Fauna, 11:28, June 30, 1896; Elliot, Proc. Biol. Soc. Washington, 18:236, December 9, 1905.
Mustela frenata goldmani, Miller, U. S. Nat. Mus. Bull., 79:100, December 31, 1912; Hall, Carnegie Instit. Washington Publ. 473:109, November 20, 1936.
Type.—Male, adult, skull and skin; no. 77519, U. S. Nat. Mus., Biol. Surv. Coll.; Pinabete, Chiapas, México; February 10, 1896; obtained by E. A. Goldman (on attached label collectors recorded as Nelson and Goldman); original no. 9279.
The skull (plates 24 and 30) has the rostrum badly injured. All the right, and part of the left nasal, the upper part of the right maxilla, the postorbital process and intervening area of frontals are missing. Each zygomatic arch is broken but the parts are present and attached to the skull. The frontal and interorbital regions are greatly malformed owing to parasites that infested the sinuses. Right I2 and I3, right and left i3, and the medial parts of the paraconid and protoconid of right m1 are missing. The light facial markings are less extensive than in any of the referred specimens. These markings consist of a separate spot between the eyes and a white line, confluent with the color of the underparts, on each side of the head, that extends from the base of the ear to above the eye. The dark color of the underparts is represented at the angles of the mouth by a spot on the left side and a similar dark area, confluent with the dark color of the face, on the right side. The large size, characters of the skull, and scrotal pouch on the skin prove the specimen to be a male as stated on the label.
Range.—Two thousand five hundred feet (El Cipres, Guatemala) to 9500 feet (near Tecpám, Guatemala), Upper Tropical Life-zone of mountains and western coasts of southern México, Guatemala and Salvador. See figure 29 on page 221.
Characters for ready recognition (characters based on males).—Differs from M. f. nicaraguae and M. f. perda by larger size (total length of adult males more than 489), least width of color of underparts not less than 26 per cent of greatest width of color of upper parts, weight of skull of adult male more than 5 grams; from M. f. macrophonius by smaller size (total length of adult males less than 540), skull of male with basilar length less than 52.5 and weight less than 6 grams; from M. f. perotae (typical specimens of same sex not available) by darker color of upper parts which are Argus Brown or darker rather than Brussels Brown; nonextension of color of underparts onto hind feet; from M. f. leucoparia in least width of color of underparts not more than 37 per cent of greatest width of color of upper parts; color of underparts not extended onto hind feet; black tip of tail two-fifths rather than one-fourth as long as tail-vertebrae; height of tympanic bulla less than four-fifths distance from its anterior margin to foramen ovale.
Description.—Size.—Male: Four adults yield average and extreme measurements as follows: Total length, 508 (500-512); length of tail, 196 (185-207); length of hind foot, 55.5 (54-58). Tail averages 63 (59-67) per cent as long as head and body. Length of hind foot more than basal length.
Female: Typical specimen unknown.
Externals.—Longest facial vibrissae black and reaching beyond ear; carpal vibrissae wholly or in part of same color as upper parts and reaching as far as hypothenar pad; hairiness of foot-soles distinctly less than that shown in figure 20 on page 60.
Color.—Spot between eyes, band, confluent with color of underparts, on each side of head extending anterodorsally anterior to each ear and posterior third of each upper lip, white; remainder of sides and top of head and neck posteriorly to or slightly behind shoulders, black; dark spots at angles of mouth usually absent; tip of tail black; remainder of upper parts Argus Brown or near (n) Argus Brown; chin, throat and breast white; remainder of underparts near (16' c) Ochraceous-Buff; color of underparts extending distally on posterior sides of forelegs onto medial toes and on hind legs to points between knees and heels. Least width of color of underparts, in five adult males, averaging 28 (extremes 26-33) per cent of greatest width of color of upper parts; black tip of tail, in four adult males, averaging 40 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on five adults): See measurements and plates 24-26, 30; weight, 5.4 (5.3-5.5) grams; basilar length, 49.9 (49.6-51.3); zygomatic breadth (except in no. 12523 from Salvador) more than or equal to distance between condylar foramen and M1 or between anterior palatine foramen and anterior margin of tympanic bulla. Mastoid breadth less than postpalatal length; postorbital breadth more or less than length of upper premolars and greater than width of basioccipital measured from median margin of one foramen lacerum posterior to its opposite; interorbital breadth less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate more or less than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of five upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and shorter than or equal to length of rostrum; anterior margin of masseteric fossa immediately behind m2.
Female: Typical skull unknown.
Comparison of male skull with that of M. f. perda made in discussion of that form. Comparison with that of M. f. nicaraguae shows similar differences, some of which are more pronounced. For example, squamosals anterior to tympanic bullae more convex ventrally and these bullae project less from braincase than in M. f. perda; thus the difference in these features is greater between goldmani and nicaraguae than between goldmani and perda.
As compared with the skull of the male of M. f. macrophonius, each one of the skulls of the adult males of M. f. goldmani is smaller in every measurement taken, with two exceptions. The width of the tympanic bullae was more in three specimens of M. f. goldmani as was also the depth of the same in three specimens. Relative to the basilar length all but two of these measurements average less in goldmani; the exceptions are the zygomatic breadth and depth of the skull at the anterior margin of the tympanic bullae which average more. Relative to the basilar length, the orbitonasal length and depth of the skull at the posterior margin of M1 are less in each skull of goldmani. Thus, excepting the width and height of the tympanic bullae and the relative zygomatic breadth and relative depth of the braincase posteriorly, the skull of goldmani is shorter and relatively as well as actually narrower and lighter throughout.
As compared with the skull of the male of M. f. leucoparia, that of M. f. goldmani averages a trifle shorter and no skull of goldmani equals that of leucoparia in actual or relative zygomatic and mastoid breadths or length or height of tympanic bullae. In depth, the skull of goldmani averages actually and relatively greater. Its teeth are smaller. The squamosal anterior to each tympanic bulla is convex ventrally whereas it is concave ventrally in leucoparia as in frenata.
Remarks.—When Merriam (1896:28) named this subspecies, he had only one specimen but he called attention to the more important diagnostic characters, which additional specimens show pertain to the race as a whole.
M. f. goldmani in typical form occurs in high mountains of the Upper Tropical Life-zone and is most closely related to M. f. frenata and M. f. macrophonius. The altitude at which the two specimens were taken, twenty miles southeast of Teopisca in Chiapas, is not known. Merriam (1896:28) states that the type specimen was obtained at "about 8200 feet." The specimen taken by Stirton in Salvador comes from 8000 feet and the one obtained by Barber in Guatemala from 9500 feet. The specimen from Dueñas, the skin alone of a young animal, is not instructive.
As regards size, goldmani is larger than the immediately adjacent subspecies from the Lower Tropical Life-zone but is smaller than M. f. leucoparia or macrophonius. As compared with M. f. frenata, goldmani is longer, has an actually as well as relatively shorter tail, and a much longer hind foot.
The most outstanding difference in externals from frenata is the naked foot soles.
Molting probably takes place twice each year although actual proof of this is lacking. In number 133254 from twenty miles southeast of Teopisca, taken on May 12, the molt is well advanced. Another specimen from the same place still retains the winter coat.
In color, goldmani is much darker than frenata, has less extensive white facial markings, longer black tip on tail, more restricted color of underparts, and lacks the extension of color of the underparts onto the hind feet.
Of the adult males from the high mountains, the type specimen from Chiapas is lightest, and the one from Salvador is darkest. This progressively darker color to the southward probably is geographic variation.
In total length and relative and actual length of tail, the specimen from Salvador is the smallest of the five adult males from the higher mountains. In addition to its darker color and smaller size, no. 12523 from Salvador shows certain distinctive cranial characters. The zygomatic breadth is less than, rather than more than, or equal to, the distance between the condylar foramen and M1 or than that between the anterior palatine foramen and the anterior margin of the tympanic bulla. This difference appears to be correlated with geographic position, since no. 15953 from Guatemala has the three distances about equal and therefore is intermediate in this respect between the specimen from Salvador and those from Chiapas, in which the zygomatic breadth is greater than the other two measurements. Also in the greater depth of the skull and smaller size of the teeth, this specimen from Salvador approaches the subspecies of the Lower Tropical Life-zone. It has, however, the longest, highest and widest tympanic bullae of any of the five specimens. The amount of ventral convexity of the squamosal in front of each tympanic bulla appears not to be greater than in the other specimens.
As indicative of intergradation with perotae, leucoparia and possibly frenata, there is the specimen from Cerro San Felipe, Oaxaca. The degree of restriction of the color of the underparts is intermediate between that of goldmani and leucoparia. The same is true as regards the amount of projection from the braincase of the anterior margins of the tympanic bullae. The squamosal immediately anterior to each tympanic bulla is flat instead of ventrally convex as in goldmani or ventrally concave as in leucoparia and frenata. In accordance with the custom adopted in this paper of referring every specimen to some one subspecies, this specimen from Cerro San Felipe is referred to Mustela frenata perotae, to the description of which it most nearly answers.
Possibly goldmani, as here constituted, is a composite form. The specimens from the high mountains closely resemble one another. However, a specimen, no. 68541 from "Finca El Cipres," Guatemala, which place Mr. G. Goodwin tells me is at an elevation of 2500 feet, approximately 5 miles north of Retalhuleu, has a basilar length of 47.3 and is correspondingly small in other parts. This suggests the existence of a small, lowland race on the western side of the central divide corresponding to perda and tropicalis on the eastern side. From only a few miles away, at San Sebastian, there is available, the adult skull of a still smaller animal. This skull only, no. 41026, in the Berlin Zoological Museum, has a basilar length of 46.1, zygomatic breadth of 27.4, and other cranial measurements notably smaller than those of specimens from the high mountains. A skin-only, no. 12038, collection of Donald R. Dickey, from La Cebia, altitude 2150 feet, near the city of San Salvador, seemingly represents an animal smaller than typical goldmani. This specimen from La Cebia has the light color of the underparts extended distally on the hind legs to the tips of the toes as in M. f. tropicalis. However, the upper parts are darker and resemble those of M. f. goldmani. A fourth specimen from only 3500 feet elevation, on the south side of Volcano Tajumulco, Guatemala, no. 41768, Field Museum of Natural History, a subadult male, measures only 490 in total length and has the least color of the underparts so restricted as to amount to only 22 per cent of the greatest width of the color of the upper parts. Both these features are suggestive of the lowland races.
These four specimens indicate that the lowland population on the western side of the divide is smaller than the mountain population. The juvenile from Carolina and a young male from Finca Cipres, however, both closely resemble individuals of goldmani from the higher mountains. All these animals here are referred to goldmani. More specimens may reveal an amount and a pattern of geographic variation in weasels of this region that will require application of another subspecific name.
The female, no. 68540, from Puebla agrees remarkably well with the skull of the female, no. 132528, of macrophonius. Differences displayed by the specimen from Puebla are its slightly narrower braincase and longer space between the foramen ovale and anterior end of the tympanic bulla. Considering the far eastern location of Puebla (just north of Río Motagua, at 89° W, according to a sketch map provided by Mr. G. G. Goodwin), this specimen might be expected to show some approach to the small lowland races. Actually, however, it displays the characters of goldmani better than does the subadult female from Volcano San Lucas, which is nearer the metropolis of goldmani, and I assume at a higher elevation than Puebla.
Concerning this weasel Merriam (1896:29) says: "Mr. E. W. Nelson writes me that this fine weasel is found sparingly in the forest about Pinabete, Chiapas, at an altitude of 7000 to 8000 feet (2100 to 2500 meters). The type specimen was shot in the afternoon while hunting on a heavily wooded hill slope. It was heard making long, slow leaps over the dry, crisp leaves. Coming to a log, it stood up and rested its fore feet on the log, in which position it was shot by Mr. Goldman."
The specimen taken by R. A. Stirton in Salvador comes from an elevation of 8000 feet in the rain forest of the Upper Tropical Life-zone. Mr. Stirton tells me that one morning on visiting his traps set for small rodents, he found in one the partly eaten remains of a Heteromys. Leaving these remains as found he placed a steel trap beside them and on the following morning found the male weasel in the trap.
At least three of the ten specimens had the frontal sinuses infested with parasites.
Specimens examined.—Total number, 15, listed by localities from north to south, and unless otherwise indicated in the American Museum of Natural History.
México: Chiapas: 20 mi. SE Teopisca, 2[91]; Pinabete, 1[91].
Guatemala: Puebla, 1; Finca Porvenir, 3500 ft., S slope Volcan Tajumulco, 1[60]; Sierra [= ? Cerro] Santa Elena, 9500 ft. (near Tecpám), 1[60]; Carolina, 1; Volcano San Lucas, 1; "Finca El Cipres," 1; "Finca Cipres," 2500 ft., 1; Finca San Isidro, San Sebastión, Dept. Retalhuleu, 1[4]; Dueñas, 1[7]; no locality more definite than Guatemala, 1[7].
El Salvador: Los Esesmiles, 8000 ft., Chalatenango, 1[59]; La Cebia, 2150 ft., near San Salvador, 1[59].
Mustela frenata macrophonius (Elliot)
Long-tailed Weasel
Plates 24, 25, 26, 30, 37, 38 and 39
Putorius macrophonius Elliot, Proc. Biol. Soc. Washington, 18:235, December 9, 1905.
Mustela macrophonius, Miller, U. S. Nat. Mus. Bull., 79:100, December 31, 1912.
Mustela frenata macrophonius, Hall, Carnegie Instit. Washington Publ. 473:109, November 20, 1936.
Type.—Male, adult, skull and skin; no. 14063, Field Mus. Nat. Hist.; Achotal, Veracruz, México; January 15, 1904; obtained by Edmund Heller and Charles M. Barber; original no. 3424.
The skull (plates 24-26, 30) is complete and unbroken. Excepting right P2, which has been aborted or broken away, all the teeth are present. The skin is well made and in good condition. As shown by the scrotal pouch, the specimen is a male.
Range.—Tropical Life-zone, probably into Boreal life-zones, of mountains along eastern border of southern Veracruz. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. frenata frenata and M. f. perotae and M. f. leucoparia in lacking color of underparts on hind feet and in larger skull (skulls of adult males with basilar length more than 52.5); from M. f. goldmani by larger size of skull (see above) and entire animal and wider tympanic bullae; from M. f. tropicalis and M. f. perda by larger size (total length of adult males more than 510), postorbital breadth amounting to less than combined length of upper premolars.
Description.—Size.—Male: External measurements of the type specimen, an adult, are: Total length, 598; length of tail, 246; length of hind foot, 59. Tail 70 per cent as long as body; length of hind foot more than basal length.
Female: The skin, without field collector's measurements, of an adult female from Pérez, Veracruz, shows this sex to be correspondingly large. Because the skin is understuffed and because the hind feet are skinned out, reliable measurements can not be obtained from the dried skin.
Externals.—As described in Mustela frenata goldmani except that all carpal vibrissae are of same color as upper parts and that hairiness of foot-soles is halfway between that shown in figures 20 and 21.
Color.—As in darkest individuals of M. f. goldmani, thus, color of upper parts on posterior part of back near (n) Argus Brown. Color of underparts near (12) Mikado Orange in a juvenile, extending distally on posterior sides of forelegs onto inner toes and on hind legs to points between knees and heels. Least width of color of underparts 28 per cent of greatest width of color of upper parts. Black tip of tail 34 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on type specimen): See measurements and plates 24-26, 30. As described in Mustela frenata frenata except that: Weight, 6.9 grams; basilar length, 54.1; zygomatic breadth less than distance between condylar foramen and M1 or that between anterior palatine foramen and anterior margin of tympanic bulla; interorbital breadth less than distance between foramen opticum and anterior margin of tympanic bulla; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4 to 6 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; anterior margin of masseteric fossa below posterior half of m2.
Female (based on no. 132528): See measurements and plates 37-39. As described in Mustela frenata frenata except that: Weight, 3.6 grams; basilar length, 43.5; zygomatic breadth less than distance between condylar foramen and M1 and more or less than (in the single specimen, equal to) that between anterior palatine foramen and anterior margin of tympanic bulla; least width of palate more or less than (about equal to) outside length of P4; tympanic bulla as far posterior to foramen ovale as width of 4 or 5 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale.
The skull of the female is 48 per cent lighter than that of the male.
Comparison of the skull with that of M. f. goldmani is made in the account of that subspecies. Similar differences probably exist between males of perotae and macrophonius. As compared with skulls of males of M. f. tropicalis and perda, the skull of the male of macrophonius is larger in every measurement taken. The postorbital constriction is less, rather than more, than the combined length of the upper premolars. Relative to the basilar length, the following measurements are less than in any specimen of tropicalis or perda: length of tooth-rows; orbitonasal length; depth of skull at posterior border of upper molars; and depth of skull at anterior margin of basioccipital.
Remarks.—This large weasel appears to have escaped the notice of naturalists until the spring of 1903 when J. Friesser obtained an adult female and juvenal male at Pérez for the collection of the United States Bureau of Biological Survey. These specimens were tentatively referred to Mustela tropicalis. In the following January, Edmund Heller and Charles M. Barber obtained the adult male that was made the type specimen by Elliot who did not see, or if he did, did not mention, the specimens from Pérez. He did, however, refer a young female from Xuchil, Veracruz, to his Putorius macrophonius. This young female is here referred to Mustela frenata tropicalis.
The extent of the geographic range of this subspecies is not well known.
Mustela frenata macrophonius and M. f. texensis are the largest American weasels. The basilar length in the type specimen is greater by one-tenth of a millimeter than in the type specimen of M. f. texensis. The other cranial measurements taken are greater in M. f. texensis. The skull of the female from Pérez is one of the largest skulls examined of that sex. The juvenal male has teeth as large as those of the type specimen and the skull is the largest for its age of any seen. Although the skin of the female is understuffed and hence does not provide reliable measurements, it shows that the female is also large.
The white bands in front of the ears are confluent with the white patch between the eyes on one side only in one specimen. It is the juvenal male. These bands are not confluent with the color of the underparts on either side in the female and on one side only in the adult male. None of the specimens has a white patch between the ears. The dark spot at each angle of the mouth is present only in the juvenile where it occurs on each side. Of the three specimens, the juvenile is the darkest and the adult male the lightest. The white facial markings are most extensive in the juvenal male and the least extensive in the adult female.
M. f. macrophonius most closely resembles M. f. goldmani but in the relatively flattened braincase, deep constriction of the postorbital region and general angularity of the skull approaches M. f. perotae and M. f. frenata.
Only one of the three skulls, that of the female, shows evidence of infestation of the frontal sinuses by parasites, and this did not result in malformation of the interorbital region.
Specimens examined.—Total number, 3, all from México, listed by localities from north to south.
Veracruz. Achotal, 1 (Field Mus. Nat. Hist.); Pérez, 2 (U. S. Nat. Mus.).
Mustela frenata tropicalis (Merriam)
Long-tailed Weasel
Plates 25, 26, 27, 30, 37, 38 and 39
Putorius tropicalis Merriam, N. Amer. Fauna, 11:30, pl. 3, figs. 5, 5a, 6, 6a, text fig. 16, June 30, 1896; Merriam, Proc. Biol. Soc. Washington, 15:68, March 22, 1902.
Putorius frenatus, Merriam, N. Amer. Fauna, 11:27, June 30, 1896.
Mustela tropicalis tropicalis, Miller, U. S. Nat. Mus. Bull., 79:100, December 31, 1912; Allen, Bull. Amer. Mus. Nat. Hist., 35:99, April 28, 1916.
Mustela frenata tropicalis, Hall, Carnegie Instit. Washington Publ. 473:109, November 20, 1936.
Type.—Male, adult, skull and skin; no. 54994, U. S. Nat. Mus., Biol. Surv. Coll.; Jico, Veracruz, México; July 9, 1893; obtained by E. W. Nelson; original no. 5195.
The skull (plates 25-27, 30) is complete. All the upper incisors, except the second and third on the left side, are missing. The right upper canine is broken. The skin is well made and in good condition.
Range.—Up to 5000 feet (as now known) in Tropical Life-zone of Veracruz, México. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. frenata and M. f. perotae in least width of color of underparts not exceeding 36 per cent of greatest width of color of upper parts and in postorbital breadth exceeding length of upper molar and premolar tooth-rows; from M. f. macrophonius and M. f. perda in least width of color of underparts averaging more than 29 per cent of greatest width of color of upper parts; and from M. f. perda by longer tympanic bullae which in males are more than 14.9; and from M. f. macrophonius by lesser basilar length (not more than 48) and in postorbital breadth exceeding length of upper molar and premolar tooth-row.
Description.—Size.—Male: The type specimen and no. 12764/11058, a subadult, from Jalapa, Veracruz, measure, respectively, as follows: Total length, 444, 442; length of tail, 175, 160; length of hind foot, 50, 47. The tail is 65 and 57 per cent as long as the head and body. The hind foot is more or less than (approximately equal to) the basilar length.
Female: Merriam (1896:31) gives the measurements of a female topotype (probably no. 54993, U. S. Nat. Mus., which has no measurements written on the attached label) as: Total length, 333; length of tail, 121; length of hind foot, 37. The length of the tail amounts to 57 per cent of the length of the body. The length of the hind foot of no. 54993, U. S. Nat. Mus. is the same as the basal length.
The differences in external measurements between the male and the female topotypes are: Total length, 111; length of tail, 54; length of hind foot, 13.
Externals.—As described in Mustela frenata frenata except that carpal vibrissae do not reach apical pad of fifth digit and hairiness of foot soles is less.
Color.—As described in M. f. frenata except that: Blackish of head extends half way or more from ears to shoulders; upper parts near (14) Brussels Brown or slightly faded tone 2 of Maroon of Oberthür and Dauthenay, pl. 341; underparts of juvenal pelage near (a) Ochraceous-Buff. Least width of color of underparts averaging (in three specimens from Jico and one from Jalapa) 34 (extremes 30-37) per cent of greatest width of color of underparts. Black tip of tail, in two male topotypes, 57.5 (55 and 60) mm. long; thus longer than hind foot and in each individual comprising 34 per cent of length of tail-vertebrae.
As compared with M. f. frenata: White facial markings slightly less extensive; blackish (not black) of head extending onto neck; upper parts slightly darker; ventral side of tail noticeably darker; color of underparts more restricted, averaging approximately one-third rather than nearly one-half width color of upper parts; black tip of tail one-third rather than one-fourth length of tail and much longer than hind foot. Similar differences of lesser amount exist between perotae and tropicalis. M. f. perda, macrophonius and goldmani bear the opposite relation to tropicalis. That is to say, in the latter: White facial markings slightly more extensive; blackish of head less extended over neck; upper parts markedly lighter; color of underparts less restricted and black tip of tail shorter.
Skull and teeth.—Male (based on type specimen and a subadult, no. 11058, from Jalapa): See measurements and plates 25-27, 30. As described in Mustela frenata perda except that: Weight 4.7 (4.6 and 4.7) grams; basilar length 46.7 (45.5 and 47.8); zygomatic breadth more or less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; least width of palate more than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4 (including I3) upper incisors; anterior margin of masseteric fossa below middle of m2 or posterior to that tooth.
Female (based on no. 54993 and no. 1060): See measurements and plates 37-39. As described in Mustela frenata perda except that: Weight (of 54993) 2.2 grams; basilar length, 37.5 (36.0-39.0); zygomatic breadth more or less than distance between anterior palatine foramen and anterior margin of tympanic bulla; least width of palate more than greatest length of P4; height of tympanic bulla equal to one-third to three-fourths of distance from its anterior margin to foramen ovale.
The skull of the adult female is 53 per cent lighter than that of the type specimen, a male.
Comparison of the skulls of males and females with those of M. f. perda, the nearest relative, has been made in the discussion of that subspecies. Some of the features that readily distinguish skulls of M. f. tropicalis from those of M. f. frenata, perotae and macrophonius are as follows: Weight less than 4.8 grams; basilar length less than 48; postorbital breadth more than length of upper M-Pm tooth-row. The skulls of male frenata, perotae and macrophonius are much larger, heavier, and are decidedly more angular with more constricted postorbital region the least width of which is less than the length of the upper premolars. In frenata the anterior margins of the tympanic bullae are protruded much farther from the braincase. The skull of the female of M. f. tropicalis is smaller, weighing less than 3 grams; basilar length less than 41; postorbital breadth more than length of upper molar and premolar tooth-row.
Remarks.—This subspecies was originally described by Merriam as a full species. Later he described Putorius tropicalis perdus as another subspecies. Allen (1916) placed P. t. perdus in synonymy but named Mustela tropicalis nicaraguae as new. In the present paper all three forms are recognized but are regarded as only subspecifically distinct from the other bridled weasels of México and Central America.
The limits of the geographic range of tropicalis are fairly well known on the south and west but the only specimen available from the tropical coastal region north of Jico, is a young female from a point 50 miles south of Victoria. Thus, how far north along the coast it ranges toward Matamoros, where M. f. frenata occurs, is not known. The three specimens from Jico, a young female from Jalapa and another adult collected by J. Potts and labeled as coming from México City, are assumed to be typical. The latter specimen certainly came from an elevation lower than that of México City because M. f. frenata occurs there. Although the female from Jalapa, agrees well with specimens from Jico, a male, no. 12764/11058, from Jalapa, has a relatively broader skull, as in perda, although the tympanic bullae are short as in tropicalis. The resemblances to perda in features of coloration are: slightly darker upper parts, and the termination just below the knees of the color of the underparts. There are three specimens labeled as from Orizaba that indicate intergradation with perotae as does also the coloration of the juvenal female from 5 kilometers north of Jalapa. The specimens labeled as from Orizaba are old, poorly-prepared skins, only two of which have partial skulls. The size and coloration of the skins suggest perotae as do also the partial skulls in some respects although the skulls show greater resemblance to those of tropicalis.
The topotype, female, no. 54993, was figured by Merriam (1896, fig. 16, p. 31) along with that of what now is the type specimen of M. f. perotae. Merriam called attention to the great difference in size between the skulls of the two sexes of M. f. tropicalis and compared the condition to that found in noveboracensis. Although the skull of the female from Jico is fully adult, it probably is exceptionally small.
The young female from Xuchil is indistinguishable in coloration from the juvenal female of M. f. perotae from Perote, but in size of skull and size of teeth is intermediate between the female of tropicalis from Jalapa and the females from Cofre de Perote.
There is then, indication of intergradation with M. f. perotae as well as with M. f. perda. M. f. tropicalis differs from M. f. perotae and M. f. frenata in about the same way that M. f. perda differs from M. f. goldmani and M. f. macrophonius. M. f. tropicalis and perda each is smaller and more intensely colored than goldmani and macrophonius, and inhabits the lowland to the east of their highland relative.
At least five of the nine skulls have the frontal sinuses infested by parasites.
Specimens examined.—Total number, 13, all from México, listed by localities from north to south. Unless otherwise indicated specimens are in the collection of the United States National Museum.
Tamaulipas. 50 mi. S Victoria, 1[71]. Near? México City, 1.
Veracruz. Jico, 3; 5 km. N Jalapa, 1[90]; Jalapa, 2 (1[2], 1[75]); Xuchil, 1[60]; Orizaba, 4 (2[75], 1[4]).
Mustela frenata perda (Merriam)
Long-tailed Weasel
Plates 25, 26, 27, 30, 37, 38 and 39
Putorius tropicalis perdus Merriam, Proc. Biol. Soc. Washington, 15:67, March 22, 1902.
Putorius (Gale) brasiliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877 (part).
Mustela tropicalis perda, Miller, U. S. Nat. Mus. Bull., 79:100, December 31, 1912.
Mustela tropicalis tropicalis, Allen, Bull. Amer. Mus. Nat. Hist., 35:99, April 28, 1916.
Mustela frenata perda, Hall, Carnegie Instit. Washington Publ. 473:109, November 20, 1936.
Type.—Male, subadult, skull and skin; no. 100041, U. S. Nat. Mus., Biol. Surv. Coll.; Teapa, Tabasco, México; March 31, 1900; obtained by E. W. Nelson and E. A. Goldman; original no., 14074.
The skull (plates 25-27, 30) is unbroken and all the teeth are present and entire. The skin is well made and in good condition.
Range.—Fifty feet (Chichén Itzá) to 4000 feet (San Vicente) in Lower Tropical Life-zone south from southern Veracruz through southern México into Guatemala. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. nicaraguae in lesser extent of color of underparts (not more than 22 per cent of greatest extent of color of upper parts), black tip of tail more than 38 per cent of length of tail, and broader skull (in adult males, mastoid breadth more than 23.9 and zygomatic breadth more than 27.4); from M. f. tropicalis in more restricted color of underparts (least width of color of underparts less than 28 per cent of greatest width of color of upper parts) and shorter tympanic bullae, which in males are less than 15; from M. f. goldmani by total length not exceeding 489, least width of color of underparts not exceeding 24 per cent of greatest width of color of upper parts, weight of adult skull less than 5 grams and basilar length less than 48.5.
Description.—Size.—Male: The type specimen and another subadult from San Vicente, Chiapas, measure, respectively, as follows: Total length, 473 and 443; length of tail, 184 and 169; length of hind foot, 51 and 51.5. The tail is 62 and 64 per cent as long as the head and body. The length of the hind foot is greater than the basal length.
Female: Estimates made from the dried skin of no. 218036 are: Total length, 375; length of tail, 140; length of hind foot, 40. The hind foot of no. 65422 from Catemaco also measures 40.
The average differences in external measurements of the two sexes are: Total length, 83; length of tail, 37; length of hind foot, 11.
Externals.—As described in Mustela frenata goldmani except that hairiness of foot soles is slightly less.
Color.—As described in Mustela frenata goldmani except that: back near (n) Argus Brown or Carbon Brown, tone 3, of Oberthür and Dauthenay, pl. 342; underparts Ochraceous-Buff. Least width of color of underparts, in four specimens, averaging 20 (extremes 18-22) per cent of greatest width of color of upper parts; black tip of tail, in two subadult males, averaging 48 (extremes 46-49) per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on type specimen and subadult no. 132997 from San Vicente): See measurements and plates 25-27, 30; weight 4.4 grams (same for each); basilar length 45.7 (45.3 and 46.1); zygomatic breadth less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth less than postpalatal length; postorbital breadth more or less than (approximately equal to) length of upper premolars and greater than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum not greater than length of tympanic bulla; least width of palate less than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4-1/2 to 5-1/2 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum; anterior margin of masseteric fossa below middle of m2.
Female (based on two subadults, nos. 65422 and 218036): See measurements and plates 36-39; weight, 2.4 (2.3-2.5) grams; basilar length, 40.5 (40.4-40.6); zygomatic breadth less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth more than length of upper premolars or than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate more than outside length of P4 and less than inside length of same; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 5 or 6 upper incisors; height of tympanic bulla equal to one-third to one-half distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and more or less than (about equal to) length of rostrum.
The skull of the female averages 48 per cent lighter than that of the male.
Comparison of the skull of the male with that of M. f. nicaraguae has been made in the account of that subspecies. The skull of the male as compared with that of M. f. tropicalis has shorter tympanic bullae, deeper braincase at anterior margin of basioccipital, lesser zygomatic and palatal breadth and smaller P4 and m1. The skull of the female is larger in every measurement taken except those reflecting width of the preorbital portion. This part is actually narrower but probably mainly because the females of perda are younger than those of tropicalis. Features in which three skulls of subadults of M. f. perda differ from the five adults of M. f. goldmani and show no overlap are: lesser basilar length, lesser weight, greater relative length of upper tooth-rows, greater relative width of rostrum, greater relative length of rostrum, lesser mastoid and zygomatic breadths, lesser width, length and height of tympanic bullae; lesser outside length of P4 and greater relative depth of braincase at anterior margin of basioccipital and at posterior margin of M1. Features in which perda averages less are: length of tooth-rows, interorbital breadth, orbitonasal length, relative zygomatic breadth, length of m1, outside and inside lengths of P4, width and length of M1, and depth of skull at posterior margin of M1. Features in which perda averages more than goldmani are: relative interorbital breadth, relative mastoid breadth and depth of skull at anterior margin of basioccipital. The length of the inner half of M1 averages the same. As compared with goldmani, the skull of the male of perda is shorter, otherwise generally smaller, but relatively broader except across the zygomatic arches, and relatively deeper. The anterior margins of the tympanic bullae project slightly less from the braincase and the squamosals immediately in front of these bullae are slightly more convex ventrally.
Remarks.—Described by Merriam in 1902 as a subspecies of Putorius tropicalis, the form perda was regarded by Allen (1916:99) as not subspecifically distinct from P. t. tropicalis.
This is the eastern, lowland subspecies of the Tropical Life-zone, corresponding to M. f. goldmani of the higher mountains just as M. f. tropicalis corresponds to M. f. frenata and perotae of the high mountains and table land. The difference in size between perda and nicaraguae and between perda and tropicalis is slight. M. f. perda is slightly less richly colored than M. f. nicaraguae but has the color of the underparts more restricted and has a longer black tip on the tail. In these respects it is second only to M. f. panamensis among Central American weasels. Evidence of intergradation with goldmani is furnished by the specimens from Cobán, Guatemala, and the nearby locality San Cristóbal in Verapaz, Guatemala. Reduced size as compared with goldmani suggests affinity with perda but the greater width of the light-colored underparts, which averages 24 (extremes 18-32) per cent of the greatest width of the color of the upper parts, shows approach to goldmani. Farther north, in Chiapas, however, specimens of perda from San Cristóbal and San Vicente are readily distinguishable from those of goldmani taken a few miles away at Pinabete and near Teopisca. The latter two localities are, however, several thousand feet higher than San Cristóbal (Chiapas) and San Vicente.
Two of the nine skulls (only 3 adult) examined for malformation of the frontal sinuses reveal infestation by parasites.
Specimens examined.—Total number, 18, listed by localities from north to south, and unless otherwise indicated in the United States National Museum.
Veracruz. Catemaco, 1.
Tabasco. Teapa, 2 (1[7]).
Chiapas. San Cristóbal, 1; San Vicente, 1; no locality more definite than state, 1.
Yucatán. Chichén-Itzá, 1[76].
Guatemala: Cobán, 2 (1[7], 1[4]); Finca la Providenci, S. Cristóbal, Verapaz, 3[76]; central Guatemala, 1; no locality more definite than Guatemala, 5 (2[7]).
Mustela frenata nicaraguae Allen
Long-tailed Weasel
Plates 1, 25, 26, 27 and 30
Mustela tropicalis nicaraguae Allen, Bull. Amer. Mus. Nat. Hist., 35:100, April 28, 1916.
Putorius tropicalis, Allen, Bull. Amer. Mus. Nat. Hist., 24:661, 1908.
Mustela frenata nicaraguae, Hall, Carnegie Instit. Washington Publ. 473:109, November 20, 1936.
Type.—Male, subadult, skull and skin; no. 30754, Amer. Mus. Nat. Hist., Matagalpa, Nicaragua; April 16, 1910; obtained by W. B. Richardson; original no., 712.
The skull (plates 25-27, 30) of the type specimen lacks the entire right zygomatic arch. Otherwise it is complete. The teeth all are present and unbroken. The skin is complete and unfaded but only partly stuffed.
Range.—Honduras and Nicaragua. Altitudinal and zonal limits unknown. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. costaricensis and M. f. goldmani in shorter black tip of tail (not more than 35 per cent of length of tail) and lesser width (usually not more than 7 mm.) of tympanic bulla; from M. f. perda in greater extent of color of underparts (22 or more per cent of greatest width of color of upper parts), shorter black tip on tail (not more than 35 per cent as long as tail) and narrower skull, the mastoid breadth in adult males being less than 23.9 and the zygomatic breadth less than 27.
Description.—Size.—Male: Average and extreme measurements of five subadults and one young (four from Matagalpa and one from San Rafel del Norte) are: Total length, 450 (420-480); length of tail, 178 (150-190); length of hind foot, 48 (46-50). Tail averages 65 (extremes 56-69) per cent as long as head and body. Length of hind foot (measurements from dried skins) more than basal length.
Female: Measurements unrecorded.
Externals.—As described in Mustela frenata goldmani, except that hairiness of foot soles (between that shown in figures 20 and 21) is less, slightly less even than in M. f. perda.
Color.—As described in Mustela frenata goldmani except that: Back near (n) Argus Brown, or Carbon Brown, tone 4 of Oberthür and Dauthenay, pl. 342. Underparts Ochraceous-Buff. Least width of color of underparts, in four males, young, subadult and adult, 24 (extremes 22-26) per cent of greatest width of color of upper parts; the corresponding per cent in one female is 32; black tip of tail, in two subadult males, averaging 29 (extremes, 28-30) per cent of length of tail-vertebrae; corresponding per cent in one female, 36.
Skull and teeth.—Male (based on type specimen, one adult topotype [?] and one subadult from San Rafel del Norte): See measurements and plates 25-27, 30. As described in Mustela frenata perda except that: Weight, 4.2 grams (estimated for adults); basilar length 45.0 (44.8-45.5); interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bulla; anterior margin of tympanic bulla as far posterior to foramen ovale as width of four to five upper incisors; length of tympanic bulla not less than length of lower molar and premolar tooth-row; anterior margin of masseteric fossa below anterior margin of m2 or posterior to that tooth.
Female: Skull unknown.
Comparison of the skull of the male with that of M. f. costaricensis is made in the account of that subspecies. As compared with that of M. f. perda, which it most closely resembles, the skull of the male has a narrower, shorter rostrum, lesser interorbital breadth, lesser mastoid and zygomatic breadths and slightly shallower braincase, measured at anterior margin of basioccipital. The tympanic bullae are slightly less projected, at their anterior margins, from the braincase and the squamosal, directly anterior to each, is a little more convex ventrally. The skull of M. f. nicaraguae is, then, slightly shorter than that of M. f. perda and relatively narrower.
Remarks.—When naming this form, Allen (1916:100) characterized it as "Similar to M. tropicalis tropicalis but general coloration much darker and the white face markings somewhat reduced in area." In the sentence preceding the one quoted, Putorius tropicalis perdus was placed as a synonym of Putorius tropicalis tropicalis. M. f. nicaraguae and M. f. perda are nearly alike in color and color pattern but differ in cranial characters. M. f. perda and M. f. tropicalis are widely different in color and more especially in color pattern but differ only slightly in cranial characters. The aggregate difference between perda and nicaraguae is less than that between perda and tropicalis. All three are lowland forms and each is smaller than the adjacent highland forms, namely, M. f. goldmani, macrophonius, perotae and frenata.
The weasels from Honduras definitely are not typical of nicaraguae as it is known from the specimens from Nicaragua itself. The specimens from the state of Tegucigalpa, Honduras, are larger. Some are darker than topotypical nicaraguae. The dorsal outline of the skull is more nearly flat (less convex) in some. In these and several other differential features studied, the average of specimens from Tegucigalpa is intermediate toward goldmani, but everything considered the animals seem best placed with nicaraguae rather than with goldmani or perda, to which latter also, they show some resemblance. With better material from Nicaragua and additional specimens from Salvador (here referred to goldmani) a restudy of all the material now referred to the three races named would be profitable. Aims of this restudy might be to determine if a highland race additional to goldmani should be recognized and if the lowland races perda and nicaraguae differ from one another in the way that the existing specimens indicate.
In the five males from Matagalpa, the narrow white band in front of each ear is confluent with the color of the underparts on one side only in one specimen and on both sides in two specimens. None of these bands is confluent with the white patch between the eyes. A dark spot at the angle of the mouth is present on one side in one specimen. The corresponding area is dark colored in all other specimens but not separated from the color of the upper parts. In the specimen from San Rafel del Norte the white bands are not confluent with the color of the underparts. The female from Mambacho has the mentioned bands confluent with the color of the underparts. This female approaches M. f. costaricensis in the dark color of the upper parts but has more extensive white facial markings than some specimens from much farther north. Like a female seen of M. f. costaricensis, this one has a "frosted" nape but the white hairs on the back of the neck are less numerous than in the female of M. f. costaricensis.
M. f. nicaraguae in typical form, then, is thought of as a small, lowland, tropical subspecies only slightly differentiated from M. f. perda. By reason of its intermediate characters, it constitutes a link between the lowland forms, and the larger animals called M. f. goldmani and M. f. costaricensis.
None of the four skulls from Nicaragua shows signs of infestation of the frontal sinuses by parasites.
Specimens examined.—Total number, 16, listed by localities from north to south. Specimens are in the American Museum of Natural History, unless otherwise indicated.
Honduras: Alto Cantoral, 2; Cerro Grande La Paz, 1. La Flor Archaga, 1[75]; Comayagüela, 1[75]; vicinity of Tegucigalpa, 2; no locality more definite than Honduras, 1[4].
Nicaragua: San Rafel del Norte, 1; Matagalpa, 6; Ma[o]mbacho, 1.
Mustela frenata costaricensis Goldman
Long-tailed Weasel
Plates 25, 26, 27, 28, 29 and 30
Mustela costaricensis Goldman, Proc. Biol. Soc. Washington, 25:9, January 23, 1912.
Mustela brasiliensis, Gray, Ann. and Mag. Nat. Hist., 14(ser. 4):374, 1874.
Putorius (Gale) brasiliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877 (part).
Putorius affinis, Merriam, N. Amer. Fauna, 11:31, June 30, 1896 (part).
Mustela affinis costaricensis, Allen, Bull. Amer. Mus. Nat. Hist., 35:101, April 28, 1916; Lönnberg, Arkiv för Zool., 14(no. 4):16, 1921.
Mustela frenata costaricensis, Hall, Carnegie Instit. Washington Publ. 473:109, November 20, 1936.
Type.—Male, young, skull and skin; no. 13770/37149, U. S. Nat. Mus.; San José, Costa Rica; obtained by C. H. Van Patten.
The skull (plates 28-30) is complete and unbroken. All teeth are present and unworn. The skin apparently has been remade. It lacks the distal two-thirds of the tail. The head is somewhat shrunken. The color is possibly faded but if so only to a slight degree. Otherwise, the skin is in good condition. The orange color of the underparts is so intense as to suggest that the full, adult pelage has not been acquired. No white markings are present on the face. There is no sex mark on the label attached to the skin but the size and proportions of the skull and the scrotal pouch on the skin prove that the specimen is a male. The presence of sutures on the dorsal face of the rostrum and the short, wide, and low sagittal crest show the specimen to be young.
Range.—Costa Rica. Altitudinal and zonal range unknown. See figure 29 on page 221.
Characters for ready recognition.—Differs from M. f. panamensis in lighter color of upper parts (tone 2 rather than tone 4 of Reddish Black of Oberthür and Dauthenay, pl. 344) and longitudinally flat interorbital region of skull; from M. f. nicaraguae in darker color of upper parts (of Oberthür and Dauthenay, tone 2 of pl. 344 rather than tone 4 of pl. 342) and greater width (more than 7) of tympanic bulla.
Description.—Size.—Male: No collector's measurements available of fully grown animals. Estimated measurements of adult males: Total length, 470; length of tail, 165; length of hind foot (taken from dried skins of 3 adults), 52 (50-52). Tail estimated to average 55 per cent as long as head and body. Length of hind foot more or less than (about equal to) basal length.
Female: A subadult or adult, from the Candelaria Mountains, and a subadult from Irazú, measure, respectively: Total length, 370, 385; length of tail, 130, 150; length of hind foot, 40, 31. Tail 59 per cent as long as head and body. Length of hind foot probably about equal to basal length.
The estimated differences in external measurements of the two sexes are: Total length, 92; length of tail, 25; length of hind foot, 16 (probably average difference is less).
Externals.—As described in M. f. panamensis (figure 21) except that foot soles are slightly more hairy.
Color.—As described in Mustela frenata panamensis except that: back is near Reddish Black, tone 2 of Oberthür and Dauthenay, pl. 344; chin, lips, and throat white or whitish; remainder of underparts near (c) Ochraceous-Buff; color of underparts rarely extending distally onto toes of forefeet. Least width of color of underparts, in eleven specimens, averaging 23 (10-36) per cent of greatest width of color of upper parts; black tip of tail, in six specimens, averaging 36 (31-38) per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on 2 adults, no. 3.2.1.6. from vicinity of San José and no. 11408, U. S. Nat. Mus., from "Costa Rica"): See measurements and plates 25-30; weight, 5.9 grams; basilar length 49 +; zygomatic breadth more than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth less than postpalatal length; postorbital breadth in undiseased skulls less than length of upper premolars (less than distance between posterior borders of P2 and P4) and less than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more or less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum more or less (about equal to) length of tympanic bulla; least width of palate less than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 5 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; tympanic bulla longer or shorter than (about equal to) lower molar and premolar tooth-row and longer or shorter than (about equal to) rostrum; anterior margin of masseteric fossa directly below posterior border of m2.
Female: Skull of adult unknown.
Comparison of the skull of the male with that of M. f. panamensis has been made in the account of that subspecies. As compared with that of M. f. nicaraguae the skull of M. f. costaricensis is heavier and in every measurement taken is larger. The skull is generally more massive and it follows that most measurements of depth and width are greater in relation to the basilar length as well as actually greater. The individual teeth are larger and the tympanic bullae wider and at their anterior ends are more projected from the braincase. Indeed the skull is more like that of M. f. goldmani than like that of M. f. nicaraguae.
Remarks.—The half dozen ill-prepared skins, with partial skulls inside, of this form in the United States National Museum long were referred either to Mustela brasiliensis or Mustela affinis. It was not until 1912 when Goldman studied these specimens that the distinctive characters of the Costa Rican weasel were recognized and made the basis of the name costaricensis.
M. f. costaricensis is well differentiated from M. f. nicaraguae and M. f. goldmani which occur to the northward and from M. f. panamensis which occurs to the southward and is a large, heavy-skulled, dark-colored animal with white facial markings restricted or absent. In the type specimen and the female from the Candelaria Mountains the white facial markings are only narrow facial bars or a few white hairs, but in the young male from Cervantes there is a well developed bar 6 millimeters wide on each side of the face and a separate nasofrontal spot, 10 x 12 mm. The young female from Cachí has a V-shaped frontonasal spot, on the right side of the face a white bar 5 mm. wide and 17 mm. long connected with the color of the underparts, and on the left side a white spot in front of the ear and another between the ear and eye. White facial markings were not recorded in the other specimens. The color of the upper parts is only a little less dark than those of M. f. panamensis. Owing to the numerous white hairs on the dorsal side of the neck, the nape of the female from the Candelaria Mountains has a frosted appearance not present in other specimens.
M. f. costaricensis is a large animal and among its geographic neighbors is approached in size only by a specimen of panamensis from Boquete, Panamá. Also the young male from Cervantes suggests panamensis in the less flattened interorbital region, but even so is more like costaricensis. The small size of two young males, one from Navarro and the other from the vicinity of San José, is suggestive of M. f. nicaraguae. However, the large size of most of the specimens and the configuration of the skull are more as in M. f. goldmani than in M. f. nicaraguae and thus suggest that the known specimens are of high mountain subspecies. The long black tip of the tail is another point of resemblance to M. f. goldmani, the high mountain subspecies to the north. Perhaps in the lowlands of Costa Rica, there are weasels of another subspecies.
Of the eight skulls examined for malformation of the frontal sinuses, each of the two adults and two subadults shows signs of having the frontal sinuses infested with parasites.
Specimens examined.—Total number, 14, listed by localities from north to south.
Costa Rica: Irazú (Frasu or Irasu on label), 3000 M., 1[4]; Cervantes, 1[2]; San José, 1[91]; vicinity of San José, 2[7]; Azahar Cartago, 1[78]; Tucurrique, 1[7]; Cachí, 1[7]; El Muñco [= Muñeco?] (Río Nivarro [= Navarro?]), 4000 ft., 10 mi. S Cartago, Caribbean Slope, 1[76]; Navarro, 1[91]; Candelaria Mts., 1[75]; no locality more definite than Costa Rica, 3[91].
Mustela frenata panamensis Hall
Long-tailed Weasel
Plates 1, 25, 26, 27, 28, 29 and 30
Mustela frenata panamensis Hall, Proc. Biol. Soc. Washington, 45:139, September 9, 1932; Hall, Carnegie Instit. Washington Publ. 473:109, November 20, 1936.
Mustela brasiliensis, Alston, Biol. Cent. Amer., Mammalia, p. 78, 1879.
Putorius affinis, Bangs, Bull. Mus. Comp. Zoöl., 39:49, April, 1902; Hollister, Proc. Biol. Soc. Washington, 28:143, July 10, 1914.
Mustela affinis, Goldman, Proc. Biol. Soc. Washington, 25:10, January 23, 1912; Hollister, Proc. Biol. Soc. Washington, 28:143, July 10, 1914.
Mustela affinis costaricensis, Allen, Bull. Amer. Mus. Nat. Hist., 35:101, April 28, 1916; Goldman, Smithsonian Miscel. Col., 69 (no. 5): 161, 1920.
Type.—Female, subadult, skull and skin; no. 170970, U. S. Nat. Mus., Biol. Surv. Coll.; Río Indio, Canal Zone, near Gatún, Panamá; February 17, 1911; obtained by E. A. Goldman; original no. 20897.
The skull is complete and unbroken. The left lower incisor is broken off but all the other teeth are present and entire. The skin is well made and seems to be in faded, worn, first, adult pelage.
Range.—Sea level (type locality) to 5800 feet (Boquete, see Bangs [1902:49]); Upper Tropical and Lower Tropical life-zones of Panamá. See figure 29 on page 221.
Characters for ready recognition.—Differs from both M. f. meridana and M. f. costaricensis in darker tone (tone 4 of Oberthür and Dauthenay, pl. 344) of color of upper parts and in convex dorsal outline of skull (Compare figures of mentioned subspecies on plates 25-27).
Description.—Size.—Male: Two adults from Boquete in the Museum of Comparative Zoölogy, nos. 10112 and 10113, measure, respectively, as follows: Total length, 480 and 400; length of tail, 170 and 143; length of hind foot, 52 and 43. Hind feet of two other adult males measure 46 on dried skins. Tail, in two specimens mentioned above, is 55 and 56 per cent as long as head and body. Length of hind foot, in each of three adults, slightly longer than basal length. Corresponding measurements of no. 178970 from Mt. Pirre are: 422, 164, 50. Tail 64 per cent (same per cent as in young male, no. 137514 from Boquete) as long as head and body, and hind foot longer than basal length.
Female: An adult and a young from Chiriquí, nos. 18434 and 18435 (Acad. Nat. Sci. Philadelphia), measure, respectively: Total length, 372, 389; length of tail, 138, 144; length of hind foot, 42, 41. The type specimen measures: Total length, 408; length of tail, 159; length of hind foot, 46.5. Tail 64 per cent as long as head and body, and hind foot longer than basal length.
The average differences in external measurements of the two sexes from the vicinity of Boquete are: Total length, 59; length of tail, 15; length of hind foot, 6.
Externals.—Longest facial vibrissae black and extending beyond posterior border of ear; carpal vibrissae wholly, or in part, black and extending as far as hypothenar pad; hairiness of foot-soles as shown in figure 21.
Color.—Usually, posterior fourth of each upper lip and sometimes few hairs in front of ear, white; sides and top of head and neck posteriorly to, or behind, shoulders, black; dark areas at angles of mouth confluent with color of upper parts; tip of tail, black; remainder of upper parts near (n) Bay of Ridgway and Reddish Black, tone 4, pl. 344 of Oberthür and Dauthenay; chin and lips, whitish; remainder of underparts Warm Buff or near (16´ c) Ochraceous-Buff; near (12) Salmon-Orange in juveniles and small young; color of underparts extending distally on posterior sides of forelegs to wrists, but not to soles, and on hind legs to or slightly below knees. Least width of color of underparts, in seven specimens, averaging 18 (extremes 11-28) per cent of greatest width of color of upper parts; black tip of tail, in five adults and subadults, averaging 45 (extremes 41-50) per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on three adults from Boquete): See measurements and plates 25-30; weight, 5 (4.5-5.4) grams; basilar length, 45.2 (42.8-48.3); zygomatic breadth more or less than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth less than postpalatal length; postorbital breadth more than length of upper premolars and more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth not less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum approximately same (more or less than) length of tympanic bulla; least width of palate less than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4-1/2 to 5-1/2 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; length of tympanic bulla more or slightly less than (approximately equal to) length of lower molar and premolar tooth-row or length of rostrum; anterior margin of masseteric fossa directly below posterior fourth of m2.
Female (based on subadult, type specimen and one adult from Siola): See measurements; weight, 3.3 and 2.1 grams; basilar length, 41.3 and 39.3; zygomatic breadth more than distance between condylar foramen and M1 and more or less than (about equal to) that between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth more than combined length of upper premolars or than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate more than length of P4 (less in the adult); anterior margin of tympanic bulla as far posterior to foramen ovale as width of five upper incisors; height of tympanic bulla less than (about half) distance from its anterior margin to foramen ovale; length of tympanic bulla less than length of lower molar and premolar tooth-row or than rostrum.
The skull of the one adult female from Chiriquí is 58 per cent lighter than the average of the two adult males.
The skull of the male of M. f. panamensis as compared with that of M. f. meridana, is heavier and averages larger in nearly every measurement taken. Relative to basilar length, tooth-rows, orbitonasal length, interorbital breadth and zygomatic breadth averaging narrower. Mastoid breadth always narrower. Tympanic bullae longer, narrower, and usually slightly less protruded. P4 and m1 larger. Dorsal outline of skull, viewed laterally, more convex. Postorbital breadth actually and relatively greater. Postorbital processes, mastoid processes, and sagittal crest not so well developed. Differences between skulls of females, in so far as known, similar to those described between males.
As compared with M. f. costaricensis, M. f. panamensis has a lighter skull averaging smaller in every measurement taken except interorbital breadth, which is greater. Relative to basilar length, width of rostrum, interorbital breadth and depth of skull at plane of upper molars, less. Tympanic bullae shorter, narrower, less protruded. P4, M1, and m1 larger. Dorsal outline of skull, viewed laterally, more convex. Postorbital breadth relatively and actually greater. Postorbital processes, mastoid processes, sagittal crest and lambdoidal crest less developed. No skull of an adult female of M. f. costaricensis is available for comparison.
Remarks.—This subspecies had not been recognized by previous workers because specimens from Panamá were supposed to be Mustela affinis Gray up until 1916, when Allen (1916:100) restricted the type locality of M. affinis to Bogotá, Colombia. At that time Allen referred specimens from Panamá to Mustela affinis costaricensis, and Goldman (1920:161) followed Allen.
The specimens examined show much variation. Part of this is geographic variation. For instance the specimens from Boquete approach M. f. costaricensis in size more than do those from farther south. Too few adult females have been seen to ascertain the amount of secondary sexual variation. Bangs (1902:49) suggested that the sex of no. 10113 was wrongly recorded and that it was not really a male. If so, this would reduce the range of apparent variation in size of males from Boquete by half and bring it into accord with the amount normally existing in adult males from one locality. No. 10113 is adult but the skin shows no mammae which would prove it to be a female instead of a pigmy male. Although even smaller than 10113, the type specimen is so much larger than females of M. f. meridana that I have wondered if it is correctly sexed. However, the fact that it was sexed by E. A. Goldman, a collector of wide experience, lessens the possibility that a mistake was made.
The color of the underparts is more restricted in panamensis than in any other subspecies of the species. Excluding the specimen from Mt. Pirre, the least width of color of the underparts averages 16 (extremes 6-24) per cent of greatest width of the color of the upper parts. This feature, together with the black color, imparts an appearance to the Panamá weasel that is strikingly like that of a mink. M. f. panamensis is one of the two blackest weasels; M. f. aureoventris is the other. Each of these subspecies occurs in a region of heavy rainfall and there clearly is a positive correlation between high humidity and intensity of color. The black tip of the tail, as regards extent, here reaches the maximum attained among Central and South American weasels. The foot soles are less hairy than in any other member of the subgenus Mustela. The tympanic bullae are lower and less inflated than in any other subspecies of the species.
Adequate specimens from central and southern Panamá may reveal the existence of one or more additional subspecies since animals from each of the three localities now represented differ from those from the other two and some of these differences are correlated with geographic position. However, specimens from all three localities agree in several features. For example all of them have the dorsal outline of the skull highly convex, transversely, and, more especially, longitudinally. In this respect they are sharply differentiated from any other American weasel. Nevertheless, M. f. panamensis is clearly a link between the North and South American subspecies and panamensis intergrades with the adjacent subspecies. The large size of the skull and teeth and the slightly more ventrally projected tympanic bullae of no. 10112 from Boquete approach features seen in M. f. costaricensis. The smaller size of skull and teeth of no. 178970 from Mt. Pirre are points of resemblance to M. f. meridana.
The type specimen was selected from a region where M. f. panamensis is thought to have its distinctive characters well developed. The specimen is not adult and, therefore, does not show as many differential characters as does a nontypical adult from Boquete. Nevertheless, the majority of the above mentioned differential characters are shown by the type specimen and an adult from the same place would, it is judged, show all the differential characters better than would an adult from Boquete.
Of the 11 skulls examined, 6 show no signs of having had the frontal sinuses infested with parasites.
Specimens examined.—Total number, 19, listed by localities from north to south and unless otherwise indicated in the United States National Museum.
Panamá: Boquete, 10 (3[75], 1[8], 1[2], 3[4], 1[7]); Río Gariche [é], 5300 ft., 1[1]; Siola, 1[1]; Chiriquí, 1[7]; Río Indio, near Gatún, 1; Mt. Pirre, 3 (2[1]); Calovebora, 1[7] (locality not found, possibly misspelling of Calovébora); no locality more definite than Panamá, 1[4].
Mustela frenata meridana Hollister
Long-tailed Weasel
Plates 25, 26, 27, 37, 38 and 39
Mustela meridana Hollister, Proc. Biol. Soc. Washington, 28:143, July 10, 1914.
Putorius affinis, Robinson and Lyon, Proc. U. S. Nat. Mus., 24:147, October, 1901; Allen, Bull. Amer. Mus. Nat. Hist., 30:256, December 2, 1911.
Mustela affinis, Osgood, Field Mus. Nat. Hist. Publ. 155, zoöl. ser. 10:61, January 10, 1912.
Putorius macrurus, Allen, Bull. Amer. Mus. Nat. Hist., 31:92, April 19, 1912.
Mustela affinis affinis, Allen, Bull. Amer. Mus. Nat. Hist., 35:100, April 28, 1916 (part).
Mustela affinis costaricensis, Allen, Bull. Amer. Mus. Nat. Hist., 35:101, April 28, 1916 (part).
Mustela frenata meridana, Hall, Carnegie Instit. Washington Publ. 473:110, November 20, 1936; Hall, Physis, 16:175, 1939.
Type.—Male, subadult, skull and skin; no. 123341, U. S. Nat. Mus., 1630 meters elevation, Montes de Mérida, near Mérida, Venezuela; August 14, 1903; obtained by S. Briceno.
The skull (plates 25 and 26) lacks the right exoccipital condyle and posterior half of the right zygomatic arch. The teeth all are present, unworn and entire. The skin is well made and complete.
Range.—Near sea level (San Julián) to 8500 feet (Montes de Culata, Mérida, Venezuela), and 9000 feet (Santa Elena, Colombia). Temperate to Subtropical life-zones of Venezuela and northern and western Colombia. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela africana stolzmanni by absence of median, longitudinal, abdominal stripe of same color as upper parts, presence of p2 and two roots, rather than one root, on P2; from M. frenata panamensis in lighter color of upper parts (tone 3 rather than tone 4, pl. 344, Reddish Black, Oberthür and Dauthenay), flat rather than convex dorsal outline of skull immediately behind postorbital processes (see pl. 27); from M. f. affinis, in males, by lesser average breadth and length of skull and greater actual and relative size (see measurements) of facial part of skull; from M. f. aureoventris, in males, by lighter upper parts (tone 3 rather than tone 4, pl. 344, Reddish Black, Oberthür and Dauthenay) and by smaller skull and teeth (basilar length less than 45, length of m1 less than 6.3, width of M1 less than 4.8, outside length of P4 less than 5.7).
Description.—Size.—Male: Average and extreme measurements of topotypes (as recorded by collectors on labels, and so uniform as to show them not to be accurate to within more than 5 mm.) are as follows: Total length, 434 (410-460); length of tail, 164 (150-180); length of hind foot, 50 (no variation in collectors' measurements). Tail averages 61 per cent as long as head and body. Length of hind foot more than basal length. Corresponding measurements of no. 22191, a young male from Mérida, measured by Osgood or Conover, are 439, 165, 54. The adult male no. 18703, from Páramo de Tama (eastern boundary of Venezuela) has the following measurements written on the label by Osgood: 404, 150, 47.
Female: Average and extreme measurements of topotypes (as recorded by collectors on labels and so uniform as to show them not to be accurate to within more than 5 mm.) are as follows: Total length, 347 (320-370); length of tail, 128 (120-130); length of hind foot, 40 (no variation in collectors' measurements). Tail averages 57 per cent as long as head and body. Length of hind foot more than basal length. Two females, adult no. 11034 and young no. 11033 from Cincinnati, Santa Marta, Colombia, measured by M. A. Carriker, Jr., measure, respectively, as follows: 371, 330; 140, 140; 38, 36. No. 14463, adult, from Río Zapata, Colombia, measured (by J. H. Batty), 315, 138, 39. No. 32182, adult, from Mira Flores, Cauca, Colombia, measured (by W. B. Richardson), 375, 150, 43.
The average differences in external measurements of the two sexes, at Mérida, are: Total length, 87; length of tail, 36; length of hind foot, 10.
Externals.—Longest facial vibrissae black (few rarely white) and extending beyond ear; carpal vibrissae colored like underparts or upper parts, and not extending beyond apical pad of fifth digit; hairiness of foot soles slightly greater than shown in figure 21.
Color.—As described in Mustela frenata panamensis except that: Posterior fourth of each upper lip rarely, and small spot in front of ear usually, white; black of head proper not extending back of ears and grading insensibly into color of upper parts; anterior half of upper parts of adults "frosted" with numerous white hairs (tick bites?), upper parts near (n) Bay or tone 2 of Reddish Black (pl. 344, Oberthür and Dauthenay) or tone 3 in freshest, unfaded pelage. Least width of color of underparts (in ten males from Mérida) 20 (17-23) per cent of greatest width of color of upper parts. Black tip of tail, in same series, 60 to 75 mm. long, thus longer than hind foot and 41 (40-44) per cent as long as tail-vertebrae.
Skull and teeth.—Male (based on type specimen and seven topotypes, five adults and three subadults): See measurements and plates 25-27; weight, 4.1 (3.8-4.3) grams; basilar length, 43.6 (42.3-44.3); zygomatic breadth more than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth less than postpalatal length; postorbital breadth greater than length of upper premolars or than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth not less than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum greater than length of tympanic bulla; least width of palate greater than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4 to 5 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; length of tympanic bulla more or less than (approximately equal to) alveolar length of lower molar and premolar tooth-row and shorter than rostrum; anterior margin of masseteric fossa posterior to m2 and confined to posterior third (34 per cent average, 32 minimum, 37 maximum) of mandible.
Female (based on four adult topotypes): See measurements and plates 37-39; weight (no. 143665), 2.3 grams; basilar length 37.2 (36.3-38.2); zygomatic breadth more or less than distance between condylar foramen and M1 or than distance between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth (sinuses badly infested with parasites) more than length of upper premolars or width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate more than length of P4; tympanic bulla as far posterior to foramen ovale as combined width of 4 to 5 upper incisors; height of tympanic bulla less than (one half to three fourths of) distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row.
The skull of the female is 44 per cent lighter than that of the average male.
Comparisons of the skull with those of M. f. panamensis and affinis have been made in the accounts of those subspecies. As compared with the skull of the male of M. f. aureoventris, that of meridana averages smaller in every measurement taken. Indeed, none of the skulls of meridana equals that of aureoventris in basilar length, length of tooth-rows, length of tympanic bulla, depth of skull at anterior margin of basioccipital or at posterior margin of upper molars, or measurements of teeth. Relative to the basilar length, most of the measurements are greater in meridana. Exceptions are the relative length of the tooth-rows, and the two measurements of depth of the skull which average less.
Remarks.—In 1914 when Hollister named this weasel he compared it with M. f. affinis and most of the differential characters which he ascribed to meridana were merely "more than" or "less than" in affinis. In affinis, Hollister included specimens from Chiriquí, Panamá, and the coast of Venezuela. The specimens from these three places were referred by Allen (1916:101) to Mustela affinis costaricensis, and he restricted (op. cit.:100) the type locality of Mustela affinis affinis to Bogotá, Colombia, and synonymized Mustela meridana with M. a. affinis. Hollister probably would not have named meridana had he had specimens from Bogotá for comparison and had he regarded them as topotypes of affinis for the difference is slight. Nevertheless, within the large geographic range of M. f. meridana there is some geographic variation. There is more of such variation in the color of the pelage than in shape and size of the skull. The specimen from San Julián is darker than the average and in this respect approaches true panamensis. San Julián is situated at a relatively low elevation on the coast of Venezuela.
M. f. meridana so closely resembles M. f. affinis that the writer has no quarrel with anyone who would synonymize meridana. However, as represented by topotypes, the two races unquestionably are, on the average, different, and specimens from the southeastern part of the range of affinis probably are individually distinguishable from topotypes of meridana.
Variation in the skulls of the series from Mérida is relatively small. This applies to both males and females. The external measurements recorded by native collectors are not accurate to within more than five millimeters but, considering this, variation in external measurements also seems to be slight. The difference in size of the two sexes appears to be uniformly greater than in weasels from Central America. The twenty-six topotypes show that the color and color pattern are relatively uniform. All are of nearly the same tone except juveniles or young which are, as in the case of panamensis, much brighter colored on the underparts. Also, the young have darker-colored upper parts. The adults, without exception, have numerous white hairs scattered over the back of the head, neck and between the shoulders. I have no trustworthy evidence to support the suggestion that these white hairs are the results of tick bites or that they are caused by other parasites which damage the hair follicles. The white facial markings vary relatively little in the 45 specimens carefully examined in this regard. Also, the variation in color pattern of the two sides of the head is small. Indeed, within rather narrow limits, the color of the two sides of the head is the same in every specimen except two. In these two the white spots anterior to the ears are confluent with the color of the underparts. Only one specimen, no. 21342, has a white spot between the eyes and this spot is small. Ten of the twenty-six specimens have a definite white spot or band in front of each ear. Two specimens have such a spot on one side only. The dark spots at the angles of the mouth are present on two sides in three specimens and on one side only in three others. The mentioned spots are, then, present nine out of a possible fifty-two times. When the spots are absent, dark color usually is present in the required area but is confluent with the color of the upper parts.
A young male from San Julián, Robinson and Lyon (1901:147) state ". . . was shot . . . as it ran over some bowlders in a ravine. Its eyes shone with the same greenish light as do the eyes of our common weasel, and it emitted the same strong odor." No. 14463, Am. Mus. Nat. Hist., from Río Zapata, Colombia, according to data on the label, was "taken in timber belt in valley in balk hills" and the native name is Cosonebi. Two specimens taken on the Páramo de Tama, head of Tachira River, Venezuela and Colombia are commented on by Osgood (1912:61) as follows: "One . . . was caught in a steel trap baited with birds and set by the side of a rushing mountain stream. . . . The other was shot in midday as it came prowling about our 'house' in the clearing. . . ."
Of the thirty-three skulls before me, twelve have the frontal sinuses malformed by parasites. These twelve include most of the adults for few of the subadults and fewer of the young show pathologic conditions in the frontal region.
Note on localities.—Several of the localities in Colombia mentioned in "Specimens examined" are described and located by Chapman (1917:640-656, pl. 41) in his "Distribution of Bird-life in Colombia." Place names for Colombia on labels, not found on any map, or duplicated names of which I can not certainly select one, are Río Barrotow, Río Oscuro, Río Zapata, Río Japata, Guasca and El Baldro. Sonson may or may not be the town of that name situated some eighty miles northwest of Bogotá and on the east flank of the Central Andes west of the Magdalena River on the drainage of the Cauca River. In Venezuela most of the specimens from Mérida are labeled 1630 meters, Montes de Mérida. San Julián is some seven miles east of La Guaira (see Robinson and Lyon, 1901:136). San Esteban is located a little way back from the coast between Puerto Cabello and Valencia. Páramo de Tama is on the Venezuelan-Colombian border near the source of the Tachira River (see Osgood, 1912:35). Mt. Duida is shown as at 3° 30´ N and 65° 40´ W by Chapman (1931:13) and Mt. Auyán-tepui as near 5° 15´ N and 62° 50´ W by Chapman (1937:760).
Specimens examined.—Total number, 78, arranged by localities from north to south and unless otherwise indicated in the British Museum of Natural History.
Venezuela: San Julián, 1[91]; Carácas, 2; Galipare, Cerro del Avila, 6500 feet, 1; San Esteban, 1[2]; Mérida, 45 (10[91], 14[2], 10[4], 2[60], 2[14], 1[78]); Páramo de Tama, 1[60].
Colombia: Páramo de Tama, 1[60]; Cincinnati, 3[9]; Valdiva, 3800 ft., 1; Medellín, 2; 7200 ft., Barro Blanco, 1[2]; Santa Elena, 9000 ft., 1[2]; Santa Elena, 1[2]; Sonson, 2 (1[91], 1[2]); Mt. Auyan-tepuy, 1[2]; Pueblo Rico, 5200 ft., 1[91]; Mira Flores, 1[2]; Jerico, near Cauca River, 1; Tornel, 20 mi. NE Quitichao, 1; Mt. Duida, 1[2]; El Tambo, Cauca, 1[78]; El Baldro, 1[2]; Río Japata, 2[2]; Río Zapata, 4500 ft., 1; Río Oscuro, 3300 ft., 1; Río Barrotow, 3300 ft., 1; Guasca, 1[75]; no locality more definite than Colombia, 1.
Mustela frenata affinis Gray
Long-tailed Weasel
Plate 30
Mustela affinis Gray, Ann. and Mag. Nat. Hist., 14(ser. 4):375, 1874.
Putorius (Gale) brasiliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877 (part).
Putorius affinis, Merriam, N. Amer. Fauna, 11:31, June 30, 1896.
Mustela affinis affinis, Allen, Bull. Amer. Mus. Nat. Hist., 35:100, April 28, 1916; Allen, Bull. Amer. Mus. Nat. Hist., 35:220, May 31, 1916.
Mustela frenata affinis, Hall, Carnegie Instit. Washington Publ. 473:110, November 20, 1936; Hall, Physis, 16:175, 1939.
Type.—Male, adult, skull with skin; no. 54.1.11.3 (skull originally numbered 195d, later 54.6.3.4), Brit. Mus. Nat. Hist.; Colombia [given as new Granada in original description]; purchased from Mr. S. Stevens. Type locality restricted by Allen (1916:99) to Bogotá, Colombia.
The skin is in a good state of preservation and has been made over into a conventional study specimen from a mount on exhibition. Exposure to light when mounted probably accounts for the faded color. The skull (plate 30) lacks the middle 9 mm. of the right zygomatic arch, occiput, basioccipital and posterior two-thirds of the left tympanic bulla. The teeth all are present and entire.
Range.—Four thousand six hundred feet (Quetame) to 9154 feet (El Carmen), Tropical to Temperate life-zones of eastern Andes of Colombia. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela africana stolzmanni by absence of median, longitudinal, abdominal stripe of same color as upper parts, by presence of p2 and by two roots rather than one root on P2; from M. frenata meridana, in case of males, by, on average, greater breadth and length of skull and lesser actual and relative size (see measurements) of facial part of skull; from M. f. aureoventris by lighter-colored upper parts (tone 2 rather than tone 4, pl. 344, Reddish Black of Oberthür and Dauthenay); from M. f. macrura by darker color (Reddish Black, tone 2, pl. 344, Ober. and Dauth., rather than Chocolate, tone 3, pl. 343, Ober. and Dauth.).
Description.—Size.—Male: Measurements in life, estimated from dried skins, are: Total length, 455; length of tail, 175; length of hind foot, 52. Proportions of parts supposedly as described in Mustela frenata meridana.
Female: Estimates from two dried skins: Total length, 365; length of tail, 135; length of hind foot, 43. Proportions of parts supposedly as described in Mustela frenata meridana.
The estimated differences in external measurements of the two sexes are: Total length, 90; length of tail, 40; length of hind foot, 9.
Externals.—As described in Mustela frenata meridana.
Color.—As described in Mustela frenata panamensis except that: posterior fourth of each upper lip and spot in front of each ear white in approximately half of the specimens; black of head proper not extending back of ears and grading insensibly into color of upper parts; upper parts near (n) Bay, or tone 2 of Reddish Black (pl. 344, Oberthür and Dauthenay). Least width of color of underparts (in five males from vicinity of Bogotá) 24 (15-29) per cent of greatest width of color of upper parts. Black tip of tail, in same series, 60 to 75 mm. long, thus longer than hind foot and averaging 38 per cent as long as tail-vertebrae.
Skull and teeth.—Male (based on three adults and two subadult topotypes): See measurements and plate 30. As described in Mustela frenata meridana except that: Weight, 4.5 grams (estimated); basilar length 45.8±; interorbital breadth not greater than distance between foramen opticum and anterior margin of tympanic bulla (type as in meridana where interorbital breadth is more than distance between foramen opticum and anterior margin of tympanic bulla); least width of palate not less than length of P4; masseteric fossa confined to posterior two-fifths (38 to 40 per cent; average 39 per cent) of mandible and not extended anteriorly to middle of m2.
Female: No adults examined.
As compared with M. f. meridana the skull of the male is larger, to the average amount of 2.2 mm. in basilar length and 1.2 mm. in zygomatic breadth of adults; length of tooth-rows and mastoid breadth average greater but relatively less; breadth of rostrum, interorbital breadth and orbitonasal length average actually and relatively less. Thus the skull of affinis is longer and broader, but the facial region is actually, as well as relatively, smaller. As compared with the skull of the male of M. f. aureoventris, that of M. f. affinis is about the same in basilar length. However, in no specimen of affinis are the measurements of length of tooth-rows or breadth of rostrum, actually, or relatively, as great as in aureoventris. The same is true of all measurements taken of M1, P4 and m1. The specimens from the vicinity of Quito and north of there, although referred to macrura, are nearly as dark as typical affinis, approach affinis in cranial characters, and indicate intergradation of affinis with macrura.
Remarks.—Mustela affinis was named by John Edward Gray in 1874 (p. 375) on the basis of a specimen from New Granada. Although usually synonymized with Mustela brasiliensis by later authors until 1896 when Merriam (1896:31) applied the name to weasels from Costa Rica, nearly all the South American and several of the Central American weasels have, at one time or another, had Gray's name, affinis, applied to them. Gray, in 1865 (p. 115) when giving measurements of Mustela aureoventris, probably mentioned the specimen, that later became the holotype. In 1916 (p. 98) Allen restricted the type locality to Bogotá, Colombia. Allen's action was a necessary procedure in clearing up the systematics of South American weasels and was based on good grounds. As set forth by Allen (loc. cit.), and more in detail by Chapman (1917:642), Bogotá has long been the shipping point for Colombian vertebrate specimens, many of which were obtained in the mountains to the east. Allen (1916A:220) quotes Thomas as saying that the type specimen was purchased from Stevens at about the same time that a number of Colombian birds were purchased from the same dealer. Also, specimens from Bogotá agree with Gray's description of the type specimen.
Mustela frenata affinis, as here defined, constitutes one of the several slight geographic variants met with, on the sides of, and between, the three north and south mountain chains of Colombia. The others are lumped under the name Mustela frenata meridana. M. f. affinis, in common with specimens from the northern part of the range of macrura has large teeth. Weasels of all of the region from Quito to Bogotá have large teeth. To the north there is the smaller-toothed meridana and to the south the smaller-toothed macrura grading into the still smaller-toothed agilis, and boliviensis.
Two skins, without corresponding skulls, from Caqueta are lighter colored than any others of affinis; possibly the skins are faded by exposure to light. Since they probably come from an elevation of less than 1000 feet in the Amazonian region, they may pertain to another subspecies.
Complete, unbroken, skulls of affinis are needed to ascertain the degree to which affinis and meridana differ in cranial features. The several specimens from the immediate region of Bogotá show well the color and the color pattern but lack collectors' measurements.
None of the ten skulls examined shows malformation of the frontal region due to infestation of the frontal sinuses by parasites. Possibly three of the four adults were infested, although not severely.
Specimens examined.—Total number, 27, arranged by localities from north to south and unless otherwise indicated in the United States National Museum.
Colombia: El Carmen, 1[2]; W. Cundinamarca, 1[7]; Muzzo [= Muzo?], 1[4]; Bogotá, 1; Castillo, near Bogotá, 1[7]; Fambrias, near Bogotá, 1[75]; Bogotá district, 1[2]; Choachí, 9 (1[75], 2[7], 1[84]); Páramo de Choachí, 2 (1[2], 1[84]); Laguna del Verjón (= City of Bogotá), 1[75]; Quetame, 2[2]; Fusagasuga, 1; Caqueta, 2[2]; no locality more definite than Colombia, 3 (1[7]).
Mustela frenata aureoventris Gray
Long-tailed Weasel
Mustela aureoventris Gray, Proc. Zoöl. Soc. London, 1864:55, pl. 8, 1864; Gray, Proc. Zoöl. Soc. London, 1865:115, 1865.
Putorius (Gale) brasiliensis var. aequatorialis Coues, Fur-bearing animals, p. 142, 1877, part? ("merely as a substitute for Gray's [supposedly] preoccupied name," that is, aureoventris).
Mustela affinis costaricensis, Allen, Bull. Amer. Mus. Nat. Hist., 35:101, April 28, 1916 (part).
Mustela macrura, Lönnberg, Arkiv för Zool., 14 (no. 4):11, 1921 (part ?).
Mustela frenata aureoventris, Hall, Carnegie Instit. Washington Publ. 473:110, November 20, 1936; Hall, Physis, 16:175, 1939.
Type.—Probably female, juvenile, skull with skin, no. 64.6.6.3 (formerly 1432a), British Mus. Nat. Hist.; probably Subtropical Life-zone of western Ecuador (locality given as Quito, probably because received from that place).
The skin, once exhibited as a mount, has lost some hair from the back and other parts of the body and is not suitable for remaking into a conventional study specimen. The skull lacks the occiput, basioccipital, premaxillae, upper incisors, two of the lower incisors, all of the canines, premolars 2/2 on both sides, right P3, left p3, and has the left jugal mesially defective. The premolars present are not all fully emerged.
Range.—Pacific coastal regions of Ecuador and Colombia; Subtropical and Tropical life-zones. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela africana stolzmanni by absence of median, longitudinal, abdominal stripe of same color as upper parts, by presence of p2 and by two rather than one root on P2; from Mustela frenata macrura by Reddish Black, tone 4, plate 344 rather than Chocolate, tone 3, pl. 343 (of Oberthür and Dauthenay), or slightly darker color of upper parts; from M. f. affinis and M. f. meridana by darker color (tone 4 rather than tone 2, Reddish Black of Ober. and Dauth.) of upper parts and larger size of teeth (M1 with length more than 2.4 and breadth more than 4.7; P4 with outside length more than 5.9; length of m1 more than 6.2).
Description.—Unless otherwise stated, information concerning this subspecies is derived from the one referred specimen available, a young male, no. 34677, Amer. Mus. Nat. Hist.
Size.—Male: Total length, 470; length of tail, 160; length of hind foot, 50. Tail 51 per cent as long as head and body.
Female: Not known.
Externals.—Longest facial vibrissae black and reaching beyond ear. Carpal vibrissae reaching to or beyond apical pad of fifth digit; hairiness of foot soles slightly less than shown in figure 20.
Color.—Sides and top of head and neck posteriorly to shoulders black; white facial markings represented by only five white hairs anterior to right ear, one anterior to left ear and three far back on forehead; dark areas at angles of mouth confluent with color of upper parts; tip of tail black; remainder of upper parts near (n) Bay or Reddish Black, tone 4 of Oberthür and Dauthenay, pl. 344; chin whitish; remainder of underparts Warm Buff, deep orange in juvenile, type specimen, according to Gray (1864, pl. 8); color of underparts extending distally on posterior sides of forelegs to wrists but not reaching foot soles and on hind legs to or slightly below knees. Least width of color of underparts equal to 15 per cent of greatest width of color of upper parts. Black tip of tail equal to 27 per cent of length of tail-vertebrae.
In color, no. 34677 is, to me as it was to Allen (1916:101), indistinguishable from the darkest specimens (nos. 178970 and 10112) of M. f. panamensis. Therefore, M. f. aureoventris is one of the two darkest subspecies of weasels.
Skull and teeth.—Male: See measurements and plates 27-29; weight, 4.3 grams; basilar length, 45.8; zygomatic breadth approximately equal to distance between condylar foramen and M1 and to distance between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth less than postpalatal length; postorbital breadth more than length of upper premolars and greater than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth greater than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less (at least in young specimen) than length of tympanic bulla; least width of palate seldom if ever greater than length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of three (including I3) upper incisors; height of tympanic bulla not greater than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and shorter than orbitonasal length; anterior margin of masseteric fossa below anterior half of m2.
Skulls of males of M. f. aureoventris, and Mustela frenata macrura from the vicinity of Quito so closely resemble one another as not to be distinguished with the material now available, although the teeth of aureoventris are larger. Comparisons of the skulls of males with those of M. f. meridana and affinis, which are readily distinguishable from those of aureoventris, have been made in the accounts of those subspecies.
Female: Skull of adult unknown.
Remarks.—This subspecies of the Tropical Life-zone, or at least the Subtropical Life-zone, of Ecuador, in certain cranial characters resembles Mustela frenata macrura of the Temperate Life-zone. The two differ markedly in color. Nevertheless, a large number of the specimens collected in Ecuador are intermediate in color as well as in zonal distribution.
The type specimen is young or a juvenile. The measurements of no. 34677 from Gualea indicate an animal similar in size to M. f. affinis. Gray (1864:55) states that the type specimen measures "Length of body and head 6 inches, of tail 4-1/2 inches." The plate (pl. 8) accompanying Gray's original description (loc. cit.) is marked one-half natural size and represents the animal as having a head and body length of eight and one-half inches. One year later Gray (1865:115) gives the measurements of this species as "Length of body and head 12, tail 8 inches." Since he had at this time another specimen, larger than the type specimen (which specimen later, probably, became the type of Mustela affinis Gray), the larger measurements probably were taken from it.
Geographically, and as regards cranial characters, Mustela frenata aureoventris is most closely related to M. f. affinis and to the northern section of M. f. macrura, but in color to M. f. panamensis. M. f. aureoventris and M. f. panamensis are the two darkest-colored subspecies and each occurs in a region of extremely heavy rainfall. There is a skin only, no. 32620, Amer. Mus. Nat. Hist., from Munchique, obtained on June 1, 1911, which is appreciably darker than specimens of M. f. affinis in corresponding pelage and is intermediate between M. f. affinis and M. f. aureoventris in color as it is geographically. The specimen measures 495, 202, 52.
The name Mustela aureoventris Gray has been regarded by most authors as preoccupied by Mustela auriventer Hodgson (1841:909). However, the writer is not of this opinion and agrees with Thomas (1920:224) that "The name aureoventris is not invalidated by the auriventer of Hodgson, as, apart from 'one-letterist' differences, its first half comes from the adjective aureus, while Hodgson's name is based on the substantive aurum, so that not only the spellings but the derivations are different." The spelling of Gray's name should be aureoventris for this is the spelling in the original description which in pagination precedes the colored plate of the animal that is labeled Mustela aureoventris. Putorius brasiliensis var. aequatorialis Coues (1877:142) is the only name known to the writer that has been proposed as a substitute for Mustela aureoventris Gray.
Thomas (1920:224) treats Mustela macrura Taczanowski as a synonym of Mustela aureoventris Gray. Allen (1916:101) also treats the two names as applying to the same kind of weasel but regards aureoventris as preoccupied and therefore uses the name macrura. Taczanowski's original description (1874:311) and plate of Mustela macrura indicate an animal that is lighter colored than M. f. affinis. Gray's original description (1864:55) and plate of aureoventris indicate an animal that is darker colored than M. f. affinis. Indeed Gray (1865:115) in speaking of the type of aureoventris as compared with an adult from New Granada [= Colombia] that probably later became the type specimen of Mustela affinis, states: "The young from Quito is much darker than the adult;. . . ." Comparison of the plates accompanying the original descriptions of aureoventris and macrura well illustrate the difference stated in the written descriptions. My examination of the type specimens of M. macrura and M. f. aureoventris shows them to have been fairly accurately portrayed in the plates accompanying the original descriptions. Accordingly the two names are used for the two kinds of animals which appear, however, to be only subspecifically distinct.
Comparison of Gray's plate (1864, pl. 8) with the available specimens from South America indicates that the name aureoventris is based on an individual that is lighter colored than no. 34677 Amer. Mus. Nat. Hist., from Gualea, Ecuador, but on one which resembles no. 34677 more than it does the lighter-colored specimens from the Temperate Zone of Ecuador and northern Perú. Because Quito, Ecuador, is in the Temperate Life-zone and because the available specimens from this zone in Ecuador and northern Perú are distinctly lighter colored than Gray's plate representing the type of aureoventris shows this specimen to be, it is judged to have come from an altitude lower than that of Quito (9350 feet, according to Chapman, 1926:717); probably it came from the Subtropical Life-zone of Ecuador. Indeed Gray (1864:55) did not say that the specimen was collected or obtained at Quito but that it was ". . . received from Quito. . . ." Chapman (1926:717) has pointed out that Quito, since 1846 has been the distributing point for bird skins which specimens ". . . come from the vicinity of the city, from the 'Napo' region on the Amazonian slopes of the Andes, and from Nanegal, Gualea, and other localities on the Pacific side rarely below the Subtropical Zone." It is also pointed out that only some of the specimens are labeled with their approximate place of capture and that even then these localities cannot be accepted as definite; they indicate mainly whether the specimen is from the eastern or western side of the Andes.
The above mentioned considerations and information gained by study of the specimens cause me to think that the type is an intergrade tending toward the lighter-colored Mustela f. macrura of the Temperate Zone although sufficiently dark to be referred to the dark subspecies represented by no. 34677 Amer. Mus. Nat. Hist., from Gualea, Ecuador.
The skull of no. 34677 shows no infestation of the frontal sinuses by parasites.
Specimens examined.—Total number, 3, as follows:
Ecuador: Gualea, 1, Amer. Mus. Nat. Hist.
Colombia: 8325 ft., Munchique, 1, Amer. Mus. Nat. Hist. In the British Museum of Natural History, the type, (1).
Mustela frenata helleri Hall
Long-tailed Weasel
Plates 27, 28 and 29
Mustela frenata helleri Hall, Proc. Biol. Soc. Washington, 48:143, August 22, 1935; Hall, Carnegie Instit. Washington Publ. 473:110, November 20, 1936; Hall, Physis, 16:175, 1939.
Type.—Male, adult, skull and skin; no. 24133, Field Mus. Nat. Hist.; 3000 feet, Hacienda San Antonio, Río Chinchao, Perú; August 22, 1922. Obtained by Edmund Heller. Original no. 6589.
The skull (plates 27-29) is complete and unbroken. The teeth all are present, entire and but slightly worn. The skin is well made, unfaded, and in good condition.
Range.—Three thousand feet (type locality) to 6700 feet (Ambo), Tropical and Subtropical life-zones of eastern Perú. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela africana stolzmanni by absence of median, longitudinal, abdominal stripe of same color as upper parts, presence of p2 and two roots rather than one root on P2; from Mustela frenata macrura by darker color (Carbon Brown, tone 3, pl. 342 rather than Chocolate, tone 3, pl. 343, Oberthür and Dauthenay) of upper parts.
Description.—Size.—Male: Measurements of the type specimen and topotype, no. 24132, are, respectively, as follows: Total length, 382, 418; length of tail, 152, 164; length of hind foot, 52, 48. Tail 66 and 65 per cent as long as head and body. Hind foot more than basal length.
Female: Measurements of two referred females, no. 24134 from Ambo and no. 24136 from Huanuco, are, respectively, as follows: Total length, 328 and 303; length of tail, 118 and 103; length of hind foot, 39 and 38.5. Tail 56 and 51 per cent as long as head and body. Hind foot shorter than basal length.
The average differences in external measurements of the two sexes are: Total length, 85; length of tail, 49; length of hind foot, 11.
Externals.—Longest facial vibrissae black and extending beyond ear; carpal vibrissae same color as upper parts and extending to apical pad of fifth digit; hairiness of foot-soles as shown in figure 20.
Color.—Rarely a few white hairs anterior to each ear; posterior fifth of each upper lip white; top of head, posteriorly to slightly behind ears, black, grading into color of upper parts of body; dark spots at angles of mouth absent; tip of tail black; remainder of upper parts near (n) Argus Brown and Carbon Brown, tone 3 (pl. 342, Oberthür and Dauthenay); chin whitish; remainder of underparts Warm Buff; color of underparts extends distally on posterior sides of forelegs to wrists but not reaching foot-soles and on hind legs to slightly below knees. Least width of color of underparts 24 per cent of greatest width of color of upper parts in each of two males and 19 to 30 per cent in three females. Black tip of tail longer than hind foot and averaging 40 (39-42) per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on type specimen and adult no. 24132): See measurements and plates 27-29. As described in Mustela frenata macrura except that: Weight, 4.5 (4.2 and 4.8); basilar length, 44.6 (44.0-45.3); zygomatic breadth more than distance between condylar foramen and M1 or than between anterior palatine foramen and anterior margin of tympanic bulla; breadth of rostrum more or less than (approximately equal to) length of tympanic bulla; height of tympanic bulla less than distance from its anterior margin to foramen ovale; length of tympanic bulla less than length of rostrum; anterior margin of masseteric fossa posterior to m2 by length of that tooth.
Female (based on nos. 24134 to 24136): See measurements. As described in Mustela frenata macrura except that: Weight, 1.7 (1.5-1.9) grams; basilar length, 36.5 (35.3-38.1); zygomatic breadth less than distance between anterior palatine foramen and anterior margin of tympanic bulla; least width of palate more or less than (approximately equal to) outside length of P4; length of tympanic bulla less than length of rostrum.
The skull of the female averages 62 per cent lighter than that of the male.
The skull of the male is generally large and heavy as are the teeth. Comparison with macrura is made in the account of that subspecies. From males of affinis those of helleri differ in: skull shorter; breadth of rostrum and interorbital breadth actually and relatively greater.
Remarks.—The five specimens examined of this subspecies were taken by Edmund Heller for the Field Museum of Natural History in 1922 and 1923. It is to honor his contributions to mammalogy that the subspecies is named helleri. No. 24135 is the specimen carried as a pet for some time by Mr. and Mrs. Heller and of which Mrs. Heller (1924:481) has given an account.
This subspecies is insufficiently known, especially as to geographic range; probably it occupies a considerable range in the Tropical Life-zone along the eastern base of the Andes. The three females, two from Ambo and one from Huanuco, come from a much higher altitude than do the two males and the climate is said to be arid at Ambo and Huanuco. The skulls of the females are 62 per cent lighter and correspondingly smaller in measurements, than those of males. This difference is more than that found in any other South American weasel and it may be that the females are of a subspecies other than helleri.
The type specimen has a broad skull with major proportions strikingly like those of Mustela stolzmanni. Possibly the similar climatic conditions under which the two live have left their impress in similar fashion in this part of each of the two species. The teeth, tympanic bullae, and certain other parts of the skull are, however, so differently proportioned as to show that the skulls represent two species. The referred male has a much longer skull than the type specimen and the relative proportions of breadth and depth of the two skulls differ widely. Judging from large series of weasels examined from localities outside the range of M. f. helleri, the two skulls probably represent almost the maximum of individual variation occurring in one subspecies.
The dark color is as might be expected since helleri inhabits the humid Tropical Zone.
None of the five skulls shows signs of having had the frontal sinuses infested by parasites.
Specimens examined.—Total number, 5, all in the Field Museum of Natural History.
Perú: 3500 ft., Hacienda Buena Vista, Río Chinchao, 1; 3000 ft., Hacienda San Antonio, Río Chinchao, 1; Huanuco, 1; Ambo, 2.
Mustela frenata agilis Tschudi
Long-tailed Weasel
Mustela agilis Tschudi, Fauna Peruana, p. 110, 1844; Gray, Proc. Zoöl. Soc. London, 1865:113, 1865; Taczanowski, Proc. Zoöl. Soc. London, 1874:311, 1874; Taczanowski, Proc. Zoöl. Soc. London, 1881:648, 1881; Allen, Bull. Amer. Mus. Nat. Hist., 35:104; April 28, 1916; Thomas, Proc. U. S. Nat. Mus., 58-224, 1920.
Mustela macrura, Allen, Bull. Amer. Mus. Nat. Hist., 35:103, April 28, 1916.
Mustela frenata agilis, Hall, Carnegie Instit. Washington Publ. 473:110, November 20, 1936; Hall, Physis, 16:176, 1939.
Type.—No type specimen, or type locality more restricted than cold, barren highlands of the Cordillera [referring to Perú] designated.
Range.—High, barren Cordillera of Perú (see Tschudi, orig. descr.); as here restricted, Temperate Life-zone and higher in western Andes and intermountain valleys of Perú. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela frenata macrura by lighter color (Chocolate, tone 2 rather than 3, pl. 343, Oberthür and Dauthenay) of upper parts; length of upper tooth-rows, in females, less than 13; inside length of P4 more than 4.6; from M. f. aureoventris by smaller teeth (maximum size just given for agilis); from M. f. boliviensis by lighter color, upper parts being Chocolate, tone 2, pl. 343, rather than tone 4 or darker of Carbon Brown, pl. 342 (Oberthür and Dauthenay).
Description.—Size.—Male: The stuffed skin of an adult, from Lima, measures: Total length, 460; length of tail, 125; length of hind foot, 45.7. A skin alone from Huarochirí has a body, as now stuffed, 277 mm. long. The tail is missing and the bones of the hind feet have been removed.
Female: The mounted specimen, no. 565, Mus. Polonais d'Hist. Nat., yields measurements, taken by me, as follows: Total length, 250; length of tail, 75; length of hind foot, 32.5. The female, no. 21147, from Macate, measures, 300, 102, 34.
Externals.—Longest facial vibrissae, either dark-or light-colored and extending beyond ear; carpal vibrissae either dark-or light-colored and extending to apical pad of fifth digit; hairiness of foot soles as shown in figure 20.
Color.—Tschudi's description of the color is, in substance, as follows: Head, back and tail reddish gray; base of hair gray, followed by broader grayish-yellow ring and then reddish-brown tip; nose simply dark brown or upper lips edged with white; throat, breast, belly and higher parts of inner sides of extremities whitish gray, at times wholly gray, bases of hairs always gray; feet darker than body, almost chestnut brown; tail darker on tip than at base; ears externally dark brown, internally whitish.
No. 565 possibly somewhat faded from exposure to light, has all the upper parts near (14´ j) Ochraceous-Tawny or Cinnamon, and tone 4 of Oberthür and Dauthenay, plate 323; posterior half of each upper lip white; no other white facial markings present; dark spot at each angle of mouth (one spot confluent with color of upper parts); tip of tail probably black (tip missing); underparts white, belly probably originally with slight tinge of yellow or allied color; color of underparts extending distally on forelegs to feet and onto upper sides of toes and on hind legs to just above heels. Least width of color of underparts equal to about one-fourth of greatest width of color of upper parts.
No. 21147, subadult, from Macate, has a white band confluent with the underparts extending anterodorsally anterior to each ear and the posterior third of each upper lip white. Top of head near (n) Mars Brown, and Carbon Brown, tone 3 (pl. 342, Ober. and Dauth.); tip of tail black; remainder of upper parts near (16" j) Tawny-Olive, and Chocolate (tone 2, of pl. 343 of Ober. and Dauth.) or Raw Umber (tone 3 of pl. 301 of Ober. and Dauth.); anterior half of underparts, including posterior sides of forelegs and antipalmar faces of forefeet, white; remainder of underparts tinged with Warm Buff and extended on posterior legs almost to ankles.
No. 8.1.10.1., male adult, from Lima, is also light colored, and as described in no. 21147, except that left side of head has a white spot rather than bar; posterior eighth of each upper lip white; white frontonasal spot present, 11 x 11 mm.; antipalmar faces of forefeet spotted with brown color of upper parts; color of underparts extending distally on hind legs along medial side of foot to point halfway between heel and tip of inner toe.
No. 13257 from Huarochirí in color and color pattern closely resembles no. 21147. It differs from no. 21147 in slightly lighter color of upper parts, entirely white underparts, less extension of color of underparts onto forefeet, few white hairs instead of white band in front of each ear; color of underparts more restricted.
In each of the four specimens, the least width of the underparts, expressed as a percentage of the upper parts, is as follows: no. 13257, 11 per cent; no. 21147, 29 per cent; no. 565, 31 per cent; no. 8.1.10.1., nineteen per cent.
Skull and teeth.—Male (based on no. 8.1.10.1.): See measurements and plates 27-29. As described in Mustela frenata macrura except that: Weight 4.1 grams; basilar length, 42.5; zygomatic breadth more than distance between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth less than postpalatal length; tympanic bullae shorter than rostrum.
Female (based on no. 21147): See measurements and plates 39 and 40. As described in Mustela frenata macrura except that: Weight (no. 21147, subadult), 1.5 grams; basilar length, 35.2; least width of palate less than outside length of P4; tympanic bulla as far posterior to foramen ovale as combined width of five upper incisors; no. 565 answers to the same description but differs from no. 21147 in greater basilar length and larger tympanic bullae which are slightly more projected, at their anterior margins, from the braincase.
To judge from the skull of the female from Macate and the skull of the male from Lima, the skull and teeth of agilis are smaller than in any other South American subspecies of Mustela frenata, except M. f. boliviensis.
Remarks.—Tschudi almost certainly used the name Mustela agilis in a composite sense. His statement (see quoted matter below) about the marked variation in color of this species, as represented by the skins carried by the Indian women as purses, indicates that the forms here designated as Mustela macrura, M. helleri and possibly others additional to the one here called agilis were included by him under the name Mustela agilis. Taczanowski took account of Mustela agilis when he described other species from Perú. Allen (1916:104) and Thomas (1920:224) were not convinced that Mustela agilis and Mustela macrura were distinct species or subspecies.
Search on August 28, 1937, in the Musée d'Histoire Naturelle, at Neuchatel, Switzerland, by Mr. Théodore Delachaux, assistant there, and the writer, revealed no trace of weasels from Tschudi's collection, although some other specimens of mammals that he figured in the "Fauna Peruana" are preserved in that Museum. Not only were the collections of specimens examined but the new catalogue and old catalogue of mammals were vainly searched for mention of weasels deposited by Tschudi. Later, at the British Museum of Natural History, on p, 105 of a personal notebook, of the late Mr. Oldfield Thomas, record was found of his fruitless search for the same specimens of Mustela in May, 1902, at Neuchatel.
Although Tschudi certainly used the name Mustela agilis in a composite sense, as subspecies are at present understood, his description most nearly applies to the light-colored animals from western Perú—the lightest colored of any South American weasels seen. They are of approximately the same color as North American subspecies inhabiting semiarid regions, for example Mustela frenata longicauda of the Great Plains.
Another, but in my opinion less weighty, justification for applying Tschudi's name agilis to these light-colored weasels of western Perú is that by one line of reasoning, Taczanowski in naming macrura (jelskii is a synonym of it) from farther eastward in Perú, and that Hall in naming helleri from still farther eastward, and boliviensis to the southeastward, geographically restricted the application of the name agilis. Hall's action did this because he recognized geographic variation and employed the subspecies concept. Taczanowski, however, proposed his name macrura for a kind of animal which he indicated was specifically (as opposed to subspecifically) distinct from agilis and his account (1881:649) of jelskii indicates that he thought Mustela agilis Tschudi might occur in the same place as the animals which he named as new kinds. Thus, we can not credit Taczanowski with intent to restrict the name agilis geographically, even though later authors may choose to rule that his naming of macrura in effect did so restrict the application of Mustela agilis Tschudi.
The equivalents in millimeters given by Allen (1916:104) for Tschudi's measurements of 9 to 10 inches entire length, and tails of 4 inches to 4 inches and 4 lines, apparently are based on the London scale in use today. If Tschudi employed the Rhine scale also of eight lines to the inch, but one which has the foot longer by an amount of 20 millimeters, or the Leipzig scale in which the foot is 22 millimeters shorter than the London foot, the measurements recorded by Tschudi differ in one direction or the other from those computed by Allen. However, knowledge of which scale Tschudi employed would not help much, if any, in more precise application of the name agilis because he does not indicate whether his measurements are of male or female animals; animals of the two sexes of the same subspecies differ more in external measurements than animals of the same sex of different subspecies of Peruvian weasels.
Specimen no. 565, in the Polish Museum of Natural History, without definite locality, is provisionally referred to this subspecies. The specimen is intermediate in several respects between the female from Macate and the one of macrura from Cutervo.
Tschudi (1844:111-112) has given the following account: "Lebensweise und geographische Verbreitung. Das peruanische Wiesel lebt auf den kalten, öden Hochebenen der Cordillera an sonnigen Steinhaufen und Felsen gewöhnlich in Gesellschaft von 8-12 Stücken. Diese Thierchen sind so ausserordentlich behende und scheu, dass bei dem leisesten Geräusche die ganze Schaar mit Blitzesschnelle verschwindet. Es ist uns auch nie gelungen, eines derselben zu erlegen. Die Indianer aber verstehen es, dieselben lebendig einzufangen und zu zähmen. Ein sehr zahmes sahen wir bei einer uns befreundeten Dame in Tarma; gegen alle Fremden biss es mit Wuth und liess sich nicht anfassen, während es sich von seiner Herrin Alles gefallen liess; sie öffnete ihm den Mund und steckte ihm den Finger hinein, ohne dass es eine böse Miene dazu machte, während es bei der geringsten Bewegung, die wir machten, es zu ergreifen, grimmig auf uns lossprang. Wenn es eingeschüchtert wurde, versteckte es sich in den Busen seiner Gebieterin und kroch ihr bald nachher zum Aermel heraus. An den Wänden und Meublen kletterte es mit grosser Behendigkeit und schlüpfte durch so kleine Ritzen und Löcher, dass wir fast an der Möglichkeit dieses Hindurchdringens gezweifelt haben würden, wenn wir es nicht selbst mit angesehen hätten. Wenn es unartig war, wurde es mit einer Schnur an seinem kleinen Halsbande festgebunden; dadurch vermehrte sich sein Zorn, so dass es zuweilen gegen die Dame auffuhr. Mehrmals verschwand es während 8-10 Tagen und kam dann plötzlich wieder zum Vorschein. Seine Nahrung bestand in Gemüse und Fleisch, besonders aber liebte es Zuckerbrod in Milch aufgeweicht; einmal machte es sich an einen Kanarienvogel, den es auch tödtete. Es erhielt seine Strafe und verschwand dann für immer. Die Indianer sollen dieses Wiesel zum Fange der Viscacha abrichten (davon weiter unten). Sie nennen es Comadreja, auch Ardilla. ([footnote] Ardilla ist spanisch und heisst Eichhörnchen. Mit diesem Namen werden sehr verschiedene Thiere bezeichnet; ausser dem Sc. variabilis und der Galictis agilis auch noch mehrere Nager und einige Didelphysarten.) Die Indianerinnen verfertigen sich aus dem kleinen Felle Geldbeutel. Des Sonntags trifft man unter den vielen tausend Punaindianerinnen die nach den grossen Dörfern der Sierra kommen, um ihre Einkäufe zu machen, kaum ein halbes Dutzend, die nicht solche Börsen mit sich führten, und dann kann man auch die verschiedensten Farbennuancen, die bei dieser Species vorkommen, beobachten."
None of the three skulls referred to this subspecies shows infestation of the frontal sinuses by parasites.
Specimens examined.—Total number, 4.
Perú: Macate, 1 (Field Mus. Nat. Hist.); Huarochirí, 1 (Mus. Comp. Zool.); Lima, 1 (British Mus. Nat. Hist.); no locality more definite than Perú, 1 (Mus. Polonais d'Hist. Nat.).
Mustela frenata macrura Taczanowski
Long-tailed Weasel
Plates 1, 27, 28, 29, 30, 37, 38, 39 and 40
Mustela macrura Taczanowski, Proc. Zoöl. Soc. London, 1874:311, pl. 48, May 19, 1874; ibid., 1881:647, May 17, 1881; ibid., 835, November 15, 1881; Lönnberg, Arkiv för Zool., 8 (no. 1):21, 1913 (?); Hollister, Proc. Biol. Soc. Washington, 28:143, July 10, 1914; Allen, Bull. Amer. Mus. Nat. Hist., 35:101, April 28, 1916; Lönnberg, Arkiv för Zool., 14 (no. 4):11, 1921.
Putorius (Gale) braziliensis frenatus, Coues, Fur-bearing animals, p. 142, 1877.
Mustela jelskii Taczanowski, Proc. Zoöl. Soc. London, 1881:647, May 17, 1881.
Mustela affinis, Lönnberg, Arkiv för Zool., 8 (no. 1):21, July 12, 1913.
Mustela aureoventris, Thomas, Proc. U. S. Nat. Mus., 58:224, 1920.
Mustela frenata macrura, Hall, Carnegie Instit. Washington Publ. 473:110, November 20, 1936; Hall, Physis, 16:176, 1939.
Type.—Male, adult, mounted skin, with skull separate; no. 561, Mus. Polonais d'Hist. Nat. (Warsaw, Poland); Lake Junín, central Perú; 1873; obtained by M. Jelski.
The skull (plates 27-29, 30), mounted with the skin but removed by me for study, lacks the right jugal, the basisphenoid, the basioccipital and parts of each exoccipital bearing the exoccipital condyles. The right tympanic bulla, although detached from the skull, is preserved separately. The teeth all are present and entire. The skin is fairly-well mounted, in a good state of preservation, and shows no fading due to exposure to light.
Range.—Altitudinally, 3200 (Guainche) to at least 12000 feet (Pichincha); Upper Subtropical and Temperate life-zones of central Perú and Ecuador north from the states of Apurimac and Cuzco, Perú, to San Antonio, northern Ecuador. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela africana stolzmanni by absence of median, longitudinal, abdominal stripe of same color as upper parts; presence of p2 and two roots rather than one root on P2; from Mustela frenata helleri, M. f. affinis and M. f. aureoventris by lighter color of upper parts which are Chocolate tone 3, pl. 343, Oberthür and Dauthenay, whereas, with reference to the same color standard, the colors are: in helleri, Carbon Brown, tone 3, pl. 342; in affinis, Reddish Black, tone 2, pl. 344; in aureoventris, Reddish Black, tone 4, pl. 344; from M. f. agilis by darker color (Chocolate, tone 3 rather than 2, pl. 343, Oberthür and Dauthenay) of upper parts, length of upper tooth-rows, in females, more than 13, inside length of P4 more than 4.6; from M. f. boliviensis by lighter color of upper parts which are as above rather than tone 4 of Carbon Brown, pl. 342 of Oberthür and Dauthenay, and larger size (in males, hind foot more than 45 and m1 more than 5.6).
Description.—Size.—Male (measurements as recorded by Taczanowski in the original description, for two specimens, type and topotype, with correction of the length of tail of his "female" [= male]): Total length, 420, 415; length of tail, 150, 145; length of hind foot, 51, 51. An adult from Yana Mayo, Río Tarma, was measured by Hendee as 394, 134. Hind foot relaxed measures, 47. Tail 55 per cent as long as head and body. Length of hind foot more than basal length.
Female (based on measurements given by Taczanowski (1881:647) of no. 564): Total length, 323; length of tail, 120; length of hind foot, 37. Tail 59 per cent as long as head and body. Length of hind foot approximately equal to basal length.
Differences in external measurements of the two sexes are: Total length, 87; length of tail, 23; length of hind foot, 13.
Externals.—Longest facial vibrissae extending beyond ear; carpal vibrissae color of either upper parts or underparts; hairiness of foot-soles as shown in figure 20.
Color.—(Based on specimens from Cutervo and south thereof). Rarely few white hairs between eyes and in front of ears; top of head posteriorly to slightly behind eyes, near (n) Chestnut-Brown (Ridgway) and Carbon Brown, tone 2 or darker (pl. 342, Oberthür and Dauthenay); posterior half of upper lip rarely white; dark spots at angles of mouth absent; tip of tail black; remainder of upper parts near (l) Russet (Ridgway) and Chocolate, tone 3 (pl. 343, Ober. and Dauth.); underparts white or whitish on medial sides of forelegs, otherwise cream color with tinge of Ochraceous-Buff; color of underparts extended distally on posterior sides of forelegs to just below elbow (in type specimen) or onto forefeet (in specimen from Yana Mayo) and on medial sides of hind legs to points between knees and ankles. Least width of color of underparts averages (in six skins) 17 (14-21) per cent of greatest width of color of upper parts. Black tip of tail longer than hind foot and averaging 36 (32-49) per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on type specimen and no. 562): See measurements and plates 27-30; weight, not known; basilar length, 43.2 (40.8 and 45.5); zygomatic breadth more or less than distance between condylar foramen and M1 and more than that between anterior palatine foramen and anterior margin of tympanic bulla; mastoid breadth more or less than postpalatal length; postorbital breadth more than length of upper premolars and greater than width of basioccipital, measured from medial margin of one foramen lacerum posterior to its opposite; interorbital breadth more than distance between foramen opticum and anterior margin of tympanic bulla; breadth of rostrum less than length of tympanic bulla; least width of palate more than inside length of P4; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 4 (including I3) upper incisors; height of tympanic bulla not more than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum; anterior margin of masseteric fossa below or behind m2.
Female (based on no. 564, from Cutervo, Perú, type specimen of Mustela jelskii Taczanowski): See measurements and plates 37-40; weight, not known; basilar length, 38±; zygomatic breadth less than distance between condylar foramen and M1 and not greater than that between anterior palatine foramen and anterior margin of tympanic bulla; postorbital breadth more than alveolar length of upper premolars and (probably) more than width of basioccipital measured from medial margin of one foramen lacerum posterior to its opposite; least width of palate more than inside length of P4; tympanic bulla as far posterior to foramen ovale as width of at least 5-1/2 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; length of tympanic bulla more than length of lower molar and premolar tooth-row and longer or shorter than rostrum.
As compared with that of helleri, the skull of the male of macrura from Junín southward has a lesser mastoid breadth, notably smaller teeth, and a flatter skull which averages lighter throughout. The skulls of females available indicate that the skull and teeth are larger than in agilis.
Remarks.—Seven years after Taczanowski named this subspecies, he applied the name jelskii to a female taken farther north than the original examples of macrura. As indicated in synonymy, various other names have been applied to animals included by the present author in this subspecies.
Mustela frenata macrura intergrades with M. f. affinis as shown by practically all the referred specimens from north of Junín. As one proceeds northward the color of the weasels becomes progressively darker and the teeth become larger until the conditions found in affinis are met with near the northern border of Ecuador. From the material available it appears that the light-colored upper parts found in macrura characterize weasels of, at least, the Temperate Zone, from Marcapata, Perú, to near Quito, Ecuador. West of the range of macrura there exists the still lighter-colored subspecies, M. f. agilis. Immediately adjacent on the north, east, and south, darker-colored weasels occur. So far as color is concerned, the geographic range of the subspecies M. f. macrura is not difficult to define. However, the small size of the teeth characterizes only that part of this light-colored subspecies from Junín southward including the subspecies boliviensis at the southern extremity of the range of the species. From Cutervo northward the light-colored weasels of the Temperate Zone have teeth similar in size to those of the darker, more northern affinis. To designate the slightly larger-toothed, light-colored animals from Ecuador as a subspecies distinct from affinis and macrura is one solution but at present it seems best to refer all of these light-colored animals to macrura.
The type specimen and topotype no. 562 differ more in the amount of inflation of the tympanic bullae than adult males of comparable ages from a given locality usually do. In other respects, the differences between the two skulls are not greater than those ordinarily found in specimens from the same locality. No. 562 has the tympanic bullae greatly, relative to the other South American weasels, inflated posteriorly. Otherwise, the bullae agree with those of the type specimen.
Specimens from southwestern Ecuador, average large, and include the largest specimens of the species Mustela frenata seen from South America. A subadult male, no. 61406, in the American Museum of Natural History, is the largest. Its external measurements are 482, 191, 56. The basilar length of the skull is 48.2 and the zygomatic breadth is 30.3. Although not so large as this specimen, the corresponding measurements of specimens from Alamor, El Chiral, and even from as far away as Sigsig also are distinctly large.
The skull of the female from Ollantaytambo and that of the male from Marcapata have teeth equally as small as do the specimens from Lake Junín.
The skin alone, no. 194328, from Ollantaytambo has the color of the underparts extended over the entire upper sides of the forefeet. The male from Marcapata has less of this color on the forefeet and is in this respect intermediate between the specimens from Lake Junín and the one from Ollantaytambo.
In size of teeth the female, type specimen of M. jelskii, from Cutervo, shows an approach to the larger-toothed weasels of the northern part of the range of macrura.
The specimens in the Riksmuseum from the vicinity of Quito, Ecuador, have been rather fully described by Lönnberg (1921:11-17) and need little comment here, except to say that they show, as he suggested, that the weasel of the Temperate Zone of Ecuador is an intermediate link between M. f. macrura and M. f. affinis.
The adult female and juvenal male labeled as from Ambato have little left of the skulls except some of the teeth and the assignment of the specimens to the subspecies macrura is made mainly on geographic grounds. These two specimens probably are part of the shipment of birds and mammals of which Chapman (1926:703) speaks as follows: "A small collection of native-made skins purchased by the American Museum from a commission merchant in New York City as from 'Ambato' proved to be from the eastern slope of the Andes." Another skin in the same Museum, labeled by a native collector as from "Baeza arriba" [= above Baeza] is so dark colored and has the color of the underparts so much restricted, as to suggest that it belongs to the race aureoventris. Possibly, therefore, it was taken not at Baeza, Ecuador, which I find to the eastward of Quito at 77° 55' W and O° 25' S, but at some place of the same name on the Pacific Slope, unless the locality has been altogether wrongly recorded on the label. If the specimen was taken near the Baeza above referred to, then it gives evidence of an unnamed race of Mustela on the eastern slope of the Andes, characterized by its dark color. Unfortunately the specimen is young and its skull therefore offers insufficient basis for the judging of its subspecific relationships.
Other specimens, in the British Museum of Natural History, recorded as taken "near Quito" and here tentatively listed under macrura, mostly, include specimens so dark colored as to lead me to think they came from country, lower than Quito, adjacent to the range of aureoventris.
Nematodes taken from the right frontal sinus of no. 562 from Junín proved to belong to the superfamily Oxyuriodea according to Professor W. B. Herms and Mr. O. L. Williams, who have independently identified them. Because these worms had been dried fifty-five years in the mounted specimen and were later boiled in cleaning the skull, a more accurate determination was impossible and whether or not they pertain to the same species found in North American weasels cannot be said. Of 18 adult skulls examined for this type of infestation, 13 were found affected as judged by the evident malformation of the frontal region.
Specimens examined.—Total number, 74, arranged by localities from north to south and unless otherwise indicated in the American Museum of Natural History.
Ecuador: Ibarra, 6600 ft., and 7500 ft., 2[7]; San Antonio, 8000-8500 ft., 5 mi. N Quito, 4 (2[7], 2[78]); Nono, 10000 ft., 1; Mindo, 1[78]; Zambiza, 8000-8100 ft., NE Quito, 4 (2[78], 2[95]); Carapungo, 8500 ft., NE Quito, 1[78]; Panecillo, 10000 ft., near Quito, 2[78]; Guapulo, 8800 ft., 3 mi. E of Quito, 1[78]; Pichincha, 10500 ft., and 12000 ft., 2 (1[78], 1[95]); San Ignacio, 11500 ft., Pichincha, 1; Santa Rosa, 9600 ft., Río Pita, 2; near Santa Rosa, 9000 ft., 1; Río San Rafel, 9000 ft., 1; N side Quito, 9000 ft., 1[78]; Quito, 1[4]; near Quito, 5[7]; Nára Papallacta, 11000 ft., 1[78]; below Papallacta, 9000 ft., 1[78]; Chillo Valley, 1[78]; "Hacienda Hda," 10000 ft., Pintag, Valencia, 1; Baeza arriba, 1; Ambato, 2; San Francisco, 8000 ft., E of Ambato, 1; Chunchí, Pagma Forest, 6400 ft., 1[1]; Canar, 2600 M., 1[7]; Malletura, 7600 ft., 1; Contrayerbas, 11000 ft., 1; Sisig, 8500 ft., 3[7]; El Chiral, 1; Almor, 1; Guainche, 3200 ft., 1; no locality more definite than Ecuador, 4[95]; "Received from Quito," 1[7]; Quisaya, 6000 ft. (locality not found), 1[7]; La Carolina (locality not found), 1[78].
Perú: La Lejía, 1; Huancabamba, 4 (2[75]); Cutervo, 9000 ft., 1[73]; Condechacha, 7000 ft., Río Utcubamba, 1[7]; San Pedro, 8600-9400 ft., S of Chachapoyas, 1; Celendín, 1[7]; Junín, 2[73]; Yana Mayo, Río Tarma, 1[7]; Ollantaytambo, 9000 ft., 3 (1[7], 2[91]); Ocabamba, 1[7]; Anta Cuzco, 3400 and 3500 M., 2[4]; Marcapata, 1[91].
Mustela frenata boliviensis Hall
Long-tailed Weasel
Mustela frenata boliviensis Hall, Proc. Biol. Soc. Washington, 51:67, March 18, 1938.
Mustela frenata macrura, Hall, Carnegie Instit. Washington Publ. 473:110, November 20, 1936; Hall, Physis, 16:176, 1939 (part).
Type.—Male, adult, skull and skin; no. 72587, Amer. Mus. Nat. Hist.; Nequejahuira, 8000 feet, Bolivia; May 19, 1926; obtained by G. H. H. Tate; original no. 4135 (see plates 28-30).
Range.—As now known 8000 to 9500 feet in the Andes from Limbaní, Perú, south to Nequejahuira, Bolivia; upper Subtropical and Temperate life-zones. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela africana stolzmanni by absence of median, longitudinal, abdominal stripe of same color as upper parts; presence of p2 and two roots rather than one root on P2; from Mustela frenata macrura by darker color of upper parts (tone 4 or darker of Carbon Brown, pl. 342 rather than tone 3 of Chocolate, pl. 343, Oberthür and Dauthenay) and lesser size (in males hind foot less than 45 and m1 less than 5.6); from Mustela frenata agilis by darker color of upper parts (as given above rather than tone 2 of Chocolate, pl. 343, of Oberthür and Dauthenay).
Description.—Size.—Male: The type and two young specimens from Limbaní, Perú, measure respectively, as follows: Total length, 383, 368, 304; length of tail, 140, 132, 115; length of hind foot, 43, 44, 41. Tail 55 per cent as long as head and body. Length of hind foot approximately equal to basal length.
Female: Unknown.
Externals.—As described in Mustela frenata macrura.
Color.—Top of head blackish posteriorly to behind ears; upper lips same color as upper parts of head; dark area at angle of mouth not separated from upper parts as a distinct spot; tip of tail black; remainder of upper parts near (n) Mars Brown of Ridgway and tone 4 or darker of Carbon Brown (pl. 342, Oberthür and Dauthenay); underparts Cream-Colored with strong wash of Ochraceous-Buff; whitish on insides of forelegs to just below elbow; color of underparts extended distally on forelegs over ankles onto antipalmar faces of inner toes, and on hind legs to knees. Least width of color of underparts averages 15 (11-19) per cent of greatest width of color of upper parts. Black tip of tail in type longer than hind foot and amounting to 36 per cent of length of tail-vertebrae.
Skull and teeth.—Male (based on the type): See measurements and plates 28-30. As described in Mustela frenata macrura except that: Weight, 2.8 grams; basilar length, 41.6; zygomatic breadth less than distance between anterior palatine foramen and anterior margin of tympanic bulla; anterior margin of tympanic bulla as far posterior to foramen ovale as width of 5 upper incisors.
Female: Skull unknown.
Remarks.—Apparently the first specimens of this race to find their way into a zoölogical collection were the two young males taken on February 17, 1904, at Limbaní, by Geo. Ockenden [sic.].
M. f. boliviensis is smaller than any other South American weasel except possibly agilis. Better material of the two races probably will show even agilis to be larger.
Early in my study of Mustela after examination of the one young specimen, from Limbaní, in the United States National Museum, an account of this race was drawn up, but the account was discarded for want of satisfactory material and the animal was referred to macrura. Then, in 1937, when the two other specimens were studied, the race was formally characterized as different from previously recognized kinds.
The collector has noted on the labels of the two young from Limbaní that they were shot in the afternoon when running together beneath bushes. The frontal sinuses of the type are malformed as a result of infestation by parasites.
Specimens examined.—Total number, 3, as follows:
Perú: Carabaya, Limbaní, 2 (one in U. S. Nat. Mus. and one in Berlin Zool. Mus.).
Bolivia: Nequejahuira, 1 Amer. Mus. Nat. Hist.
Mustela frenata (?) gracilis (Brown)
Putorius gracilis Brown, Mem. Amer. Mus. Nat. Hist., 9(pt. 4):182, pl. 17, 1908.
Mustela gracilis, Hay, Iowa Geol. Surv. Bull., 23:32, 1914; Hay, Carnegie Instit. Washington Publ. 322A:252, October 15, 1924; Hay, Carnegie Instit. Washington Publ. 390(vol. 2):528, 1930.
Mustela frenata gracilis, Hall, Carnegie Instit. Washington Publ. 473:112, November 20, 1936.
Type.—Adult skull without lower jaws, probably of a female, no. 12431, Amer. Mus. Nat. Hist.; from Conard Fissure, four miles west of Willcockson, Newton County, Arkansas; obtained sometime in the period 1903 to 1905 inclusive. (See plates 39 and 40.)
Range.—Known only from the Pleistocene deposit in Conard Fissure, at the type locality in northern Arkansas.
Description.—Skull. Probably female (based on the type): See measurements and plates 39 and 40; weight unknown; basilar length, 38.1; least width of palate less than greatest length of P4; tympanic bulla as far posterior to foramen ovale as width of 3 to 5 upper incisors; height of tympanic bulla less than distance from its anterior margin to foramen ovale; length of tympanic bulla less than length of rostrum.
Comparison and remarks.—The type specimen was the only individual referred by Brown (1908) to this species. The remaining material of weasels from this deposit was referred by Brown to his Putorius cicognanii angustidens. Examination of the original materials convinces the writer, too, that the specimens, except no. 12431, are of the species erminea [= cicognanii of Brown]. No. 12431 itself may possibly be erminea but is far more probably of the species frenata. The uncertainty is due to the fact that an occasional skull alone of a subadult male erminea is extremely difficult certainly to distinguish from a skull alone of an adult female frenata. This is true among Recent specimens in the northern Mississippi Valley today; more exactly in Iowa and southern Minnesota the females of frenata, oftentimes intergrades between the subspecies Mustela frenata longicauda, M. f. noveboracensis and M. f. primulina, by only the skulls are next to indistinguishable from certain, unusually slender skulls of male erminea. At other places where the ranges of the two species meet, this difficulty is not so often encountered. Also, the type of gracilis has the skull broken in such a way that the postglenoid length in relation to the length of the skull as a whole could not be accurately determined in this particular skull.
The type specimen of gracilis surely is an adult and because of its small size is thought to be a female. Of known long-tailed weasels of the species frenata, gracilis is structurally nearest to M. f. primulina which occurs in the same region today and to M. f. noveboracensis, the long-tailed weasel of the eastern United States. M. gracilis differs from noveboracensis and agrees with primulina in possessing well-marked temporal ridges which fuse to form a low sagittal crest, in having the mastoid processes projecting farther, laterally, beyond the braincase, in having the anterior ends of the tympanic bullae produced below the squamosal rather than on the same plane with the squamosal, and in having the bullae more inflated anteromedially. M. gracilis differs from both noveboracensis (97 ♂ and 56 ♀ with skulls of comparable age) and primulina (64 ♂ and 24 ♀ with skulls of comparable age) in that the zygomatic breadth amounts to less than 58 per cent of the basilar length. Another difference from any one of the skulls of females of primulina is the longer rostrum, which, when measured from the posterior base of the postorbital process of the frontal to the anterior end of the nasal on the same side, amounts to more than 35 per cent of the basilar length. As pointed out by Brown (1908:182) this specimen represents the extreme of slender skull among known kinds of American weasels.
Selected measurements of no. 12431, the type specimen of Mustela gracilis, are as follows: Basilar length of Hensel, 38.1 mm.; length of upper tooth-rows, 14.3 to 14.4; breadth of rostrum, 11.0; interorbital breadth, 8.5; orbitonasal length, 13.6; mastoid breadth, 18.2; length of tympanic bulla, 13.0; breadth of tympanic bulla, 6.3; depth of tympanic bulla, 3.25; outside length of P4, 4.5; inside length of P4, 4.7; breadth of M1, 3.4; length of inner moiety of M1, 1.8; depth of skull at anterior margin of basioccipital, 12.2; depth of skull at posterior borders of last upper molars, 11.3; distance from foramen ovale to tympanic bulla, 3.6 mm.
MUSTELA AFRICANA Desmarest
Tropical Weasel
(Synonymy under subspecies)
Type.—Mustela africana Desmarest, Nouv. Dict. d'Hist. Nat., vol. 19, p. 376. 1818.
Range.—Known from the headwaters of the Amazon in eastern Perú and from near the mouth of the same river, on its southern side in Brazil, all within the Tropical Life-zone. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela frenata, the only geographically adjacent species of the genus, by: presence of thenar pad on forefoot; presence of a longitudinal, median, abdominal stripe of same color as upper parts; upper lips being broadly edged, entirely round, with color of underparts; failure of longest facial vibrissae to reach posterior margin of ear; absence of p2; relative flatness (see pl. 29, fig. i and pl. 39, fig. h) of tympanic bullae.
Characters of the species.—Size large (total length of adults approximately 500 mm.); foot-soles naked; thenar pad present on forefoot; length of claws, measured on concave sides, less than one and one-fourth times depth of claws measured at bases; longest facial vibrissae not reaching posterior margin of ear; tail relatively long haired; tail at all ages terminating in point as is characteristic of only juveniles and very young of Mustela frenata and M. erminea; tip of tail, and muzzle, only slightly darker than remainder of upper parts; upper lips broadly edged with color of underparts; pelage coarse, harsh and sparse; longitudinal, median, abdominal stripe of same color as upper parts present; skull broad and deep; braincase large, rounded, and much inflated anteriorly; palatal region wide; tympanic bullae less inflated than in any other American species of the subgenus; angle of lower jaw reduced; dental formula
3 1 2-3 1 -, -, ---, -; 3 1 2 2
teeth heavy; medial lobe of M1 but slightly larger than lateral lobe. See plates 28, 29, 30, 39 and cranial measurements.
Geographic variation.—The reddish versus chocolate color of the upper parts constitutes the only variation of a geographic nature so far detected.
Remarks.—One of the most noteworthy of the several unique characters of this large, tropical weasel is the longitudinal, median, abdominal band. The species exhibits the minimum degree of development of certain features that become progressively less apparent as one proceeds southward from Central America. The relative uniformity of the coloration of the upper parts (reduction in intensity of black color on the muzzle and tip of the tail) and the reduction of the tympanic bullae are two cases in point. Viewed dorsally the general outline of the skull is most nearly matched by that of the skull of Mustela frenata meridana from Venezuela or that of M. f. helleri from Perú. However, the resemblance is not close. The tympanic bullae, although unique among American weasels, are more like those of M. f. meridana from Venezuela than like those of any other kind. The great postorbital width (relatively less in M. africana than in several South American subspecies of Mustela frenata) and small angular process of the mandible are characters, in varying degrees, also common to all South American weasels. Structurally M. africana clearly is more nearly like other subspecies of M. frenata from South America than it is like any species or subspecies from North America.
Mustela africana is the most primitive of the American weasels. The distinctive cranial and dental characters, excepting the reduction in number of premolars, are of a primitive nature. For example, the relatively wide postorbital region, the large braincase that is inflated anteriorly, and the flattened, tympanic bullae, are points of resemblance to the holarctic Mustela erminea, which species is regarded as nearest the original stem form; also the mentioned characters correspond to ontogenetic stages passed through by other weasels. Mostly on these accounts, one is led to look upon M. africana as a migrant from North America. It may have become isolated from its original stock, by a water barrier in the Central American region, for a length of time sufficient to permit of a degree of differentiation to develop between it and the North American weasels which prevented crossbreeding with the frenata stock when that stock, at a later time, reached South America. This assumption is suggested only by evidence from the Recent specimens. No remains of true weasels (subgenus Mustela) have been recorded from deposits in South America older than the Recent period. The alternate possibility, that M. africana intergrades with some race of M. frenata in western or northern South America, has been considered and regarded as highly improbable.
Cabrera (1940:15) has made the distinctive structural characters of Mustela africana basis of the generic name Grammogale to include the one species africana. I am inclined to accord Grammogale only subgeneric rank.
It is possibly significant that Mustela africana is intermediate in several respects between Lyncodon and typical Mustela. The median, longitudinal, abdominal band of the same color as the upper parts in M. africana and the relative uniformity of the coloration of its upper parts might be considered as an intermediate stage between the dark, bicolored (black muzzle and tail tip and brown body) upper parts and light-colored underparts of the North American weasels on the one hand and the light, unicolored upper parts and dark-colored underparts of the Patagonian weasel (Lyncodon) on the other hand. The number of premolars,
2-3 ---, 2
is also intermediate between the numbers
3 2 - and - 3 2
of the North American Mustela and the Patagonian Lyncodon, respectively. The American Mustela and the Patagonian Lyncodon, respectively. The more medially, as opposed to anteriorly, directed medial cusp of P4 (characteristic of approximately half of the specimens examined), and the structure of the skull in general of M. africana also seem to be morphologically intermediate between those parts of Mustela and Lyncodon.
On the chance that Lyncodon is closely enough related to Mustela, to be included in a group with Mustela rather than in a group with Grisonella, it is worth noting that Lyncodon lujanensis Ameghino (1889:324, 325), from the villa of Lujan and at the city of Córdoba, at each place in the Pampean [= Pleistocene] formation of Argentine (see also Cabrera, 1928:263) is the first and only fossil form of this group recorded from the whole of South America. Actually, however, Lyncodon seems to me to be as nearly related to Grisonella, if not more so, than to Mustela. If Lyncodon is more closely related to Grisonella and Grison than to Mustela, then the above remarked intermediacy in characters of M. africana has more of interest as a tendency to parallelism than it has of phylogenetic import. Appraisal of phylogenetic relationships would require appraisal of the ancestral stem forms of the Grison stock and the Mustela stock. None of either is known from deposits of the Pliocene, the period of time immediately preceding the Pleistocene.
None of the skulls of Mustela africana seen or figured has the nasomaxillary sutures entirely obliterated and the specimens would, judged on this character alone and by analogy with North American species, be regarded as young and subadult. However, the sutures close at what seems to be a later age than in M. frenata and M. erminea. The condition of the mammae in the type specimen of M. stolzmanni and in the specimen from Moyobamba, indicate that they have borne young. North American weasels old enough to bear young lack visible traces of the nasomaxillary sutures. I have examined no skulls of africana with greatly worn teeth and hence cannot say if the sutures are obliterated in advanced age.
If available data be correct, this species is unique within the genus in that the two sexes are of approximately the same actual size and of the same relative proportions in the body and in the skull. There was no difference between individuals said to be of different sexes from Pará, described and figured by Goeldi (1904:61-62, pls. 1, 2). The undoubted female, type specimen of Mustela africana stolzmanni, is as large as the undoubted male, no. 37475, of the same species, but of a different subspecies, from Pará, Brazil. All the specimens of M. a. africana that I have handled are labeled male and those of M. a. stolzmanni female. More material may show that the female is smaller than the male, as is the case in all near relatives of M. africana.
Little has been recorded concerning the habits of this species. Tate (1931:254) states that a live individual which he saw in a cage at Pará had been captured "swimming in the salt water of the estuary about half a mile away from the shore." On the label of the specimen from Moyobamba, there appears: "caught in Willow tree."
Subspecies examined.—All described forms, of which there are two.
Mustela africana africana Desmarest
Tropical Weasel
Mustela africana Desmarest, Nouv. Diction. d'Hist. Nat., 19:376, 1818; Cabrera, Bol. Real Soc. Españ. de Hist. Nat., 13:429, November, 1913; Cabrera, Bol. Real Soc. Españ. de Hist. Nat., 14:175, pl. 1, March, 1914.
Putorius (Mustela) brasiliensis paraensis, Goeldi, Zool. Jahrb. abt. f. systematik, geogr. u., Biol., 10:556, pl. 21, September 15, 1897, type from Pará, Brazil, near Pará, Ward of Marco da Legoa, Brazil; Goeldi, Bol. do Mus. Paraense, 3:195 [translation of orig. descr.], August, 1901.
Putorius paraensis, Goeldi, Bol. do Museu Goeldi, 4:61, pls. 1, 2, 1904.
Mustela affinis paraensis, Hollister, Proc. Biol. Soc. Washington, 28:143, July 10, 1914.
Mustela paraensis, Allen, Bull. Amer. Mus. Nat. Hist., 35:105, April 28, 1916; Tate, Journ. Mamm., 12:253, August 24, 1931.
Mustela stolzmanni paraensis, Hall, Carnegie Instit. Washington Publ. 473:111, November 20, 1936; Hall, Physis, 16:167, pl. 1, figs. 1-4, 1939.
Type.—Male, adult or subadult, mounted; no. 848, Paris Museum; from the "Cabinet de Lisbonne 1808," originally from South America as determined from the characters of the animal; probably came from Brazil, and for the present assumed to be from Pará.
On August 25, 1937, the skull was in the mounted skin and the specimen was in the position shown in the figure published by Cabrera (1914, pl. 1). Except for the loss of the distal part of the tail, and fading because of exposure to light, the specimen was in good condition. See also under remarks.
Range.—Known from the south side of the Amazon River, near its mouth at Pará and Cametá, Río Tocantins, in the Tropical Life-zone of Brazil. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela frenata, the only other geographically adjacent species of the genus, in presence of median, longitudinal, abdominal stripe of same color as upper parts and naked foot-soles, in absence of p2 and in reduced size of tympanic bullae (see pls. 29 and 30) and from Mustela africana stolzmanni by lighter color of upper parts which although near Chestnut-Brown are in adults 10' l (darker in yg. M. C. Z., no. 30802), instead of 11' n as in M. a. stolzmanni.
Description.—Size.—This is a relatively large weasel. Goeldi (1897:559) gives the total length of the type specimen of his P. b. paraensis, a female, as 520 mm. (495 in the flesh) and, by computation from his figures, the length of the tail as 200 (205 in the flesh). These measurements probably include the hairs on the tip of the tail as probably also do the measurements given of two other specimens (see Goeldi, 1904:62). One of these specimens, a female, measured: Total length, 520; length of tail, 200. The other specimen, a male, measured: Total length, 510; length of tail, 200. The skin of no. 37475, Amer. Mus. Nat. Hist., a male, has the following measurements written on the attached label: Total length, 548; length of tail, 234; length of hind foot, 56. The hairs project 20 mm. beyond the tip of the last vertebra of the tail and probably are included in the measurements of total length and length of tail. Collectors' measurements of a young male from Cametá, and a subadult labeled as male, from Pará Murutucu, are respectively as follows: 500, 430; 210, 190; 50 and 54.
Externals.—Foot-soles naked, except for a few scattered hairs on ventral sides of interdigital membranes; length of claws, measured on concave sides, not more than one and one-fifth times depth of claws measured at bases; carpal vibrissae not extending beyond apical pad of first digit (not beyond hypothenar pad except in one young specimen); longest facial vibrissae not extending to posterior margin of ear; superior genal tuft not found, hairiness of foot-soles as shown in figure 22.
Color.—Upper parts near (10 l) Chestnut-Brown and relatively uniform since tip of tail and muzzle are only slightly darker than remainder; underparts with longitudinal stripe of same color as upper parts extending along median line of belly from throat or breast posteriorly to within 40 to 50 millimeters of anus. Underparts otherwise near (20" a) Olive-Ocher (lips and chin whiter in one young specimen). Color of underparts extends distally on median sides of forelegs to bare foot-soles and on median side of hind legs two-thirds of distance from knee to ankle. Upper lips broadly edged with whitish, which color passes posteriorly below and not touching eye to ventral margin of concha of ear. An inverted, basally broad, V-shaped extension passes upward 4 millimeters, just posterior to the eye.
Skull and teeth.—See measurements (plates 28-30). Male: (based on 3 adult and subadult topotypes and figures and descriptions published by Goeldi, 1897 and 1904.) Weight, 7.0 grams; basilar length, 45.8 (44.6-47.8); skull broad and deep; braincase large, rounded, and much inflated anteriorly; distance from postorbital process to anterior, nasal notch approximately equal to breadth across exoccipital condyles; palatal region wide; tympanic bullae less inflated than in any other species; mastoid bone, laterally, concave; length of upper tooth-rows in adults and subadults less than breadth of palate measured between two outer margins of fourth upper premolars; alveolar distance between C1 and P4 less than length of P4; teeth heavy; medial lobe of M1 only slightly larger than lateral lobe; deuterocone of P4 heavy and often inclined mesially; p2 absent (P2 present above on both sides in only one of seven specimens seen or described); lower jaw heavy; masseteric fossa not extending anteriorly to posterior fourth of talonid of m1; paraconid of m1 low and base of cleft between it and protoconid relatively low on tooth.
Female: No skull examined but from figures published by Goeldi (1904, pl. 2), apparently as described in the male.
Remarks.—Desmarest in 1818 gave a remarkably good description of this animal which he named as a new species, Mustela africana, but mistakenly indicated that the single specimen known to him came originally from Africa. Until 1913 the name was applied, wrongly, to weasels of northern Africa or to those of the Azores Islands and St. Thomas Island. In that year Cabrera (1913:429) identified the species with the one later named Putorius (Mustela) brasiliensis paraensis by Goeldi (1897:556, pl. 21) from Pará, Brazil. Despite Cabrera's clear identification in 1913, and his later mention of the correct application of the name Mustela africana, it was not correctly employed by other authors, including myself who even as late as 1936 (p. 111) instead used Goeldi's name. In 1937 Mr. Cabrera called my attention to his published account of Mustela africana and so permitted me to examine the type specimen in the Paris Museum, whither I was bound when I received Mr. Cabrera's letter. My own examination of the specimen fully confirmed the conclusions published by Cabrera (1913:429).
As a matter of historical interest, however, it is worth noting that Cabrera (1913) originally supposed the type specimen to have been taken as booty of war from Portugal by the French and that Cabrera later, at the request of P. Trouessart, pointed out (1914:176) that the specimen had been acquired in exchange ("a cambio") since according to Dr. Trouessart the Museum register showed that offer had been made to Portugal to return this and other specimens but that Portugal had replied that it had nothing to reclaim. Dr. P. Rode in August, 1937, at the Paris Museum, gave it to me as his opinion that the specimen had been an outright gift from the "Cabinet de Lisbonne" to E. Geoffroy St.-Hilaire on his trip to Portugal in 1808 when he was given also from the same cabinet several primates, all from Brazil. Of the labels attached to the pedestal on which the specimen is mounted, that of most ancient appearance is glued to the bottom of the stand and bears in a hand apparently written before Trouessart's entries on the same label, the information "Du Cabinet de Lisbonne 1808" and "J. H. S. 1809."
The opened mouth of the mounted specimen permits one to determine that P2 is absent on each side above. The stuffed scrotal pouch and hair projecting downward about the preputial opening clearly show the animal to have been a male. The least faded portions of the mounted specimen, its sides, are of the same reddish color as characterizes adults from Pará and not of the darker chocolate color of specimens of M. stolzmanni from Perú. The specimen is indistinguishable from topotypes of P. paraensis of Goeldi and his name will have to fall as a synonym of Mustela africana Desmarest.
Goeldi gave an extended description, with figures of the skull, head, and entire animal, when he named paraensis. As his account shows, he was unaware that Taczanowski had described a similar weasel from the headwaters of the Amazon, or for that matter that any weasel excepting Mustela affinis Gray, had been found in South America. Goeldi's later account of additional specimens (1904:61, pls. 1, 2) gives much useful information about the animal. Photographs of several specimens and photographs and detailed measurements of several skulls are presented by him.
Pará, and Cametá, Brazil, places from which Mustela africana africana is known, are nearly 2000 miles from the localities in eastern Perú and eastern Ecuador from which M. a. stolzmanni is known, and no specimens, from intermediate localities, are available to show actual intergradation of the two. However, the similarity in structure of the two weasels is so great as to indicate close affinity. Furthermore, it is understood that environmental conditions at and between the two localities are similar. These considerations, in the light of our knowledge of actual intergradation of geographic races of weasels in other places, cause me to treat, with a feeling of assurance, M. africana [= P. paraensis Goeldi] and M. stolzmanni Taczanowski as subspecies of a single species. M. Rodolpho Legueira Rodríguez writes me, under date of June 16, 1928, that the type specimen of Putorius (Mustela) brasiliensis paraensis Goeldi is stuffed and preserved in a "vitrine" at the Museum Goeldi (Museum Paraense) De Historia Natural e Ethnographia, Pará, Brazil.
The one young specimen seen, that from Cametá, is darker colored than any of the four older specimens examined. It is almost exactly the Chestnut Brown of Ridgway (1912) and therefore approaches closely in color the adult specimens of M. a. stolzmanni. This same tendency to greater richness of color in young than in adults is seen also in Mustela frenata.
Specimens examined.—Total number, 5, all from eastern Brazil, as follows: Pará, 2 (1[2], 1[7]); Pará Murutucu, 1[7]; Río Tocantins, Cametá, 1[75]; type specimen, 1[84].
Mustela africana stolzmanni Taczanowski
Tropical Weasel
Mustela stolzmanni Taczanowski, Proc. Zoöl. Soc. London, 1881:835, November 15, 1881; Allen, Bull. Amer. Mus. Nat. Hist., 35:105, April 28, 1916.
Mustela stolzmanni stolzmanni, Hall, Carnegie Instit. Washington Publ. 473:111, November 20, 1936; Hall, Physis, 16:167, pl. 1, figs. 5, 6, 1939.
Type.—Female, adult, mounted skin, with skull separate; no. 563, Mus. Polonais d'Hist. Nat. (Warsaw, Poland); Yurimaguas, Perú; 1880; obtained by J. Stolzmann.
The skull (plate 40), mounted with the skin but removed by me for study, consists of the premaxillae, maxillae, two halves of the lower jaw and dentition. Of these parts, right m2, left coronoid process, right P4 and M1 and adjacent part of maxilla are lost. The skin is well mounted, in a good state of preservation and shows no fading due to exposure to light. Inguinal mammae are distinctly shown on the skin and prove that the specimen is a female. Except for a few scattered hairs on the lower throat, a spot six by eight millimeters on the medial side of the region of the olecranon of the left foreleg and another of similar size in the left axilla, the underparts are, excepting the ventral longitudinal, abdominal stripe, unmarked by color of the upper parts.
Range.—Known from the Tropical Life-zone of eastern Ecuador and Perú from Jatun Yacu south to Valle del Perené. See figure 29 on page 221.
Characters for ready recognition.—Differs from Mustela frenata, the only other geographically adjacent species of the genus, in presence of median, longitudinal, abdominal stripe of same color as upper parts and naked foot soles, in absence of p2 and in reduced size of tympanic bullae (see pls. 28, 29, 30, 39 and 40) and from Mustela africana africana by darker color of upper parts which, although near Chestnut Brown, are 11' n instead of 10' l as in M. a. africana.
Description.—Size.—Male: unknown.
Female: Taczanowski (1881:836) gives, among others, the following measurements of the type specimen: Total length, 523, length of body, 260; length of tail without hair, 190 (with hair 224); length of hind foot, 54. Whether or not the measurements were taken from the animal when in the flesh I do not know. Allowing for shrinkage of hind feet and changes due to the posture of the now mounted specimen, I get from it essentially the same measurements. Collectors' measurements of a subadult from Moyobamba and a young female from Valle del Perené, are respectively, as follows: 469, 415; 184, 160; 57, 52. My own measurements of the dry hind feet on the skins are respectively, 48 and 49.
Externals.—As described in M. a. africana except that the length of the concave sides of the claws are approximately one and one-fourth times the depth; thus the claws are relatively longer than in M. a. africana.
Color.—As described in M. a. africana with the following noted exceptions: Upper parts near (11' n) Chestnut-Brown; area of lighter ventral coloration on the throat and sides of head less strongly tinged with yellow; pelage more dense, finer and softer than in M. a. africana.
Skull and teeth.—Male: Skull unknown.
Female: See measurements and plate 39 and 40. As described in male of Mustela africana africana except that: Weight, 4.7 grams. As contrasted with M. a. africana, the dentition of the lower jaw is lighter; the transverse diameter of m2 is 1.2 mm. in the type and also in the specimen from Moyobamba as against 1.5 to 1.7 in three male topotypes of M. a. africana.
Remarks.—After the Polish naturalist, Stolzmann, in the course of his explorations in Perú, obtained the single specimen which was made the type, no other naturalist, so far as known, visited the type locality until thirty-two years later when Wilfred H. Osgood and M. P. Anderson spent more than a month collecting at Yurimaguas (see Osgood, 1914:147), but secured no topotypes of this little-known weasel. C. O. Schunke took the second specimen in the Valle del Perené in April, 1921; L. Rutter on January 25, 1924, took the third specimen, and W. Clark-MacIntyre took the fourth specimen on the Jatun Yacu. This obscure place name is shown on the map (fig. 4, p. 827) published by Brown (1941) and is the stream flowing from the west to the town of Napo. Napo is situated at approximately 1° 2' S and 77° 49' W.
In the female from Moyobamba there are only 3 pairs of mammae. One pair is inguinal and two pairs are on the posterior part of the abdomen.
Taczanowski (1881:836) relates that this species was taken in the forest to which it appears to be restricted since the inhabitants of the village did not know of the animal. He points out also that the previously known Peruvian species [M. f. macrura and M. f. agilis] live in the treeless territory of eight to eleven thousand feet altitude whereas M. stolzmanni was found in the humid forest of the great plain of the Maynas at an elevation of 500 feet or less above sea level. The frontal sinuses of the specimens seen reveal no malformation as a result of infestation by parasites.
Specimens examined.—Total number, 4, as follows:
Ecuador: R. Tatun [= Jatun] = Yacu, 1, Mus. Comp. Zoöl.
Perú: Yurimaguas, 1 in Mus. Polonais d'Hist. Naturelle, Warsaw; Moyobamba, 2700 ft. [6° S, 77° W], 1 in Brit. Mus. Nat. Hist.; Valle del Perené, 1200 meters, 1 in Amer. Mus. Nat. Hist.
EXPLANATION OF CRANIAL MEASUREMENTS APPEARS ON PAGE 417
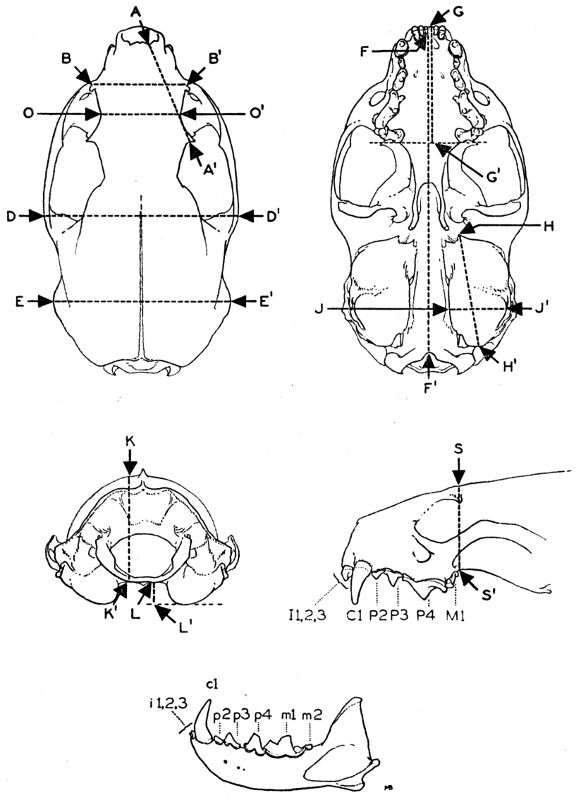
Fig. 31. Four views of the skull and a lateral view of the left lower jaw to show points between which measurements of the skull were taken. Based on M. f. primulina, from 3 mi. E Bergman, Boone County, Arkansas, obtained December 12, 1933, by B. G. Roberts; ad. ♀. 62854 Mus. Vert. Zoöl. × 1-2/5.
EXPLANATION OF CRANIAL MEASUREMENTS
Basilar length (of Hensel).—From the anteriormost border of the foramen magnum to a line connecting the posterior margins of the alveoli of the first upper incisors. F to F' on fig. 31.
Condylobasal length.—Least distance from a line connecting the posteriormost parts of the exoccipital condyles to a line connecting the anteriormost projections of the premaxillary bones.
Length of tooth-rows.—Least distance between a line connecting posterior borders of upper molars and a line connecting anterior faces of middle upper incisors. G to G' on fig. 31.
Breadth of rostrum.—Least distance from lateral base of hamular process of lacrimal bone to corresponding point on opposite side of skull. B to B' on fig. 31.
Interorbital breadth.—Least distance across top of skull between orbits (eye sockets). O to O' on fig. 31.
Orbitonasal length.—Distance on anterior part of skull from posterior margin of base of postorbital process of frontal bone to posteriormost part of anterior border of nasal bone on same side of skull. A to A' on fig. 31.
Mastoid breadth.—Greatest distance across mastoid bones perpendicular to long axis of skull. E to E' on fig. 31.
Zygomatic breadth.—Greatest distance across zygomatic arches of cranium perpendicular to long axis of skull. D to D' on fig. 31.
Tympanic bulla:
Length.—From posterior face to most anterior part of anterior border. H to H' on fig. 31.
Breadth.—From bottom of pit immediately posterior to external auditory meatus to medial face of bulla at right angle with longitudinal axis of skull. J to J' on fig. 31.
Depth.—Least distance from ventral face of basioccipital, excluding median ridge, to line touching ventralmost points of the two bullae. L to L' on fig. 31.
m1, Length.—Greatest length which rarely or never is alveolar length.
P4.—
Lateral.—Length from posterior margin of tooth to anteriormost part of the protocone (anterolateral cusp).
Medial.—Length from the posterior margin of tooth to anteriormost part of the deuterocone (anterointernal cusp).
M1.—
Breadth.—Distance from medial edge of crown to lateral margin of crown, approximately at a right angle with longitudinal axis of the skull.
Length.—Greatest diameter, anteroposteriorly, of the inner lobe or inner half of the tooth.
Depth of skull at anterior margin of basioccipital.—Measured from anterior end of ventral face of basioccipital, excluding median ridge, vertically to dorsal face of parietal excluding sagittal crest. K to K' on fig. 31.
Depth of skull at posterior borders of Ms1.—Measured from ventral face of palatine bones at posterior edge of upper molars to dorsal face of frontals in plane of postorbital processes of frontals. S to S' on fig. 31.
TABLE OF CRANIAL MEASUREMENTS
| Collection | Catalog Number | Sex and age | Locality | Basilar length (of Hensel) | Length of tooth rows | Breadth of rostrum | Interorbital breadth | Orbitonasal length | Mastoid breadth | Zygomatic breadth | Length | Breadth | Depth | Length m1 | Lateral | Medial | Breadth | Length | Depth of Skull at Ant. margin of basioccipital | Depth of Skull at posterior borders of Msl |
| Mustela frenata noveboracensis | ||||||||||||||||||||
| average} | ♂ ad. 10 | Adirondacks, New York | {45.1 | 16.6 | 13.7 | 10.9 | 15.3 | 23.3 | 26.7 | 15.4 | 7.8 | 3.5 | 5.9 | 5.3 | 5.5 | 4.3 | 2.4 | 14.1 | 12.7 | |
| maximum} | {47.0 | 17.4 | 14.6 | 11.9 | 15.9 | 24.7 | 28.0 | 16.2 | 8.4 | 4.0 | 6.2 | 5.7 | 5.9 | 4.5 | 2.7 | 14.5 | 13.3 | |||
| minimum} | {43.2 | 15.9 | 13.0 | 10.4 | 14.8 | 22.0 | 25.3 | 14.6 | 7.0 | 3.1 | 5.5 | 4.9 | 4.9 | 4.0 | 1.9 | 13.5 | 12.2 | |||
| average} | ♂ ad. 10 | Massachusetts | {44.7 | 16.3 | 13.7 | 11.3 | 15.7 | 23.2 | 26.5 | 15.4 | 7.7 | 3.4 | 5.9 | 5.2 | 5.5 | 4.4 | 2.4 | 14.1 | 12.7 | |
| maximum} | {47.0 | 16.8 | 14.9 | 12.0 | 16.7 | 25.6 | 28.2 | 16.4 | 8.2 | 3.7 | 6.3 | 5.5 | 5.9 | 4.7 | 3.0 | 14.7 | 13.0 | |||
| minimum} | {43.3 | 15.9 | 13.1 | 10.5 | 14.8 | 21.8 | 25.1 | 14.3 | 7.3 | 3.1 | 5.6 | 5.0 | 5.2 | 4.0 | 2.0 | 13.5 | 12.0 | |||
| average} | ♂ ad. 10 | Liberty Hill, Connecticut | {44.0 | 16.3 | 13.2 | 10.9 | 15.3 | 22.4 | 26.3 | 15.0 | 7.5 | 3.3 | 5.8 | 5.1 | 5.4 | 4.2 | 2.2 | 14.3 | 12.6 | |
| maximum} | {46.0 | 17.4 | 14.4 | 12.0 | 16.4 | 23.5 | 28.0 | 15.9 | 8.0 | 3.6 | 6.3 | 5.6 | 6.1 | 5.0 | 2.5 | 15.2 | 13.2 | |||
| minimum} | {41.5 | 15.2 | 12.1 | 9.7 | 14.2 | 20.8 | 24.0 | 14.4 | 6.9 | 3.0 | 5.3 | 4.6 | 5.0 | 3.7 | 2.0 | 13.4 | 11.7 | |||
| average} | ♂ ad. 10 | Beaver Dam, Wisconsin | {44.4 | 16.5 | 13.3 | 10.6 | 15.6 | 23.0 | 26.3 | 15.2 | 8.0 | 5.8 | 5.3 | 5.5 | 4.3 | 2.4 | 13.9 | 12.6 | ||
| maximum} | {46.1 | 17.7 | 14.1 | 11.3 | 16.4 | 23.8 | 27.4 | 16.2 | 8.2 | 6.1 | 5.7 | 5.8 | 4.6 | 2.7 | 14.5 | 13.2 | ||||
| minimum} | {40.6 | 15.6 | 12.3 | 9.8 | 13.7 | 22.1 | 23.9 | 14.2 | 7.7 | 5.6 | 4.7 | 5.1 | 3.9 | 2.1 | 13.0 | 11.8 | ||||
| average} | ♂ ad. 10 | Washtenaw Co., Michigan | {43.0 | 15.6 | 13.0 | 10.2 | 14.9 | 22.0 | 24.9 | 14.7 | 7.6 | 5.6 | 5.0 | 5.3 | 4.2 | 2.2 | 13.4 | 11.9 | ||
| maximum} | {45.4 | 16.5 | 13.4 | 11.0 | 15.7 | 22.8 | 26.1 | 15.2 | 7.9 | 6.0 | 5.3 | 5.6 | 4.5 | 2.5 | 14.2 | 12.3 | ||||
| minimum} | {39.7 | 14.7 | 11.6 | 9.6 | 13.8 | 21.1 | 23.5 | 14.1 | 7.3 | 5.0 | 4.7 | 4.9 | 3.8 | 1.9 | 12.6 | 11.5 | ||||
| average} | ♀ ad. 8 | Adirondacks, New York | {37.5 | 13.4 | 10.7 | 8.7 | 12.9 | 18.7 | 20.9 | 13.5 | 6.5 | 2.8 | 4.8 | 4.3 | 4.6 | 3.5 | 1.7 | 11.4 | 10.2 | |
| maximum} | {39.8 | 14.5 | 11.2 | 9.3 | 13.4 | 19.3 | 22.1 | 14.3 | 7.0 | 2.9 | 5.2 | 4.7 | 5.0 | 3.7 | 1.9 | 12.1 | 10.8 | |||
| minimum} | {36.4 | 12.8 | 9.8 | 7.9 | 12.1 | 17.5 | 20.0 | 12.6 | 5.9 | 2.5 | 4.5 | 4.0 | 4.2 | 3.2 | 1.7 | 10.3 | 9.4 | |||
| average} | ♀ ad. 5 | Massachusetts | {36.5 | 13.4 | 10.5 | 8.5 | 12.9 | 18.5 | 20.1 | 13.3 | 6.3 | 2.3 | 5.0 | 4.3 | 4.5 | 3.5 | 1.8 | 11.4 | 10.0 | |
| maximum} | {38.1 | 14.0 | 11.0 | 8.6 | 13.3 | 19.6 | 20.8 | 13.9 | 6.9 | 2.7 | 5.1 | 4.5 | 4.7 | 3.7 | 2.0 | 11.6 | 10.4 | |||
| minimum} | {35.2 | 13.0 | 10.0 | 8.3 | 12.1 | 17.2 | 19.3 | 12.8 | 5.6 | 2.0 | 4.9 | 4.2 | 4.4 | 3.3 | 1.4 | 11.0 | 9.6 | |||
| average} | ♀ ad. 6 | Maryland | {37.3 | 13.5 | 10.8 | 8.6 | 13.0 | 18.8 | 21.0 | 13.1 | 6.6 | 4.9 | 4.4 | 4.7 | 3.6 | 1.9 | 11.9 | 10.9 | ||
| maximum} | {37.9 | 14.1 | 11.2 | 8.9 | 13.5 | 19.6 | 21.7 | 13.5 | 6.8 | 5.2 | 4.6 | 4.9 | 4.1 | 2.3 | 12.6 | 11.4 | ||||
| minimum} | {36.8 | 13.0 | 10.4 | 8.5 | 12.8 | 18.2 | 20.1 | 13.0 | 6.4 | 4.7 | 4.2 | 4.5 | 3.1 | 1.4 | 11.6 | 10.3 | ||||
| average} | ♀ ad. 5 | Beaver Dam, Wisconsin | {37.2 | 13.4 | 10.6 | 8.8 | 13.0 | 18.7 | 21.0 | 13.5 | 6.5 | 4.9 | 4.5 | 4.6 | 3.5 | 1.8 | 11.6 | 10.2 | ||
| maximum} | {37.6 | 13.7 | 11.3 | 9.7 | 13.5 | 19.5 | 21.7 | 13.8 | 6.8 | 5.0 | 4.5 | 4.8 | 3.7 | 2.1 | 12.3 | 10.3 | ||||
| minimum} | {37.0 | 12.9 | 10.0 | 8.0 | 12.0 | 17.6 | 20.2 | 13.1 | 6.1 | 4.9 | 4.4 | 4.3 | 3.3 | 1.5 | 10.8 | 9.8 | ||||
| average} | ♀ ad. 9 | Washtenaw Co., Michigan | {36.5 | 13.1 | 10.5 | 8.4 | 12.6 | 18.5 | 20.5 | 13.2 | 6.3 | 4.8 | 4.3 | 4.5 | 3.5 | 1.8 | 11.8 | 10.3 | ||
| maximum} | {37.5 | 13.7 | 11.0 | 8.8 | 13.1 | 19.1 | 21.3 | 13.8 | 6.7 | 5.0 | 4.5 | 4.7 | 3.7 | 2.0 | 12.2 | 10.7 | ||||
| minimum} | {35.9 | 12.8 | 9.8 | 7.8 | 12.0 | 18.1 | 19.5 | 12.9 | 6.0 | 4.6 | 4.1 | 4.3 | 3.3 | 1.5 | 11.3 | 10.0 | ||||
| Mustela frenata occisor | ||||||||||||||||||||
| A. N. S. P. | 4137 | ♂ ad. | Washington Co., Maine | 45.8 | 16.5 | 13.3 | 11.0 | 16.2 | 24.5 | 27.5 | 15.7 | 8.5 | 3.5 | 6.2 | 5.6 | 5.9 | 4.6 | 2.8 | 13.3 | 13.0 |
| M. C. Z. | 7267 | ♂ ad. | Moose Head Lake, Maine | 44.9 | 16.6 | 14.5 | 11.8 | 15.6 | 24.2 | 27.7 | 15.2 | 7.7 | 3.1 | 6.0 | 5.2 | 5.6 | 4.2 | 2.4 | 14.2 | 13.0 |
| M. C. Z. | 5501 | ♂ ad. | Bucksport, Maine | 46.9 | 17.1 | 14.4 | 11.0 | 15.8 | 24.0 | 28.0 | 16.4 | 8.0 | 4.0 | 6.0 | 5.8 | 6.1 | 4.5 | 2.6 | 14.7 | 13.2 |
| M. C. Z. | 9142 | ♂ ad. | Bucksport, Maine | 45.0 | 16.1 | 14.0 | 11.2 | 15.3 | 24.2 | 27.4 | 15.8 | 7.3 | 2.8 | 6.1 | 5.3 | 5.6 | 4.3 | 2.4 | 14.5 | 12.6 |
| av | 45.7 | 16.6 | 14.1 | 11.3 | 15.7 | 24.2 | 27.7 | 15.8 | 7.9 | 3.4 | 6.1 | 5.5 | 5.8 | 4.4 | 2.6 | 14.2 | 13.0 | |||
| M. C. Z. | 9101 | ♀ sad. | Bucksport, Maine | 37.7 | 14.0 | 11.6 | 9.1 | 13.0 | 19.0 | 21.7 | 14.1 | 6.3 | 2.6 | 5.2 | 4.6 | 4.6 | 3.8 | 2.1 | 12.5 | 10.5 |
| M. C. Z. | 9122 | ♀ sad. | Bucksport, Maine | 38.2 | 13.8 | 11.5 | 9.2 | 13.6 | 19.5 | 21.8 | 13.6 | 6.5 | 2.5 | 4.6 | 4.3 | 4.5 | 3.5 | 1.7 | 12.3 | 10.3 |
| Mustela frenata primulina | ||||||||||||||||||||
| average} | ♂ ad. 10 | Douglas Co., Kansas | {44.8 | 16.8 | 13.5 | 10.6 | 14.9 | 24.0 | 27.2 | 15.5 | 8.3 | 3.4 | 6.1 | 5.5 | 5.8 | 4.6 | 2.4 | 14.2 | 12.8 | |
| maximum} | {46.0 | 17.8 | 14.1 | 11.5 | 15.4 | 24.9 | 28.2 | 16.3 | 8.8 | 3.9 | 6.6 | 5.9 | 6.1 | 4.7 | 2.6 | 14.8 | 13.2 | |||
| minimum} | {43.8 | 16.2 | 12.9 | 10.0 | 14.3 | 23.2 | 26.2 | 14.4 | 8.0 | 3.0 | 5.8 | 5.2 | 5.5 | 4.3 | 2.2 | 13.6 | 12.0 | |||
| average} | ♂ ad. an d sad. 8 | Boone Co., Arkansas | {44.7 | 16.5 | 13.2 | 10.5 | 15.0 | 24.1 | 26.9 | 15.5 | 8.1 | 3.6 | 6.0 | 5.5 | 5.6 | 4.3 | 2.2 | 14.2 | 12.3 | |
| maximum} | {46.5 | 17.1 | 14.0 | 11.8 | 16.3 | 24.8 | 27.7 | 16.4 | 8.8 | 4.2 | 6.5 | 6.0 | 6.2 | 4.9 | 2.5 | 14.6 | 13.0 | |||
| minimum} | {42.5 | 15.6 | 12.6 | 9.7 | 14.1 | 22.8 | 26.1 | 14.9 | 7.5 | 3.1 | 5.8 | 5.2 | 5.2 | 4.0 | 2.0 | 13.9 | 11.7 | |||
| average} | ♀ ad. an d sad. 11 | Douglas Co., Kansas | {38.9 | 14.4 | 11.3 | 8.6 | 12.9 | 20.3 | 22.6 | 13.5 | 6.9 | 2.9 | 5.2 | 4.8 | 5.0 | 3.5 | 1.8 | 12.6 | 11.2 | |
| maximum} | {40.7 | 15.3 | 12.0 | 9.2 | 13.4 | 21.4 | 23.8 | 15.1 | 7.5 | 3.3 | 5.7 | 5.2 | 5.4 | 4.2 | 2.1 | 13.5 | 11.7 | |||
| minimum} | {37.6 | 13.8 | 10.8 | 7.9 | 12.4 | 18.8 | 21.1 | 13.0 | 6.3 | 2.5 | 4.8 | 4.5 | 4.6 | 3.4 | 1.5 | 11.7 | 10.0 | |||
| average} | ♀ ad. an d sad. 6 | Arkansas | {39.3 | 14.1 | 11.4 | 8.7 | 13.1 | 20.3 | 23.1 | 14.0 | 6.9 | 2.9 | 5.2 | 4.8 | 5.0 | 3.8 | 1.9 | 12.5 | 10.7 | |
| maximum} | {40.1 | 14.6 | 11.9 | 8.8 | 13.7 | 21.0 | 23.8 | 14.5 | 7.2 | 3.0 | 5.5 | 5.0 | 5.3 | 4.2 | 2.0 | 13.0 | 11.8 | |||
| minimum} | {38.8 | 13.7 | 11.0 | 8.5 | 12.6 | 19.8 | 22.5 | 13.5 | 6.5 | 2.8 | 5.0 | 4.5 | 4.7 | 3.5 | 1.7 | 12.3 | 10.3 | |||
| Mustela frenata arthuri | ||||||||||||||||||||
| U. S. N. M. | 246345 | ♂ ad. | Convent, Louisiana | 43.4 | 15.4 | 13.7 | 11.5 | 14.5 | 22.3 | 26.6 | 15.4 | 7.7 | 3.3 | 5.4 | 4.9 | 5.1 | 3.7 | 2.0 | 14.8 | 12.4 |
| M. V. Z. | 37515 | ♂ sad. | Remy, Louisiana | 43.8 | 16.4 | 12.7 | 10.9 | 15.0 | 22.3 | 25.5 | 15.5 | 7.5 | 3.7 | 4.9 | 5.7 | 5.8 | 4.2 | 2.3 | 14.2 | 12.4 |
| M. V. Z. | 38901 | ♂ sad. | Springville, Louisiana | *45.0 | *16.5 | 15.5 | 16.0 | 8.3 | 5.8 | 5.5 | 5.7 | 4.6 | 2.5 | |||||||
| Mustela frenata olivacea | ||||||||||||||||||||
| M. V. Z. | 47165 | ♂ ad. | Talbot Co., Georgia | 50.1 | 18.3 | 15.0 | 12.5 | 16.5 | 26.7 | 30.8 | 17.2 | 9.2 | 4.0 | 6.8 | 6.2 | 6.2 | 4.7 | 2.5 | 15.0 | 13.8 |
| M. V. Z. | 47144 | ♂ ad. | Talbot Co., Georgia | 49.2 | 18.3 | 13.8 | 11.7 | 16.9 | 26.0 | 29.0 | 17.9 | 9.4 | 4.2 | 6.8 | 6.0 | 6.3 | 4.8 | 2.8 | 15.5 | 13.4 |
| M. V. Z. | 47166 | ♂ ad. | Talbot Co., Georgia | 48.5 | 17.6 | 14.6 | 11.5 | 16.5 | 27.0 | 30.9 | 17.5 | 9.8 | 4.3 | 6.8 | 5.7 | 6.0 | 4.5 | 2.5 | 15.6 | 13.2 |
| M. V. Z. | 47167 | ♂ ad. | Talbot Co., Georgia | 45.8 | 17.3 | 14.2 | 11.5 | 16.2 | 25.7 | 29.4 | 15.9 | 8.7 | 4.1 | 6.1 | 5.6 | 6.0 | 4.9 | 2.3 | 15.4 | 13.7 |
| M. V. Z. | 47147 | ♂ ad. | Talbot Co., Georgia | 48.1 | 18.0 | 14.0 | 10.9 | 16.9 | 26.2 | 28.9 | 17.2 | 8.8 | 4.2 | 6.1 | 5.7 | 6.2 | 4.6 | 2.3 | 14.5 | 13.2 |
| av. 5 | 48.3 | 17.9 | 14.3 | 11.6 | 16.6 | 26.3 | 29.8 | 17.1 | 9.2 | 4.2 | 6.5 | 5.8 | 6.1 | 4.7 | 2.5 | 15.2 | 13.5 | |||
| M. V. Z. | 41023 | ♂ ad. | Thomas Co., Georgia | 48.8 | 17.5 | 14.9 | 11.9 | 16.0 | 27.3 | 31.2 | 17.9 | 8.7 | 4.2 | 6.4 | 5.9 | 6.3 | 4.9 | 2.5 | 17.0 | 13.4 |
| M. V. Z. | 41025 | ♂ ad. | Grady Co., Georgia | 44.8 | 16.9 | 13.2 | 10.9 | 15.5 | 24.7 | 28.3 | 16.8 | 8.8 | 3.9 | 6.2 | 5.7 | 5.8 | 4.3 | 2.4 | 15.0 | 13.3 |
| M. V. Z. | 40934 | ♂ ad. | Grady Co., Georgia | 47.8 | 17.4 | 14.1 | 12.1 | 15.5 | 25.1 | 29.1 | 17.9 | 8.8 | 4.5 | 6.6 | 6.0 | 6.0 | 4.7 | 2.6 | 15.0 | 14.2 |
| M. V. Z. | 40935 | ♂ ad. | Grady Co., Georgia | 47.4 | 18.0 | 13.6 | 11.7 | 16.2 | 24.1 | 29.1 | 15.8 | 8.5 | 4.5 | 6.4 | 5.7 | 6.0 | 4.6 | 2.2 | 15.0 | 13.5 |
| av. 4 | 47.2 | 17.5 | 14.0 | 11.7 | 15.8 | 25.3 | 29.4 | 17.1 | 8.7 | 4.3 | 6.4 | 5.8 | 6.0 | 4.6 | 2.4 | 15.5 | 13.6 | |||
| U. S. N. M. | 223880 | ♂ ad. | Okefinokee Swamp | 49.0 | 18.8 | 14.2 | 11.6 | 14.6 | 26.1 | 30.4 | 16.5 | 8.6 | 4.4 | 6.1 | 5.7 | 6.0 | 4.6 | 2.4 | 15.6 | 13.2 |
| Cornell | 198 | ♂ ad. | Okefinokee Swamp | 47.3 | 18.0 | 14.0 | 11.4 | 16.3 | 26.8 | 29.8 | 17.2 | 8.8 | 4.3 | 6.5 | 5.8 | 6.3 | 4.6 | 2.5 | 14.8 | 13.2 |
| Cornell | 652 | ♂ ad. | Okefinokee Swamp | 47.0 | 17.0 | 13.8 | 11.8 | 16.0 | 25.8 | 30.5 | 17.3 | 8.6 | 4.5 | 6.2 | 5.8 | 6.0 | 4.5 | 2.5 | 15.5 | 13.6 |
| U. S. N. M. | 180802 | ♂ ad. | Autaugaville, Alabama | 46.5 | 17.3 | 13.4 | 10.7 | 16.3 | 25.5 | 29.2 | 15.6 | 8.3 | 3.7 | 6.8 | 5.8 | 6.1 | 4.9 | 2.6 | 14.5 | 13.2 |
| M. V. Z. | 51527 | ♀ ad. | Talbot Co., Georgia | 43.5 | 16.4 | 12.5 | 9.8 | 14.5 | 22.4 | 25.7 | 15.5 | 7.8 | 3.8 | 5.8 | 5.6 | 5.8 | 4.2 | 2.2 | 13.5 | 12.3 |
| M. V. Z. | 41024 | ♀ ad. | Thomas Co., Georgia | 42.7 | 16.0 | 12.0 | 9.6 | 14.7 | 22.8 | 26.0 | 15.8 | 8.0 | 3.9 | 5.8 | 5.0 | 5.4 | 4.1 | 2.1 | 14.4 | 12.2 |
| M. V. Z. | 41022 | ♀ ad. | Thomas Co., Georgia | 44.0 | 16.1 | 12.8 | 10.6 | 15.3 | 23.4 | 25.8 | 15.3 | 8.2 | 3.6 | 6.1 | 5.7 | 5.8 | 4.4 | 2.1 | 13.2 | 12.0 |
| Mustela frenata peninsulae | ||||||||||||||||||||
| F. S. M. | 49387 | ♂ ad. | Apopka, Florida | 49.8 | 18.6 | 15.1 | 12.2 | 15.8 | 27.8 | 31.4 | 17.6 | 10.2 | 4.8 | 6.3 | 5.9 | 6.2 | 4.7 | 2.4 | 15.3 | 14.6 |
| A. N. S. P. | 9379 | ♀ ad. | Tarpon Springs, Florida | 44.2 | 16.6 | 13.8 | 11.0 | 15.8 | 23.7 | 27.1 | 16.4 | 8.4 | 4.3 | 6.3 | 5.7 | 5.7 | 4.1 | 1.9 | 13.3 | 12.9 |
| A. N. S. P. | 8515 | ♀ yg. | Pasco Co., Florida | 15.9 | 11.8 | 9.6 | 14.2 | 6.4 | 5.7 | 5.9 | 4.6 | 2.0 | 12.3 | |||||||
| Mustela frenata spadix | ||||||||||||||||||||
| M. V. Z. | 53795 | ♂ ad. | Elk River, Minnesota | 49.2 | 19.1 | 15.9 | 11.9 | 17.1 | 28.0 | 31.7 | 15.9 | 9.5 | 3.6 | 6.9 | 6.5 | 6.7 | 5.4 | 2.7 | 15.5 | 14.0 |
| Walker | A23 | ♂ ad. | Elk River, Minnesota | 49.0 | 18.7 | 15.9 | 13.5 | 16.5 | 28.1 | 32.1 | 16.4 | 9.7 | 4.0 | 6.8 | 6.2 | 6.7 | 4.9 | 2.0 | 15.9 | 15.0 |
| Walker | A37 | ♂ sad. | Elk River, Minnesota | 48.7 | 18.9 | 15.2 | 11.9 | 16.5 | 27.0 | 29.9 | 15.5 | 9.2 | 3.8 | 6.6 | 6.1 | 6.5 | 5.0 | 3.0 | 15.0 | 13.8 |
| Dickey | A865 | ♂ sad. | Elk River, Minnesota | 46.8 | 17.6 | 15.4 | 16.7 | 26.1 | 15.2 | 8.4 | 3.3 | 6.2 | 5.5 | 5.9 | 4.3 | 2.3 | ||||
| Dickey | A846 | ♂ sad. | Elk River, Minnesota | 46.1 | 18.0 | 14.2 | 10.6 | 15.5 | 24.9 | 28.2 | 14.8 | 8.5 | 3.7 | 6.6 | 5.9 | 6.6 | 4.8 | 2.9 | 15.1 | 12.8 |
| Dickey | 11548 | ♀ ad. | Elk River, Minnesota | 42.3 | 16.4 | 13.1 | 10.7 | 15.0 | 22.9 | 27.0 | 13.8 | 7.7 | 3.1 | 6.6 | 5.6 | 5.6 | 4.4 | 2.1 | 13.0 | 12.3 |
| Walker | A174 | ♀ ad. | Elk River, Minnesota | 43.2 | 16.2 | 13.8 | 10.8 | 15.3 | 24.5 | 26.8 | 14.5 | 8.6 | 3.7 | 5.8 | 5.3 | 5.5 | 4.5 | 2.1 | 13.9 | 13.2 |
| Dickey | 9688 | ♀ ad. | Elk River, Minnesota | 43.2 | 16.8 | 12.7 | 10.5 | 15.0 | 23.3 | 25.6 | 15.2 | 7.6 | 3.2 | 5.9 | 5.4 | 5.7 | 4.1 | 2.0 | 14.0 | 12.2 |
| U. S. N. M. | 188410 | ♀ ad. | Elk River, Minnesota | 43.0 | 16.2 | 13.2 | 10.7 | 14.9 | 23.3 | 26.0 | 14.7 | 8.1 | 3.3 | 5.7 | 5.3 | 5.6 | 4.3 | 2.4 | 13.8 | 12.8 |
| av. 4 | 42.9 | 16.4 | 13.2 | 10.7 | 15.1 | 23.4 | 26.4 | 14.6 | 8.0 | 3.3 | 6.0 | 5.4 | 5.6 | 4.3 | 2.2 | 13.7 | 12.6 | |||
| Mustela frenata longicauda | ||||||||||||||||||||
| A. M. N. H. | 15875 | ♂ ad. | Red Deer, Alberta | 46.5 | 18.6 | 15.1 | 12.1 | 15.3 | 25.3 | 30.8 | 15.2 | 8.9 | 3.4 | 6.9 | 6.3 | 6.5 | 4.9 | 2.6 | 15.0 | 14.6 |
| N. M. C. | 8060 | ♂ ad. | Sweet Grass Hills, Alberta | 45.5 | 18.1 | 15.5 | 12.2 | 15.5 | 31.0 | 6.9 | 6.3 | 6.6 | 5.0 | 2.5 | 13.8 | |||||
| F. M. N. H. | 7021 | ♂ ad. | Canadian Nat. Park, Alberta | 46.8 | 17.8 | 13.8 | 11.4 | 16.0 | 25.7 | 30.4 | 15.7 | 8.7 | 3.9 | 6.3 | 5.7 | 5.9 | 4.4 | 1.9 | 15.5 | 13.4 |
| F. M. N. H. | 8567 | ♂ ad. | Calgary, Alberta | 44.7 | 17.2 | 13.9 | 11.3 | 14.8 | 24.8 | 29.7 | 15.5 | 8.7 | 3.6 | 6.0 | 5.4 | 5.6 | 4.3 | 2.4 | 15.0 | 13.4 |
| U. S. N. M. | 75725 | ♂ ad. | St. Albert, Alberta | 46.5 | 18.3 | 15.0 | 12.4 | 15.6 | 25.0 | 29.4 | 15.2 | 8.3 | 3.4 | 6.6 | 6.2 | 6.4 | 4.6 | 2.0 | 16.0 | 13.6 |
| av. 5 | 46.0 | 17.9 | 14.7 | 11.9 | 15.4 | 25.2 | 30.3 | 15.4 | 8.6 | 3.6 | 6.5 | 6.0 | 6.2 | 4.6 | 2.3 | 15.4 | 13.8 | |||
| N. M. C. | 6968 | ♀ ad. | Daysland, Alberta | 43.7 | 16.8 | 13.1 | 14.9 | 24.0 | 26.1 | 14.7 | 8.3 | 3.1 | 5.7 | 5.2 | 5.4 | 4.3 | 2.0 | 13.3 | 12.2 | |
| U. S. N. M. | 68731 | ♀ ad. | S. Edmonton, Alberta | 42.5 | 16.0 | 13.4 | 10.3 | 14.3 | 24.1 | 26.7 | 14.5 | 8.8 | 3.3 | 5.6 | 5.4 | 5.5 | 4.3 | 2.3 | 14.0 | 12.5 |
| A. M. N. H. | 16044 | ♀ ad. | Blindman River, Alberta | 40.0 | 15.1 | 12.2 | 8.8 | 13.2 | 22.5 | 24.5 | 13.4 | 8.2 | 2.8 | 5.6 | 5.0 | 5.0 | 4.0 | 2.0 | 12.8 | 11.5 |
| M. V. Z. | 53792 | ♀ ad. | Grafton, North Dakota | 42.8 | 16.5 | 13.6 | 10.5 | 14.9 | 23.2 | 26.1 | 15.3 | 8.0 | 2.8 | 6.1 | 5.4 | 5.6 | 4.3 | 2.4 | 14.0 | 12.3 |
| U. S. N. M. | 75483 | ♀ ad. | Wingard, Sask. | 42.3 | 16.9 | 12.9 | 9.8 | 15.5 | 23.3 | 25.9 | 14.8 | 8.3 | 3.2 | 6.4 | 5.9 | 6.1 | 4.8 | 2.4 | 14.3 | 12.1 |
| av. 5 | 42.3 | 16.3 | 13.0 | 9.9 | 14.6 | 23.4 | 25.9 | 14.6 | 8.3 | 3.0 | 5.9 | 5.4 | 5.5 | 4.3 | 2.2 | 13.7 | 12.1 | |||
| Mustela frenata oribasus | ||||||||||||||||||||
| M. V. Z. | 44568 | ♂ sad. | Barkerville reg., B. C. | 48.8 | 19.1 | 17.2 | 13.0 | 17.7 | 28.5 | 32.2 | 17.4 | 9.8 | 3.2 | 6.8 | 6.2 | 6.4 | 5.3 | 2.8 | 15.5 | 14.6 |
| M. V. Z. | 43817 | ♂ sad. | Isaacs Lake, British Columbia | 48.8 | 19.5 | 15.8 | 12.0 | 17.3 | 28.4 | 31.7 | 15.9 | 9.7 | 4.0 | 6.9 | 6.3 | 6.4 | 5.3 | 2.9 | 15.5 | 13.7 |
| Cowan | 443 | ♂ ad. | Quesnel, British Columbia | 46.6 | 17.7 | 14.5 | 10.1 | 15.9 | 26.4 | 30.7 | 15.8 | 9.2 | 3.9 | 6.2 | 5.8 | 6.1 | 4.6 | 2.5 | 15.0 | 13.1 |
| N. M. C. | 2676 | ♂ ad. | Lillooet, British Columbia | 47.5 | 18.5 | 14.3 | 10.3 | 15.9 | 25.5 | 30.1 | 15.2 | 9.1 | 3.8 | 6.4 | 5.9 | 6.3 | 4.6 | 2.5 | 15.0 | 13.1 |
| N. M. C. | 2695 | ♂ ad. | Lillooet, British Columbia | 45.0 | 17.5 | 15.9 | 11.8 | 15.9 | 25.6 | 31.0 | 15.2 | 8.9 | 3.0 | 6.2 | 5.6 | 6.1 | 4.7 | 2.9 | 15.0 | 13.4 |
| M. C. Z. | 9058 | ♀ ad. | Source Kettle River, B. C. | 41.7 | 16.4 | 12.8 | 10.5 | 14.8 | 24.9 | 26.7 | 14.3 | 8.2 | 2.8 | 6.1 | 5.5 | 5.7 | 4.5 | 2.5 | 14.0 | 12.6 |
| M. V. Z. | 62791 | ♀ ad. | Beaverfoot Range, B. C. | 42.0 | 16.4 | 13.5 | 10.2 | 15.1 | 24.4 | 27.0 | 14.3 | 8.1 | 3.1 | 5.9 | 5.5 | 5.6 | 4.3 | 2.2 | 14.0 | 12.3 |
| Mustela frenata alleni | ||||||||||||||||||||
| U. S. N. M. | 186451 | ♂ ad. | Custer, South Dakota | 40.9 | 15.0 | 13.8 | 11.0 | 14.1 | 22.0 | 27.7 | 13.7 | 7.9 | 3.0 | 5.7 | 4.9 | 5.3 | 3.9 | 2.5 | 14.3 | 12.3 |
| A. M. N. H. | 7440/9136 | ♂ ad. | Hill City, South Dakota | 41.0 | 15.6 | 13.7 | 11.0 | 13.9 | 23.3 | 25.7 | 13.6 | 8.3 | 2.3 | 5.1 | 4.8 | 5.2 | 4.2 | 1.9 | 13.7 | 12.0 |
| A. M. N. H. | 7441 | ♀ ad. | Black Hills, South Dakota | 37.6 | 14.1 | 12.2 | 9.1 | 13.2 | 22.3 | 23.1 | 13.8 | 7.3 | 3.1 | 5.5 | 4.9 | 5.0 | 3.6 | 1.8 | 12.2 | 10.5 |
| Mustela frenata arizonensis | ||||||||||||||||||||
| M. V. Z. | 55211 | ♂ ad. | near Parks, Arizona | 40.4 | 15.5 | 12.5 | 14.2 | 23.3 | 26.0 | 14.3 | 8.9 | 3.1 | 5.7 | 5.2 | 5.5 | 4.0 | 2.2 | 13.5 | 12.1 | |
| M. V. Z. | 65231 | ♂ ad. | Alpine, Arizona | 39.6 | 15.1 | 12.2 | 9.7 | 13.7 | 22.4 | 25.6 | 13.7 | 8.3 | 3.2 | 5.4 | 4.9 | 5.1 | 4.0 | 2.1 | 14.0 | 11.4 |
| U. S. N. M. | 248993 | ♂ ad. | Kaibab Plat., Arizona | 40.4 | 15.6 | 12.9 | 9.8 | 14.5 | 22.9 | 26.3 | 14.1 | 8.4 | 3.2 | 5.8 | 5.5 | 5.5 | 4.0 | 1.7 | 14.8 | 12.0 |
| A. M. N. H. | 2490/1886 | ♀ ad. | S. F. Forest, Arizona | 35.5 | 13.8 | 10.8 | 8.6 | 12.9 | 19.9 | 21.7 | 13.3 | 7.7 | 2.9 | 4.8 | 4.6 | 4.7 | 3.6 | 1.6 | 12.3 | 10.3 |
| Mustela frenata nevadensis | ||||||||||||||||||||
| average} | ♂ ad. 25 | Sierra Nevada, California | {43.6 | 16.6 | 13.7 | 10.7 | 15.1 | 23.9 | 28.0 | 15.0 | 8.4 | 3.4 | 5.9 | 5.4 | 5.6 | 4.3 | 2.2 | 14.4 | 12.5 | |
| maximum} | {46.1 | 17.6 | 14.9 | 12.0 | 16.2 | 26.1 | 31.4 | 15.9 | 9.0 | 4.0 | 6.4 | 5.8 | 6.1 | 4.8 | 2.7 | 15.2 | 14.8 | |||
| minimum} | {40.6 | 15.2 | 12.5 | 9.9 | 14.0 | 22.1 | 25.0 | 14.4 | 7.8 | 2.9 | 5.5 | 4.9 | 5.1 | 3.9 | 1.8 | 13.7 | 11.6 | |||
| average} | ♂ ad. 10 | S and SW Colorado | {43.7 | 16.5 | 13.9 | 10.6 | 15.0 | 24.2 | 27.9 | 15.1 | 8.6 | 3.5 | 6.1 | 5.5 | 5.7 | 4.5 | 2.1 | 14.5 | 12.6 | |
| maximum} | {44.6 | 17.3 | 14.8 | 11.1 | 16.4 | 25.3 | 29.3 | 15.8 | 9.2 | 4.0 | 6.8 | 5.9 | 6.0 | 4.8 | 2.6 | 15.6 | 13.0 | |||
| minimum} | {41.6 | 16.0 | 12.8 | 9.9 | 13.8 | 23.1 | 26.5 | 14.4 | 8.2 | 3.0 | 5.7 | 5.2 | 5.3 | 4.0 | 1.9 | 13.9 | 12.1 | |||
| average} | ♀ ad. 10 | California | {38.2 | 14.7 | 11.8 | 9.2 | 13.4 | 20.9 | 23.1 | 13.4 | 7.3 | 2.9 | 5.2 | 4.8 | 5.0 | 3.8 | 1.7 | 12.3 | 11.0 | |
| maximum} | {39.5 | 15.1 | 12.4 | 9.9 | 14.2 | 21.8 | 23.4 | 14.1 | 7.9 | 3.2 | 5.6 | 5.2 | 5.4 | 4.1 | 2.0 | 13.0 | 11.8 | |||
| minimum} | {36.7 | 13.9 | 11.0 | 8.6 | 12.6 | 20.1 | 22.4 | 12.7 | 6.8 | 2.6 | 4.9 | 4.5 | 4.7 | 3.6 | 1.5 | 11.4 | 10.0 | |||
| average} | ♀ ad. 14 | Colorado | {38.5 | 14.8 | 12.2 | 9.3 | 13.4 | 20.6 | 23.1 | 13.4 | 7.6 | 2.8 | 5.4 | 4.9 | 5.1 | 3.9 | 1.9 | 12.9 | 11.1 | |
| maximum} | {39.7 | 15.4 | 13.1 | 10.2 | 14.4 | 22.1 | 24.6 | 13.9 | 8.1 | 3.2 | 5.6 | 5.3 | 5.4 | 4.1 | 2.3 | 13.8 | 11.8 | |||
| minimum} | {36.1 | 14.0 | 11.1 | 8.5 | 12.5 | 19.8 | 22.0 | 12.9 | 7.0 | 2.5 | 5.1 | 4.3 | 4.6 | 3.4 | 1.7 | 12.0 | 10.6 | |||
| Mustela frenata effera | ||||||||||||||||||||
| average} | ♂ ad. 6 | NE Oregon | {40.5 | 15.2 | 12.3 | 9.6 | 13.7 | 22.1 | 25.6 | 14.1 | 7.8 | 3.3 | 5.5 | 5.0 | 5.3 | 3.9 | 2.0 | 13.5 | 11.8 | |
| maximum} | {41.8 | 16.4 | 12.7 | 10.0 | 14.4 | 23.3 | 27.3 | 15.0 | 8.4 | 3.5 | 5.9 | 5.3 | 5.8 | 4.1 | 2.6 | 14.3 | 12.2 | |||
| minimum} | {39.3 | 14.4 | 11.9 | 9.2 | 13.1 | 20.5 | 25.0 | 12.3 | 7.2 | 3.2 | 5.0 | 4.7 | 5.0 | 3.6 | 1.6 | 12.6 | 11.4 | |||
| U. S. N. M. | 212423 | ♀ ad. | Vale, Oregon | 37.4 | 9.2 | 19.5 | 22.0 | 13.1 | 6.9 | 3.0 | 5.4 | 4.9 | 5.0 | 3.7 | 1.8 | 12.3 | ||||
| Mustela frenata washingtoni | ||||||||||||||||||||
| average} | ♂ ad. 22 | Mt. Adams, Washington | {43.7 | 16.7 | 13.7 | 10.5 | 15.5 | 23.4 | 27.0 | 14.6 | 8.0 | 3.1 | 5.9 | 5.4 | 5.6 | 4.3 | 2.2 | 14.4 | 12.6 | |
| maximum} | {47.7 | 18.0 | 15.4 | 12.0 | 16.5 | 26.4 | 29.6 | 15.8 | 8.7 | 3.4 | 6.5 | 6.0 | 6.1 | 4.8 | 2.6 | 15.8 | 13.7 | |||
| minimum} | {40.0 | 15.6 | 12.5 | 9.0 | 14.5 | 22.1 | 24.6 | 13.5 | 7.6 | 2.7 | 5.4 | 4.9 | 5.0 | 4.0 | 1.7 | 13.1 | 11.5 | |||
| average} | ♀ ad. 11 | Mt. Adams, Washington | {37.7 | 14.3 | 11.5 | 8.8 | 12.8 | 20.2 | 22.5 | 12.9 | 7.2 | 2.8 | 5.1 | 4.7 | 4.8 | 3.9 | 1.9 | 12.2 | 10.4 | |
| maximum} | {39.0 | 14.9 | 12.0 | 9.3 | 13.2 | 21.1 | 24.5 | 13.6 | 7.7 | 3.0 | 5.6 | 4.9 | 5.0 | 4.2 | 2.1 | 13.0 | 11.0 | |||
| minimum} | {37.1 | 13.3 | 10.8 | 8.2 | 12.1 | 19.4 | 21.3 | 12.3 | 6.8 | 2.3 | 4.7 | 4.4 | 4.2 | 3.4 | 1.7 | 11.2 | 9.8 | |||
| Mustela frenata saturata | ||||||||||||||||||||
| U. O. | 3709 | ♂ ad. | Mt. Ashland, Oregon | 45.8 | 17.2 | 14.1 | 11.1 | 15.8 | 26.0 | 27.9 | 15.1 | 8.9 | 3.9 | 6.1 | 5.5 | 5.9 | 4.3 | 2.3 | 14.2 | 12.6 |
| U. S. N. M. | 65930 | ♂ ad. | Siskiyou, Oregon | 42.6 | 15.9 | 14.0 | 10.9 | 14.4 | 24.5 | 27.7 | 14.7 | 8.5 | 4.0 | 5.7 | 5.0 | 5.5 | 4.1 | 1.9 | 14.8 | 12.4 |
| M. V. Z. | 13778 | ♂ ad. | Jackson Lake, California | 45.5 | 18.0 | 13.2 | 10.0 | 16.2 | 24.7 | 27.4 | 14.6 | 8.7 | 3.2 | 6.3 | 5.9 | 6.2 | 4.6 | 2.5 | 14.6 | 12.8 |
| M. V. Z. | 13779 | ♂ ad. | Jackson Lake, California | 43.8 | 16.9 | 12.7 | 9.9 | 15.3 | 24.2 | 26.9 | 15.1 | 8.6 | 3.3 | 5.6 | 5.2 | 5.5 | 4.0 | 2.2 | 14.2 | 12.3 |
| av. 4 | 44.4 | 17.0 | 13.5 | 10.2 | 15.4 | 24.9 | 27.5 | 14.9 | 8.7 | 3.6 | 5.9 | 5.4 | 5.8 | 4.3 | 2.2 | 14.5 | 12.5 | |||
| M. V. Z. | 52144 | ♀ ad. S | . Fork Mt., California | 38.2 | 14.7 | 10.8 | 8.1 | 13.5 | 19.8 | 21.8 | 12.4 | 7.2 | 2.7 | 5.0 | 4.6 | 4.9 | 3.7 | 1.8 | 12.2 | 10.4 |
| Mustela frenata altifrontalis | ||||||||||||||||||||
| average} | ♂ ad. 10 | Tillamook Co., Oregon | {45.8 | 17.5 | 14.2 | 11.2 | 15.9 | 25.1 | 29.2 | 15.6 | 8.5 | 3.5 | 6.1 | 5.5 | 5.7 | 4.5 | 2.5 | 15.4 | 13.6 | |
| maximum} | {48.0 | 18.9 | 15.0 | 12.0 | 16.8 | 26.0 | 31.6 | 16.5 | 9.0 | 3.6 | 6.5 | 6.0 | 6.0 | 4.9 | 2.8 | 16.2 | 14.0 | |||
| minimum} | {42.4 | 16.1 | 13.2 | 10.0 | 14.8 | 23.9 | 26.0 | 15.0 | 7.7 | 3.2 | 5.6 | 5.0 | 5.2 | 4.0 | 2.1 | 14.5 | 12.4 | |||
| Walker | 392 | ♀ ad. | Blaine, Oregon | 39.7 | 15.1 | 12.8 | 10.1 | 13.7 | 21.8 | 24.0 | 13.8 | 7.7 | 5.2 | 5.0 | 5.0 | 3.7 | 1.9 | 13.5 | 11.8 | |
| Walker | 185 | ♀ ad. | Blaine, Oregon | 37.8 | 13.9 | 11.9 | 9.5 | 13.4 | 20.8 | 22.7 | 13.4 | 7.3 | 2.8 | 4.9 | 4.5 | 4.7 | 3.2 | 1.5 | 12.6 | 10.7 |
| Walker | 89 | ♀ ad. | Blaine, Oregon | 38.3 | 14.3 | 11.3 | 9.5 | 12.7 | 19.8 | 22.7 | 13.2 | 7.0 | 3.1 | 4.4 | 4.7 | 3.8 | 1.9 | 13.4 | 11.3 | |
| Walker | 45 | ♀ ad. | Tillamook, Oregon | 37.8 | 14.1 | 11.0 | 8.6 | 12.5 | 20.1 | 23.2 | 13.0 | 7.3 | 4.9 | 4.7 | 4.8 | 3.8 | 2.0 | 13.5 | 11.0 | |
| Mustela frenata oregonensis | ||||||||||||||||||||
| M. V. Z. | 11747 | ♂ ad. | Eureka, California | 44.0 | 16.8 | 13.2 | 10.7 | 15.3 | 24.3 | 28.0 | 14.6 | 8.3 | 3.4 | 6.0 | 5.4 | 5.8 | 4.3 | 2.2 | 13.6 | 12.7 |
| C. A. C. | 3907 | ♂ ad. | Requa, California | 41.8 | 16.0 | 13.5 | 10.9 | 14.0 | 22.9 | 26.6 | 14.2 | 7.6 | 3.1 | 5.8 | 5.3 | 5.4 | 4.0 | 2.2 | 13.4 | 12.3 |
| F. M. N. H. | 9595 | ♂ ad. | Gold Beach, Oregon | 43.0 | 16.9 | 13.1 | 10.5 | 15.5 | 23.4 | 26.2 | 15.1 | 8.2 | 3.3 | 5.8 | 5.2 | 5.5 | 4.4 | 2.0 | 14.7 | 12.5 |
| U. S. N. M. | 32019 | ♂ sad. | Grants Pass, Oregon | 42.9 | 16.3 | 13.7 | 10.5 | 15.7 | 23.2 | 26.5 | 15.2 | 8.8 | 4.0 | 5.5 | 4.8 | 5.2 | 4.3 | 2.1 | 14.0 | 12.5 |
| M. V. Z. | 34325 | ♀ ad. | Carlotta, California | 37.8 | 14.7 | 10.8 | 8.8 | 13.0 | 20.7 | 23.2 | 13.4 | 7.5 | 2.2 | 5.3 | 5.0 | 5.5 | 4.0 | 1.8 | 13.7 | 11.0 |
| U. O. | 1413 | ♀ ad. | 13 mi. S Grants Pass, Oregon | 39.4 | 14.7 | 12.0 | 9.7 | 13.9 | 21.5 | 24.0 | 14.2 | 7.6 | 2.7 | 4.8 | 4.4 | 4.7 | 3.7 | 1.9 | 12.6 | 10.8 |
| Mustela frenata munda | ||||||||||||||||||||
| M. V. Z. | 19720 | ♂ ad. | Point Arena, California | 49.0 | 18.5 | 15.2 | 11.7 | 16.8 | 27.5 | 33.2 | 16.2 | 9.3 | 3.6 | 6.5 | 5.6 | 6.0 | 4.8 | 2.6 | 14.2 | 14.2 |
| M. V. Z. | 19722 | ♂ ad. | Point Arena, California | 48.5 | 18.9 | 14.4 | 10.5 | 17.2 | 26.8 | 32.+ | 16.0 | 8.5 | 3.8 | 6.5 | 5.6 | 6.2 | 5.0 | 2.9 | 14.7 | 13.8 |
| M. V. Z. | 19718 | ♂ ad. | Gualala, California | 45.7 | 17.2 | 13.7 | 10.5 | 15.5 | 26.4 | 31.7 | 15.5 | 8.5 | 3.1 | 6.5 | 5.7 | 5.8 | 4.9 | 2.3 | 13.4 | 14.2 |
| av. 3 | 47.7 | 18.2 | 14.4 | 10.9 | 16.5 | 26.9 | 32.7 | 15.9 | 8.8 | 3.5 | 6.5 | 5.7 | 6.0 | 4.9 | 2.6 | 14.1 | 14.1 | |||
| M. V. Z. | 19714 | ♂ ad. | 6 mi. W Inverness, California | 48.2 | 18.4 | 13.9 | 11.3 | 17.2 | 26.2 | 30.5 | 15.7 | 8.5 | 3.5 | 6.5 | 5.6 | 6.0 | 4.8 | 2.6 | 14.2 | 14.2 |
| M. V. Z. | 19715 | ♂ ad. | 6 mi. W Inverness, California | 48.2 | 18.9 | 14.5 | 11.4 | 16.6 | 26.5 | 30.0 | 16.3 | 8.7 | 3.5 | 6.3 | 5.6 | 6.2 | 5.0 | 2.9 | 14.7 | 13.8 |
| M. V. Z. | 19716 | ♂ ad. | 5 mi. W Inverness, California | 46.5 | 17.5 | 13.8 | 11.0 | 15.9 | 25.2 | 30.3 | 15.4 | 7.9 | 3.5 | 6.1 | 5.7 | 5.8 | 4.9 | 2.3 | 13.4 | 14.2 |
| av. 3 | 47.6 | 18.3 | 14.1 | 11.2 | 16.6 | 26.0 | 30.3 | 15.8 | 8.4 | 3.5 | 6.3 | 5.7 | 6.0 | 4.9 | 2.6 | 14.1 | 14.1 | |||
| F. M. N. H. | 9598 | ♂ ad. | Nicasio, California | 46.5 | 18.5 | 14.0 | 10.5 | 15.4 | 25.2 | 30.5 | 15.0 | 8.8 | 3.5 | 6.9 | 5.9 | 6.3 | 5.2 | 2.7 | 15.2 | 13.0 |
| M. C. Z. | 8632 | ♂ ad. | Nicasio, California | 44.2 | 17.8 | 14.0 | 11.4 | 16.9 | 24.4 | 27.6 | 15.7 | 8.2 | 3.7 | 6.4 | 5.7 | 5.9 | 5.0 | 2.6 | 12.6 | 13.7 |
| M. C. Z. | 5459 | ♂ ad. | Point Reyes, California | 39.7 | 15.5 | 13.3 | 9.0 | 13.4 | 23.1 | 26.5 | 13.7 | 7.7 | 3.2 | 6.1 | 5.1 | 5.5 | 4.4 | 2.5 | 13.6 | 12.1 |
| M. V. Z. | 40302 | ♂ ad. | 4 mi. N Vallejo, California | 45.8 | 17.7 | 13.0 | 10.7 | 15.9 | 24.4 | 26.9 | 15.1 | 8.0 | 2.8 | 6.3 | 5.7 | 6.2 | 4.8 | 2.3 | 14.5 | 13.0 |
| av. 4 | 44.1 | 17.4 | 13.6 | 10.4 | 15.4 | 24.3 | 27.9 | 14.9 | 8.2 | 3.3 | 6.4 | 5.6 | 6.0 | 4.9 | 2.5 | 14.0 | 13.0 | |||
| M. V. Z. | 19723 | ♀ ad. | Point Arena, California | 42.3 | 16.0 | 12.3 | 12.2 | 14.0 | 23.6 | 25.5 | 14.5 | 8.3 | 2.9 | 5.0 | 5.2 | 4.0 | 1.9 | 13.4 | 11.3 | |
| U. S. N. M. | 135010 | ♀ ad. | Point Reyes, California | 38.7 | 15.3 | 11.0 | 8.7 | 14.5 | 21.0 | 23.7 | 13.0 | 7.5 | 3.0 | 5.1 | 4.9 | 5.4 | 3.9 | 2.3 | 12.9 | 10.8 |
| F. M. N. H. | 9597 | ♀ ad. | Point Reyes, California | 39.5 | 15.2 | 11.1 | 8.2 | 13.1 | 20.2 | 22.8 | 12.7 | 6.8 | 2.7 | 5.3 | 4.9 | 5.0 | 3.9 | 1.9 | 14.7 | 10.8 |
| U. S. N. M. | 91764 | ♀ ad. | Point Reyes, California | 38.7 | 15.4 | 12.0 | 9.7 | 12.7 | 21.7 | 24.7 | 13.7 | 7.4 | 2.9 | 5.5 | 5.0 | 5.3 | 4.2 | 2.0 | 13.2 | 11.7 |
| Mustela frenata xanthogenys | ||||||||||||||||||||
| Walker | 1440 | ♂ ad. | 5 mi. W Fresno, California | 43.9 | 16.5 | 13.8 | 10.4 | 14.1 | 23.8 | 28.5 | 14.7 | 7.9 | 3.2 | 5.7 | 5.2 | 5.3 | 4.2 | 2.1 | 14.8 | 12.6 |
| A. N. S. P. | 11863 | ♂ ad. | Fresno, California | 43.4 | 16.8 | 12.9 | 10.0 | 14.8 | 24.0 | 27.5 | 14.5 | 7.5 | 5.8 | 5.3 | 5.5 | 4.4 | 2.0 | 13.7 | 12.7 | |
| Wisconsin U. | 4232 | ♂ ad. | Selma, California | 43.7 | 16.2 | 13.3 | 9.9 | 14.5 | 23.7 | 27.1 | 15.2 | 8.5 | 3.7 | 5.8 | 5.3 | 5.4 | 4.2 | 2.2 | 13.8 | 12.3 |
| Snyder | 2626 | ♀ ad. | Selma, California | 39.4 | 15.0 | 12.0 | 8.9 | 13.2 | 21.3 | 24.3 | 13.7 | 7.2 | 5.3 | 4.0 | 13.0 | 11.4 | ||||
| M. V. Z. | 79640 | ♀ ad. | Tegner School, California | 43.4 | 16.5 | 12.6 | 9.3 | 15.1 | 22.8 | 24.9 | 15.0 | 7.5 | 3.0 | 5.6 | 5.5 | 5.9 | 4.5 | 2.0 | 13.3 | 12.2 |
| Mustela frenata nigriauris | ||||||||||||||||||||
| Stanford U. | 863 | ♂ ad. | Palo Alto, California | 48.1 | 18.7 | 15.0 | 11.2 | 17.5 | 32.9 | 15.5 | 6.5 | 6.4 | 6.7 | 5.2 | 2.4 | 14.3 | ||||
| F. M. N. H. | 6559 | ♂ ad. | Palo Alto, California | 48.0 | 18.7 | 13.9 | 11.0 | 16.2 | 27.0 | 29.6 | 15.7 | 8.7 | 3.4 | 6.2 | 5.8 | 6.0 | 4.8 | 2.7 | 14.0 | 14.1 |
| Stanford U. | 1651 | ♂ ad. | Menlo Park, California | 47.1 | 17.8 | 13.3 | 9.8 | 14.9 | 25.6 | 30.0 | 15.0 | 8.0 | 3.3 | 6.5 | 6.0 | 6.5 | 4.6 | 2.4 | 14.6 | 13.8 |
| Stanford U. | 487 | ♂ ad. | Palo Alto, California | 46.5 | 18.3 | 13.4 | 11.0 | 16.1 | 25.2 | 31.1 | 15.1 | 8.0 | 2.9 | 6.3 | 5.7 | 6.0 | 4.9 | 2.5 | 14.5 | 14.3 |
| F. M. N. H. | 7031 | ♂ ad. | Palo Alto, California | 46.1 | 18.1 | 14.5 | 10.7 | 15.1 | 26.0 | 29.5 | 14.9 | 8.3 | 2.4 | 6.3 | 6.0 | 6.1 | 4.6 | 2.6 | 15.0 | 13.2 |
| Stanford U. | 236 | ♂ ad. | Menlo Park, California | 46.1 | 17.8 | 13.5 | 10.4 | 15.5 | 29.6 | 15.6 | 8.5 | 6.0 | 5.6 | 5.7 | 4.8 | 2.5 | 14.0 | |||
| av. 6 | 47.0 | 18.2 | 13.9 | 10.7 | 15.9 | 26.0 | 30.5 | 15.3 | 8.3 | 3.0 | 6.3 | 5.9 | 6.2 | 4.8 | 2.5 | 14.5 | 14.0 | |||
| M. V. Z. | 5851 | ♀ ad. | Hayward, California | 40.7 | 15.3 | 11.8 | 8.8 | 13.9 | 21.8 | 24.9 | 14.2 | 7.9 | 3.0 | 5.2 | 4.9 | 4.9 | 4.1 | 1.8 | 12.4 | 11.0 |
| M. V. Z. | 30327 | ♀ ad. | Palo Alto, California | 41.2 | 16.2 | 11.1 | 8.6 | 14.3 | 21.7 | 24.8 | 13.4 | 7.8 | 3.0 | 5.3 | 5.0 | 5.4 | 4.1 | 2.1 | 12.6 | 11.0 |
| U. S. N. M. | 43574 | ♀ ad. | Morro, California | 42.2 | 16.1 | 12.2 | 8.9 | 14.2 | 22.7 | 24.3 | 15.1 | 8.1 | 2.9 | 5.5 | 5.4 | 5.6 | 4.2 | 2.0 | 13.2 | 11.4 |
| Mustela frenata latirostra | ||||||||||||||||||||
| M. V. Z. | 3257 | ♂ ad. | San Diego, California | 44.4 | 17.8 | 13.4 | 10.2 | 15.0 | 23.5 | 27.6 | 15.6 | 8.4 | 3.9 | 6.0 | 5.5 | 5.5 | 4.3 | 2.2 | 14.0 | 12.8 |
| U. S. N. M. | 52701 | ♂ ad. | El Vido, California | 43.7 | 16.9 | 12.7 | 9.9 | 14.3 | 24.0 | 14.7 | 8.7 | 4.0 | 5.7 | 5.6 | 5.6 | 4.2 | 2.0 | 14.2 | 13.4 | |
| U. S. N. M. | 52702 | ♂ ad. | El Cajon, California | 43.2 | 16.7 | 13.9 | 10.0 | 15.0 | 24.0 | 27.2 | 15.1 | 8.2 | 4.2 | 5.8 | 5.3 | 5.5 | 4.2 | 2.5 | 14.0 | 12.5 |
| M. V. Z. | 3258 | ♂ ad. | San Diego, California | 42.5 | 16.6 | 14.1 | 12.1 | 14.7 | 24.0 | 28.7 | 15.1 | 8.1 | 3.9 | 5.9 | 5.6 | 5.5 | 4.4 | 2.5 | 14.3 | 13.5 |
| Stephens | 20 | ♂ ad. | San Jacinto Plain, California | 41.9 | 16.9 | 13.1 | 10.6 | 14.0 | 24.0 | 28.2 | 14.7 | 8.0 | 3.2 | 6.0 | 5.5 | 5.7 | 4.2 | 2.1 | 14.0 | 12.9 |
| S. D. M. | 7098 | ♂ ad. | Jamacha, California | 47.0 | 18.5 | 15.6 | 11.3 | 16.8 | 24.8 | 29.2 | 16.7 | 8.9 | 3.8 | 6.0 | 5.8 | 6.2 | 4.8 | 2.2 | 14.0 | 13.2 |
| av. 6 | 43.8 | 17.2 | 13.8 | 10.7 | 15.0 | 24.1 | 28.2 | 15.3 | 8.4 | 3.8 | 5.9 | 5.6 | 5.7 | 4.4 | 2.3 | 14.1 | 13.1 | |||
| Stephens | 22 | ♀ ad. | Santa Ysabel, California | 40.1 | 15.5 | 11.7 | 9.3 | 13.1 | 21.9 | 24.5 | 14.2 | 8.0 | 3.0 | 5.3 | 4.8 | 5.1 | 4.1 | 2.0 | 14.0 | 11.3 |
| Stephens | 19 | ♀ ad. | Ballena, California | 40.0 | 15.0 | 12.3 | 9.3 | 13.5 | 21.6 | 23.5 | 13.7 | 8.1 | 3.2 | 5.1 | 4.9 | 5.0 | 4.0 | 2.1 | 12.0 | 11.2 |
| S. D. M. | 6748 | ♀ ad. | Santa Ysabel, California | 42.0 | 16.1 | 12.9 | 9.4 | 14.2 | 22.0 | 24.5 | 14.0 | 7.8 | 3.0 | 5.8 | 5.3 | 5.7 | 4.5 | 2.0 | 13.4 | 11.5 |
| S. D. M. | 7194 | ♀ ad. | Jamacha, California | 39.8 | 15.0 | 11.8 | 9.1 | 13.7 | 20.2 | 23.7 | 13.7 | 7.0 | 2.9 | 5.2 | 4.8 | 4.9 | 3.9 | 1.9 | 12.6 | 11.5 |
| av. 4 | 40.5 | 15.4 | 12.2 | 9.3 | 13.6 | 21.4 | 24.1 | 13.9 | 7.7 | 3.0 | 5.4 | 5.0 | 5.2 | 4.1 | 2.0 | 13.0 | 11.4 | |||
| Mustela frenata pulchra | ||||||||||||||||||||
| C. A. S. | 335 | ♂ ad. | Buttonwillow, California | 48.6 | 18.8 | 15.2 | 11.6 | 17.2 | 27.4 | 31.4 | 16.7 | 9.1 | 3.6 | 6.6 | 5.9 | 6.4 | 5.1 | 2.7 | 16.2 | 13.7 |
| C. A. S. | 337 | ♂ ad. | Buttonwillow, California | 48.6 | 18.8 | 15.0 | 11.8 | 17.2 | 27.1 | 32.7 | 16.6 | 9.0 | 3.5 | 6.6 | 6.3 | 6.3 | 5.0 | 2.4 | 16.5 | 14.3 |
| M. V. Z. | 16668 | ♂ ad. | Buttonwillow, California | 48.1 | 18.8 | 14.8 | 12.0 | 17.1 | 27.7 | 31.2 | 16.4 | 9.2 | 3.6 | 6.4 | 5.7 | 5.9 | 5.1 | 2.8 | 15.2 | 14.0 |
| U. S. N. M. | 137935 | ♂ ad. | Buttonwillow, California | 47.2 | 18.2 | 14.0 | 10.3 | 16.0 | 26.1 | 29.5 | 15.6 | 8.5 | 3.3 | 6.0 | 5.6 | 5.9 | 4.1 | 2.0 | 15.0 | 12.9 |
| C. A. S. | 336 | ♂ ad. | Buttonwillow, California | 47.0 | 18.3 | 14.5 | 11.5 | 16.5 | 27.0 | 29.5 | 15.5 | 9.1 | 3.2 | 6.3 | 5.6 | 5.9 | 5.0 | 2.3 | 15.0 | 13.0 |
| C. A. S. | 338 | ♂ ad. | Buttonwillow, California | 46.0 | 17.4 | 14.7 | 11.3 | 16.1 | 26.3 | 32.6 | 15.9 | 8.9 | 3.7 | 6.1 | 5.7 | 5.8 | 4.6 | 2.2 | 15.0 | 13.3 |
| av. 6 | 47.6 | 18.4 | 14.7 | 11.4 | 16.7 | 26.9 | 31.1 | 16.1 | 9.0 | 3.5 | 6.3 | 5.8 | 6.0 | 4.8 | 2.4 | 15.5 | 13.5 | |||
| Mustela frenata inyoensis | ||||||||||||||||||||
| M. V. Z. | 25907 | ♂ ad. | 2 mi. N Independence, Calif. | 44.7 | 17.3 | 13.3 | 10.8 | 15.9 | 25.3 | 29.5 | 15.9 | 9.0 | 3.7 | 6.0 | 5.7 | 5.9 | 4.6 | 2.4 | 15.0 | 13.2 |
| Mustela frenata texensis | ||||||||||||||||||||
| A. M. N. H. | 14821 | ♂ ad. | Kerr Co., Texas | 54.0 | 19.2 | 16.0 | 12.6 | 17.2 | 28.6 | 35.1 | 17.5 | 8.0 | 3.5 | 6.9 | 6.5 | 6.7 | 5.0 | 2.5 | 16.7 | 15.1 |
| M. C. Z. | 15476 | ♂ yg. | Kerr Co., Texas | 53.3 | 18.9 | 16.3 | 12.8 | 18.3 | 28.2 | 34.8 | 18.0 | 8.3 | 4.0 | 6.4 | 6.3 | 6.5 | 4.8 | 2.4 | 16.0 | 15.0 |
| Baylor U. | 2017 | ♂ sad. | 5 mi. N Waco, Texas | 52.0 | 18.3 | 16.5 | 13.5 | 16.7 | 29.2 | 18.0 | 9.1 | 6.6 | 6.3 | 6.4 | 4.9 | 2.6 | 16.3 | 15.6 | ||
| Mustela frenata neomexicana | ||||||||||||||||||||
| Kansas U. | 1485 | ♂ ad. | Liberal, Kansas | 50.5 | 17.9 | 15.4 | 12.4 | 16.4 | 28.3 | 35.0 | 17.0 | 9.0 | 4.0 | 6.3 | 5.9 | 6.0 | 4.9 | 2.4 | 15.5 | 14.3 |
| U. S. N. M. | 131582 | ♂ ad. | Berino, New Mexico | 47.7 | 17.5 | 15.0 | 11.1 | 15.9 | 26.3 | 31.2 | 16.5 | 7.9 | 3.4 | 6.4 | 5.7 | 6.3 | 4.7 | 2.5 | 15.8 | 13.5 |
| U. S. N. M. | 36482 | ♀ ad. | Tombstone, Arizona | 45.5 | 16.5 | 12.8 | 9.7 | 15.5 | 22.3 | 26.6 | 15.2 | 7.5 | 3.3 | 5.9 | 5.6 | 5.9 | 4.5 | 2.2 | 13.8 | 12.0 |
| U. S. N. M. | 230973 | ♀ ad. | Willcox, Arizona | 42.5 | 15.1 | 12.9 | 9.9 | 14.0 | 22.6 | 26.5 | 14.5 | 7.0 | 3.1 | 5.5 | 5.3 | 5.6 | 4.1 | 1.8 | 14.0 | 12.0 |
| U. S. N. M. | 225629 | ♀ ad. | Albuquerque, New Mexico | 40.8 | 15.0 | 12.4 | 9.4 | 13.3 | 21.6 | 24.5 | 14.5 | 7.5 | 3.0 | 5.2 | 4.7 | 5.0 | 3.9 | 1.8 | 13.0 | 11.0 |
| Mustela frenata frenata | ||||||||||||||||||||
| M. C. Z. | 240 | ♂ ad. | Brownsville, Texas | 49.4 | 17.4 | 14.9 | 11.6 | 15.4 | 25.9 | 33.0 | 16.7 | 7.9 | 4.3 | 5.9 | 5.9 | 6.1 | 4.5 | 2.1 | 15.0 | 13.8 |
| A. N. S. P. | 724 | ♂ ad. | Brownsville, Texas | 50.1 | 17.9 | 15.5 | 12.3 | 15.5 | 27.0 | 32.2 | 16.5 | 8.5 | 4.1 | 6.5 | 6.2 | 6.3 | 4.8 | 2.7 | 16.0 | 14.0 |
| U. S. N. M. | 58684 | ♂ ad. | Brownsville, Texas | 48.2 | 17.3 | 14.2 | 11.0 | 15.3 | 27.2 | 31.0 | 16.6 | 8.3 | 4.2 | 6.3 | 5.9 | 6.1 | 4.8 | 2.7 | 15.5 | 13.5 |
| U. S. N. M. | 63857 | ♂ ad. | Brownsville, Texas | 48.6 | 18.0 | 13.7 | 11.1 | 16.5 | 26.0 | 31.0 | 16.0 | 8.2 | 4.8 | 6.5 | 5.7 | 6.1 | 4.8 | 2.6 | 15.0 | 13.6 |
| U. S. N. M. | 44976 | ♂ ad. | Brownsville, Texas | 50.9 | 18.0 | 16.9 | 26.9 | 16.6 | 7.9 | 3.4 | 6.5 | 5.6 | 6.1 | 4.7 | 2.3 | 16.0 | 13.4 | |||
| av. 5 | 49.4 | 17.7 | 14.6 | 11.5 | 15.9 | 26.6 | 31.8 | 16.3 | 8.2 | 4.2 | 6.4 | 5.9 | 6.2 | 4.7 | 2.5 | 15.5 | 13.7 | |||
| A. M. N. H. | 24405 | ♀ ad. | Brownsville, Texas | 47.3 | 16.2 | 12.5 | 10.0 | 14.6 | 5.9 | 5.4 | 5.4 | 4.1 | 2.1 | 12.2 | ||||||
| U. S. N. M. | 58685 | ♀ sad. | Brownsville, Texas | 41.3 | 15.0 | 11.8 | 9.5 | 12.8 | 22.7 | 27.0 | 14.0 | 6.9 | 3.3 | 5.5 | 5.2 | 5.4 | 4.1 | 2.0 | 14.0 | 11.7 |
| U. S. N. M. | 36362/4873 | 2 ♀ sad. | Brownsville, Texas | 42.5 | 15.4 | 12.5 | 10.0 | 14.3 | 23.8 | 26.7 | 14.3 | 7.5 | 3.2 | 5.9 | 5.5 | 5.7 | 4.2 | 2.0 | 13.5 | 11.8 |
| av. 3 | 43.7 | 15.5 | 12.3 | 9.8 | 13.9 | 23.3 | 26.9 | 14.2 | 7.2 | 3.3 | 5.8 | 5.4 | 5.5 | 4.1 | 2.0 | 13.8 | 11.9 | |||
| B. Z. M. | 991 | ♀ ad. | México | 15.5 | 12.2 | 13.8 | 22.8 | 27.0 | 13.7 | 5.7 | 5.2 | 5.5 | 4.4 | 2.2 | 12.7 | |||||
| B. Z. M. | 992 | ♀ ad. | México | 13.9 | 10.7 | 8.9 | 12.9 | 21.1 | 13.0 | 6.6 | 2.9 | 4.9 | 4.5 | 4.7 | 3.7 | 1.7 | 12.3 | 10.8 | ||
| M. C. Z. | 20841 | ♂ ad. | Miquihana, Nuevo León | 50.2 | 18.0 | 15.3 | 12.1 | 16.3 | 28.0 | 32.0 | 16.9 | 8.8 | 3.3 | 4.6 | 2.2 | 15.0 | ||||
| U. S. N. M. | 50826 | ♂ ad. | Tlalpam, D. F. | 51.3 | 18.3 | 15.1 | 12.1 | 17.5 | 27.7 | 33.5 | 16.3 | 8.4 | 3.5 | 6.7 | 5.9 | 6.4 | 4.7 | 2.7 | 15.3 | 14.6 |
| Mustela frenata leucoparia | ||||||||||||||||||||
| U. S. N. M. | 125972 | ♂ ad. | Los Reyes, Michoacán | 51.2 | 17.5 | 15.0 | 12.0 | 16.0 | 28.3 | 32.9 | 16.0 | 7.7 | 3.5 | 5.9 | 5.7 | 5.7 | 4.3 | 2.2 | 15.5 | 14.0 |
| U. S. N. M. | 34914/4717 | 9 ♂ ad. | Pátzcuaro, Michoacán | 18.7 | 14.3 | 16.8 | 6.8 | 6.5 | 6.8 | 5.0 | 2.6 | 14.3 | ||||||||
| A. M. N. H. | 26153 | ♀ ad. | Artenkiki, Jalisco | 44.5 | 16.0 | 12.7 | 10.0 | 14.4 | 22.4 | 26.3 | 15.0 | 7.0 | 3.2 | 5.9 | 5.5 | 6.0 | 4.5 | 2.1 | 14.0 | 11.9 |
| Mustela frenata perotae | ||||||||||||||||||||
| U. S. N. M. | 68197 | ♂ ad. | Cerro San Felipe, Oaxaca | 49.2 | 17.3 | 13.9 | 11.7 | 15.9 | 25.0 | 29.2 | 15.5 | 6.8 | 2.5 | 6.1 | 5.3 | 5.8 | 4.1 | 2.1 | 15.5 | 13.3 |
| U. S. N. M. | 54278 | ♀ ad. | Type specimen | 43.5 | 15.5 | 12.3 | 10.3 | 14.0 | 23.2 | 25.5 | 15.0 | 7.0 | 2.0 | 5.7 | 5.2 | 5.7 | 4.1 | 2.1 | 12.0 | 13.7 |
| Mustela frenata goldmani | ||||||||||||||||||||
| U. S. N. M. | 133253 | ♂ ad. | 20 mi. SE Teopisca, Chiapas | 50.4 | 18.0 | 15.0 | 12.5 | 15.9 | 26.5 | 32.3 | 15.6 | 7.1 | 2.9 | 6.4 | 5.9 | 6.2 | 5.0 | 2.5 | 16.5 | 13.7 |
| U. S. N. M. | 133254 | ♂ ad. | 20 mi. SE Teopisca, Chiapas | 49.6 | 17.5 | 13.8 | 10.9 | 16.2 | 26.4 | 31.8 | 15.8 | 7.4 | 3.0 | 6.1 | 5.5 | 5.8 | 4.5 | 2.5 | 15.0 | 13.7 |
| U. S. N. M. | 77519 | ♂ ad. | Pinabete, Chiapas | 50.7 | 18.3 | 26.5 | 31.8 | 15.3 | 7.5 | 2.9 | 6.6 | 5.8 | 6.1 | 5.0 | 2.4 | 16.0 | ||||
| F. M. N. H. | 15953 | ♂ ad. | near Tecpám, Guatemala | 50.0 | 17.0 | 13.5 | 12.0 | 16.1 | 26.2 | 31.5 | 15.5 | 7.0 | 5.9 | 5.6 | 6.0 | 4.5 | 2.2 | |||
| Dickey | 12523 | ♂ ad. | Los Esesmiles, Salv. | 51.3 | 17.5 | 14.5 | 11.7 | 16.2 | 26.7 | 30.6 | 15.8 | 7.7 | 3.1 | 6.3 | 5.5 | 5.8 | 4.5 | 2.3 | 16.5 | 14.1 |
| av. 5 | 50.4 | 17.7 | 14.2 | 11.8 | 16.1 | 26.5 | 31.6 | 15.6 | 7.3 | 3.0 | 6.3 | 5.7 | 6.0 | 4.7 | 2.4 | 16.0 | 13.8 | |||
| Mustela frenata macrophonius | ||||||||||||||||||||
| F. M. N. H. | 14063 | ♂ ad. | Achotal, Veracruz | 54.1 | 19.2 | 15.6 | 12.9 | 17.8 | 28.5 | 33.6 | 16.8 | 7.6 | 2.9 | 7.1 | 6.4 | 6.8 | 5.2 | 2.9 | 16.8 | 15.3 |
| U. S. N. M. | 132528 | ♀ ad. | Pérez, Veracruz | 43.5 | 15.3 | 12.5 | 10.2 | 14.5 | 23.1 | 26.5 | 15.0 | 6.5 | 2.6 | 5.5 | 5.2 | 5.7 | 4.1 | 1.9 | 14.2 | 12.2 |
| Mustela frenata tropicalis | ||||||||||||||||||||
| U. S. N. M. | 54994 | ♂ ad. | Jico, Veracruz | 47.8 | 17.2 | 13.7 | 10.7 | 15.8 | 24.5 | 28.2 | 15.5 | 6.3 | 2.9 | 6.2 | 5.6 | 5.9 | 4.5 | 2.2 | 15.5 | 13.7 |
| A. M. N. H. | 12764/1105 | 8 ♂ sad. | Jalapa, Veracruz | 45.5 | 16.8 | 13.7 | 11.4 | 16.0 | 24.0 | 30.0 | 15.4 | 6.7 | 3.0 | 6.4 | 5.7 | 5.9 | 4.7 | 2.2 | 15.2 | 13.6 |
| M. C. Z. | 6514 | ♂ sad. | Orizaba, Veracruz | *46.0 | 16.4 | 13.2 | 15.5 | 15.0 | 8.0 | 6.3 | 5.7 | 6.0 | 5.0 | 2.5 | 16.5 | 14.5 | ||||
| U. S. N. M. | 54993 | ♀ ad. | Jico, Veracruz | 36.0 | 13.0 | 11.0 | 9.2 | 12.1 | 19.8 | 22.6 | 12.4 | 6.1 | 2.5 | 5.0 | 4.5 | 4.7 | 4.0 | 1.7 | 12.5 | 11.6 |
| U. S. N. M. | 1060 | ♀ ad. | México | 39.0 | 14.0 | 11.0 | 9.6 | 21.0 | 22.5 | 4.9 | 4.6 | 4.9 | 3.9 | 1.9 | 11.5 | |||||
| M. C. Z. | 2605 | ♀ yg. | Jalpa, Veracruz | 38.7 | 13.8 | 10.3 | 8.3 | 12.6 | 19.4 | 12.7 | 5.8 | 4.9 | 4.5 | 4.6 | 3.5 | 1.5 | 13.7 | 10.9 | ||
| F. M. N. H. | 14050 | ♀ yg. | Xuchil, Veracruz | 39.0 | 14.2 | 11.6 | 9.3 | 13.0 | 20.5 | 23.5 | 13.5 | 6.1 | 5.3 | 4.9 | 5.3 | 4.4 | 2.0 | 15.3 | 11.5 | |
| Mustela frenata perda | ||||||||||||||||||||
| U. S. N. M. | 100041 | ♂ sad. | Teapa, Tabasco | 46.1 | 17.0 | 14.4 | 11.1 | 15.3 | 24.4 | 28.5 | 14.9 | 6.6 | 2.2 | 6.4 | 5.5 | 6.0 | 4.6 | 2.5 | 16.4 | 13.8 |
| U. S. N. M. | 132997 | ♂ sad. | San Vicente, Chiapas | 45.3 | 16.7 | 13.9 | 12.0 | 15.5 | 24.0 | 27.4 | 14.5 | 6.7 | 2.8 | 5.5 | 5.1 | 5.4 | 4.2 | 2.2 | 15.8 | 13.0 |
| U. S. N. M. | 132996 | ♂ sad. | San Cristóbal, Chiapas | 16.9 | 13.3 | 10.6 | 16.0 | 6.4 | 5.5 | 6.0 | 4.5 | 2.5 | 13.0 | |||||||
| av. 3 | 45.7 | 16.9 | 13.9 | 11.2 | 15.6 | 24.2 | 28.0 | 14.7 | 6.7 | 2.5 | 6.1 | 5.4 | 5.8 | 4.4 | 2.4 | 16.1 | 13.3 | |||
| U. S. N. M. | 218036 | ♀ sad. | State of Chiapas | 40.0 | 14.6 | 11.6 | 9.4 | 13.5 | 20.2 | 23.2 | 13.0 | 5.6 | 2.1 | 5.2 | 4.8 | 5.0 | 3.7 | 1.7 | 13.2 | 11.4 |
| U. S. N. M. | 65422 | ♀ sad. | Catemaco, Veracruz | 40.4 | 14.5 | 11.5 | 9.1 | 13.2 | 21.2 | 23.0 | 13.7 | 6.5 | 2.1 | 4.8 | 4.7 | 5.2 | 3.8 | 1.9 | 14.2 | 10.8 |
| Mustela frenata nicaraguae | ||||||||||||||||||||
| A. M. N. H. | 30754 | ♂ sad. | Matagalpa, Nicaragua | 44.8 | 17.2 | 12.8 | 10.5 | 15.0 | 23.4 | 25.5 | 14.0 | 6.5 | 2.9 | 6.3 | 6.0 | 6.3 | 4.6 | 2.5 | 15.5 | 13.4 |
| A. M. N. H. | 28331 | ♂ ad. | Matagalpa, Nicaragua | 44.8 | 16.7 | 13.6 | 11.5 | 15.2 | 22.7 | 26.8 | 15.2 | 6.7 | 5.8 | 5.1 | 5.5 | 4.4 | 2.4 | 14.7 | 13.4 | |
| A. M. N. H. | 29280 | ♂ sad. | San Rafel Del Norte | 45.5 | 17.2 | 13.5 | 11.0 | 15.5 | 23.5 | 27.4 | 14.2 | 6.8 | 6.3 | 6.0 | 6.3 | 4.7 | 2.5 | 15.5 | 13.1 | |
| Mustela frenata costaricensis | ||||||||||||||||||||
| U. S. N. M. | 11408 | ♂ ad. | Costa Rica | *49.0 | 18.3 | 15.2 | 9.4 | *18.0 | 26.0 | 30.5 | 15.0 | 7.5 | 6.5 | 5.9 | 6.4 | 5.0 | 2.6 | 16.7 | 14.2 | |
| B. M. | 3216 | ♂ ad. | Vic. San José, Costa Rica | 49.3 | 18.6 | 15.0 | 12.7 | 16.9 | 25.9 | 31.3 | 15.3 | 7.3 | 2.9 | 6.6 | 6.0 | 6.5 | 4.9 | 2.5 | 16.8 | 15.1 |
| U. S. N. M. | 13770/3714 | 9 ♂ yg. | San José, Costa Rica | 48.2 | 18.0 | 14.0 | 11.7 | 16.0 | 24.8 | 29.0 | 15.0 | 7.2 | 2.7 | 7.0 | 6.0 | 6.5 | 4.8 | 2.9 | 16.5 | 14.0 |
| N. H. R. S. | 1-138 | ♂ ad. | Azahar Cartago, Costa Rica | 47.8 | 17.4 | 14.9 | 11.7 | 15.5 | 24.3 | *30.1 | 15.1 | 7.5 | 6.6 | 5.9 | 6.2 | 4.8 | 2.5 | |||
| B. Z. M. | A 59.13 | ♀ sad. | Irazú, 3000M., Costa Rica | 38.8 | 14.4 | 12.5 | 10.1 | 14.2 | 20.0 | 23.6 | 12.8 | 6.3 | 2.9 | 4.9 | 4.7 | 5.0 | 3.7 | 1.8 | 13.7 | 11.5 |
| Mustela frenata panamensis | ||||||||||||||||||||
| M. C. Z. | 10112 | ♂ ad. | Boquete, Panamá | 48.3 | 17.1 | 15.0 | 12.5 | 15.7 | 25.1 | 28.3 | 14.1 | 7.0 | 2.7 | 6.3 | 5.7 | 5.9 | 4.5 | 2.5 | 16.7 | 14.0 |
| A. M. N. H. | 18848 | ♂ ad. | Boquete, Panamá | 44.5 | 16.3 | 14.0 | 12.0 | 15.0 | 22.5 | 29.1 | 14.4 | 6.1 | 2.7 | 6.2 | 5.7 | 6.0 | 4.5 | 2.3 | 16.0 | 13.5 |
| M. C. Z. | 10113 | ♂ ad. | Boquete, Panamá | 42.8 | 15.8 | 14.0 | 12.0 | 15.5 | 27.1 | 13.6 | 5.5 | 6.1 | 5.4 | 5.7 | 4.3 | 2.2 | 15.8 | 13.5 | ||
| av. 3 | 45.2 | 16.4 | 14.3 | 12.2 | 15.4 | 23.8 | 28.2 | 14.0 | 6.2 | 2.7 | 6.2 | 5.6 | 5.9 | 4.4 | 2.3 | 16.3 | 13.7 | |||
| U. S. N. M. | 170970 | ♀ sad. ne | ar Gatún, Panamá | 41.3 | 15.3 | 12.5 | 10.4 | 14.3 | 23.0 | 26.8 | 12.5 | 6.1 | 5.7 | 5.3 | 5.5 | 4.1 | 2.1 | 15.5 | 12.5 | |
| A. N. S. P. | 18434 | ♀ ad. Si | ola, Panamá | 39.3 | 14.2 | 11.3 | 9.4 | 12.5 | 20.1 | 22.5 | 13.0 | 6.0 | 5.0 | 4.6 | 5.0 | 3.7 | 2.0 | 13.2 | 10.8 | |
| Mustela frenata meridana | ||||||||||||||||||||
| A. M. N. H. | 33154 | ♂ ad. | Mérida, Venezuela | 44.3 | 16.7 | 14.2 | 12.0 | 15.0 | 23.9 | 28.5 | 13.0 | 6.6 | 3.0 | 6.2 | 5.4 | 5.6 | 4.4 | 2.5 | 15.0 | 13.4 |
| U. S. N. M. | 137517 | ♂ ad. | Mérida, Venezuela | 43.4 | 16.0 | 14.1 | 11.8 | 15.2 | 23.5 | 27.4 | 13.8 | 6.5 | 2.4 | 6.0 | 5.5 | 5.7 | 4.1 | 2.3 | 15.6 | 13.3 |
| U. S. N. M. | 172959 | ♂ ad. | Mérida, Venezuela | 44.0 | 16.5 | 14.1 | 11.6 | 15.4 | 23.6 | 28.0 | 13.9 | 7.0 | 6.2 | 5.6 | 5.7 | 4.7 | 2.4 | 15.7 | 12.9 | |
| A. M. N. H. | 24309 | ♂ ad. | Mérida, Venezuela | 44.2 | 16.1 | 13.7 | 11.5 | 15.4 | 23.6 | 27.3 | 14.2 | 6.4 | 2.3 | 5.9 | 5.2 | 5.6 | 4.5 | 2.3 | 15.3 | 13.0 |
| A. M. N. H. | 33155 | ♂ ad. | Mérida, Venezuela | 42.3 | 16.7 | 13.8 | 11.5 | 15.5 | 23.0 | 27.6 | 13.5 | 6.5 | 2.6 | 5.8 | 5.4 | 5.6 | 4.3 | 2.1 | 15.5 | 13.7 |
| av. 5 | 43.6 | 16.4 | 14.0 | 11.7 | 15.3 | 23.5 | 27.8 | 13.7 | 6.6 | 6.0 | 5.4 | 5.6 | 4.4 | 2.3 | 16.0 | 13.3 | ||||
| U. S. N. M. | 123341 | ♂ sad. | Mérida, Venezuela | 43.5 | 16.9 | 13.8 | 12.1 | 15.1 | 23.0 | 27.5 | 13.2 | 6.0 | 5.5 | 5.7 | 4.5 | 2.5 | ||||
| U. S. N. M. | 137516 | ♂ sad. | Mérida, Venezuela | 43.6 | 16.2 | 13.5 | 11.4 | 15.5 | 23.4 | 27.9 | 13.4 | 6.4 | 2.2 | 5.6 | 5.2 | 5.7 | 4.2 | 2.3 | 16.0 | 13.0 |
| U. S. N. M. | 143667 | ♂ sad. | Mérida, Venezuela | 15.4 | 12.5 | 10.5 | 15.4 | 25.0 | 13.2 | 5.5 | 5.0 | 5.3 | 4.2 | 2.3 | 15.0 | 13.2 | ||||
| U. S. N. M. | 143666 | ♀ ad. | Mérida, Venezuela | 38.2 | 14.1 | 11.9 | 9.7 | 13.2 | 20.3 | 23.0 | 12.4 | 5.9 | 1.8 | |||||||
| U. S. N. M. | 143665 | ♀ ad. | Mérida, Venezuela | 36.4 | 13.4 | 11.5 | 9.9 | 13.0 | 20.1 | 23.4 | 12.1 | 5.9 | 2.0 | |||||||
| A. M. N. H. | 24308 | ♀ ad. | Mérida, Venezuela | 36.3 | 13.4 | 11.4 | 9.7 | 11.8 | 18.8 | 22.0 | 11.7 | 5.5 | 2.0 | 4.9 | 4.5 | 4.6 | 3.5 | 1.6 | 10.5 | |
| A. M. N. H. | 24311 | ♀ ad. | Mérida, Venezuela | 37.7 | 13.9 | 11.4 | 9.8 | 13.8 | 19.5 | 12.1 | 5.9 | 1.8 | 4.9 | 4.7 | 4.9 | 3.7 | 1.8 | 13.0 | 11.2 | |
| av. 4 | 37.2 | 13.7 | 11.6 | 9.8 | 13.0 | 19.7 | 22.8 | 12.1 | 5.8 | 1.9 | ||||||||||
| A. M. N. H. | 21343 | ♀ sad. | Mérida, Venezuela | 37.3 | 13.6 | 11.1 | 9.2 | 13.2 | 19.2 | 22.5 | 11.9 | 5.6 | 2.1 | 5.1 | 4.6 | 4.8 | 3.5 | 1.7 | 13.5 | 11.1 |
| A. M. N. H. | 24310 | ♀ sad. | Mérida, Venezuela | 35.0 | 12.6 | 11.1 | 8.7 | 12.0 | 19.3 | 21.8 | 11.5 | 5.8 | 1.8 | 4.7 | 4.4 | 4.7 | 3.5 | 1.6 | 13.8 | 11.4 |
| Mustela frenata affinis | ||||||||||||||||||||
| U. S. N. M. | 241314 | ♂ ad. | Choachí, Colombia | *46.0 | 17.4 | 13.6 | 11.8 | 15.6 | 29.5 | 6.1 | 5.9 | 6.1 | 4.4 | 2.3 | 13.9 | |||||
| U. S. N. M. | 239946 | ♂ ad. | Choachí, Colombia | 16.1 | 12.7 | 10.8 | 14.3 | 27.7 | 6.0 | 5.3 | 5.7 | 4.5 | 2.1 | 13.0 | ||||||
| A. M. N. H. | 35805 | ♂ ad. | Quetame, Colombia | 45.5 | 16.6 | 13.7 | 11.0 | 15.3 | 6.4 | 5.8 | 5.5 | 5.7 | 4.4 | 2.0 | 13.3 | |||||
| av. 3 | 45.8 | 16.7 | 13.3 | 11.2 | 15.1 | 28.6 | 6.0 | 5.6 | 5.8 | 4.4 | 2.1 | 13.4 | ||||||||
| U. S. N. M. | 241313 | ♂ yg. | Bogotá, Columbia | 44.7 | 16.7 | 13.9 | 12.3 | 15.6 | 23.9 | 29.0 | 5.9 | 5.7 | 5.7 | 4.4 | 2.5 | |||||
| U. S. N. M. | 241315 | ♂ yg. | Choachí, Columbia | 45.1 | 16.7 | 13.5 | 11.5 | 15.2 | 23.9 | 28.7 | 14.0 | 6.9 | 5.9 | 5.6 | 5.8 | 4.5 | 2.1 | 13.5 | ||
| Mustela frenata aureoventris | ||||||||||||||||||||
| A. M. N. H. | 34677 | ♂ yg. | Gualea, Ecuador | 45.6 | 17.6 | 14.0 | 11.8 | 15.4 | 23.8 | 28.3 | 15.1 | 6.8 | 2.7 | 6.7 | 6.0 | 6.4 | 5.0 | 3.0 | 17.0 | 14.3 |
| Mustela frenata helleri | ||||||||||||||||||||
| F. M. N. H. | 24133 | ♂ ad. | Rio Chinchao | 44.0 | 16.4 | 14.5 | 12.5 | 14.9 | 24.5 | 29.1 | 14.4 | 6.5 | 2.4 | 6.2 | 5.8 | 5.9 | 4.6 | 2.5 | 16.0 | 14.0 |
| F. M. N. H. | 24132 | ♂ ad. | Rio Chinchao | 45.3 | 17.0 | 13.9 | 11.7 | 15.5 | 24.5 | 29.0 | 14.4 | 6.5 | 2.4 | 5.8 | 5.5 | 5.8 | 4.5 | 2.5 | 16.0 | 13.7 |
| F. M. N. H. | 24136 | ♀ ad. | Huanuco, Perú | 35.3 | 13.0 | 10.5 | 8.9 | 12.1 | 17.9 | 20.8 | 11.8 | 4.7 | 2.0 | 4.7 | 4.4 | 4.6 | 3.5 | 1.6 | 11.8 | 10.0 |
| F. M. N. H. | 24135 | ♀ sad. | Ambo, Perú | 36.1 | 13.6 | 10.9 | 9.0 | 12.4 | 18.8 | 22.4 | 11.8 | 5.0 | 1.9 | 4.7 | 4.4 | 4.6 | 3.6 | 1.7 | 12.6 | 10.5 |
| F. M. N. H. | 24134 | ♀ sad. | Ambo, Perú | 38.1 | 14.0 | 10.6 | 9.1 | 12.6 | 19.8 | 22.2 | 12.0 | 5.4 | 2.0 | 4.7 | 4.6 | 4.6 | 3.5 | 1.5 | 13.9 | 11.0 |
| Mustela frenata agilis | ||||||||||||||||||||
| B. M. 8 | .1.10.1 | ♂ ad. | Lima, Perú | 42.5 | 16.0 | 13.4 | 11.1 | 14.9 | 23.1 | 28.2 | 13.8 | 6.9 | 2.6 | 5.8 | 5.5 | 5.8 | 4.5 | 2.2 | 14.8 | 12.9 |
| F. M. N. H. | 21147 | ♀ ad. | Macate, Perú | 35.2 | 12.7 | 10.0 | 8.4 | 12.0 | 18.0 | 20.6 | 12.4 | 5.0 | 1.7 | 4.4 | 4.1 | 4.4 | 3.2 | 1.5 | 13.0 | 10.3 |
| M. P. H. N. | 565 | ♀ ad. | Perú | *36.0 | 12.6 | 10.7 | 9.2 | 12.5 | 18.1 | 21.5 | 13.0 | 5.8 | 2.2 | 4.8 | 4.1 | 4.5 | 3.5 | 1.9 | 12.0 | 11.7 |
| Mustela frenata macrura | ||||||||||||||||||||
| M. P. H. N. | 561 | ♂ ad. | Junín, Perú | 45.5 | 16.6 | 14.0 | 11.6 | 15.1 | 23.8 | 29.0 | 14.5 | 6.2 | 5.5 | 5.4 | 5.5 | 4.3 | 2.1 | 15.7 | 13.2 | |
| M. P. H. N. | 562 | ♂ ad. | Junín, Perú | 40.8 | 15.0 | 13.6 | 11.3 | 15.1 | 22.9 | 26.5 | 14.5 | 7.5 | 2.7 | 5.8 | 5.0 | 5.1 | 4.0 | 2.0 | 14.6 | 12.9 |
| B. M. 2 | 6.2.1.2 | ♂ ad. | Yana Mayo, Perú | 42.6 | 15.9 | 13.0 | 10.2 | 15.0 | 21.8 | 26.2 | 13.5 | 7.0 | 2.5 | 5.8 | 5.5 | 5.6 | 4.4 | 2.3 | 14.6 | 12.3 |
| U. S. N. M. | 148528 | ♂ ad. | Marcapata, Perú | 16.0 | 14.5 | 5.7 | 5.0 | 5.2 | 4.3 | 2.1 | ||||||||||
| M. P. H. N. | 564 | ♀ ad. | Cutervo, Perú | *38.0 | 13.8 | 11.2 | 9.5 | 13.0 | 18.1 | *23.0 | 4.9 | 4.6 | 4.9 | 3.6 | 1.9 | 11.7 | ||||
| A. M. N. H. | 60508 | ♂ ad. | El Chiral, Ecuador | 45.5 | 17.1 | 13.8 | 11.5 | 14.5 | 24.2 | 23.2 | 15.0 | 7.0 | 2.3 | 5.9 | 5.6 | 6.0 | 4.8 | 2.6 | 16.4 | 13.8 |
| A. M. N. H. | 61406 | ♂ sad. | Guainche, Ecuador | 48.2 | 17.3 | 14.5 | 12.3 | 15.7 | 25.6 | 30.3 | 15.6 | 7.2 | 3.0 | 6.1 | 5.2 | 5.4 | 4.5 | 2.3 | 15.7 | 13.9 |
| N. H. R. S. | 2 | ♂ ad. | Panecillo, Ecuador | 48.0 | 16.8 | 13.3 | 14.8 | 14.5 | 7.0 | 6.0 | 5.4 | 5.7 | 4.3 | 2.2 | ||||||
| N. H. R. S. | 5 | ♂ ad. | San Antonio, Ecuador | 44.0 | 16.3 | 14.0 | 11.9 | 14.8 | 23.5 | 28.0 | 14.2 | 7.0 | 2.9 | 5.8 | 5.1 | 5.4 | 4.3 | 2.1 | 16.8 | 14.1 |
| N. H. R. S. | 7 | ♂ ad. | Carapungo, Ecuador | 46.8 | 17.4 | 13.5 | 11.1 | 14.9 | 23.7 | 29.0 | 14.6 | 7.0 | 2.9 | 6.2 | 5.8 | 6.2 | 4.5 | 2.3 | 16.0 | 14.2 |
| N. H. R. S. | 14 | ♂ ad. | Nára Papallacta, Ecuador | 45.4 | 17.3 | 14.1 | 12.6 | 15.8 | 24.5 | 29.9 | 14.3 | 7.1 | 2.6 | 6.1 | 5.4 | 5.6 | 4.3 | 2.5 | 17.5 | 9.5 |
| N. H. R. S. | 10 | ♀ ad. | Guapulo, Ecuador | 37.9 | 13.5 | 11.3 | 9.5 | 12.7 | 19.7 | 22.5 | 12.4 | 5.9 | 2.0 | 5.1 | 4.5 | 4.7 | 3.7 | 1.7 | 12.9 | 11.3 |
| Mustela frenata boliviensis | ||||||||||||||||||||
| A. M. N. H. | 72587 | ♂ ad. | Nequejahuira, Bolivia | 41.6 | 15.3 | 12.2 | 10.0 | 14.7 | 22.2 | 25.0 | 13.4 | 7.4 | 2.2 | 5.3 | 5.0 | 5.2 | 4.4 | 2.2 | 14.8 | 12.4 |
| B. Z. M. | 602 | ♂ yg. | Limbaní, Perú | 42.4 | 15.5 | 5 | .5 | 5.0 | 5.4 | 4.2 | 2.2 | |||||||||
| U. S. N. M. | 137513 | ♂ yg. | Limbaní, Perú | 40.3 | 15.2 | 5 | .1 | 4.8 | 5.1 | 3.9 | 1.9 | |||||||||
| Mustela africana africana | ||||||||||||||||||||
| A. M. N. H. | 374.75 | ♂ sad. | Pará, Brazil | 47.8 | 17.2 | 14.3 | 12.9 | 15.4 | 26.4 | 32.2 | 16.9 | 8.2 | 2.8 | 6.6 | 5.8 | 6.2 | 4.7 | 2.4 | 17.8 | 14.9 |
| B. M. 5. | 1.25.1. | ♂? sad. | Pará, Brazil | 44.9 | 16.4 | 13.0 | 10.2 | 14.2 | 23.3 | 26.8 | 14.4 | 7.0 | 2.7 | 5.9 | 5.4 | 5.5 | 4.5 | 1.8 | 16.4 | 13.7 |
| B. M. 26. | 1.8.10. | ♂? ad. | Pará, Brazil | 44.6 | 16.4 | 13.3 | 10.9 | 14.4 | 24.4 | 29.4 | 14.4 | 6.7 | 2.5 | 6.0 | 5.6 | 5.7 | 4.5 | 1.8 | 15.5 | 13.6 |
| Mustela africana stolzmanni | ||||||||||||||||||||
| B. M. 24.12 | .12.24. | ♀ ad. | Moyobamba, Perú | 45.8 | 16.9 | 13.5 | 12.0 | 14.6 | 23.5 | 28.8 | 14.5 | 6.2 | 2.6 | 5.9 | 5.4 | 4.9 | 4.3 | 1.8 | 16.5 | 13.9 |
| M. P. H. N. | 563 | ♀ ad. | Yurimaguas, Perú | 17.5± | 6.0 | 5.8 | 5.6 | 4.4 | 2.3 | |||||||||||
| A. M. N. H. | 61813 | ♀ yg. | Val. d. Perené, Perú | 44.6 | 16.0 | 13.0 | 11.0 | 15.4 | 24.1 | 28.9 | 15.7 | 7.0 | 2.8 | 6.2 | 5.6 | 5.9 | 4.5 | 2.2 | 16.8 | 13.4 |
| Mustela erminea arctica | ||||||||||||||||||||
| M. C. Z. | 10012 | ♂ ad. | Pt. Barrow | 43.3 | 15.9 | 15.2 | 12.0 | 16.2 | 23.5 | 27.6 | 15.5 | 8.4 | 5.6 | 5.1 | 5.1 | 3.9 | 2.0 | 13.3 | 13.2 | |
| F. M. N. H. | 35894 | ♂ ad. | Pt. Barrow | 41.8 | 15.6 | 15.3 | 13.3 | 16.0 | 23.2 | 27.6 | 15.1 | 8.5 | 6.0 | 5.2 | 5.3 | 4.0 | 2.2 | 14.1 | 13.5 | |
| A. N. S. P. | 6909 | ♂ ad. | Pt. Barrow | 42.3 | 15.1 | 14.8 | 11.6 | 15.3 | 22.9 | 26.2 | 15.2 | 8.4 | 5.9 | 5.1 | 5.3 | 4.1 | 2.4 | 14.5 | 12.5 | |
| A. N. S. P. | 6910 | ♂ ad. | Pt. Barrow | 16.2 | 16.0 | 12.7 | 16.7 | 5.7 | 5.4 | 5.7 | 4.3 | 2.1 | ||||||||
| N. M. C. | 2445 | ♂ ad. | Salirochet River | 42.8 | 15.7 | 15.4 | 13.0 | 16.0 | 23.5 | 28.5 | 15.2 | 8.0 | 5.6 | 5.0 | 5.2 | 4.0 | 2.0 | 14.3 | 13.3 | |
| av. 5 | 42.5 | 15.7 | 15.3 | 12.5 | 16.0 | 23.3 | 27.5 | 15.3 | 8.3 | 5.8 | 5.2 | 5.3 | 4.1 | 2.1 | 14.1 | 13.1 | ||||
| F. M. N. H. | 35895 | ♀ ad. | Pt. Barrow | 35.4 | 13.1 | 12.7 | 10.3 | 13.3 | 19.3 | 23.0 | 12.7 | 7.2 | 5.0 | 4.5 | 4.8 | 3.5 | 1.7 | 12.0 | 10.5 | |
| M. V. Z. | 43286 | ♀ ad. | Pt. Barrow | 35.4 | 12.9 | 12.0 | 10.0 | 13.2 | 18.8 | 21.8 | 12.6 | 6.7 | 4.6 | 4.2 | 4.3 | 3.2 | 1.8 | 11.4 | 10.3 | |
| U. S. N. M. | 243489 | ♀ sad. | Alatna River | 37.0 | 13.4 | 11.9 | 9.4 | 13.5 | 19.4 | 22.0 | 13.2 | 7.2 | 4.7 | 4.3 | 4.5 | 3.3 | 1.7 | 11.9 | 10.6 | |
| U. S. N. M. | 243493 | ♀ sad. | 16 mi. below Bettles | 36.2 | 13.0 | 12.0 | 9.4 | 13.0 | 19.1 | 21.8 | 12.9 | 7.2 | 5.0 | 4.2 | 4.5 | 3.3 | 1.8 | 11.9 | 10.3 | |
| U. S. N. M. | 180459 | ♀ ad. | N. Fk. Kuskokim | 35.3 | 12.9 | 11.2 | 8.8 | 12.8 | 18.9 | 20.7 | 12.7 | 7.0 | 4.7 | 4.3 | 4.5 | 3.3 | 1.8 | 10.5 | 9.7 | |
| U. S. N. M. | 242205 | ♀ ad. | Fairbanks | 34.5 | 12.4 | 11.5 | 9.3 | 13.0 | 18.0 | 12.7 | 6.8 | 4.8 | 4.3 | 4.5 | 3.3 | 1.7 | 11.8 | 10.2 | ||
| U. S. N. M. | 157306 | ♀ sad. | Bear Creek | 36.9 | 13.5 | 12.6 | 9.5 | 13.6 | 18.7 | 20.9 | 13.0 | 6.9 | 4.8 | 4.4 | 4.5 | 3.3 | 1.8 | 11.4 | 10.2 | |
| U. S. N. M. | 157305 | ♀ sad. | Bear Creek | 35.2 | 12.5 | 11.4 | 9.6 | 12.8 | 17.8 | 20.5 | 12.6 | 6.3 | 4.8 | 4.2 | 4.4 | 3.3 | 1.6 | 11.2 | 10.1 | |
| av. 8 | 35.8 | 12.9 | 11.8 | 9.3 | 13.1 | 18.7 | 21.2 | 12.9 | 6.9 | 4.8 | 4.3 | 4.5 | 3.3 | 1.7 | 11.5 | 10.2 | ||||
| Mustela erminea polaris | ||||||||||||||||||||
| M. C. Z. | 29831 | ♂ ad. | Ymer Is. | 41.6 | 15.6 | 15.3 | 12.0 | 13.0 | 22.3 | 26.1 | 14.8 | 8.0 | 5.8 | 5.2 | 5.7 | 4.1 | 2.2 | 13.7 | 12.3 | |
| C. Z. M. | 1245 | ♂ ad. | Danmarks Havn. | 40.7 | 15.0 | 14.7 | 11.9 | 14.9 | 22.3 | 25.5 | 14.6 | 7.8 | 5.9 | 5.1 | 5.6 | 3.9 | 2.0 | 13.4 | 12.2 | |
| C. Z. M. | 1246 | ♂ ad. | Danmarks Havn. | 39.0 | 14.1 | 13.8 | 10.9 | 14.2 | 21.3 | 24.7 | 14.5 | 7.3 | 5.3 | 4.8 | 5.0 | 3.7 | 1.9 | 12.0 | 11.2 | |
| C. Z. M. | 1247 | ♂ ad. | Danmarks Havn. | 41.0 | 15.2 | 14.5 | 11.5 | 14.4 | 22.3 | 26.7 | 14.4 | 7.6 | 5.7 | 5.1 | 5.5 | 4.1 | 2.0 | 13.1 | 12.1 | |
| C. Z. M. | 1248 | ♂ ad. | Danmarks Havn. | 42.3 | 15.5 | 15.5 | 12.0 | 15.3 | 23.2 | 27.9 | 15.1 | 8.1 | 5.8 | 5.5 | 5.8 | 4.0 | 2.3 | 13.8 | 12.8 | |
| C. Z. M. | 1249 | ♂ ad. | Danmarks Havn. | 42.2 | 15.8 | 15.0 | 12.2 | 15.1 | 23.2 | 28.0 | 15.2 | 8.1 | 5.9 | 5.2 | 5.7 | 4.2 | 2.3 | 13.7 | 12.4 | |
| C. Z. M. | 1871 | ♂ ad. | Scoresby Sd. | 42.4 | 15.3 | 15.3 | 12.3 | 15.6 | 15.4 | 8.3 | 5.7 | 5.1 | 5.3 | 3.9 | 2.0 | 14.4 | 12.8 | |||
| av. 7 | 41.3 | 15.2 | 14.9 | 11.8 | 14.9 | 22.4 | 26.5 | 14.9 | 7.9 | 5.7 | 5.2 | 5.5 | 4.0 | 2.1 | 13.4 | 12.3 | ||||
| C. Z. M. | 1060 | ♀ ad. | Turner Sd. | 35.9 | 13.0 | 12.5 | 10.1 | 12.9 | 19.6 | 22.6 | 13.6 | 7.1 | 5.0 | 4.5 | 4.7 | 3.6 | 1.7 | 12.3 | 10.3 | |
| B. Z. M. | 43965 | ♀ ad. | Kap Hoegh | 37.8 | 13.4 | 12.7 | 10.4 | 13.2 | 19.3 | 22.4 | 13.9 | 7.0 | 4.8 | 4.2 | 4.5 | 3.7 | 1.8 | 12.1 | 10.9 | |
| Mustela erminea semplei | ||||||||||||||||||||
| C. M. | 6688 | ♂ ad. | Prairie Point | *39.9 | 14.5 | 13.5 | 11.5 | 14.6 | 21.5 | 24.8 | 14.1 | 7.9 | 5.0 | 4.6 | 4.7 | 3.6 | 2.0 | 14.0 | 11.6 | |
| average} | ♂ 1 ad. and 10 sad . | Southampton Isl. | {37.5 | 13.8 | 13.1 | 10.6 | 13.8 | 20.2 | 23.6 | 13.6 | 7.2 | 5.1 | 4.7 | 4.9 | 3.6 | 2.0 | 13.4 | 11.0 | ||
| maximum} | {39.9 | 14.5 | 13.5 | 11.5 | 14.8 | 21.5 | 24.8 | 15.0 | 7.9 | 5.3 | 4.9 | 5.0 | 3.7 | 2.3 | 14.0 | 11.6 | ||||
| minimum} | {35.7 | 13.1 | 12.6 | 9.7 | 12.7 | 19.2 | 22.2 | 12.9 | 6.4 | 4.8 | 4.4 | 4.5 | 3.4 | 1.8 | 12.3 | 10.5 | ||||
| C. M. | 8474 | ♀ ad. | Minnimunnek Pt. | 32.6 | 11.9 | 11.3 | 9.2 | 12.2 | 18.5 | 21.2 | 12.2 | 6.6 | 4.4 | 3.9 | 4.2 | 3.0 | 1.5 | 11.9 | 9.8 | |
| average} | ♀ 1 ad. and 4 sad . | Southampton Isl. | {34.2 | 12.4 | 11.7 | 9.5 | 12.6 | 18.8 | 20.9 | 12.8 | 6.7 | 4.5 | 4.1 | 4.3 | 3.1 | 1.6 | 12.1 | 10.1 | ||
| maximum} | {35.1 | 13.0 | 12.7 | 9.9 | 13.1 | 19.4 | 21.5 | 13.4 | 7.0 | 4.3 | 4.3 | 4.5 | 3.3 | 1.7 | 12.4 | 10.4 | ||||
| minimum} | {32.6 | 11.9 | 11.3 | 9.2 | 12.2 | 18.1 | 20.1 | 12.2 | 6.6 | 4.6 | 3.9 | 4.2 | 3.0 | 1.5 | 11.9 | 9.6 | ||||
| Mustela erminea kadiacensis | ||||||||||||||||||||
| U. S. N. M. | 107496 | ♂ ad. | Kadiak | 43.2 | 15.4 | 14.9 | 11.1 | 14.9 | 22.1 | 26.0 | 15.1 | 7.6 | 5.5 | 5.1 | 5.1 | 3.8 | 2.0 | 14.0 | 12.2 | |
| F. M. N. H. | 7290 | ♂ ad. | Kadiak Id. | 42.1 | 14.9 | 14.0 | 10.9 | 14.4 | 21.6 | 26.0 | 14.1 | 7.1 | 5.7 | 5.2 | 5.2 | 3.8 | 1.7 | 13.5 | 11.5 | |
| U. S. N. M. | 98042 | ♀ ad. | Kadiak | 33.0 | 12.0 | 10.2 | 8.0 | 11.3 | 16.9 | 19.4 | 11.5 | 6.0 | 4.4 | 4.0 | 4.0 | 2.8 | 1.4 | 11.5 | 8.9 | |
| Mustela erminea richardsonii | ||||||||||||||||||||
| average} | ♂ ad. 6 | 3 mi. S Big Isl. | {40.9 | 14.3 | 12.5 | 10.2 | 13.5 | 21.2 | 24.2 | 14.9 | 7.9 | 5.3 | 4.7 | 4.8 | 3.8 | 2.1 | 13.0 | 11.9 | ||
| maximum} | {43.7 | 15.0 | 13.2 | 11.1 | 14.0 | 22.2 | 25.5 | 15.5 | 8.3 | 6.0 | 5.0 | 5.2 | 4.2 | 2.3 | 14.2 | 11.4 | ||||
| minimum} | {39.6 | 13.8 | 11.7 | 9.4 | 12.9 | 20.0 | 22.9 | 14.2 | 7.2 | 4.9 | 4.5 | 4.6 | 3.7 | 2.0 | 12.5 | 12.3 | ||||
| average} | ♂ sad. 7 | 3 mi. S Big Isl. | {40.2 | 14.1 | 12.2 | 9.8 | 14.0 | 20.6 | 23.3 | 14.6 | 7.7 | 5.2 | 4.7 | 4.9 | 3.7 | 2.1 | 13.6 | 11.8 | ||
| maximum} | {41.5 | 14.7 | 12.6 | 10.4 | 14.2 | 21.5 | 24.4 | 15.1 | 8.2 | 5.5 | 4.9 | 5.0 | 3.8 | 2.5 | 14.5 | 12.2 | ||||
| minimum} | {38.4 | 13.6 | 12.0 | 9.5 | 13.7 | 20.0 | 22.0 | 13.7 | 7.0 | 5.0 | 4.5 | 4.7 | 3.5 | 2.0 | 12.8 | 11.4 | ||||
| U. S. N. M. | 136112 | ♀ ad. | Willow River | 33.0 | 11.5 | 8.8 | 7.6 | 11.4 | 15.9 | 18.0 | 12.5 | 5.7 | 4.0 | 3.8 | 3.9 | 2.7 | 1.5 | 11.6 | 8.9 | |
| M. C. Z. | 242866 | ♀ sad. | 3 mi. S Big Isl. | 32.3 | 11.0 | 9.0 | 7.2 | 11.7 | 16.6 | 17.7 | 12.3 | 6.5 | 4.0 | 3.7 | 3.9 | 3.0 | 1.5 | 10.0 | 8.8 | |
| U. S. N. M. | 129703 | ♀ ad. | Ft. Resolution | 34.2 | 12.0 | 10.4 | 8.2 | 11.5 | 17.2 | 19.7 | 12.3 | 6.5 | 4.2 | 4.0 | 4.2 | 3.2 | 1.7 | 12.0 | 9.6 | |
| U. S. N. M. | 110682 | ♀ sad. | 15 mi. above Smith Landing | 33.8 | 11.3 | 9.8 | 7.4 | 11.0 | 16.5 | 18.3 | 12.1 | 6.0 | 4.2 | 3.8 | 3.9 | 2.9 | 1.5 | 11.2 | 9.3 | |
| U. S. N. M. | 235959 | ♀ ad. | Athabasca Delta | 33.1 | 11.7 | 9.3 | 7.3 | 11.0 | 15.7 | 18.1 | 11.6 | 5.6 | 4.0 | 3.8 | 3.8 | 3.0 | 1.6 | 10.5 | 8.6 | |
| M. C. Z. | 18776 | ♀ ad. | Athabasca Delta | 31.5 | 10.8 | 8.9 | 7.6 | 11.2 | 16.3 | 18.3 | 11.6 | 6.3 | 3.8 | 3.7 | 3.8 | 3.1 | 1.6 | 11.0 | 8.7 | |
| av. 6 | 33.0 | 11.4 | 9.4 | 7.6 | 11.3 | 16.4 | 18.3 | 12.1 | 6.3 | 4.0 | 3.8 | 3.9 | 3.0 | 1.6 | 11.1 | 9.0 | ||||
| Mustela erminea cicognanii | ||||||||||||||||||||
| M. V. Z. | 53789 | ♂ ad. | Ogdensburg, N. Y. | 36.6 | 12.9 | 11.5 | 9.1 | 13.2 | 18.5 | 21.7 | 13.3 | 7.0 | 4.7 | 4.4 | 4.5 | 3.4 | 1.8 | 12.3 | 10.4 | |
| U. S. N. M. | 32240/4406 | 6 ♂ sad. | Amsterdam, N. Y. | 36.7 | 12.9 | 11.5 | 9.4 | 13.3 | 19.1 | 21.5 | 13.7 | 6.9 | 4.6 | 4.4 | 4.5 | 3.4 | 2.0 | 13.2 | 10.8 | |
| A. M. N. H. | 67869 | ♂ ad. | Berlin, N. Y. | 36.2 | 12.5 | 10.2 | 8.9 | 12.7 | 18.8 | 20.6 | 12.9 | 6.7 | 4.3 | 3.8 | 4.0 | 3.3 | 1.9 | 11.5 | 10.0 | |
| A. M. N. H. | 67868 | ♂ ad. | Berlin, N. Y. | 34.8 | 12.1 | 9.8 | 8.2 | 12.7 | 17.4 | 19.8 | 12.3 | 6.3 | 4.5 | 3.9 | 4.1 | 2.9 | 1.7 | 11.5 | 9.7 | |
| A. M. N. H. | 15841 | ♂ ad. | Schoharie, N. Y. | 33.8 | 11.7 | 9.7 | 7.7 | 11.9 | 17.3 | 20.0 | 11.8 | 6.4 | 4.2 | 3.8 | 4.0 | 2.9 | 1.5 | 11.8 | 9.5 | |
| Cornell | 494 | ♂ sad. | Cascadilla Creek, N. Y. | 34.8 | 12.2 | 10.4 | 8.3 | 12.5 | 17.9 | 19.3 | 12.4 | 6.6 | 4.4 | 4.1 | 4.2 | 3.0 | 1.6 | 12.2 | 10.3 | |
| C. M. | 7461 | ♂ sad. | Pymatuning Swamp | 34.2 | 12.0 | 9.6 | 8.1 | 12.8 | 17.3 | 19.0 | 11.9 | 6.4 | 4.3 | 3.9 | 4.2 | 3.0 | 1.9 | 11.5 | 9.2 | |
| C. M. | 10264 | ♂ sad. | 3-1/2 mi. W Linesville | 37.6 | 13.2 | 11.0 | 8.9 | 12.7 | 18.8 | 20.6 | 12.9 | 7.2 | 4.9 | 4.3 | 4.5 | 3.5 | 2.0 | 12.6 | 10.6 | |
| M. V. Z. | 53788 | ♂ sad. | Lopez, Penn. | 36.5 | 12.6 | 10.7 | 8.6 | 12.6 | 18.6 | 20.3 | 13.1 | 6.8 | 4.5 | 4.1 | 4.3 | 3.2 | 1.6 | 10.9 | 9.9 | |
| av. 9 | 35.7 | 12.5 | 10.5 | 8.6 | 12.7 | 18.2 | 20.3 | 12.7 | 6.7 | 4.5 | 4.1 | 4.3 | 3.2 | 1.8 | 11.9 | 10.0 | ||||
| U. S. N. M. | 135570 | ♀ ad. | Lake George | 33.3 | 11.8 | 9.3 | 7.8 | 11.4 | 11.8 | 5.8 | 4.4 | 3.8 | 4.0 | 3.0 | 1.8 | 9.1 | ||||
| B. S. N. | 994 | ♀ sad. | Cattaraugus | 31.4 | 10.8 | 9.2 | 7.2 | 10.0 | 15.3 | 10.7 | 5.7 | 3.8 | 3.7 | 3.6 | 2.7 | 1.5 | 10.3 | 8.2 | ||
| C. M. | 7460 | ♀ sad. | Pymatuning Swamp | 32.0 | 10.9 | 9.2 | 7.5 | 11.0 | 15.9 | 17.5 | 11.1 | 5.9 | 3.7 | 3.4 | 3.6 | 2.8 | 1.5 | 10.3 | 8.2 | |
| C. M. | 10252 | ♀ sad. | 3 mi. NW Linesville | 32.7 | 11.2 | 9.5 | 7.6 | 11.3 | 16.0 | 18.0 | 11.8 | 6.2 | 3.9 | 3.6 | 3.7 | 3.1 | 1.7 | 10.9 | 8.8 | |
| av. 4 | 32.4 | 11.2 | 9.4 | 7.5 | 10.9 | 15.7 | 17.8 | 11.4 | 5.9 | 3.9 | 3.6 | 3.7 | 2.9 | 1.6 | 10.5 | 8.6 | ||||
| Mustela erminea bangsi | ||||||||||||||||||||
| F. M. N. H. | 18134 | ♂ ad. | Aitkin, Minn. | 40.3 | 14.1 | 12.7 | 10.6 | 14.6 | 20.6 | 23.5 | 14.3 | 7.7 | 4.9 | 4.6 | 4.9 | 3.6 | 1.9 | 13.3 | 11.8 | |
| F. M. N. H. | 18135 | ♂ ad. | Aitkin, Minn. | 14.1 | 12.5 | 10.4 | 14.4 | 24.3 | 5.3 | 4.5 | 4.6 | 3.7 | 2.1 | 12.2 | ||||||
| F. M. N. H. | 18130 | ♂ ad. | Aitkin, Minn. | 40.7 | 15.0 | 12.2 | 9.7 | 14.6 | 20.2 | 22.8 | 14.6 | 7.5 | 5.2 | 5.0 | 5.1 | 3.8 | 2.6 | 13.5 | 12.0 | |
| F. M. N. H. | 18133 | ♂ ad. | Aitkin, Minn. | 39.3 | 13.9 | 11.2 | 9.0 | 13.0 | 19.5 | 22.0 | 14.3 | 7.2 | 5.1 | 4.5 | 4.6 | 3.5 | 2.0 | 13.0 | 11.2 | |
| F. M. N. H. | 18131 | ♂ ad. | Aitkin, Minn. | 38.5 | 13.4 | 11.2 | 9.0 | 12.6 | 18.8 | 21.0 | 13.7 | 7.3 | 4.8 | 4.4 | 4.5 | 3.4 | 1.8 | 12.0 | 10.5 | |
| F. M. N. H. | 7222 | ♂ ad. | Aitkin, Minn. | 13.6 | 12.4 | 9.5 | 13.3 | 14.4 | 7.9 | 4.8 | 4.4 | 4.4 | 3.5 | 1.8 | 11.2 | |||||
| F. M. N. H. | 18127 | ♂ sad. | Aitkin, Minn. | 38.4 | 13.1 | 11.1 | 9.0 | 13.0 | 19.5 | 22.0 | 14.3 | 6.9 | 4.6 | 4.5 | 4.5 | 3.5 | 1.9 | 13.4 | 11.0 | |
| F. M. N. H. | 18129 | ♂ sad. | Aitkin, Minn. | 39.4 | 13.7 | 11.1 | 9.1 | 13.3 | 19.0 | 21.6 | 13.6 | 7.2 | 4.7 | 4.3 | 4.5 | 3.4 | 2.0 | 12.8 | 11.1 | |
| F. M. N. H. | 18132 | ♂ sad. | Aitkin, Minn. | 39.3 | 14.3 | 11.5 | 9.5 | 13.2 | 20.2 | 22.8 | 14.3 | 7.2 | 4.9 | 4.7 | 4.7 | 3.5 | 2.1 | 12.5 | 11.2 | |
| F. M. N. H. | 18441 | ♂ sad. | Aitkin, Minn. | 40.2 | 14.0 | 12.0 | 9.9 | 14.6 | 19.6 | 22.6 | 15.0 | 7.1 | 5.2 | 4.7 | 4.8 | 3.7 | 2.0 | 13.3 | 11.5 | |
| F. M. N. H. | 18440 | ♂ sad. | Aitkin, Minn. | 38.8 | 13.8 | 11.3 | 9.3 | 13.2 | 19.7 | 22.0 | 13.8 | 7.6 | 5.1 | 4.5 | 4.7 | 3.7 | 2.0 | 13.0 | 11.1 | |
| F. M. N. H. | 7219 | ♂ sad. | Aitkin, Minn. | 13.9 | 11.9 | 10.0 | 13.5 | 24.7 | 14.6 | 7.3 | 5.3 | 4.6 | 4.8 | 4.0 | 2.1 | 11.8 | ||||
| av. 12 | 39.4 | 13.9 | 11.8 | 9.6 | 13.6 | 19.7 | 22.7 | 14.3 | 7.4 | 5.0 | 4.6 | 4.7 | 3.6 | 2.0 | 13.0 | 11.4 | ||||
| average} | ♂ ad. 5 and sad. 5 | Elk River | {37.9 | 13.2 | 11.4 | 9.1 | 13.1 | 19.3 | 21.6 | 14.4 | 7.2 | 4.7 | 4.3 | 4.4 | 3.4 | 1.9 | 12.7 | 10.8 | ||
| maximum} | {39.5 | 13.9 | 12.6 | 9.8 | 14.2 | 20.5 | 22.8 | 15.3 | 8.0 | 5.1 | 4.4 | 4.6 | 3.6 | 2.0 | 14.0 | 11.7 | ||||
| minimum} | {34.8 | 12.0 | 10.5 | 8.5 | 12.2 | 18.0 | 20.8 | 12.4 | 6.3 | 4.3 | 4.1 | 4.1 | 3.1 | 1.7 | 12.0 | 10.0 | ||||
| Walker | 377 | ♀ ad. | Deer | 31.8 | 11.0 | 9.0 | 7.5 | 10.9 | 16.6 | 18.4 | 11.7 | 5.9 | 4.2 | 3.7 | 3.8 | 2.8 | 1.6 | 11.2 | 9.3 | |
| Walker | 11 | ♀ ad. | Grand Maris | 32.7 | 10.8 | 11.0 | 15.9 | 17.3 | 12.2 | 5.6 | 3.9 | 3.8 | 3.8 | 2.9 | 1.6 | 10.7 | 8.5 | |||
| Wisc. U. | 8681 | ♀ ad. | T. 61N, R. 26W | 32.9 | 10.8 | 9.2 | 7.4 | 11.3 | 15.8 | 16.8 | 12.3 | 6.2 | 3.8 | 3.6 | 3.6 | 2.8 | 1.4 | 10.0 | 8.3 | |
| Wisc. U. | 8679 | ♀ ad. | T. 61N, R. 26W | 33.6 | 11.6 | 9.8 | 7.6 | 12.2 | 15.7 | 17.1 | 11.9 | 5.7 | 4.0 | 3.6 | 3.7 | 3.0 | 1.6 | 9.6 | 8.9 | |
| Walker | A 58 | ♀ ad. | Elk River | 32.8 | 11.5 | 9.9 | 8.1 | 11.3 | 16.8 | 18.3 | 11.8 | 6.4 | 4.0 | 3.8 | 4.0 | 2.9 | 1.7 | 10.5 | 9.2 | |
| av. 5 | 32.8 | 11.1 | 9.5 | 7.7 | 11.3 | 16.2 | 17.6 | 12.0 | 6.0 | 4.0 | 3.7 | 3.9 | 2.9 | 1.6 | 10.4 | 8.8 | ||||
| Wisc. U. | 8691 | ♀ ad. | Fisher Lake | 31.5 | 10.6 | 9.5 | 7.7 | 10.8 | 15.8 | 17.4 | 11.2 | 5.6 | 3.8 | 3.5 | 3.7 | 2.6 | 1.4 | 10.4 | 8.5 | |
| Wisc. U. | 8674 | ♀ ad. | Gordon | 32.8 | 11.5 | 9.8 | 7.2 | 11.4 | 15.7 | 16.7 | 11.6 | 5.7 | 4.2 | 3.8 | 3.8 | 2.8 | 1.6 | 9.6 | 8.9 | |
| Snyder | 2637 | ♀ ad. | Beaver Dam | 32.9 | 11.0 | 9.8 | 7.8 | 11.7 | 16.9 | 18.9 | 12.2 | 6.4 | 4.1 | 3.8 | 3.8 | 3.2 | 1.5 | 11.3 | 9.7 | |
| Snyder | 993 | ♀ ad. | Beaver Dam | 34.1 | 11.3 | 9.4 | 7.5 | 11.8 | 16.7 | 18.0 | 12.8 | 6.1 | 4.4 | 3.8 | 3.8 | 3.0 | 1.7 | 10.8 | 9.0 | |
| Snyder | 2999 | ♀ ad. | Beaver Dam | 31.9 | 10.8 | 9.2 | 7.4 | 11.2 | 15.9 | 17.5 | 12.4 | 5.8 | 4.0 | 3.6 | 3.8 | 3.0 | 1.6 | 11.4 | 9.2 | |
| av. 5 | 32.6 | 11.0 | 9.5 | 7.5 | 11.4 | 16.2 | 17.7 | 12.0 | 5.9 | 4.1 | 3.7 | 3.8 | 2.9 | 1.6 | 10.7 | 9.1 | ||||
| Mustela erminea invicta | ||||||||||||||||||||
| average} | ♂ ad. 5 | Idaho Co | {37.0 | 12.8 | 11.4 | 9.1 | 13.1 | 19.0 | 21.3 | 13.6 | 7.0 | 4.5 | 4.1 | 4.2 | 3.2 | 1.7 | 11.7 | 10.5 | ||
| maximum} | {39.8 | 14.1 | 13.0 | 10.0 | 14.2 | 19.7 | 22.6 | 14.2 | 7.1 | 4.9 | 4.3 | 4.5 | 3.5 | 1.9 | 12.3 | 11.3 | ||||
| minimum} | {35.8 | 12.2 | 10.6 | 8.6 | 12.0 | 18.2 | 20.5 | 13.3 | 6.8 | 4.2 | 4.0 | 4.0 | 3.1 | 1.4 | 10.9 | 9.7 | ||||
| M. V. Z. | 90763 | ♀ ad. | Pilot Creek, Idaho | 31.6 | 10.8 | 9.2 | 7.0 | 10.5 | 15.6 | 16.5 | 10.9 | 5.6 | 4.0 | 3.6 | 3.7 | 2.9 | 1.4 | 9.0 | 8.2 | |
| average} | ♀ ad. 1 and sad. 4 | Idaho Co | {32.2 | 10.6 | 9.0 | 7.1 | 11.1 | 16.3 | 17.2 | 12.0 | 5.9 | 3.9 | 3.7 | 3.7 | 2.8 | 1.4 | 9.5 | 8.3 | ||
| maximum} | {32.8 | 11.2 | 9.2 | 7.2 | 12.2 | 17.0 | 17.8 | 12.7 | 6.5 | 4.3 | 3.9 | 3.9 | 2.9 | 1.5 | 10.0 | 8.7 | ||||
| minimum} | {31.6 | 10.8 | 8.5 | 7.0 | 10.5 | 15.6 | 16.5 | 10.9 | 5.6 | 3.5 | 3.6 | 3.6 | 2.7 | 1.3 | 9.0 | 8.0 | ||||
| Mustela erminea alascensis | ||||||||||||||||||||
| average} | ♂ ad. 8 | Windham | {37.5 | 13.1 | 11.5 | 9.4 | 13.2 | 19.4 | 21.9 | 13.2 | 6.9 | 4.8 | 4.2 | 4.4 | 3.5 | 1.9 | 12.5 | 10.7 | ||
| maximum} | {38.9 | 13.7 | 12.3 | 10.1 | 14.3 | 20.5 | 23.7 | 13.7 | 7.4 | 5.0 | 4.4 | 4.7 | 3.9 | 2.2 | 13.8 | 11.6 | ||||
| minimum} | {36.5 | 12.3 | 11.0 | 8.6 | 12.0 | 18.5 | 20.4 | 12.9 | 6.6 | 4.5 | 4.1 | 4.2 | 3.3 | 1.8 | 11.8 | 10.4 | ||||
| U. S. N. M. | 74422 | ♀ ad. | Juneau | 33.2 | 11.4 | 10.5 | 8.3 | 10.5 | 16.2 | 18.3 | 11.5 | 5.7 | 3.9 | 3.5 | 3.7 | 2.9 | 1.6 | 10.0 | 8.7 | |
| M. V. Z. | 995 | ♀ ad. | Juneau | 33.1 | 11.3 | 9.4 | 7.8 | 11.3 | 16.3 | 17.8 | 12.0 | 5.8 | 3.9 | 3.7 | 3.8 | 2.9 | 1.7 | 8.7 | ||
| M. V. Z. | 473 | ♀ ad. | Helm Bay | 32.9 | 11.2 | 9.5 | 8.5 | 11.6 | 16.3 | 17.6 | 11.9 | 6.0 | 4.0 | 3.7 | 3.9 | 2.6 | 1.4 | 10.5 | 9.3 | |
| U. S. N. M. | 74773 | ♀ ad. | Wrangel | 32.2 | 11.3 | 9.2 | 7.5 | 16.0 | 17.6 | 11.9 | 5.8 | 4.0 | 3.8 | 4.0 | 3.2 | 1.7 | 11.5 | 8.7 | ||
| M. V. Z. | 78243 | ♀ sad. | Windham | 31.9 | 11.1 | 10.1 | 7.7 | 11.5 | 16.8 | 18.0 | 11.5 | 5.7 | 4.0 | 3.6 | 3.8 | 3.1 | 1.7 | 10.6 | 8.8 | |
| av. 5 | 32.7 | 11.3 | 9.7 | 8.0 | 11.2 | 16.3 | 17.9 | 11.8 | 5.8 | 4.0 | 3.7 | 3.8 | 2.9 | 1.6 | 10.6 | 8.8 | ||||
| Mustela erminea salva | ||||||||||||||||||||
| average} | ♂ ad. to sad. 12 | Mole Harbor | {37.8 | 13.0 | 11.9 | 9.6 | 13.3 | 19.2 | 22.0 | 12.8 | 6.8 | 4.6 | 4.3 | 4.4 | 3.5 | 1.8 | 11.7 | 10.7 | ||
| maximum} | {39.5 | 13.7 | 13.0 | 10.8 | 14.2 | 20.0 | 23.2 | 13.8 | 7.2 | 5.0 | 4.6 | 4.8 | 3.9 | 2.0 | 13.1 | 11.4 | ||||
| minimum} | {36.4 | 12.5 | 10.7 | 8.4 | 12.4 | 18.0 | 20.4 | 12.0 | 6.2 | 4.4 | 4.0 | 4.0 | 3.1 | 1.7 | 11.2 | 10.0 | ||||
| average} | ♀ ad. 2 and sad. 4 | Mole Harbor | {33.0 | 11.3 | 9.9 | 8.1 | 11.6 | 16.5 | 18.2 | 11.5 | 5.8 | 4.0 | 3.8 | 3.8 | 3.0 | 1.5 | 10.1 | 9.1 | ||
| maximum} | {33.6 | 11.9 | 10.2 | 8.7 | 12.3 | 17.1 | 18.7 | 12.0 | 6.2 | 4.2 | 3.9 | 3.9 | 3.2 | 1.6 | 10.9 | 9.4 | ||||
| minimum} | {32.0 | 10.9 | 9.5 | 7.5 | 11.1 | 15.4 | 17.1 | 11.0 | 5.3 | 3.8 | 3.6 | 3.6 | 2.9 | 1.5 | 8.9 | 8.4 | ||||
| Mustela erminea initis | ||||||||||||||||||||
| M. V. Z. | 289 | ♂ ad. | Saook Bay | 40.5 | 13.9 | 12.8 | 10.6 | 14.4 | 22.1 | 24.5 | 14.8 | 7.6 | 5.2 | 4.7 | 5.0 | 4.1 | 1.9 | 12.8 | 11.0 | |
| M. V. Z. | 286 | ♂ ad. | Saook Bay | 39.6 | 13.5 | 13.1 | 11.4 | 15.0 | 21.0 | 24.3 | 13.6 | 7.6 | 5.0 | 4.7 | 4.9 | 3.6 | 2.1 | 12.3 | 12.0 | |
| Mustela erminea celenda | ||||||||||||||||||||
| average} | ♂ ad. 5 | Prince of Wales Id | {39.5 | 14.0 | 13.6 | 11.5 | 14.7 | 20.9 | 24.2 | 13.6 | 7.5 | 5.1 | 4.7 | 4.8 | 3.7 | 1.9 | 12.9 | 11.6 | ||
| maximum} | {40.7 | 14.4 | 14.5 | 12.1 | 15.6 | 21.7 | 25.8 | 14.2 | 7.9 | 5.1 | 4.9 | 4.9 | 3.9 | 2.2 | 13.6 | 12.5 | ||||
| minimum} | {38.9 | 13.9 | 13.1 | 10.9 | 13.8 | 19.9 | 23.2 | 13.2 | 7.0 | 5.0 | 4.6 | 4.6 | 3.6 | 1.7 | 12.3 | 10.8 | ||||
| average} | ♂ ad. 5 and sad. 1 5 | Prince of Wales Id | {38.7 | 13.6 | 13.2 | 11.2 | 14.4 | 20.3 | 23.3 | 13.2 | 7.3 | 5.0 | 4.6 | 4.7 | 3.6 | 1.8 | 12.9 | 11.4 | ||
| maximum} | {40.9 | 14.4 | 14.5 | 12.1 | 15.6 | 21.7 | 25.8 | 14.2 | 7.9 | 5.3 | 5.0 | 4.9 | 3.9 | 2.2 | 13.6 | 12.5 | ||||
| minimum} | {36.7 | 13.0 | 11.8 | 10.2 | 13.4 | 19.0 | 21.3 | 12.3 | 6.8 | 4.6 | 4.3 | 4.2 | 3.3 | 1.6 | 12.1 | 10.8 | ||||
| M. V. Z. | 31223 | ♀ sad. | Prince of Wales Id | 12.2 | 11.5 | 9.8 | 12.6 | 4.5 | 4.2 | 4.2 | 3.2 | 1.5 | 10.0 | |||||||
| Mustela erminea seclusa | ||||||||||||||||||||
| M. V. Z. | 31232 | ♂ ad. | Suemez Id | 34.3 | 12.6 | 12.6 | 10.6 | 13.9 | 20.2 | 22.7 | 12.7 | 6.9 | 5.1 | 4.7 | 5.0 | 3.8 | 1.8 | 12.3 | 11.5 | |
| Mustela erminea haidarum | ||||||||||||||||||||
| U. S. N. M. | 94430 | ♂ ad. | Massett | 36.7 | 13.4 | 12.7 | 10.5 | 13.9 | 19.3 | 22.6 | 12.4 | 6.4 | 5.0 | 4.3 | 4.5 | 3.3 | 1.9 | 12.4 | 11.4 | |
| average} | ♂ ad. 7 | Graham Id | {36.7 | 13.4 | 12.7 | 10.9 | 14.3 | 18.9 | 21.8 | 12.6 | 6.8 | 5.0 | 4.3 | 4.6 | 3.4 | 1.7 | 12.6 | 11.3 | ||
| maximum} | {37.5 | 13.6 | 12.9 | 11.2 | 14.8 | 19.6 | 22.4 | 13.0 | 7.1 | 4.8 | 4.4 | 4.7 | 3.4 | 1.9 | 13.1 | 11.7 | ||||
| minimum} | {35.6 | 13.0 | 12.2 | 10.5 | 14.0 | 18.0 | 21.1 | 12.3 | 6.4 | 5.1 | 4.2 | 4.4 | 3.3 | 1.6 | 12.1 | 10.8 | ||||
| M. V. Z. | 31209 | ♀ ad. | Massett | 34.2 | 12.5 | 11.3 | 9.8 | 13.3 | 17.3 | 19.8 | 11.5 | 6.1 | 4.7 | 4.1 | 4.2 | 3.0 | 1.5 | 11.5 | 10.2 | |
| U. S. N. M. | 100624 | ♀ ad. | Cumsheva Inlet | 34.2 | 12.3 | 11.5 | 9.8 | 13.1 | 17.0 | 19.8 | 11.8 | 6.1 | 4.6 | 4.0 | 4.3 | 3.2 | 1.7 | 11.2 | 10.0 | |
| Mustela erminea anguinae | ||||||||||||||||||||
| average} | ♂ 13 ad. | Vancouver Id | {34.0 | 11.7 | 10.8 | 9.0 | 12.0 | 17.1 | 19.3 | 11.9 | 6.1 | 4.3 | 3.8 | 4.0 | 3.1 | 1.7 | 11.6 | 10.0 | ||
| maximum} | {35.6 | 12.2 | 11.3 | 9.6 | 12.5 | 17.9 | 20.6 | 12.5 | 6.7 | 4.6 | 4.0 | 4.1 | 3.3 | 1.9 | 12.5 | 10.5 | ||||
| minimum} | {32.5 | 11.0 | 10.1 | 8.5 | 11.3 | 16.5 | 18.8 | 11.2 | 5.7 | 4.0 | 3.6 | 3.7 | 2.9 | 1.6 | 10.7 | 9.8 | ||||
| average} | ♀ 5 ad. | Vancouver Id | {31.5 | 10.9 | 9.8 | 8.2 | 11.5 | 15.8 | 17.5 | 10.8 | 5.5 | 4.0 | 3.6 | 3.8 | 2.9 | 1.6 | 10.5 | 9.3 | ||
| maximum} | {31.8 | 11.1 | 10.0 | 8.8 | 12.4 | 16.1 | 17.8 | 11.1 | 5.7 | 4.2 | 3.8 | 4.0 | 3.0 | 1.6 | 11.5 | 10.0 | ||||
| minimum} | {30.9 | 10.5 | 9.6 | 7.9 | 10.6 | 15.6 | 17.3 | 10.4 | 5.4 | 3.8 | 3.5 | 3.6 | 2.8 | 1.5 | 10.0 | 8.9 | ||||
| Mustela erminea fallenda | ||||||||||||||||||||
| average} | ♂ ad. 7 | Topotypes | {35.7 | 12.6 | 11.1 | 9.2 | 12.5 | 18.3 | 20.8 | 13.1 | 6.8 | * | *4.7 | **4.2 | **4.4 | **3.4 | **1.9 | 11.7 | 10.5 | |
| maximum} | {38.2 | 13.1 | 11.6 | 9.9 | 13.0 | 19.6 | 22.8 | 14.1 | 7.6 | 5.1 | 4.6 | 4.7 | 3.6 | 2.1 | 12.4 | 11.0 | ||||
| minimum} | {34.3 | 12.0 | 10.5 | 8.3 | 12.0 | 17.0 | 19.4 | 12.2 | 6.2 | 4.3 | 3.9 | 3.9 | 3.2 | 1.7 | 11.0 | 10.0 | ||||
| N. M. C. | 7284 | ♀ ad. | Topotype | 29.4 | 10.1 | 9.1 | 7.1 | 10.5 | 15.4 | 17.4 | 11.1 | 5.5 | 3.7 | 3.5 | 3.6 | 2.8 | 1.5 | 10.5 | 8.6 | |
| N. M. C. | 7516 | ♀ ad. | Topotype | 31.1 | 10.1 | 9.6 | 8.0 | 11.0 | 16.0 | 18.5 | 11.5 | 5.6 | 4.1 | 3.7 | 3.8 | 2.9 | 1.5 | 10.0 | 9.0 | |
| M. C. Z. | 6852 | ♀ ad. | Sumas | 31.3 | 10.3 | 9.2 | 7.3 | 10.1 | 15.8 | 17.2 | 11.0 | 5.3 | 3.8 | 3.5 | 3.7 | 2.7 | 1.5 | 11.0 | 8.7 | |
| M. C. Z. | 3645 | ♀ ad. | Sumas | 29.4 | 10.2 | 8.6 | 7.3 | 10.7 | 14.7 | 15.4 | 10.5 | 5.2 | 3.8 | 3.5 | 3.8 | 2.7 | 1.5 | 9.5 | 8.3 | |
| M. C. Z. | 10728 | ♀ ad. | Sumas | 31.7 | 10.2 | 9.1 | 7.1 | 10.7 | 15.7 | 17.4 | 11.5 | 5.5 | 3.8 | 3.4 | 3.7 | 2.6 | 1.2 | 9.3 | 8.2 | |
| Mustela erminea olympica | ||||||||||||||||||||
| U. S. N. M. | 90738 | ♂ ad. | Type | 31.9 | 11.6 | 10.0 | 7.9 | 11.9 | 15.3 | 17.9 | 11.6 | 5.3 | 4.0 | 3.6 | 3.9 | 2.9 | 2.1 | 9.9 | 8.9 | |
| U. S. N. M. | 241941 | ♂ ad. | N. Fk. Quinault River | 32.5 | 11.7 | 10.2 | 8.2 | 12.0 | 16.3 | 18.2 | 11.7 | 5.6 | 4.2 | 3.6 | 3.7 | 2.9 | 1.8 | 10.0 | 9.2 | |
| U. S. N. M. | 231829 | ♂ ad. | Duckabush | 30.6 | 10.9 | 9.2 | 7.4 | 10.3 | 15.0 | 17.2 | 10.7 | 5.2 | 4.2 | 3.5 | 3.7 | 2.7 | 1.7 | 10.0 | 8.6 | |
| U. S. N. M. | 231830 | ♂ ad. | Duckabush | 32.1 | 11.1 | 10.0 | 8.2 | 12.3 | 16.6 | 18.6 | 11.2 | 5.9 | 4.0 | 3.7 | 3.9 | 3.0 | 1.7 | 10.2 | 9.3 | |
| M. Z. | 53700 | ♂ ad. | Lake Cushman | 32.0 | 11.4 | 9.8 | 8.0 | 11.0 | 16.9 | 18.8 | 11.4 | 6.3 | 4.3 | 3.8 | 3.9 | 3.1 | 1.7 | 10.3 | 9.2 | |
| av. 5 | 31.8 | 11.3 | 9.8 | 7.9 | 11.5 | 16.0 | 18.1 | 11.3 | 5.7 | 4.2 | 3.7 | 3.9 | 2.9 | 1.8 | 10.1 | 9.0 | ||||
| C. R. C. M. | 96 | ♀ ad. | Elwha River | 27.5 | 9.4 | 8.3 | 6.9 | 9.3 | 13.2 | 15.3 | 10.2 | 4.6 | 3.4 | 3.0 | 3.3 | 2.5 | 1.3 | 8.5 | 7.2 | |
| C. R. C. M. | 1164 | ♀ ad. | 12 mi. S Port Angeles | 26.7 | 9.0 | 8.1 | 6.7 | 9.0 | 13.1 | 14.4 | 9.7 | 4.8 | 3.4 | 3.1 | 3.4 | 2.3 | 1.3 | 9.0 | 7.3 | |
| U. S. N. M. | 242133 | ♀ ad. | Hayes Creek | 27.2 | 9.2 | 8.4 | 7.2 | 9.2 | 13.7 | 15.4 | 9.5 | 4.8 | 3.3 | 3.0 | 3.1 | 2.1 | 1.2 | 8.3 | 7.2 | |
| av. 3 | 27.1 | 9.2 | 8.3 | 6.9 | 9.2 | 13.3 | 15.0 | 9.8 | 4.7 | 3.4 | 3.1 | 3.3 | 2.3 | 1.2 | 8.6 | 7.2 | ||||
| Mustela erminea streatori | ||||||||||||||||||||
| average} | ♂ ad. 12 | Tillamook Co | {33.2 | 11.7 | 10.5 | 8.5 | 11.7 | 17.0 | 19.2 | 11.8 | 6.4 | 4.3 | 3.8 | 3.9 | 3.1 | 1.8 | 10.8 | 9.8 | ||
| maximum} | {33.8 | 12.1 | 11.1 | 9.1 | 12.5 | 18.0 | 19.8 | 12.6 | 6.9 | 4.4 | 4.0 | 4.1 | 3.5 | 2.1 | 11.4 | 10.3 | ||||
| minimum} | {32.5 | 11.3 | 10.0 | 8.2 | 11.1 | 16.1 | 18.5 | 11.1 | 6.0 | 4.1 | 3.6 | 3.8 | 2.9 | 1.6 | 10.3 | 9.5 | ||||
| average} | ♀ ad. 7 | Tillamook Co | {28.5 | 9.9 | 8.9 | 7.3 | 10.1 | 14.3 | 15.9 | 10.6 | 5.2 | 3.6 | 3.2 | 3.5 | 2.6 | 1.6 | 9.4 | 8.1 | ||
| maximum} | {29.5 | 10.2 | 9.2 | 7.6 | 10.2 | 14.8 | 16.3 | 11.2 | 5.4 | 3.7 | 3.3 | 3.6 | 2.7 | 1.7 | 10.0 | 8.4 | ||||
| minimum} | {27.6 | 9.6 | 8.7 | 7.0 | 9.8 | 14.1 | 15.5 | 10.0 | 5.0 | 3.5 | 3.1 | 3.3 | 2.5 | 1.5 | 9.2 | 7.9 | ||||
| Mustela erminea gulosa | ||||||||||||||||||||
| U. S. N. M. | 82177 | ♂ ad. | Trout Lake | 32.0 | 11.3 | 10.1 | 8.5 | 11.2 | 16.3 | 18.3 | 11.3 | 5.9 | 4.2 | 3.8 | 4.1 | 2.9 | 1.6 | 10.5 | 9.4 | |
| U. S. N. M. | 64768 | ♂ ad. | Trout Lake | 33.3 | 12.0 | 9.9 | 8.3 | 12.2 | 16.9 | 18.5 | 11.8 | 5.9 | 4.2 | 3.7 | 3.7 | 2.8 | 1.8 | 10.6 | 9.4 | |
| average} | ♂ ad. 2, sad. 13 | Trout Lake | {32.3 | 11.5 | 10.0 | 8.3 | 11.5 | 16.4 | 18.4 | 11.5 | 5.9 | 4.2 | 3.7 | 3.9 | 2.9 | 1.7 | 10.3 | 9.2 | ||
| maximum} | {33.4 | 12.0 | 10.7 | 8.8 | 12.4 | 17.2 | 19.3 | 12.1 | 6.3 | 4.5 | 3.8 | 4.1 | 3.2 | 2.0 | 11.2 | 9.7 | ||||
| minimum} | {30.9 | 10.8 | 9.0 | 7.4 | 10.8 | 15.6 | 17.8 | 10.8 | 5.5 | 3.9 | 3.5 | 3.6 | 2.7 | 1.6 | 9.6 | 8.5 | ||||
| U. S. N. M. | 232741 | ♀ ad. | Reflection Lakes | 28.4 | 9.6 | 8.7 | 7.4 | 9.3 | 14.1 | 15.8 | 10.9 | 5.4 | 3.7 | 3.2 | 3.5 | 2.7 | 1.6 | 8.4 | 7.4 | |
| U. S. N. M. | 90727 | ♀ ad. | Mt. St. Helens | 28.0 | 9.7 | 8.1 | 6.9 | 9.5 | 13.6 | 15.6 | 10.0 | 4.8 | 3.6 | 3.3 | 3.5 | 2.5 | 1.6 | 9.0 | 7.6 | |
| U. S. N. M. | 81919 | ♀ ad. | Trout Lake | 28.1 | 9.7 | 8.8 | 7.0 | 10.0 | 13.8 | 15.4 | 10.1 | 4.9 | 3.6 | 3.2 | 3.3 | 2.3 | 1.4 | 8.1 | 7.3 | |
| U. S. N. M. | 87039 | ♀ ad. | Trout Lake | 28.4 | 9.8 | 8.7 | 7.1 | *10.8 | 14.5 | 15.6 | 10.0 | 5.0 | 3.5 | 3.2 | 3.4 | 2.5 | 1.4 | 8.6 | 7.5 | |
| U. S. N. M. | 77370 | ♀ ad. | Trout Lake | 27.8 | 9.6 | 8.6 | 6.6 | *9.3 | 13.6 | 15.2 | 9.7 | 4.7 | 3.8 | 3.4 | 3.5 | 2.7 | 1.5 | 8.1 | 7.2 | |
| av. 5 | 28.1 | 9.7 | 8.6 | 7.0 | 9.8 | 13.9 | 15.5 | 10.1 | 5.0 | 3.6 | 3.3 | 3.4 | 2.5 | 1.5 | 8.4 | 7.4 | ||||
| Mustela erminea muricus | ||||||||||||||||||||
| U. S. N. M. | 231397 | ♂ ad. | Donovan, Mont | *31.2 | 10.5 | 9.5 | 8.0 | 11.3 | *16.0 | 17.5 | 5.7 | 4.0 | 3.6 | 3.7 | 2.8 | 1.6 | 11.2 | 8.8 | ||
| U. S. N. M. | 206991 | ♂ ad. | Mill Creek, Oreg | *30.9 | 10.8 | 9.0 | 7.2 | 11.0 | 17.4 | 11.3 | 5.6 | 4.3 | 3.7 | 4.0 | 3.0 | 1.6 | 8.7 | |||
| M. V. Z. | 34746 | ♂ ad. | Black Butte, Calif | 30.8 | 11.1 | 9.4 | 7.7 | 11.1 | 15.9 | 17.5 | 10.8 | 5.6 | 4.2 | 3.5 | 3.6 | 2.8 | 1.8 | 9.6 | 8.4 | |
| M. V. Z. | 41501 | ♂ ad. | Wheeler Peak, Nev | 29.8 | 10.4 | 9.3 | 7.2 | 10.6 | 15.1 | 17.1 | 10.7 | 5.4 | 4.0 | 3.5 | 3.5 | 2.7 | 1.5 | 9.8 | 8.0 | |
| E. R. W. | 3050 | ♂ ad. | Crested Butte, Colo. | *30.4 | 11.1 | 9.8 | 7.7 | 11.2 | 16.2 | 18.3 | 10.8 | 5.7 | 4.3 | 3.6 | 3.9 | 2.9 | 1.6 | 8.5 | ||
| av. 5 | 30.6 | 10.8 | 9.4 | 7.6 | 11.0 | 15.8 | 17.6 | 10.9 | 5.6 | 4.2 | 3.6 | 3.7 | 2.8 | 1.6 | 10.2 | 8.5 | ||||
| M. Z. | 62111 | ♀ ad. | Teton Co., Wyoming | 28.0 | 9.7 | 8.3 | 6.7 | 10.0 | 14.9 | 16.0 | 10.9 | 5.3 | 3.6 | 3.2 | 3.3 | 2.7 | 1.7 | 8.8 | 7.2 | |
| M. Z. | 62112 | ♀ ad. | Teton Co., Wyoming | 27.3 | 9.7 | 8.1 | 6.5 | 9.2 | 14.0 | 15.6 | 10.1 | 5.0 | 3.5 | 3.2 | 3.3 | 2.5 | 1.4 | 8.3 | 7.2 | |
| M. V. Z. | 13776 | ♀ ad. | Rush Creek, California | 28.1 | 9.5 | 8.8 | 7.4 | 9.7 | 14.3 | 16.5 | 10.2 | 4.7 | 3.6 | 3.2 | 3.4 | 2.7 | 1.5 | 9.1 | 7.8 | |
| M. V. Z. | 13777 | ♀ ad. | Castle Lake, California | 29.4 | 9.9 | 8.8 | 7.3 | 10.8 | 15.2 | 17.1 | 10.0 | 5.3 | 3.6 | 3.1 | 3.4 | 2.6 | 1.5 | 8.2 | ||
| M. V. Z. | 41502 | ♀ ad. | Wheeler Peak, Nev | 27.3 | 9.3 | 8.0 | 6.3 | 9.5 | 13.9 | 15.8 | 10.0 | 5.0 | 3.4 | 2.9 | 3.1 | 2.4 | 1.3 | 8.8 | 7.2 | |
| F. M. N. H. | 11440 | ♀ ad. | Camp Albion, Colo | 27.8 | 9.4 | 8.4 | 6.9 | 9.5 | 14.1 | 15.5 | 10.5 | 5.1 | 3.6 | 3.2 | 3.3 | 2.6 | 1.4 | 8.9 | 7.5 | |
| av. 6 | 28.0 | 9.6 | 8.4 | 6.9 | 9.8 | 14.4 | 16.1 | 10.3 | 5.1 | 3.6 | 3.1 | 3.3 | 2.6 | 1.5 | 8.8 | 7.5 | ||||
| Mustela erminea angustidens | ||||||||||||||||||||
| A. M. N. H. | 12432 | ♀ ad. | Conard Fissure, Ark | 35.1 | 12.7 | 10.4 | 8.1 | 12.1 | 18.7 | 12.1 | 6.2 | 4.5 | 4.4 | 3.3 | 1.65 | 11.0 | 9.6 | |||
| A. M. N. H. | 12433 | ♀ ad. | Conard Fissure, Ark | 12.6 | 9.9 | 8.3 | 11.4 | 4.2 | 4.5 | 3.4 | 1.5 | 9.9 | ||||||||
| A. M. N. H. | 12435 | ♀ ad. | Conard Fissure, Ark | *32.5 | 12.4 | 10.0 | *7.5 | 12.2 | 17.1 | *19.0 | 11.4 | 5.8 | 3.8 | 4.0 | 2.9 | 1.4 | 8.6 | |||
| A. M. N. H. | 11766 | ♀ ad. | Conard Fissure, Ark | 34.5 | 12.3 | 10.7 | 8.4 | 11.8 | 18.2 | 12.5 | 6.6 | 4.1 | 4.3 | 3.2 | 1.5 | 10.0 | 9.5 | |||
| av | 34.0 | 12.5 | 10.3 | 8.1 | 11.9 | 18.0 | 12.0 | 6.2 | 4.2 | 4.3 | 3.2 | 1.5 | 10.5 | 9.4 | ||||||
| A. M. N. H. | 12437 | ♂ sad. | Conard Fissure, Ark | 39.2 | 14.5 | 11.7 | 9.6 | 13.6 | 20.4 | 13.0 | 6.7 | 4.6 | 4.9 | 3.8 | 2.1 | 13.2 | 11.0 | |||
| A. M. N. H. | 12441 | ♂ ad. | Conard Fissure, Ark | 38.5 | 13.9 | 11.3 | 9.1 | 13.0 | 20.0 | 13.5 | 6.9 | 4.7 | 4.9 | 3.6 | 1.5 | 12.1 | 10.7 | |||
| A. M. N. H. | 12436 | ♂ ad. | Conard Fissure, Ark | 13.5 | 11.7 | 8.9 | 13.5 | 4.0 | 4.3 | 3.2 | 1.6 | 10.4 | ||||||||
| A. M. N. H. | 12444 | ♂ ad. | Conard Fissure, Ark | 14.3 | 11.6 | 9.2 | 13.8 | 4.7 | 4.6 | 3.2 | 1.8 | 10.8 | ||||||||
| A. M. N. H. | 11769 | ♂ sad. | Conard Fissure, Ark | 4.5 | 4.9 | 3.9 | 1.9 | |||||||||||||
| A. M. N. H. | 12438 | ♂ yg. | Conard Fissure, Ark | 36.6 | 13.5 | 12.2 | 9.3 | 12.8 | 13.0 | 6.6 | 4.4 | 4.6 | 3.5 | 1.7 | 13.7 | 10.9 | ||||
| av | 38.1 | 13.9 | 11.7 | 9.2 | 13.3 | 20.2 | 13.2 | 6.7 | 4.5 | 4.7 | 3.5 | 1.8 | 13.0 | 10.8 | ||||||
| Mustela rixosa eskimo | ||||||||||||||||||||
| average} | ♂ ad. 6 | Point Barrow | {29.5 | 10.1 | 9.1 | 7.4 | 10.1 | 15.6 | 17.8 | 11.3 | 5.4 | 3.8 | 3.6 | 3.6 | 2.7 | 1.4 | 9.5 | 8.4 | ||
| maximum} | {30.1 | 10.6 | 9.9 | 7.8 | 10.6 | 16.3 | 18.0 | 11.9 | 5.8 | 4.2 | 3.9 | 3.9 | 2.9 | 1.6 | 10.0 | 8.7 | ||||
| minimum} | {27.6 | 9.3 | 8.6 | 7.1 | 9.3 | 14.5 | 17.0 | 10.1 | 5.0 | 3.5 | 3.2 | 3.3 | 2.5 | 1.1 | 8.5 | 7.7 | ||||
| average} | ♀ ad. an d sad. 4 | Point Barrow | {27.8 | 9.3 | 8.3 | 6.9 | 9.6 | 14.1 | 15.7 | 10.5 | 5.1 | 3.4 | 3.2 | 3.2 | 2.5 | 1.2 | 9.5 | 7.7 | ||
| maximum} | {28.5 | 9.5 | 8.5 | 7.2 | 9.7 | 15.0 | 16.5 | 11.1 | 5.6 | 3.7 | 3.3 | 3.4 | 2.6 | 1.3 | 10.0 | 8.0 | ||||
| minimum} | {27.0 | 9.0 | 7.9 | 7.0 | 9.5 | 13.6 | 15.0 | 10.2 | 4.8 | 3.2 | 3.0 | 3.0 | 2.3 | 1.1 | 9.2 | 7.4 | ||||
| Mustela rixosa rixosa | ||||||||||||||||||||
| average} | ♂ ad. 2, and sad. 4 | Shaunavon | {29.5 | 10.1 | 8.2 | 6.6 | 9.9 | 15.1 | 16.4 | 11.0 | 5.2 | 3.7 | 3.3 | 3.6 | 2.6 | 1.4 | 10.0 | 8.4 | ||
| maximum} | {30.4 | 10.5 | 9.0 | 6.9 | 10.5 | 16.1 | 17.1 | 11.5 | 5.5 | 3.9 | 3.5 | 3.8 | 2.7 | 1.5 | 10.4 | 8.8 | ||||
| minimum} | {28.4 | 9.6 | 7.4 | 6.3 | 9.2 | 14.0 | 15.2 | 10.7 | 5.0 | 3.5 | 3.1 | 3.3 | 2.4 | 1.3 | 9.4 | 8.0 | ||||
| average} | ♀ ad. 3, and sad. 1 | Regina and Shaunavon | {26.1 | 8.9 | 7.2 | 5.5 | 8.9 | 13.1 | 14.1 | 9.7 | 4.9 | 3.3 | 3.0 | 3.2 | 2.3 | 1.2 | 8.6 | 7.1 | ||
| maximum} | {27.0 | 9.2 | 7.5 | 5.9 | 9.5 | 13.6 | 14.6 | 10.0 | 5.0 | 3.5 | 3.1 | 3.3 | 2.4 | 1.3 | 9.0 | 7.2 | ||||
| minimum} | {24.7 | 8.5 | 6.9 | 5.2 | 8.3 | 12.3 | 13.7 | 9.5 | 4.7 | 3.1 | 2.8 | 2.9 | 2.3 | 1.1 | 8.2 | 7.0 | ||||
| Mustela rixosa campestris | ||||||||||||||||||||
| Swenk, Mr. | 5 | ♂ ad. | 1 mi. E Inland | 11.6 | 8.8 | 7.6 | 11.1 | 16.1 | 18.0 | 4.1 | 3.6 | 3.8 | 2.7 | 1.6 | 11.5 | 9.7 | ||||
| Swenk, Mr. | 8 | ♂ ad. | Inland | 30.7 | 10.5 | 8.2 | 7.0 | 10.5 | 15.9 | 17.9 | 10.9 | 5.7 | 3.8 | 3.4 | 3.5 | 2.6 | 1.5 | 8.6 | ||
| Swenk, Mr. | 10 | ♀ ad. | Inland | 28.0 | 9.8 | 7.6 | 5.8 | 9.4 | 14.2 | 10.3 | 5.4 | 1.7 | 3.5 | 3.1 | 3.3 | 2.5 | 1.5 | 9.3 | 7.8 | |
| U. S. N. M. | 171490 | ♀ ad. | Type | 28.8 | 7.7 | 6.1 | 9.1 | 14.1 | 15.0 | 10.2 | 5.1 | 1.6 | 3.8 | 3.2 | 3.4 | 2.5 | 1.5 | 9.3 | 7.7 | |
| Mustela rixosa allegheniensis | ||||||||||||||||||||
| U. S. N. M. | 249285 | ♂ sad. | Finleyville, Pa | 29.7 | 10.2 | 10.1 | 15.0 | 16.5 | 10.5 | 5.2 | 4.1 | 3.4 | 3.7 | 2.7 | 1.5 | 9.6 | 8.1 | |||
| U. S. N. M. | 203173 | ♂ sad. | Waynesburg, Pa | 28.6 | 9.5 | 7.7 | 6.7 | 9.5 | 14.7 | 16.1 | 10.5 | 5.4 | 3.4 | 3.5 | 3.2 | 2.5 | 1.3 | 10.2 | 8.0 | |
| U. S. N. M. | 206340 | ♂ ad. | Huttonsville, W. Va | 28.5 | 9.9 | 8.5 | 7.1 | 10.3 | 15.1 | 16.7 | 10.2 | 5.1 | 3.3 | 3.0 | 3.2 | 2.4 | 1.3 | 10.5 | 8.4 | |
| C. M. | 7543 | ♀ ad. | Pymatuning Swamp | 28.0 | 9.2 | 8.1 | 9.5 | 13.6 | 10.0 | 5.2 | 3.4 | 3.0 | 3.1 | 2.4 | 1.3 | |||||
| A. N. S. P. | 11279 | ♀ ad. | Beallville, Pa | 28.0 | 9.5 | 7.5 | 6.2 | 9.7 | 13.5 | 14.6 | 10.0 | 5.1 | 3.7 | 3.3 | 3.5 | 2.6 | 1.4 | 8.7 | 7.8 | |
| U. S. N. M. | 245843 | ♀ ad. | near Marshall, N. C | 27.5 | 9.4 | 7.3 | 6.4 | 9.4 | 15.0 | 9.3 | 5.0 | 3.6 | 3.2 | 3.4 | 2.5 | 1.3 | 7.7 | |||
(Abbreviations used for names of collections in the table of measurements of Mustela)
| A. M. N. H. | American Museum of Natural History | M. C. Z. | Museum of Comparative Zoölogy |
| A. N. S. P. | Academy of Natural Sciences of Philadelphia | M. P. H. N. | Musée Polonais d'Histoire Naturelle (Warsaw, Poland) |
| Baylor U. | Baylor University | M. V. Z. | Museum of Vertebrate Zoölogy, University of California |
| B. M. | British Museum of Natural History | M. Z. | Museum of Zoölogy, University of Michigan |
| B. S. N. | Boston Society of Natural History | N. H. R. S. | Naturhistoriska Riksmuseum |
| B. Z. M. | Berlin Zoological Museum | N. M. C. | National Museum of Canada |
| C. A. C. | California Academy of Sciences | S. D. M. | San Diego Society of Natural History |
| C. M. | Carnegie Museum | Snyder | W. E. Snyder, Beaver Dam, Wisconsin |
| C. R. C. M. | Charles R. Conner Museum, Washington State College | Stan. U. | Leland Stanford Junior University |
| C. Z. M. | University Zoological Museum, Copenhagen, Denmark | Stephens | Frank Stephens, private collection |
| Cornell | Cornell University | Swenk, Mr. | Myron H. Swenk, private collection |
| Cowan | Ian McTaggart-Cowan, private collection | U. O. | University of Oregon, Eugene, Oregon |
| Dickey | Donald R. Dickey (deceased), private collection | U. S. N. M. | United States National Museum |
| E. R. W. | Edward R. Warren, private collection | Walker | Alex Walker, private collection |
| F. M. N. H. | Field Museum of Natural History | Wisc. U. | University of Wisconsin |
| F. S. M. | Florida State Museum | * | Approximate |
| Kans. U. | University of Kansas, Museum of Natural History | ** | Average of 14 |
LITERATURE CITED
Abbot, C. C.
1884. A naturalist's rambles about home. D. Appleton and Co., New York, 485 pp.
Addy, E.
1939. A weasel trails a rabbit. Jour. Mamm., 20:372-373, August 14, 1939.
Aldous, S. E., and Manweiler, J.
1942. The winter food habits of the short-tailed weasel in northern Minnesota. Jour. Mamm., 23:250-255, August 13, 1942.
Allen, D. L.
1938. Notes on the killing technique of the New York weasel. Jour. Mamm., 19:225-229, May 14, 1938.
1940. Two recent mammal records from Allegan County, Michigan. Jour. Mamm., 21:459-460, November 14, 1940.
Allen, G. M.
1933. The least weasel a circumboreal species. Jour. Mamm., 14:316-319, November 13, 1933.
Allen, J. A.
1889. Notes on a collection of mammals from southern México, with descriptions of new species of the Genera Sciurus, Tamias, and Sigmodon. Bull. Amer. Mus. Nat. Hist., 2:165-181, October 21, 1889.
1891. On a collection of mammals from southern Texas and northeastern México. Bull. Amer. Mus. Nat. Hist., 3:219-228, April 17, 1891.
1894. On the mammals of Aransas County, Texas, with descriptions of new forms of Lepus and Oryzomys. Bull. Amer. Mus. Nat. Hist., 6:165-198, 1 map, May 31, 1894.
1896. On mammals collected in Bexar County and vicinity, Texas, by Mr. H. P. Attwater, with field notes by the collector. Bull. Amer. Mus. Nat. Hist., 8:47-80, April 22, 1896.
1904. Mammals from southern México and Central and South America. Bull. Amer. Mus. Nat. Hist., 20:29-80, text figs. 1-18, February 29, 1904.
1906. Mammals from the states of Sinaloa and Jalisco, México, collected by J. H. Batty during 1904 and 1905. Bull. Amer. Mus. Nat. Hist., 22:191-262, pls. 22-33, 3 figs. in text, July 25, 1906.
1908. Mammals from Nicaragua. Bull. Amer. Mus. Nat. Hist., 24:647-670, 12 figs. in text, October 13, 1908.
1911. Mammals from Venezuela collected by Mr. M. A. Carriker, Jr., 1909-1911. Bull. Amer. Mus. Nat. Hist., 30:239-273, December 2, 1911.
1912. Mammals from western Colombia. Bull. Amer. Mus. Nat. Hist., 31:71-95, April 19, 1912.
1916. The neotropical weasels. Bull. Amer. Mus. Nat. Hist., 35:89-111, April 28, 1916.
1916A. List of mammals collected in Colombia by the American Museum of Natural History Expeditions, 1910-1915. Bull. Amer. Mus. Nat. Hist., 35:191-238, 1 map, May 31, 1916.
Alston, A. R.
1879-1882. Biologia Centrali-Americana. Mammalia, xx + 220 pp., pls. 1-22.
Ameghino, F.
1889. Contribución al conocimiento de los mamiferos fosiles de la República Argentina. Imprenta de Pablo E. Coni É Hijos, Especial para obras, xxxii + 1027 pp., pls. 1-94, numerous figures in text.
Anderson, R. M.
1945. Three mammals of the weasel family (Mustelidae) added to the Quebec list with descriptions of two new forms. Ann. Rept. Provancher Soc., 25th Anniversary, pp. 56-61, November 2, 1945.
Arthur, S. C.
1928. The fur animals of Louisiana. State of Louisiana, Dept. Conservation Bull., 18:1-433, illustrated, November, 1928.
Audubon, J. J., and Bachman, J.
1845-1853. The viviparous quadrupeds of North America: pls. in 3 vols., elephant folios, each of 50 pls., vol. 1, 1845; vol. 2, 1846; vol. 3, 1848. Text in 3 vols.: vol. 1, xiv + 389; vol. 2, 1-334; vol. 3, iv + 257.
1851. The quadrupeds of North America, vol. 2, pp. 1-334, pls. 51-100. Publ. by V. G. Audubon, New York.
1856. The quadrupeds of North America, vol. 2, pp. xiii-xiv + i-viii + 2-383, pls. 1-50. Publ. by V. G. Audubon, New York.
Bachman, J.
1839. Observations on the changes of colour in birds and quadrupeds. Trans. Amer. Philos. Soc., 6:197-239.
Bailey, B.
1929. The mammals of Sherburne County, Minnesota. Jour. Mamm., 10:153-164, May 9, 1929.
Bailey, H. H.
1930. Correcting inaccurate ranges of certain Florida mammals and others of Virginia and the Carolinas. The Bailey Mus. and Library of Nat. Hist., Bull. no. 3; 4 pages (Miami, Florida), December 1, 1930.
Bailey, V.
1905. Biological survey of Texas. N. Amer. Fauna, 25:1-222, 16 pls., 24 figs. in text, October 24, 1905.
1928. Animal life of the Carlsbad Cavern. Monograph, Amer. Soc. Mammalogists, no. 3, pp. xiii + 195 pp., 67 figs. Williams and Wilkins Co., Baltimore [Md.].
1932. Mammals of New Mexico. N. Amer. Fauna, 53:1-412, 22 pls., 58 figs. in text, March 1, 1932.
Baird, S. F.
1858. General report upon the zoölogy of the several Pacific Railroad Routes. Part I, Mammals, xlvii + 757 pp., 60 pls., July 14, 1858.
Bangs, O.
1896. A review of the weasels of eastern North America. Proc. Biol. Soc. Washington, 10:1-24, 3 pls., February 25, 1896.
1899. Three new weasels from North America. Proc. New England Zoöl. Club, 1:53-57, June 9, 1899.
1899B. Description of a new weasel from the Rocky Mountains of British Columbia. Proc. New England Zoöl. Club, 1:81-82, December 27, 1899.
1902. Chiriquí Mammalia. Bull. Mus. Comp. Zoöl., 39:17-51, 27 figs. in text, April, 1902.
Barber, C. M., and Cockerell, T. D. A.
1898. A new weasel from New Mexico. Proc. Acad. Nat. Sci. Philadelphia, 1898:188-189.
Barrett-Hamilton, G. E. H.
1903. Abstract of a physiological hypothesis to explain the winter whitening of mammals and birds inhabiting snowy countries, and the more striking points in the distribution of white in vertebrates generally. Proc. Royal Irish Acad., 24:303-314.
1904. Notes and descriptions of some new species and subspecies of Mustelidae. Ann. and Mag. Nat. Hist., 13(ser. 7):388-395, May, 1904.
Bell, T.
1874. A history of British quadrupeds. John Van Voorst, London, xi + 474 pp., illustrated.
Belcher, E.
1843. Narrative of a voyage round the world, ... in her majesty's ship Sulphur, during the years 1836-1842.... London, H. Colburn, 2 vols.: v. 1, xxxviii + 387; v. 2, vi + 474, illustrated.
Bishop, S. C.
1923. Note on the nest and young of the small brown weasel. Jour. Mamm., 4:26-27, 1 pl., February 9, 1923.
Bissonnette, T. H., and Bailey, E. E.
1940. Den and runway system for weasels and other small mammals in the laboratory. Amer. Midland Nat., 24:761-763, November, 1940.
1944. Experimental modification and control of molts and changes of coat-color in weasels by controlled lighting. Ann. New York Acad. Sci., 45:221-260, 7 pls., 1 fig., April 7, 1944.
Bonaparte, C. L.
1838. Remarks on the species of the genus Mustela. Charlesworth's Mag. Nat. Hist., 2:37-38.
Booth, E. S.
1946. Account of a weasel in a tree. Jour. Mamm., 26:439, February 12, 1946.
Borell, A. E., and Ellis, R.
1934. Mammals of the Ruby Mountains region of northeastern Nevada. Jour. Mamm., 15:12-44, 6 pls., 1 fig. in text, February 15, 1934.
Boyer, R. H.
1943. Weasel versus squirrel in Sequoia National Park. Jour. Mamm., 24:99-100, February 20, 1943.
Brown, B.
1908. The Conard Fissure, a Pleistocene bone deposit in northern Arkansas: with descriptions of two new genera and twenty new species of mammals. Mem. Amer. Mus. Nat. Hist., 9(pt. 4):155-208, pls. 14-25, 3 figs. in text.
Brown, M.
1941. A gazetteer of entomological stations in Ecuador. Ann. Ento. Soc. Amer., 34:809-851, 10 figs. in text, December 19, 1941.
Burroughs, J.
1900. Squirrels and other fur-bearers. Houghton, Mifflin and Company, 149 pp., illustrated.
Burroughs, R. D.
1939. New York weasel preying on the cottontail rabbit. Jour. Mamm., 20:253, May 14, 1939.
Cabrera, A.
1913. Sobre algunas formas del género "Mustela." Bol. d. l. Real Soc. Español d. Hist. Nat., 13:429-435, November, 1913.
1914. Sobre algunas formas del género "Mustela." Bol. d. l. Real Soc. Español d. Hist. Nat., 14:175-176, 1 pl., March, 1914.
1928. Sobre Lyncodon patagonicus con descripción de una nueva subespecie. Revista Chilean d. Hist. Nat., 32:259-263.
1940. Sobre carnívoros Sudamericanos. Inst. d. Mus. d. l. Univ. Nac. d. La Plata, 5(no. 29):1-22.
Chapman, F. M.
1917. The distribution of bird-life in Colombia; a contribution to a biological survey of South America. Bull. Amer. Mus. Nat. Hist., 36: x + 729 pp., 41 pls., 21 figs. in text.
1926. The distribution of bird-life in Ecuador. Bull. Amer. Mus. Nat. Hist., 55: xiii + 784 pp., 30 pls., 20 figs. in text.
1931. The upper zonal bird-life of mts. Roraima and Duida. Bull. Amer. Mus. Nat. Hist., 63:1-135, 42 figs.
1937. The Phelps Venezuela Expedition. Nat. Hist., 40(no. 5):760-761, December, 1937.
Coues, E.
1877. Fur-bearing animals: A monograph of North American Mustelidae. Dept. Int., U. S. Geol. Surv. Terr., Miscl. Publ., No. 8, xiv + 348 pp., 20 pls., 1 fig. in text.
Criddle, N., and Criddle, S.
1925. The weasels of southern Manitoba. Canadian Field-nat., 39:142-148, September, 1925.
Criddle, S.
1926. The habits of Microtus minor in Manitoba. Jour. Mamm., 7:193-200, August 9, 1926.
1930. The prairie pocket gopher, Thomomys talpoides rufescens. Jour. Mamm., 11:265-280, 1 pl., August 9, 1930.
1947. A nest of the least weasel. Canadian Field-nat., 61:69, April, 1947.
Dalquest, W. W.
1948. Mammals of Washington. Univ. Kansas Publ., Mus. Nat. Hist., 2:1-444, 140 figs. in text, April 9, 1948.
Danforth, C. N.
1925. Hair with special reference to hypertrichosis. Amer. Med. Assoc., N Dearborn St., Chicago, Illinois, 152 pp., 64 figs.
Davenport, C. B.
1905. Evolution without mutation. Jour. Exp. Zoöl., 2:139-143, April, 1905.
1906. The mutation theory in animal evolution. Science, N. S., 24:556-558, November 2, 1906.
Davis, W. B.
1944. Notes on Mexican mammals. Jour. Mamm., 25:370-403, 1 fig. in text, December 12, 1944.
Davis, W. B., and Robertson, J. L., Jr.
1944. The mammals of Culberson County, Texas. Jour. Mamm., 25:254-273, September 8, 1944.
Deanesly, R.
1944. The reproductive cycle of the female weasel (Mustela nivalis). Proc. Zoöl. Soc. London, 114(pt. 3):339-349.
Dearborn, N.
1932. Foods of some predatory fur-bearing animals in Michigan. Univ. Michigan School Forestry and Conservation, Bull. No. 1:1-52, 8 figs. in text, 22 charts, 10 maps.
1932B. Occurrence of the least weasel in Michigan. Jour. Mamm., 13:277, August 9, 1932.
Degerbøl, M.
1935. Mammals. Part 1. Systematic notes. Rept. 5th Thule Exped...., 2(no. 4):1-67, 12 figs. in text.
Desmarest, A. G.
1818. Nouveau Dictionnaire d' Histoire Naturelle.... Tome XIX, 619 pp., Paris.
Dice, L. R.
1921. Notes on the mammals of interior Alaska. Jour. Mamm., 2:20-28, February 21, 1921.
Dixon, J.
1931. Pikas versus weasel. Jour. Mamm., 12:72, February 12, 1931.
Dunk, F. A.
1946. Least weasel in Saskatchewan. Jour. Mamm., 27:392, November 25, 1946.
Elliot, D. G.
1905. Descriptions of apparently new species and subspecies of mammals from México and San Domingo. Proc. Biol. Soc. Washington, 18:233-236, December 9, 1905.
Errington, P. L.
1935. Food habits of mid-west foxes. Jour. Mamm., 16:192-200, August 12, 1935.
1936. Food habits of a weasel family. Jour. Mamm., 17:406-407, November 14, 1936.
Farurick, B.
1873. Ein Albino des kleiner Wiesels (Mustela vulgaris). Der Zool. Garten, 14:17-18.
Feilden, H. W.
1878. See as included under Nares, G. S., 1878.
Flintoff, R. J.
1935. Stoats and weasels, brown and white. North Western Naturalist [England], 10:214-229, September 30, 1935.
Follett, W. I.
1937. Prey of weasel and mink. Jour. Mamm., 18:365, August 14, 1937.
Fremont, J. C.
1856. A narrative of the exploring expedition to Oregon and North California, pp. 190-493, in The Life of Col. John Charles Fremont, and his narrative of Explorations and Adventures, in Kansas, Nebraska, Oregon and California. By Samuel M. Smucker, New York and Auburn.
Glover, F. A.
1942. Spring color change of the New York weasel. Pennsylvania Game News, 13(no. 7):18, 32, October, 1942.
1943A. Killing techniques of the New York weasel. Pennsylvania Game News, 13(no. 10):11, 23, January, 1943, issue.
1943B. A study of the winter activities of the New York weasel. Pennsylvania Game News, 14(no. 6):8, 9, September, 1943.
Goeldi, E. A.
1897 (for 1898). Ein erstes authentisches Exemplar eines echten Wiesels aus Brasilien. Zoöl. Jahrb., Abt. f. systematik, geogr. u. Biol., 10:556-562, pl. 21, September 15, 1897.
1901. O primeiro exemplar authentico deuna genuina dominha do Brazil. Bol. do Mus. Paraense, 3:195-203, August, 1901.
1904. Catalogo de mammiferos. Bol. do Mus. Goeldi, 4:41-122, 6 pls., February, 1904.
Goble, F. C.
1942. The Guinea-worm in a Bonaparte weasel. Jour. Mamm., 23:221, June 3, 1942.
Goldman, E. A.
1912. A new weasel from Costa Rica. Proc. Biol. Soc. Washington, 25:9-10, January 23, 1912.
1920. Mammals of Panamá. Smithsonian Miscl. Coll., 69(no. 5):1-309, pls. 1-39, 1 map.
Gray, J. E.
1844A. The zoölogy of the voyage of H. M. S. Sulphur, under the command of Captain Sir Edward Belcher, R. N., C. B., F. R. G. S., etc., during the years 1836-42. Mammalia, pp. 9-36, pls. 1-18.
1864. Description of a new Mustela from Quito. Proc. Zoöl. Soc. London, 1864:55, pl. 8.
1865. Revision of the genera and species of Mustelidae contained in the British Museum. Proc. Zoöl. Soc. London, 1865:100-154, 1 pl., 2 figs. in text.
1874. Notes on the varieties of the western-American weasels. Ann. and Mag. Nat. Hist., 14(ser. 4):374-375.
Green, C. V.
1936. Observations on the New York weasel. Jour. Mamm., 17:247-249, August 14, 1936.
Grinnell, J.
1913. A distributional list of the mammals of California. Proc. California Acad. Sci., 3(ser. 4):265-390, pls. 15, 16, August 28, 1913.
1917. Field tests or theories concerning distributional control. Amer. Nat., 51:115-128, February, 1917.
1917A. The niche-relationships of the California Thrasher. Auk, 34:427-433, October, 1917.
1922. A geographical study of the kangaroo rats of California. Univ. California Publ. Zoöl., 24:1-124, pls. 1-7, text figs. A-X, June 17, 1922.
1926. Geography and evolution in the pocket gopher. Univ. California Chronicle. July, 1926, pp. 247-262, 1 pl., 3 figs. in text.
Grinnell, J., and Dixon, J.
1919. Natural history of the ground squirrels of California. California State Comm. Hortic., Monthly Bull., 7:4 + 597-708, 5 col. plates, 30 figs. in text, January 27, 1919.
Grinnell, J., Dixon, J., and Linsdale, J. M.
1937. Fur-bearing mammals of California.... Univ. California Press, 2 vols., xii + 375 pp., pls. 1-7, figs. 1-138, xiv + 377-777 pp., pls. 8-13, figs. 139-345, July 22, 1937.
Grinnell, J., and Swarth, H. S.
1913. An account of the birds and mammals of the San Jacinto Area of southern California with remarks upon the behavior of geographic races on the margins of their habitats. Univ. California Publ. Zoöl., 10:197-406, pls. 6-10, 3 text figs., October 31, 1913.
Grosjean, M. S.
1942. A persistent weasel. Jour. Mamm., 23:443, December 30, 1942.
Gunn, C. K.
1932. Color and primeness in variable mammals. Amer. Nat, 66:546-559, 4 figs. in text, December, 1932.
Hadwen, S.
1929. Color changes in Lepus americanus and other animals. Pp. 1-12, pls. 1-5. Reprint, Canad. Jour. Research, 1:189-200, July, 1929.
Hall, E. R.
1926. Systematic notes on the subspecies of Bassariscus astutus with description of one new form from California. Univ. California Publ. Zoöl., 30:39-50, pls. 2 and 3, September 8, 1926.
1926A. Changes during growth in the skull of the rodent Otospermophilus grammurus beecheyi. Univ. California Publ. Zoöl., 21:355-404, 43 figs. in text, March 9, 1926.
1927. A new weasel from Louisiana. Proc. Biol. Soc. Washington, 40:193-194, December 2, 1927.
1930. Three new genera of Mustelidae from the Later Tertiary of North America. Jour. Mamm., 11:146-155, 2 pls., May 9, 1930.
1936. Mustelid mammals from the Pleistocene of North America with systematic notes on some Recent members of the genera Mustela, Taxidea and Mephitis. Carnegie Institution Publ., no. 473:41-119, 5 pls., 6 figs. in text, November 20, 1936.
1937. Mustela cicognanii, the short tailed weasel, incorrectly ascribed to Ohio. Amer. Midland Nat., 18:304, March, 1937.
1944. Classification of the ermines of eastern Siberia. Proc. California Acad. Sci., 23(ser. 4):555-560, 1 fig. in text, August 22, 1944.
1946. Mammals of Nevada. Univ. California Press, Berkeley, xi + 710, frontispiece, colored, 11 pls., 485 figs. in text, unnumbered silhouettes, July 1, 1946.
Hamilton, W. J., Jr.
1928. Weasels eat shrews. Jour. Mamm., 9:249-250, August 9, 1928.
1933. The weasels of New York. Amer. Midland Nat., 14:289-344, 2 maps, 3 figs., 4 pls., July, 1933.
Handley, C. O., Jr.
1949. Least weasel, prey of barn owl. Jour. Mamm., 30:431, November 14, 1949.
Harper, J.
1927. The mammals of the Okefinokee Swamp region of Georgia. Proc. Boston Soc. Nat. Hist., 38(7):191-396, pls. 4-7, March, 1927.
Heller, H. H.
1924. Peruvian pets. Natural History, 24:479-493, 14 figs. in text, July-August, 1924.
Henninger, W. F.
1921. Two mammals new for Ohio. Jour. Mamm., 2:239, November 29, 1921.
Hensel, R.
1881. Craniologische Studien. Nova Acta d. Ksl. Leop.-Carol. Deutschen Akad. d. Nat., 42:127-195, pls. 6-13, tables A-T.
Hodgson, B. H.
1841. Classified catalogue of mammals of Nepal (corrected to end of 1841, first printed in 1832). Jour. Asiatic Soc. Bengal, 10:907-916.
Hollister, N.
1913. A synopsis of the American minks. Proc. U. S. Nat. Mus., 44:471-480, April 18, 1913.
1914. Descriptions of four new mammals from tropical America. Proc. Biol. Soc. Washington, 28:141-144, July 10, 1914.
Holmes, S. J.
1925. "Age and Area" in relation to extinction. Science, N. S., 61:77-79, January 23, 1925.
Howard, W. J.
1935. Apparently neutral relations of weasel and squirrel. Jour. Mamm., 16:322-323, November 15, 1935.
Howell, A. B.
1943. An apparent mustelid trait. Jour. Mamm., 24:98-99, February 20, 1943.
Huxley, J. S.
1939. Ecology and taxonomic differentiation. Jour. Ecology, 27:408-420, August, 1939.
Ingles, L. G.
1939. Observations on a nest of the long-tailed weasel. Jour. Mamm., 20:253-254, May 14, 1939.
1942. Observations on the short-tailed weasel in California. Jour. Mamm., 23:446-448, 1 pl., December 30, 1942.
Jackson, H. H. T.
1913. Two new weasels from the United States. Proc. Biol. Soc. Washington, 26:123-124, May 21, 1913.
Jäckel, A. J.
1873. Ueber Leucismen unter den Mustelen. Der Zool. Garten, 14:456-459.
Jaeckel, O.
1902. Uber verschiedene Wege phylogenetischer Entwicklung [original not seen—only pp. 289-290 of V. L. Kellogg's Darwinism today].
Kellogg, V. L.
1907. Darwinism today. Henry Holt and Co., New York, xii + 403 pp.
Kennicott, R.
1859. The quadrupeds of Illinois, injurious and beneficial to the farmer. Agric. Rept. for 1858, U. S. Patent Office Rept., pp. 241-256.
Kirk, G. L.
1921. Shrews and weasels. Jour. Mamm., 2:111, May 2, 1921.
Lantz, D. E.
1905. A list of Kansas mammals. Trans. Kansas Acad. Sci., 19:171-178.
Leche, W.
1915. Zur Frage nach der stammesgeschichtlichen Bedeutung des Milchgebisses bei den Säugetieren. Zool. Jahrb., Abt. f. Syst., 38:275-370, 126 figs. in text.
Leopold, A.
1937. Killing technique of the weasel. Jour. Mamm., 18:98-99, February 14, 1937.
Lichtenstein, M. H. C.
1832. Darstellung neuer oder wenig bekannter Säugethiere, pl. 42 and corresponding text (unpaged), 1832 [not seen].
Llewellyn, L. M.
1942. Notes on the Alleghenian least weasel in Virginia. Jour. Mamm., 23:439-441, December 30, 1942.
Long, W. S.
1938. The weasel an enemy of the pika. Jour. Mamm., 19:250, May 14, 1938.
Lönnberg, E.
1913. Mammals from Ecuador and related forms. Arkiv för Zool., 8(no. 1):1-36, 1 pl., 1 text fig., July 12, 1913.
1921. A second contribution to the mammalogy of Ecuador with some remarks on Caenolestes. Arkiv för Zool., 14(no. 4):1-104, pl. 1, 8 figs. in text.
Lyman, C. P.
1943. Control of coat color in the varying hare Lepus americanus Erxleben. Bull. Mus. Comp. Zoöl., 93:393-461, 11 pls., December, 1943.
Lyon, M. W., Jr., and Osgood, W. H.
1909. Catalogue of the type-specimens of mammals in the United States National Museum, including the Biological Survey Collection. U. S. Nat. Mus. Bull., 62: ix + 325.
Macgillivary, W.
1848? The naturalists library, Vol. XVII. Mammalia, pp. 1-309, J. Ogden and Co., London.
Manniche, A. L. V.
1910. The terrestrial mammals and birds of northeast Greenland. Meddelelser om Grønland, 45:1-200, 6 pls., 20 figs. in text, 1910.
Manning, T. H.
1943. Notes on the mammals of south and central west Baffin Island. Jour. Mamm., 24:47-59, 1 fig. in text, February 20, 1943.
Matthew, W. D.
1902. On the skull of Bunaelurus, a musteline from the White River Oligocene. Bull. Amer. Mus. Nat. Hist., 16:137-140, April 7, 1902.
Mearns, E. A.
1891. Description of a new species of weasel, and a new subspecies of the gray fox, from Arizona. Bull. Amer. Mus. Nat. Hist, 3:234-238, June 5, 1891.
Merriam, C. H.
1896. Synopsis of the weasels of North America. N. Amer. Fauna, 11:1-33, 5 pls., 16 figs. in text, June 30, 1896.
1902. Five new mammals from México. Proc. Biol. Soc. Washington, 15:67-69, March 22, 1902.
1906. Is mutation a factor in the evolution of higher vertebrates. Science, N. S., 23:241-257, February, 1906.
1919. Criteria for the recognition of species and genera. Jour. Mamm., 1:6-9, November 28, 1919.
Metchnikoff, E.
1901. On the process of hair turning white. Proc. Royal Soc. London, 69:156.
Miller, F. W.
1930. The spring molt of Mustela longicauda. Jour. Mamm., 11:471-473, November 11, 1930.
1931A. The fall moult of Mustela longicauda. Jour. Mamm., 12:150-156, 1 fig. in text, May 14, 1931.
1931B. A feeding habit of the long-tailed weasel. Jour. Mamm., 12:164, May 14, 1931.
Miller, G. S., Jr.
1912. Catalogue of the mammals of western Europe.... British Museum of Natural History, London, xv + 1019 pp., 213 figs. in text.
1924. List of North American Recent mammals, 1923. Bull. U. S. Nat. Mus., 128: xvi + 673 pp., April 29, 1924.
Moore, J. C.
1945. Life history notes on the Florida weasel. Proc. Florida Academy of Science, 7(no. 4):247-263, for 1944.
Morse, M.
1939. A local study of predation upon hares and grouse during the cyclic decimation. Jour. Wildlife Management, 3:203-211, July, 1939.
Mulaik, S.
1938. Notes on Mustela frenata frenata. Jour. Mamm., 19:104, February 14, 1938.
Murie, A.
1935. A weasel goes hungry. Jour. Mamm., 16:321-322, November 15, 1935.
Nares, G. S.
1877. Arctic Expedition/1875-6/Journals and Proceedings/of the/Arctic Expedition, 1875-6,/under the command of/Captain Sir George S. Nares, R. N., K. C. B./[in continuation of Parliamentary Papers C 1153 of 1875, and C 1560 of 1876.]/presented to both Houses of Parliament by Command of her Majesty, 1877./London:/printed for her Majesty's stationary office, by/Harrison & Sons, printers in ordinary to her majesty,/St. Martin's Lane,/[c.-1636.] Price £1 1s.
1878. Narrative of a voyage to the Polar Sea during 1875-6 in H. M. ships "Alert" and "Discovery" by Captn. Sir G. S. Nares, R. N.... commander of the expedition with notes on the Natural History edited by H. W. Feilden, F. G. S., ... naturalist to the expedition. In 2 vols., 2 ed., London, Sampson Low, Marston, Searle, & Rivington, 1878. Vol. 1, xl + 395 pp.; vol. 2, vii + 378 pp., both vols. illustrated.
Nelson, A. W.
1934. Notes on Wisconsin mammals. Jour. Mamm., 15:252-253, August 10, 1934.
Nelson, E. W.
1909. The rabbits of North America. N. Amer. Fauna, 29:1-314, 13 pls., 19 figs. in text, August 31, 1909.
1918. Smaller mammals of North America. Nat'l Geographic, 33(5):371-493, with abundant illustrations, many in color, May, 1918.
Nichols, D. G., and Nichols, J. T.
1935. Notes on the New York weasel (Mustela noveboracensis). Jour. Mamm., 16:297-299, November 15, 1935.
Noback, C. V.
1935. Observations on the seasonal hair moult in a New York State weasel (Mustela noveboracensis). Bull. New York Zoöl. Soc., 38(no. 1):25-27, February, 1935.
Oberthür, R., and Dauthenay, H.
1905. Répertoire de Couleurs. Rennes, Impr. Oberthür; Paris, Librairie horticole. 5p. 1., [9]-82p. illus., 3 pl. (2 col.) and 365 col. pl. in 2 portfolios.
Oehler, C.
1944. Notes on the temperament of the New York weasel. Jour. Mamm., 25:198, May 26, 1944.
Ognev, S. I.
1935. The mammals of USSR and adjacent countries, vol. 3: viii + 752 pp., 8 pls., 299 figs. in text, 19 maps, 52 tables. Moskow.
Osgood, F. L.
1935. Fluctuations in small mammal populations. Jour. Mamm., 16:156, May 15, 1935.
1936. Earthworms as a supplementary food for weasels. Jour. Mamm., 17:64, February 14, 1936.
Osgood, W. H.
1901. Natural history of the Queen Charlotte Islands, British Columbia; natural history of the Cook Inlet Region, Alaska. N. Amer. Fauna, 21:1-87, 8 pls., 1 fig. in text, September 26, 1901.
1909. Revision of the mice of the American genus Peromyscus. N. Amer. Fauna, 28:1-267, 8 pls., 12 figs. in text, April 17, 1909.
1909B. Biological Investigations in Alaska and Yukon Territory. N. Amer. Fauna, 30:1-96, 5 pls., 2 figs. in text, October 7, 1909.
1912. Mammals from western Venezuela and eastern Colombia. Field Mus. Nat. Hist., Publ. no. 155, zoöl. ser., 10:33-66, pls. 4, 5, January 10, 1912.
1914. Mammals of an expedition across northern Perú. Field Mus. Nat. Hist., Publ. 176, zoöl. ser., 10(no. 12):143-185, April 20, 1914.
Pearce, J.
1937. A captive New York weasel. Jour. Mamm., 18:483-488, November 14, 1937.
Pearson, K., Nettleship, E., and Usher, C. H.
1913. A monograph on albinism in man. Drapers Co., Research Memoirs, Biometric series, VIII, Dept. Applied Statistics, Univ. College, Univ. London. Text, P II, vii + 264-524 pp., London.
Pearson, O. P., and Pearson, A. K.
1947. Owl predation in Pennsylvania, with notes on the small mammals of Delaware County. Jour. Mamm., 28:137-147, June 1, 1947.
Pitt, F.
1921. Notes on the genetic behavior of certain characters in the polecat, ferret, and in polecat-ferret hybrids. Jour. Genetics, 11:99-115, 2 pls., 5 figs. in text, September, 1921.
Polderboer, E. B.
1942. Habits of the least weasel (Mustela rixosa) in northeastern Iowa. Jour. Mamm., 23:145-147, June 3, 1942.
1948. Late fall sexual activity in an Iowa least weasel. Jour. Mamm., 29:296, August 31, 1948.
Polderboer, E. B., Kuhn, L. W., and Hendrickson, G. O.
1941. Winter and spring habits of weasels in central Iowa. Jour. Wildlife Management, 5:115-119, 1 pl., January, 1941.
Preble, E. A.
1908. A biological investigation of the Athabasca-Mackenzie region. N. Amer. Fauna, 27:1-574, 25 pls., 13 figs. in text, October 26, 1908.
Quick, H. F.
1944. Habits and economics of the New York weasel in Michigan. Jour. Wildlife Management, 8:71-78, January, 1944.
Rhoads, S. N.
1894. Contributions to the mammalogy of Florida. Proc. Acad. Nat. Sci. Philadelphia, pp. 152-161, June 19, 1894.
Ridgway, R.
1912. Color standards and color nomenclature. Published by the author, Washington, D. C., iii + 43 pp., 53 pls.
Robinson, W., and Lyon, M. W.
1901. An annotated list of mammals collected in the vicinity of La Guaira, Venezuela. Proc. U. S. Nat. Mus., 24:135-162, October, 1901.
Rothschild, M.
1942. Change of pelage in the stoat Mustela erminea L. Nature, 149:78, January 17, 1942.
Russell, W. C.
1930. Weasel badly injured by king snake. Jour. Mamm., 11:504-505, November 11, 1930.
Sanderson, G. C.
1949. Growth and behavior of a litter of captive long-tailed weasels. Jour. Mamm., 30:412-415, 1 fig. in text, November 14, 1949.
Schlosser, M.
1888. Die Affen, Lemuren, Chiropteren, Insectivoren, Marsupialier, Creodonten und Carnivoren des Europäischen Tertiärs. Alfred Hölder, K. K. Hof.-und Univ.-Buchhändler, Wien. Part 2, pp. 225-386, pls. 6-9.
Schumacher, S.
1928. Wie kommt die stellenweise Gelbfärbung des winterweissen Wiesels (Mustela erminea L.) zustande? Zeitschr. f. Morph, u. Ökologie der Tiere, 11:229-234, 1 fig., July 3, 1928.
Schwalbe, G.
1893. Ueber den Farbenwechsel winterweisser Thiere. Morphologische Arbeiten, 2:483-600, Jena.
Seton, E. T.
1929. Lives of game animals. Vol. 2, 1929, xvii + 746 pp., illustrated; vol. 4, xxii + 949 pp., Doubleday, Doran & Co., New York.
Shaw, W. T.
1921. The nest of the Washington weasel (Mustela washingtoni). Jour. Mamm., 2:167-168, August 19, 1921.
Sheldon, W. G.
1932. Mammals collected or observed in the vicinity of Laurier Pass, B. C. Jour. Mamm., 13:196-203, August 9, 1932.
Simpson, G. G.
1946. Palaeogale and allied early mustelids. Amer. Mus. Novitates, 1320:1-14, 4 figs. in text, May 28, 1946.
Snyder, L. L., and Logier, E. B. S.
1930. No. 3. A faunal investigation of King Township, York County, Ontario. Trans. Royal Canadian Institute, 17(pt. 2):167-208.
Soper, J. D.
1919. Notes on Canadian weasels. Canadian Field-nat., 33:43-47, September, 1919.
1921. Curious palatal obstruction in Mustela longicauda. Jour. Mamm., 2:37-38, 1 fig. in text, February 10, 1921.
1942. Mammals of Wood Buffalo Park, northern Alberta and District of Mackenzie. Jour. Mamm., 23:119-145, 2 pls., 1 map in text, June 3, 1942.
1946. Mammals of the northern Great Plains along the International Boundary in Canada. Jour. Mamm., 27:127-153, 1 fig. in text, May 14, 1946.
1948. Mammal notes from the Grand Prairie-Peace River region, Alberta. Jour. Mamm., 29:49-64, 1 pl., 1 fig. in text, February 13, 1948.
Sowls, L. K.
1948. The Franklin ground squirrel, Citellus franklinii (Sabine), and its relationship to nesting ducks. Jour. Mamm., 29:113-137, 3 pls., 3 figs. in text, May 14, 1948.
Stanford, J. S.
1931. Notes on small mammals of Utah. Journ. Mamm., 12:356-363, November 11, 1931.
Stephens, F.
1906. California mammals. West Coast Publishing Co., San Diego, California, pp. 1-351, numerous figs. and maps.
Strecker, J. K.
1924. The mammals of McLennan County, Texas. The Baylor Bull., Baylor Univ., Waco, Texas, 27(no. 1):1-20, September, 1924.
1926. A check-list of the mammals of Texas exclusive of the Sirenia and Cetacea. The Baylor Bull., Baylor Univ., Waco, Texas, 29(no. 3):1-48, August, 1926.
Strong, W. D.
1930. Notes on mammals of the Labrador Interior. Jour. Mamm., 11:1-10, February 11, 1930.
Sumner, F. B.
1932. Genetic, distributional, and evolutionary studies of the subspecies of deer mice (Peromyscus). Bibliographia Genetica, 9:1-106, 24 figs. in text.
Surber, T.
1932. The mammals of Minnesota. Minnesota Dept. Conservation, Div. Game and Fish, Saint Paul, Minnesota, pp. 1-84, illustrated.
Sutton, G. K.
1929. The Alleghenian least weasel in Pennsylvania. Jour. Mamm., 252-254, 1 fig. in text, August 10, 1929.
Svihla, A.
1931. Habits of the New York weasel in captivity. Jour. Mamm., 12:67-68, February 12, 1931.
Swanson, G., and Fryklund, P. O.
1935. The least weasel in Minnesota and its fluctuation in numbers. Amer. Midland Nat., 16:120-126, figs. 1-6, 1935.
Swenk, M. H.
1926. Notes on Mustela campestris Jackson, and on the American forms of least weasels. Jour. Mamm., 7:313-330, 1 fig. in text, November 23, 1926.
Taczanowski, L.
1874. Description d' une nouvelle espéce de Mustela du Pérou central. Proc. Zool. Soc. London, 1874:311-312, pl. 48, May 19, 1874.
1881. Description d' une nouvelle belette du Pérou septentrional. Proc. Zool. Soc. London, 1881:647-649, May 17, 1881.
1881. Description d' une nouvelle espéce du genre Mustela du Pérou nord-oriental. Proc. Zool. Soc. London, 1881:835-836, November 15, 1881.
Tate, G. H. H.
1931. Random observations on habits of South American mammals. Jour. Mamm., 12:248-256, August 24, 1931.
Thomas, O.
1911. The mammals of the tenth edition of Linnaeus; an attempt to fix the types of the genera and the exact bases and localities of the species. Proc. Zoöl. Soc. London, 1911:120-158, March, 1911.
1920. Report on the Mammalia collected by Mr. Edmund Heller during the Peruvian expedition of 1915 under the auspices of Yale University and the National Geographic Society. Proc. U. S. Nat. Mus., 58:217-249, pls. 14-15.
Thurber, W. A.
1940. A weasel attacks a varying hare. Jour. Mamm., 21:356, August 14, 1940.
Tschudi, J. J.
1844. Untersuchungen über die Fauna Peruana. Therologie. Druck und verlag von Scheitlin und Zollikofer, St. Gallen, xxx + 262 pp., 18 pls.
Vestal, E. H.
1937. Activities of a weasel at a wood rat colony. Jour. Mamm., 18:364, August 14, 1937.
1938. Biotic relations of the wood rat (Neotoma fuscipes) in the Berkeley Hills. Jour. Mamm., 19:1-36, figs. 1-4, February 14, 1938.
Warren, E. R.
1924. Ground squirrels and weasels. Jour. Mamm., 5:265-266, November 15, 1924.
1932. When do weasels mate? Jour. Mamm., 13:71-72, February 9, 1932.
Wight, H. M.
1932. A weasel attacks a man. Jour. Mamm., 13:163-164, May 11, 1932.
Willis, J. C.
1922. Age and Area: A study in geographic distribution and origin of species. University Press, Cambridge, England, x + 259 pp., illustrated.
Winecoff, T. E.
1930. Least weasel in Pennsylvania. Jour. Mamm., 11:312-313, August, 1930.
Woldrich, J. N.
1884. Diluviale Fauna von Zuzlawitz bei Winterberg im Bohmerwalde. Dritter Theil.... Sitzungsberich. d. K. Akad. d. Wissen. Math.—Natur.—Classe, 88(heft 3, erste abth.):978-1057, 3 pls., 2 figs. in text.
Wright, P. L.
1942A. Delayed implantation in the long-tailed weasel (Mustela frenata), the short-tailed weasel (Mustela cicognani), and the marten (Martes americana). Anat. Rec., 83:341-353, 2 pls., July, 1942.
1942B. A correlation between the spring molt and spring changes in the sexual cycle in the weasel. Jour. Exp. Zoöl., 91:103-110, October, 1942.
1947. The sexual cycle of the male long-tailed weasel (Mustela frenata). Jour. Mamm., 28:343-352, 1 pl., 3 figs. in text, November 19, 1947.
1948A. Breeding habits of captive long-tailed weasels (Mustela frenata). Amer. Midland Nat., 39:338-344, March, 1948.
1948B. Preimplantation stages in the long-tailed weasel (Mustela frenata). Anat. Rec., 100:595-607, 2 pls., April, 1948.
1950. Effects of gonadotropic hormone on the pelage of white winter long-tailed weasels; abstract. Anat. Rec., 106(no. 2):130, February, 1950.
Zimmermann, K.
1943. Zur Kenntnis deutscher Maus-und Zwerg-Wiesel. Zeitschr. fur Säugetierkunde, 15(3):289-298, 2 pls., 2 figs. in text, 3 tables, January 11, 1943.
Transmitted May 22, 1950.
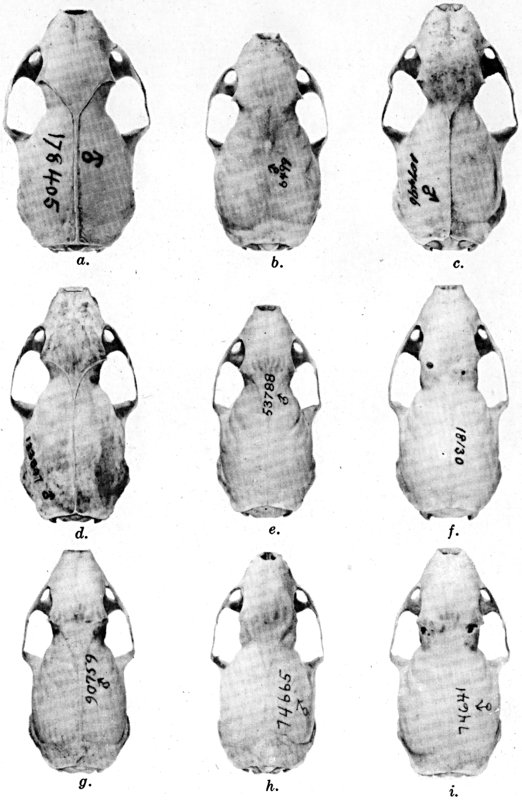
Plate 2. Photographs, retouched, of skulls in dorsal view of nine subspecies of Mustela erminea. Natural size.
a. Mustela erminea arctica, ♂ ad., 178405, U. S. Nat. Mus., Tanana, Alaska.
b. Mustela erminea semplei, ♂ sad., 6499, Carnegie Mus., Southhampton Island.
c. Mustela erminea kadiacensis, ♂ ad., 107496, U. S. N. M., Kodiak Island, Alaska.
d. Mustela erminea richardsonii, ♂ ad., 133847, U. S. N. M., Ft. Franklin, MacK.
e. Mustela erminea cicognanii, ♂ ad., 53788, Mus. Vert. Zoöl., Lopez, Pennsylvania.
f. Mustela erminea bangsi, ♂ ad., 18130, Field Mus. Nat. Hist., Aitkin, Minn.
g. Mustela erminea invicta, ♂ ad., 90759, Mus. Vert. Zoöl., Pilot Creek, Idaho.
h. Mustela erminea alascensis, ♂ ad., 74665, Mus. Vert. Zoöl., Windham, Alaska.
i. Mustela erminea salva, ♂ ad., 74641, M. V. Z., Mole Harbor, Admiralty Id., Alaska.
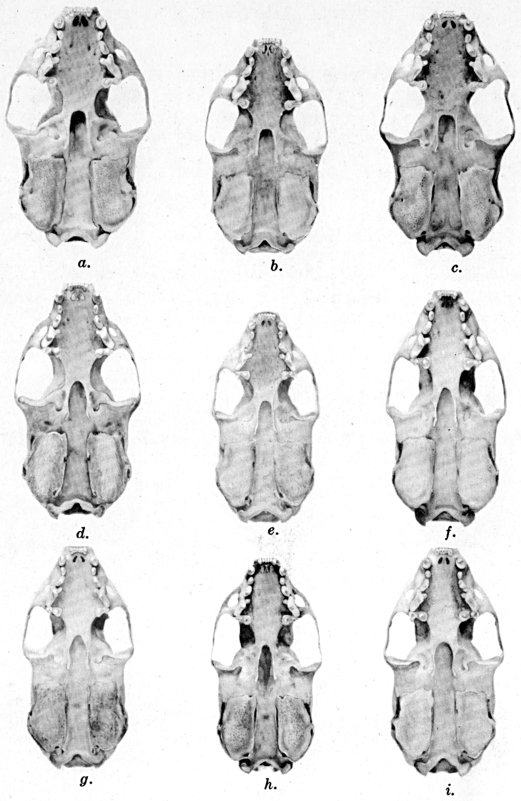
Plate 3. Photographs, retouched, of skulls in ventral view of nine subspecies of Mustela erminea. Natural size.
a. Mustela erminea arctica, ♂ ad., 178405, U. S. Nat. Mus., Tanana, Alaska.
b. Mustela erminea semplei, ♂ sad., 6499, Carnegie Mus., Southampton Island.
c. Mustela erminea kadiacensis, ♂ ad., 107496, U. S. N. M., Kodiak Island, Alaska.
d. Mustela erminea richardsonii, ♂ ad., 133847, U. S. N. M., Ft. Franklin, MacK.
e. Mustela erminea cicognanii, ♂ ad., 53788, Mus. Vert. Zoöl., Lopez, Pennsylvania.
f. Mustela erminea bangsi, ♂ ad., 18130, Field Mus. Nat. Hist., Aitkin, Minn.
g. Mustela erminea invicta, ♂ ad., 90759, Mus. Vert. Zoöl., Pilot Creek, Idaho.
h. Mustela erminea alascensis, ♂ ad., 74665, Mus. Vert. Zoöl., Windham, Alaska.
i. Mustela erminea salva, ♂ ad., 74641, M. V. Z., Mole Harbor, Admiralty Id., Alaska.
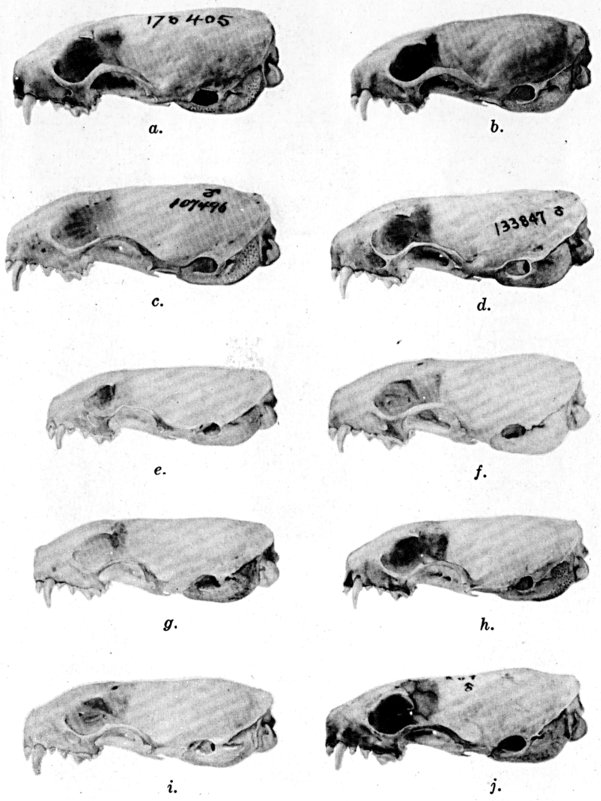
Plate 4. Photographs, retouched, of skulls in lateral view of ten subspecies Mustela erminea. Natural size.
a. Mustela erminea arctica, ♂ ad., 178405, U. S. Nat. Mus., Tanana, Alaska.
b. Mustela erminea semplei, ♂ sad., 6499, Carnegie Mus., Southampton Island.
c. Mustela erminea kadiacensis, ♂ ad., 107496, U. S. N. M., Kodiak Island, Alaska.
d. Mustela erminea richardsonii, ♂ ad., 133847, U. S. N. M., Ft. Franklin, MacK.
e. Mustela erminea cicognanii, ♂ ad., 53788, Mus. Vert. Zoöl., Lopez, Pennsylvania.
f. Mustela erminea bangsi, ♂ ad., 18130, Field Mus. Nat. Hist., Aitkin, Minn.
g. Mustela erminea invicta, ♂ ad., 90759, Mus. Vert. Zoöl., Pilot Creek, Idaho.
h. Mustela erminea alascensis, ♂ ad., 74665, Mus. Vert. Zoöl., Windham, Alaska.
i. Mustela erminea salva, ♂ ad., 74641, M. V. Z., Mole Harbor, Admiralty Id., Alaska.
j. Mustela erminea initis, ♂ ad., 289, Mus. Vert. Zoöl., Saook Bay, Alaska.
Plate 5. Photos, retouched, of skulls of 9 subspecies of Mustela erminea, × 1.
a. Mustela erminea initis, ♂ ad., 289, Mus. Vert. Zoöl., Saook Bay, Alaska.
b. Mustela erminea celenda, ♂ ad., 1053, Los Angeles Mus., Craig, Alaska.
c. Mustela erminea seclusa, ♂ ad., 31232, M. V. Z., Port Santa Cruz, Alaska.
d. Mustela erminea haidarum, ♂ ad., 230777, U. S. N. M., Graham Island, B. C.
e. Mustela erminea anguinae, ♂ ad., 13508, Nat. Mus. Canada, Cape Scott, V. I., B. C.
f. Mustela erminea fallenda, ♂ ad., 7096, Nat. Mus. Canada, Huntingdon, B. C.
g. Mustela e. olympica, ♂ ad., 90738, U. S. N. M., near head of Soleduc Riv., Wash.
h. Mustela erminea streatori, ♂ ad., 133, Coll. of Alex Walker, Blaine, Oregon.
i. Mustela erminea gulosa, ♂ ad., 82177, U. S. Nat. Mus., Trout Lake, Wash.
Plate 6. Photos, retouched, of skulls of 9 subspecies of Mustela erminea, × 1.
a. Mustela erminea initis, ♂ ad., 289, Mus. Vert. Zoöl., Saook Bay, Alaska.
b. Mustela erminea celenda, ♂ ad., 1053, Los Angeles Mus., Craig, Alaska.
c. Mustela erminea seclusa, ♂ ad., 31232, M. V. Z., Port Santa Cruz, Alaska.
d. Mustela erminea haidarum, ♂ ad., 230777, U. S. N. M., Graham Island, B. C.
e. Mustela erminea anguinae, ♂ ad., 13508, Nat. Mus. Canada, Cape Scott, V. I., B. C.
f. Mustela erminea fallenda, ♂ ad., 7096, Nat. Mus. Canada, Huntingdon, B. C.
g. Mustela e. olympica, ♂ ad., 90738, U. S. N. M., near head of Soleduc Riv., Wash.
h. Mustela erminea streatori, ♂ ad., 133, Coll. of Alex Walker, Blaine, Oregon.
i. Mustela erminea gulosa, ♂ ad., 82177, U. S. Nat. Mus., Trout Lake, Wash.
Plate 7. Photos, retouched, of skulls, of subspecies of Mustela erminea. Natural size.
a. Mustela erminea celenda, ♂ ad., 1053, Los Angeles Mus., Craig, Alaska.
b. Mustela erminea seclusa, ♂ ad., 31232, M. V. Z., Port Santa Cruz, Alaska.
c. Mustela erminea haidarum, ♂ ad., 230777, U. S. N. M., Graham Island, B. C.
d. Mustela erminea anguinae, ♂ ad., 13508, Nat. Mus. Canada, Cape Scott, V. I., B. C.
e. Mustela erminea fallenda, ♂ ad., 7096, Nat. Mus. Canada, Huntingdon, B. C.
f. Mustela erminea olympica, ♂ ad., 90738, U. S. N. M., near head of Soleduc Riv., Wash.
g. Mustela erminea streatori, ♂ ad., 133, Coll. of Alex Walker, Blaine, Oregon.
h. Mustela erminea gulosa, ♂ ad., 82177, U. S. Nat. Mus., Trout Lake, Wash.
i, j, k. Mustela erminea muricus, ♂ ad., 41501, M. V. Z., Baker Creek, 8675 ft., Nev.
l, m. Mustela erminea angustidens, ♂?, sad., 12437, A. M. N. H., Conard Fissure, Ark.
n. Mustela erminea angustidens, ♂?, ad., 12441, A. M. N. H., Conard Fissure, Ark.
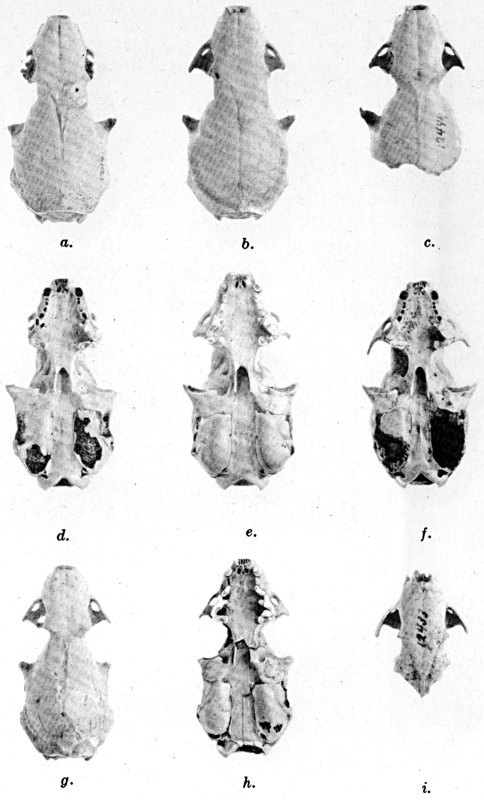
Plate 8. Photos, retouched, of Mustela erminea angustidens. All in Amer. Mus. Nat. Hist., from Conard Fissure, Arkansas. Pleistocene in age, × 1.
a. Adult, probably male, 12441.
b. Subadult, probably male, 12437.
c. Adult, probably male, 12444.
d. Adult, probably male, 12441.
e. Subadult, probably male, 12437.
f. Young, probably male, 12438.
g, h. Adult, type, probably female, 12432.
i. Adult, probably female, 12433.
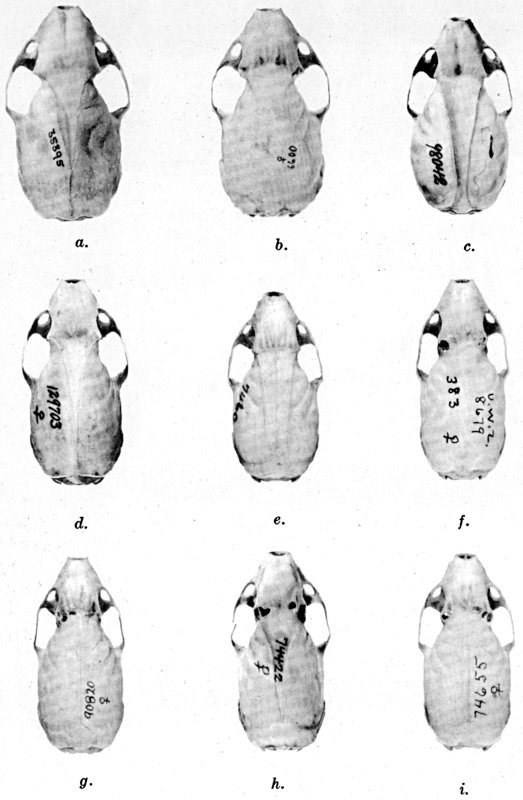
Plate 9. Photographs, retouched, of skulls in dorsal view of 9 subspecies of Mustela erminea. Natural size.
a. Mustela erminea arctica, ♀ ad., 35895, Field Mus. Nat. Hist., Point Barrow, Alaska.
b. Mustela erminea semplei, ♀ ad., 6600, Carnegie Mus., Southhampton Island.
c. Mustela erminea kadiacensis, ♀ ad., 98042, U. S. Nat. Mus., Kadiak, Alaska.
d. Mustela erminea richardsonii, ♀ ad., 129703, U. S. N. M., Fort Resolution, MacK.
e. Mustela erminea cicognanii, ♀ ad., 7460, Carnegie Mus., Pymatuning Swamp, Pa.
f. Mustela erminea bangsi, ♀ ad., 8679, Univ. Wisconsin, T. 61N, R. 26W, Minn.
g. Mustela erminea invicta, ♀ ad., 90820, M. V. Z., 1-1/2 mi. W Iron Mtn., Idaho.
h. Mustela erminea alascensis, ♀ ad., 74422, U. S. Nat. Mus., Juneau, Alaska.
i. Mustela erminea salva, ♀ ad., 74655, Mus. Vert. Zoöl., Mole Harbor, Alaska.
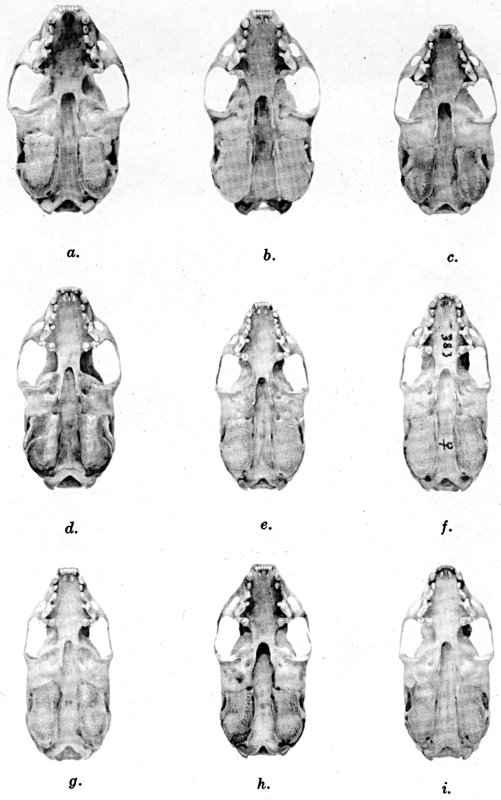
Plate 10. Photographs, retouched, of skulls in dorsal view of 9 subspecies of Mustela erminea. Natural size.
a. Mustela erminea arctica, ♀ ad., 35895, Field Mus. Nat. Hist., Point Barrow, Alaska.
b. Mustela erminea semplei, ♀ ad., 6600, Carnegie Mus., Southhampton Island.
c. Mustela erminea kadiacensis, ♀ ad., 98042, U. S. Nat. Mus., Kadiak, Alaska.
d. Mustela erminea richardsonii, ♀ ad., 129703, U. S. N. M., Fort Resolution, MacK.
e. Mustela erminea cicognanii, ♀ ad., 7460, Carnegie Mus., Pymatuning Swamp, Pa.
f. Mustela erminea bangsi, ♀ ad., 8679, Univ. Wisconsin, T. 61N, R. 26W, Minn.
g. Mustela erminea invicta, ♀ ad., 90820, M. V. Z., 1-1/2 mi. W Iron Mtn., Idaho.
h. Mustela erminea alascensis, ♀ ad., 74422, U. S. Nat. Mus., Juneau, Alaska.
i. Mustela erminea salva, ♀ ad., 74655, Mus. Vert. Zoöl., Mole Harbor, Alaska.
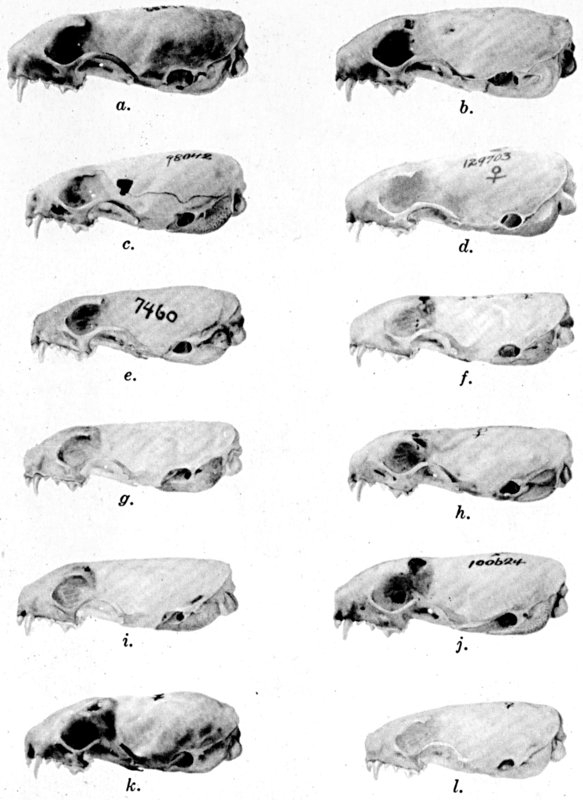
Plate 11. Photographs, retouched, of skulls in lateral view of twelve subspecies of Mustela erminea. Natural size.
a. Mustela erminea arctica, ♀ ad., 35895, Field Mus. Nat. Hist., Point Barrow, Alaska.
b. Mustela erminea semplei, ♀ ad., 6600, Carnegie Mus., Southhampton Island.
c. Mustela erminea kadiacensis, ♀ ad., 98042, U. S. Nat. Mus., Kadiak, Alaska.
d. Mustela erminea richardsonii, ♀ ad., 129703, U. S. Nat. Mus., Fort Resolution, MacK.
e. Mustela erminea cicognanii, ♀ ad., 7460, Carnegie Mus., Pymatuning Swamp, Pa.
f. Mustela erminea bangsi, ♀ ad., 8679, Univ. Wisconsin, T. 61N, R. 26W, Minn.
g. Mustela erminea invicta, ♀ ad., 90820, M. V. Z., 1-1/2 mi. W Iron Mtn., Idaho.
h. Mustela erminea alascensis, ♀ ad., 74422, U. S. Nat. Mus., Juneau, Alaska.
i. Mustela erminea salva, ♀ ad., 74655, Mus. Vert. Zoöl., Mole Harbor, Alaska.
j. Mustela erminea haidarum, ♀ ad., 100624, U. S. Nat. Mus., Moresby Island, B. C.
k. Mustela erminea anguinae, ♀ ad., 13673, Nat. Mus. Canada, Cape Scott, V. I., B. C.
l. Mustela erminea fallenda, ♀ ad., 7284, Nat. Mus. Canada, Huntingdon, B. C.

Plate 12. Photographs, retouched, of skulls in dorsal view of eight subspecies of Mustela erminea. Natural size.
a. Mustela erminea haidarum, ♀ ad., 100624, U. S. N. M., Moresby Island, B. C.
b. Mustela erminea anguinae, ♀ ad., 13673, N. M. Canada, Cape Scott, V. I., B. C.
c. Mustela erminea fallenda, ♀ ad., 7284, Nat. Mus. Canada, Huntingdon, B. C.
d. Mustela erminea olympica, ♀ ad., 242133, U. S. Nat. Mus., Hayes Creek, Wash.
e. Mustela erminea streatori, ♀ ad., 9040, D. R. Dickey Coll., Blaine, Oregon.
f. Mustela erminea gulosa, ♀ ad., 77370, U. S. Nat. Mus., Trout Lake, Wash.
g. Mustela erminea muricus, ♀ ad., 41502, Mus. Vert. Zoöl., Baker Creek, Nevada.
h. Mustela erminea angustidens, ♀?, ad., 12435, A. M. N. H., Conard Fissure, Ark.
i. Mustela erminea angustidens, ♀?, ad., 11766, A. M. N. H., Conard Fissure, Ark.
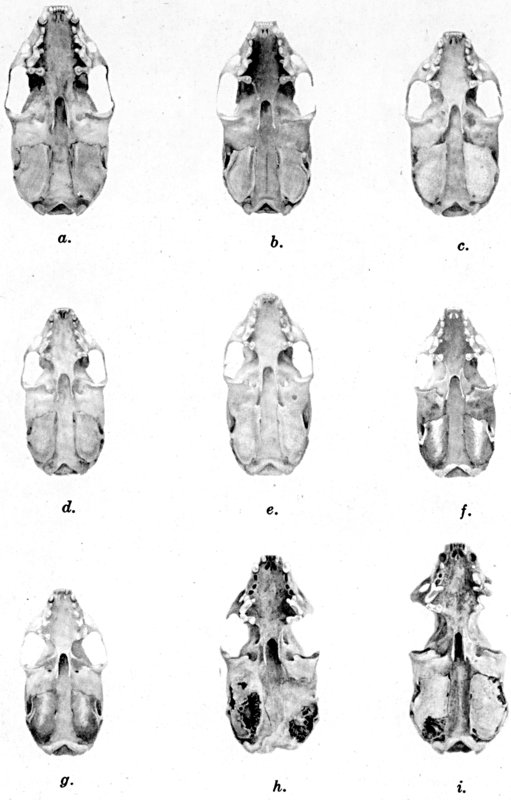
Plate 13. Photographs, retouched, of skulls in ventral view of eight subspecies of Mustela erminea. Natural size.
a. Mustela erminea haidarum, ♀ ad., 100624, U. S. N. M., Moresby Island, B. C.
b. Mustela erminea anguinae, ♀ ad., 13673, Nat. Mus. Canada, Cape Scott, B. C.
c. Mustela erminea fallenda, ♀ ad., 7284, Nat. Mus. Canada, Huntingdon, B. C.
d. Mustela erminea olympica, ♀ ad., 242133, U. S. Nat. Mus., Hayes Creek, Wash.
e. Mustela erminea streatori, ♀ ad., 9040, D. R. Dickey Coll., Blaine, Oregon.
f. Mustela erminea gulosa, ♀ ad., 77370, U. S. Nat. Mus., Trout Lake, Wash.
g. Mustela erminea muricus, ♀ ad., 41502, Mus. Vert. Zoöl., Baker Creek, Nevada.
h. Mustela erminea angustidens, ♀?, ad., 12435, A. M. N. H., Conard Fissure, Ark.
i. Mustela erminea angustidens, ♀?, ad., 11766, A. M. N. H., Conard Fissure, Ark.
Plate 14. Photographs, retouched, of M. erminea and M. minuta. Natural size.
a. Mustela erminea olympica, ♀ ad., 242133, U. S. Nat. Mus., Hayes Creek, Wash.
b. Mustela erminea streatori, ♀ ad., 9040, D. R. Dickey Coll., Blaine, Oregon.
c. Mustela erminea gulosa, ♀ ad., 77370, U. S. Nat. Mus., Trout Lake, Wash.
d. Mustela erminea muricus, ♀ ad., 41502, Mus. Vert. Zoöl., Baker Creek, Nevada.
e. Mustela erminea angustidens, adult, probably female, type, 12432, Amer. Mus. Nat. Hist., with lower jaw, Conard Fissure, Ark.
f. M. e. angustidens, ♀?, ad., 12435, A. M. N. H., Conard Fissure, Ark.
g. Mustela rixosa eskimo, ♂ sad., 43288, Mus. Vert. Zoöl., Barrow, Alaska.
h. Mustela rixosa eskimo, ♀ sad., 40059, Mus. Vert. Zoöl., Barrow, Alaska.
i. Mustela rixosa rixosa, ♂ ad., 11743, Nat. Mus. Canada, Shaunavon, Sask.
j. Mustela rixosa rixosa, ♀ ad., 12679, Nat. Mus. Canada, south of Shaunavon, Sask.
k. Mustela r. allegheniensis, ♂ ad., 35381, Field M. N. H., Portage Twp., Ohio.
l. Mustela rixosa allegheniensis, ♀ ad., 33021, Field M. N. H., Stryker, Ohio.
Plate 15. Photographs, retouched, of Mustela rixosa. Natural size.
a. Mustela rixosa eskimo, ♂ sad., 43288, Mus. Vert. Zoöl., Barrow, Alaska.
b. Mustela rixosa rixosa, ♂ ad., 11743, Nat. Mus. Canada, Shaunavon, Saskatchewan.
c. Mustela rixosa allegheniensis, ♂ ad., 33581, Field Mus. Nat. Hist., Portage Township, Wood County, Ohio.
d. Mustela rixosa campestris, ♂ ad., 261830, U. S. Nat. Mus., shore of Sand Lake, South Dakota.
e. Mustela rixosa eskimo, ♂ sad., same specimen shown in a.
f. Mustela rixosa rixosa, ♂ same specimen shown in b.
g. Mustela rixosa allegheniensis, ♂ ad., same specimen shown in c. c.
h. Mustela rixosa campestris, ♂ ad., same specimen shown in d.
i. Mustela rixosa eskimo, ♀ sad., 40059, Mus. Vert. Zoöl., Barrow, Alaska.
j. Mustela rixosa rixosa, ♀ ad., 12679, Nat. Mus. Canada, south of Shaunavon, Saskatchewan.
k. Mustela rixosa allegheniensis, ♀ ad., 33021, Field Mus. Nat. Hist., Stryker, Ohio.
l. Mustela rixosa allegheniensis, ♀ ad., same specimen shown in k.
m. Mustela rixosa eskimo, ♀ sad., same specimen shown in i.
n. Mustela rixosa rixosa, ♀ ad., same specimen shown in j.
o. Mustela rixosa campestris, ♂ ad., same specimen shown in d and h.
Plate 16. Photographs, retouched, of skulls in dorsal view of nine subspecies of Mustela frenata. Natural size.
a. Mustela frenata noveboracensis, ♂ ad., 77112, U. S. Nat. Mus., Wilmington, Massachusetts.
b. Mustela frenata occisor, ♂ ad., 7267, Mus. Comp. Zool., Moosehead Lake, Maine.
c. Mustela frenata primulina, ♂ ad., 3325, Mus. Nat. Hist., Univ. Kansas, Clinton, Kansas.
d. Mustela frenata arthuri, ♂ sad., 37515, Mus. Vert. Zoöl., type, Remy, Louisiana.
e. Mustela frenata olivacea, ♂ ad., 180802, U. S. Nat. Mus., type, Biological Surveys Collection, Autaugaville, Alabama.
f. Mustela frenata peninsulae, ♂ ad., 49387, Florida State Mus., Apopka, Florida.
g. Mustela frenata spadix, ♂ ad., 53745, Mus. Vert. Zoöl., Elk River, Minnesota.
h. Mustela frenata longicauda, ♂ ad., 15875, Amer. Mus. Nat. Hist., Red Deer, Alberta.
i. Mustela frenata oribasa, ♂ ad., 43817, Mus. Vert. Zoöl., Isaacs Lake, British Columbia.
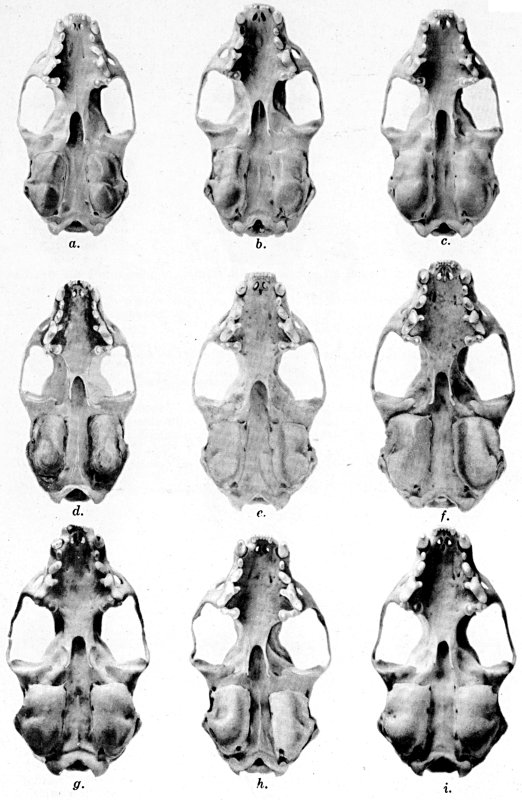
Plate 17. Photos, retouched, of skulls of males, in ventral view, of 9 subspecies of Mustela frenata, × 1. Data for a to i are given on Plate 18.
a. M. f noveboracensis
b. M. f. occisor
c. M. f. primulina
d. M. f. arthuri
e. M. f. olivacea
f. M. f. peninsulae
g. M. f. spadix
h. M. f. longicauda
i. M. f. oribasus
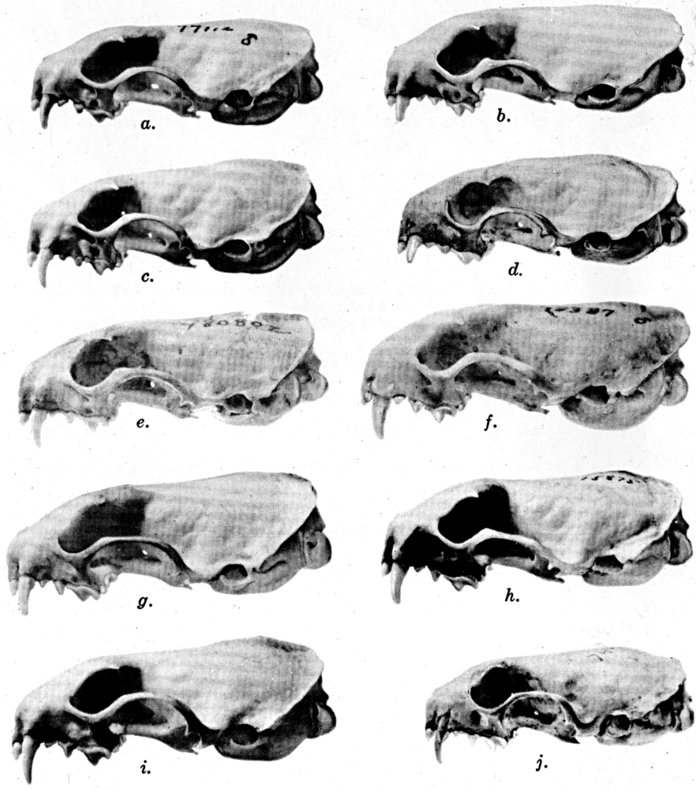
Plate 18. Photographs, retouched, of skulls in lateral view of ten subspecies of Mustela frenata. Natural size.
a. Mustela frenata noveboracensis, ♂ ad., 77112, U. S. Nat. Mus., Wilmington, Mass.
b. Mustela frenata occisor, ♂ ad., 7267, M. C. Z., Moosehead Lake, Maine.
c. Mustela frenata primulina, ♂ ad., 3325, Mus. Nat. Hist., Univ. Kansas, Clinton, Kans.
d. Mustela frenata arthuri, ♂ ad., 37515, Mus. Vert. Zoöl., type, Remy, Louisiana.
e. Mustela frenata olivacea, ♂ ad., 180802, U. S. Nat. Mus., type, Biological Surveys Collection, Autaugaville, Alabama.
f. Mustela frenata peninsulae, ♂ ad., 49387, Florida State Mus., Apopka, Florida.
g. Mustela frenata spadix, ♂ ad., 53795, Mus. Vert. Zoöl., Elk River, Minnesota.
h. Mustela frenata longicauda, ♂ ad., 15875, Amer. Mus. N. H., Red Deer, Alberta.
i. Mustela frenata oribasus, ♂ ad., 43817, Mus. Vert. Zoöl., Isaacs Lake, B. C.
j. Mustela frenata alleni, ♂ ad., 7440/9136, A. M. N. H., Hill City, S. D.
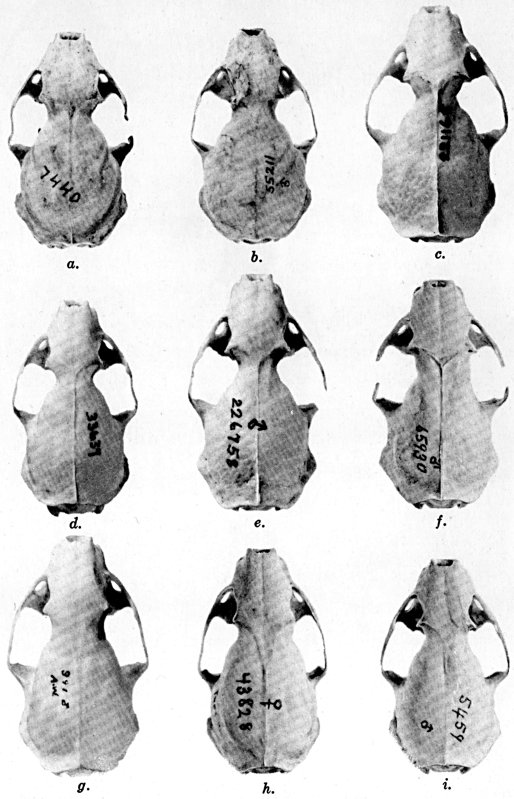
Plate 19. Photographs, retouched, of skulls in dorsal view of nine subspecies of Mustela frenata. Natural size.
a. Mustela frenata alleni, ♂ ad., 7440/9136, Amer. Mus. Nat. Hist., Hill City, South Dakota.
b. Mustela frenata arizonensis, ♂ ad., 55211, Mus. Vert. Zoöl., Government Prairie, Arizona.
c. Mustela frenata nevadensis, ♂ ad., 22116, Mus. Vert. Zoöl., Chinquapin, California.
d. Mustela frenata effera, ♂ ad., 33637, Amer. Mus. Nat. Hist., Ironside, Oregon.
e. Mustela frenata washingtoni, ♂ ad., 226758, U. S. Nat. Mus., Gotchen Creek, Washington.
f. Mustela frenata saturata, ♂ ad., 65930, U. S. Nat. Mus., type, Siskiyou, Oregon.
g. Mustela frenata altifrontalis, ♂ ad., 391, Coll. Alex Walker, Blaine, Oregon.
h. Mustela frenata oregonensis, ♂ sad., 43828/32019, U. S. Nat. Mus., Grants Pass, Oregon.
i. Mustela frenata munda, ♂ ad., 5459, Mus. Comp. Zool., type, Point Reyes, California.

Plate 20. Photos, retouched, of skulls of males in ventral view of 9 subspecies of Mustela frenata, × 1. Data for a to i on Plates 18 and 21.
a. M. f. alleni
b. M. f. arizonensis
c. M. f. nevadensis
d. M. f. effera
e. M. f. washingtoni
f. M. f. saturata
g. M. f. altifrontalis
h. M. f. oregonensis
i. M. f. munda, 5459.
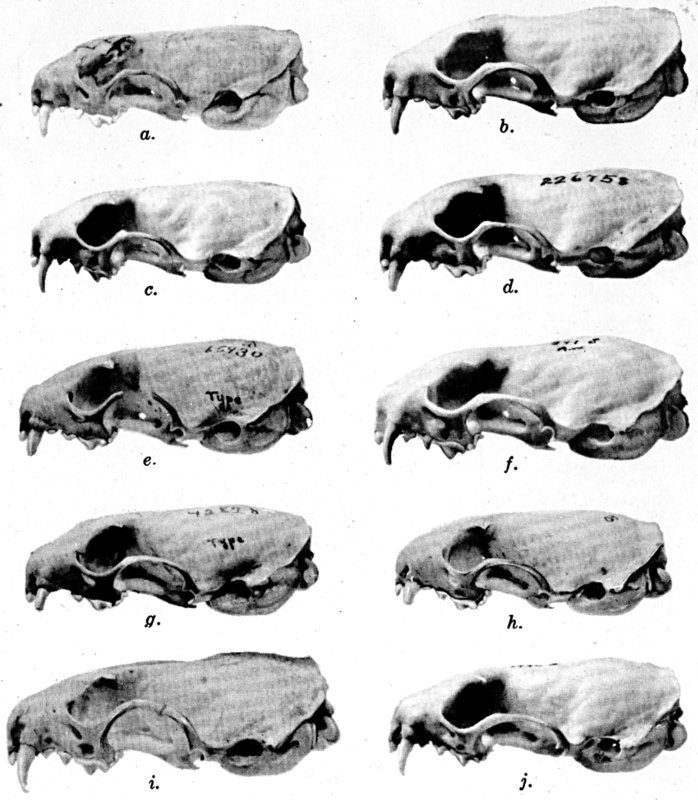
Plate 21. Photographs, retouched, of skulls, in lateral view, of nine subspecies of Mustela frenata. Natural size.
a. Mustela frenata arizonensis, ♂ ad., 55211, Mus. Vert. Zoöl., Government Prairie, Ariz.
b. Mustela frenata nevadensis, ♂ ad., 22116, Mus. Vert. Zoöl., Chinquapin, California.
c. Mustela frenata effera, ♂ ad., 33637, Amer. Mus. Nat. Hist., Ironside, Oregon.
d. Mustela frenata washingtoni, ♂ ad., 226758, U. S. Nat. Mus., Gotchen Creek, Wash.
e. Mustela frenata saturata, ♂ ad., 65930, U. S. Nat. Mus., type, Siskiyou, Oregon.
f. Mustela frenata altifrontalis, ♂ ad., 391, Coll. Alex Walker, Blaine, Oregon.
g. Mustela frenata oregonensis, ♂ sad., 43828/32019, U. S. N. M., Grants Pass, Ore.
h. Mustela frenata munda, ♂ ad., 5459, M. C. Z., type, Point Reyes, Calif.
i. Mustela frenata munda, ♂ ad., 19722, Mus. Vert. Zoöl., Point Arena, Calif.
j. Mustela frenata xanthogenys, ♂ ad., 1440, Coll. Alex Walker, 5 mi. W Fresno, Calif.
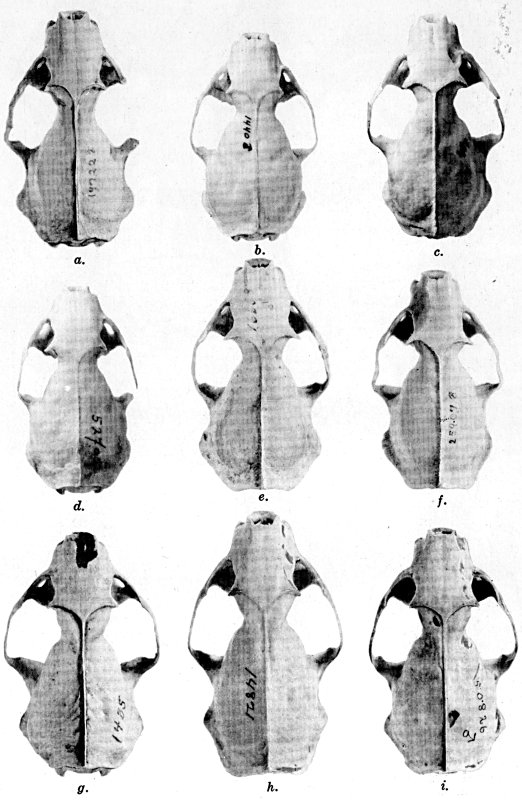
Plate 22. Photographs, retouched, of skulls, in dorsal view, of nine subspecies of Mustela frenata. Natural size.
a. Mustela frenata munda, ♂ ad., 19722, Mus. Vert. Zoöl., Point Arena, California.
b. Mustela frenata xanthogenys, ♂ ad., 1440, col. Alex Walker, 5 mi. W Fresno, California.
c. Mustela frenata nigriauris, ♂ ad., 487, Stanford Univ., Palo Alto, California.
d. Mustela frenata latirostra, ♂ ad., 52702, U. S. Nat. Mus., El Cajon, California.
e. Mustela frenata pulchra, ♂ ad., 16668, Mus. Vert. Zoöl., type, Buttonwillow, California.
f. Mustela frenata inyoensis, ♂ ad., 25907, Mus. Vert. Zoöl., 2 mi. N Independence, California.
g. Mustela frenata neomexicana, ♂ ad., 1485, Mus. Nat. Hist., Univ., Kansas, Liberal, Kansas.
h. Mustela frenata texensis, ♂ ad., 14821, Amer. Mus. Nat. Hist., Kerr County, Texas.
i. Mustela frenata frenata, ♂ ad., 50826, U. S. Nat. Mus., Tlalpam, México, D. F.
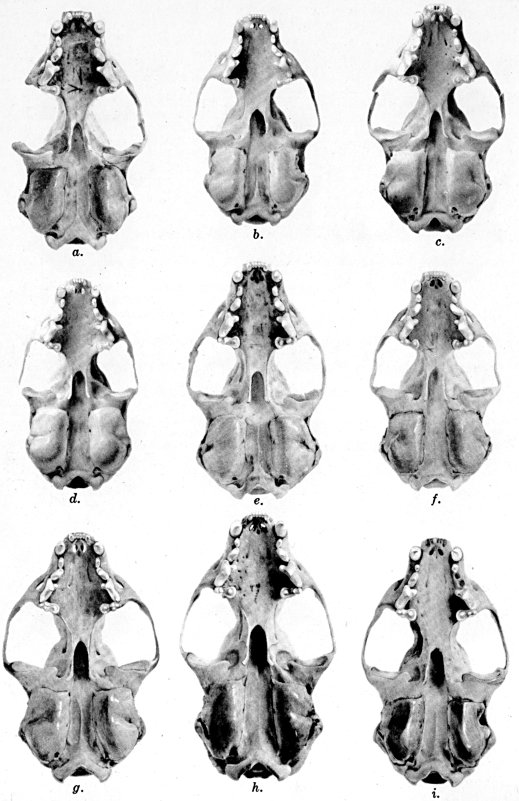
Plate 23. Ventral views of same skulls shown in Plate 22.
a. munda;
b. xanthogenys;
c. nigriauris;
d. latirostra;
e. pulchra;
f. inyoensis;
g. neomexicanus;
h. texensis;
i. frenata.
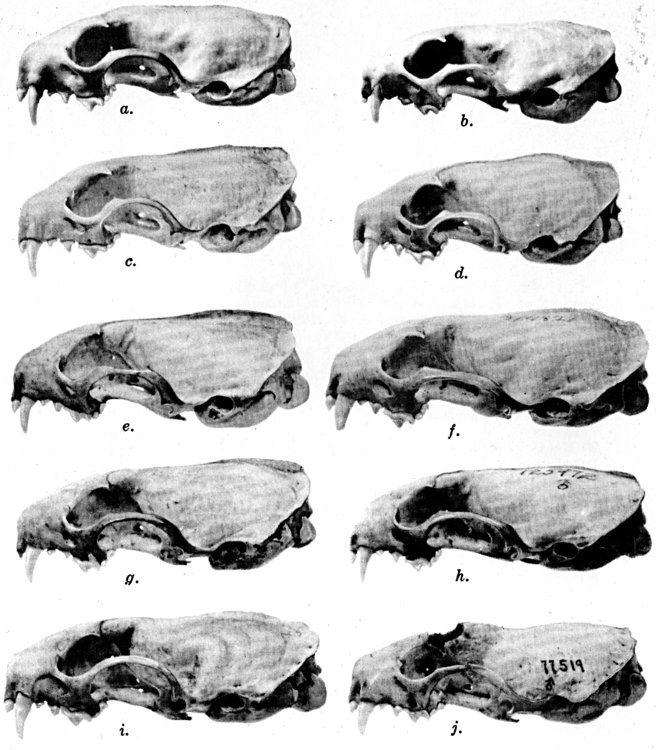
Plate 24. Photographs, retouched, of skulls, in lateral view, of ten subspecies of Mustela frenata. Natural size.
a. Mustela frenata nigriauris, ♂ ad., 487, Stanford Univ., Palo Alto, California.
b. Mustela frenata latirostra, ♂ ad., 52702, U. S. Nat. Mus., El Cajon, California.
c. Mustela frenata pulchra, ♂ ad., 16668, Mus. Vert. Zoöl., type, Buttonwillow, Calif.
d. Mustela frenata inyoensis, ♂ ad., 25907, Mus. Vert. Zoöl., type, 2 mi. N Independence, California.
e. Mustela frenata neomexicana, ♂ ad., 1485, M. N. H., Univ. Kansas, Liberal, Kansas.
f. Mustela frenata texensis, ♂ ad., 14821, A. M. N. H., Kerr County, Texas.
g. Mustela frenata frenata, ♂ ad., 50826, U. S. Nat. Mus., Tlalpam, México, D. F.
h. Mustela frenata leucoparia, ♂ ad., 125972, U. S. N. M., Los Reyes, Michoacán.
i. Mustela frenata macrophonius, ♂ ad., [14063,] Field Mus. N. H., type, Achotal, Veracruz.
j. Mustela frenata goldmani, ♂ ad., 77519, U. S. Nat. Mus., Pinabete, Veracruz.

Plate 25. Photographs, retouched, of skulls, in dorsal view of nine subspecies of Mustela frenata. Natural Size.
a. Mustela frenata leucoparia, ♂ ad., 125972, U. S. Nat. Mus., Los Reyes, Michoacán.
b. Mustela frenata macrophonius, ♂ ad., 14063, Field Mus. Nat. Hist., type, Achotal, Veracruz.
c. Mustela frenata goldmani, ♂ ad., 133253, U. S. Nat. Mus., 20 mi. SE Teopisca, Chiapas.
d. Mustela frenata tropicalis, ♂ ad., 54994, U. S. Nat. Mus., type, Jico, Veracruz.
e. Mustela frenata perda, ♂ sad., 100041, U. S. Nat. Mus., type, Teapa, Tabasco.
f. Mustela frenata nicaraguae, ♂ sad., 30754, Amer. Mus. Nat. Hist., type, Matagalpa, Nicaragua.
g. Mustela frenata costaricensis, ♂ ad., 3.2.1.6., British Mus. Nat. Hist., San José, Costa Rica.
h. Mustela frenata panamensis, ♂ ad., 18848, Amer. Mus. Nat. Hist., Boquete, Panamá.
i. Mustela frenata meridana, ♂ ad., 123341, U. S. Nat. Mus., type, Mérida, Venezuela.
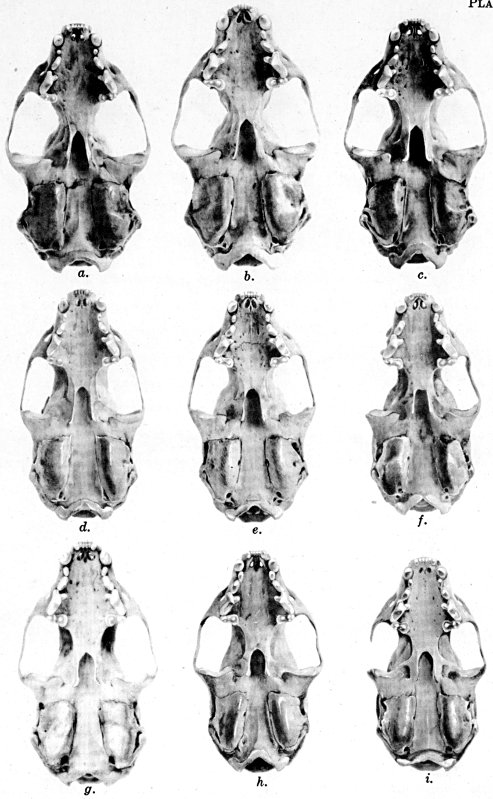
Plate 26. Ventral views of same skulls shown in Plate 25.
a. leucoparia;
b. macrophonius;
c. goldmani;
d. tropicalis;
e. perda;
f. nicaraguae;
g. costaricensis;
h. panamensis;
i. meridana.
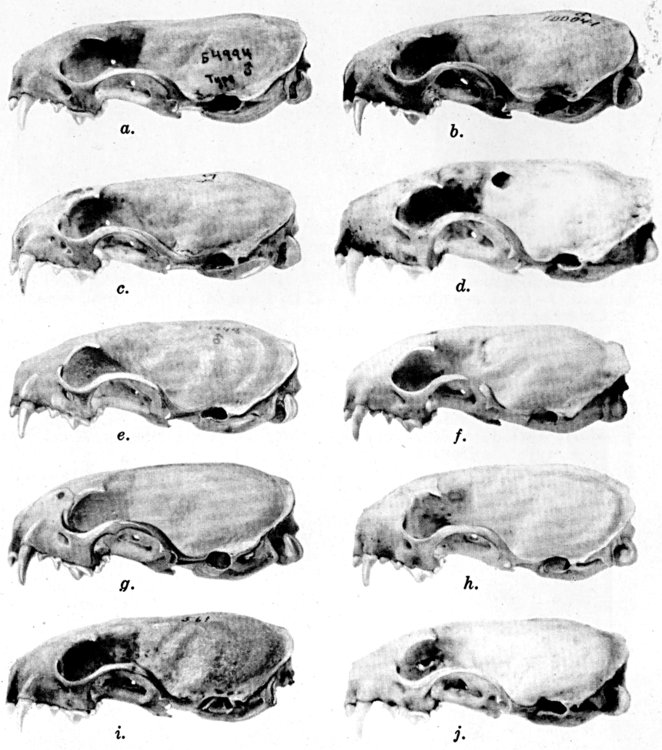
Plate 27. Photographs, retouched, of skulls, in lateral view of ten subspecies of Mustela frenata. Natural size.
a. Mustela frenata tropicalis, ♂ ad., 54994, U. S. Nat. Mus., type, Jico, Veracruz.
b. Mustela frenata perda, ♂ sad., 100041; U. S. Nat. Mus., type, Teapa, Tabasco.
c. Mustela frenata nicaraguae, ♂ sad., 30754, Amer. Mus. Nat. Hist., type, Matagalpa, Nicaragua.
d. Mustela frenata costaricensis, ♂ ad., 3.2.1.6., British Mus. Nat. Hist., San José, Costa Rica.
e. Mustela frenata panamensis, ♂ ad., 18848, Amer. Mus. Nat. Hist., Boquete, Panamá.
f. Mustela frenata meridana, ♂ ad., 24309, Amer. Mus. Nat. Hist., Mérida, Venezuela.
g. Mustela frenata aureoventris, ♂ yg., 34677, Amer. Mus. Nat. Hist., Gualea, Ecuador.
h. Mustela frenata helleri, ♂ ad., 24133, Field Mus. Nat. Hist., type, Rio Chinchao, Perú.
i. Mustela frenata macrura, ♂ ad., 561, Mus. Polonais d' Hist., Nat., type, Junín, Perú.
j. Mustela frenata agilis, ♂ ad., 8.1.10.1., British Mus. Nat. Hist., Lima, Perú.
Plate 28. Photographs, retouched (except f), of skulls, in dorsal view of nine kinds (species and subspecies) of Mustela. Natural size.
a. Mustela frenata aureoventris, ♂ yg., 34677, Amer. Mus. Nat. Hist., Gualea, Ecuador.
b. Mustela frenata helleri, ♂ ad., 24133, Field Mus. Nat. Hist., type, Rio Chinchao, Perú.
c. Mustela frenata macrura, ♂ ad., 561, Mus. Polonais d' Hist. Nat., type, Junín, Perú.
d. Mustela frenata agilis, ♂ ad., 8.1.10.1., British Mus. Nat. Hist., Lima, Perú.
e. Mustela frenata boliviensis, ♂ ad., 72587, Amer. Mus. Nat. Hist., type, Nequejahuira, Bolivia.
f. Mustela frenata xanthogenys, ♂ ad., 43.6.4.55., British Mus., Nat. Hist., type, California.
g. Mustela frenata costaricensis, ♂ yg., 37149, U. S. Nat. Mus., type, San José, Costa Rica.
h. Mustela frenata panamensis, ♂ yg., 178970, U. S. Nat. Mus., Mt. Pirre, Panamá.
i. Mustela africana africana, ♂ yg., 37475, Amer. Mus. Nat. Hist., Pará, Brazil.
Plate 29. Photographs, retouched, of skulls, in ventral view, of nine kinds (species and subspecies) of Mustela. Natural size.
a. Mustela frenata aureoventris, ♂ yg., 34677, Amer. Mus. Nat. Hist., Gualea, Ecuador.
b. Mustela frenata helleri, ♂ ad., 24133, Field Mus. Nat. Hist., type, Rio Chinchao, Perú.
c. Mustela frenata macrura, ♂ ad., 561, Mus. Polonais d' Hist. Nat., type, Junín, Perú.
d. Mustela frenata agilis, ♂ ad., 8.1.10.1., British Mus. Nat. Hist., Lima, Perú.
e. Mustela frenata boliviensis, ♂ ad., 72587, Amer. Mus. Nat. Hist., type, Nequejahuira, Bolivia.
f. Mustela frenata leucoparia, ♂ ad., 47179/34914, U. S. Nat. Mus., type, Pátzcuaro, Michoacán.
g. Mustela frenata costaricensis, ♂ yg., 37149, U. S. Nat. Mus., type, San José, Costa Rica.
h. Mustela frenata panamensis, ♂ yg., 178970, U. S. Nat. Mus., Mt. Pirre, Panamá.
i. Mustela africana africana, ♂ yg., 37475, Amer. Mus. Nat. Hist., Pará, Brazil.
Plate 30. Photographs, retouched (except e and q) of skulls and lower jaws of Mustela.
a. Mustela frenata boliviensis, ♂ ad., 72587, Amer. Mus. Nat. Hist., type, Nequejahuira, Bolivia.
b. Mustela frenata xanthogenys, ♂ ad., 43.4.6.55, British Mus. Nat. Hist., type, California.
c. Mustela frenata costaricensis, ♂ yg., 37149, U. S. Nat. Mus., type, San José, Costa Rica.
d. Mustela frenata panamensis, ♂ yg., 178970, U. S. Nat. Mus., Mt. Pirre, Panamá.
e. Mustela frenata affinis, ♂ ad., 54.6.3.4, British Mus. Nat. Hist., type, New Granada [= Colombia].
f. Mustela africana africana, ♂ yg., 37475, Amer. Mus. Nat. Hist., Pará, Brazil.
g. Mustela frenata saturata, ♂ ad., 65930, U. S. Nat. Mus., type, Siskiyou, Oregon.
h. Mustela frenata oregonensis, ♂ ad., 43828/32019, U. S. Nat. Mus., type, Grants Pass, Oregon.
i. Mustela frenata munda, ♂ ad., 5459, Mus. Comp. Zool., type, Point Reyes, California.
j. Mustela frenata leucoparia, ♂ ad., 47179/34914, U. S. Nat. Mus., type, Pátzcuaro, Michoacán.
k. Mustela frenata macrophonius, ♂ ad., 14963, Field Mus. Nat. Hist., type, Achotal, Veracruz.
l. Mustela frenata goldmani, ♂ ad., 77519, U. S. Nat. Mus., type, Pinabete, Chiapas.
m. Mustela frenata tropicalis, ♂ ad., 54994, U. S. Nat. Mus., type, Jico, Veracruz.
n. Mustela frenata perda, ♂ sad., 100041, U. S. Nat. Mus., type, Teapa, Tabasco.
o. Mustela frenata nicaraguae, ♂ sad., 30754, Amer. Mus. Nat. Hist., type, Matagalpa, Nicaragua.
p. Mustela frenata costaricensis, ♂ yg., 37149, U. S. Nat. Mus., type, San José, Costa Rica.
q. Mustela frenata affinis, ♂ ad., 54.6.3.4, British Mus. Nat. Hist., type, New Granada [= Colombia].
r. Mustela frenata macrura, ♂ ad., 561., Mus. Polonais d' Hist. Nat., type, Junín, Perú.
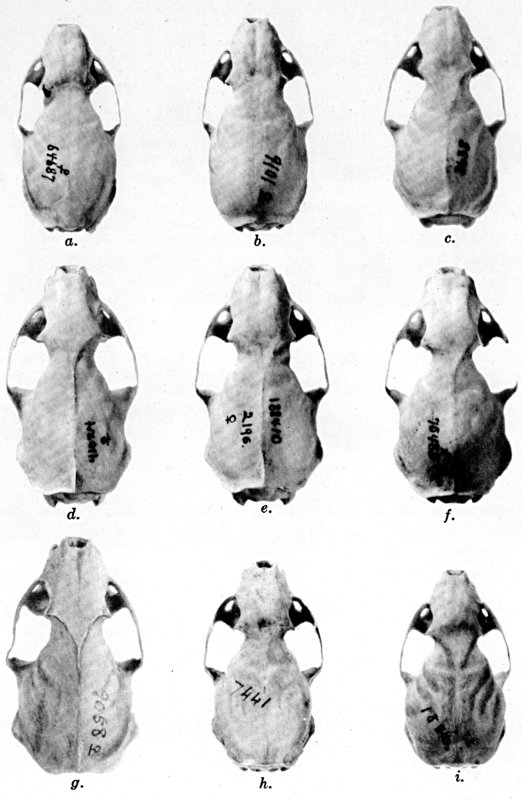
Plate 31. Photos, retouched, of skulls of 9 subspecies of M. frenata, x 1.
a. Mustela frenata noveboracensis, ♀ ad., 64687, U. S. N. M., Wilmington, Mass.
b. Mustela frenata occisor, ♀ ad., 9101, Mus. Comp. Zool., Bucksport, Maine.
c. Mustela frenata primulina, ♀ ad., 3638, U. K. M. N. H., 7 mi. SW Lawrence, Kans.
d. Mustela frenata olivacea, ♀ ad., 41024, Mus. Vert. Zoöl., Sinkola Plantation, Ga.
e. Mustela frenata spadix, ♀ ad., 188410, U. S. Nat. Mus., Elk River, Minn.
f. Mustela frenata longicauda, ♀ ad., 75483, U. S. Nat. Mus., Wingard, Sask.
g. Mustela frenata oribasus, ♀ ad., 9058, M. C. Z., type, source of Kettle River, B. C.
h. Mustela frenata alleni, ♀?, ad., 7441, A. M. N. H., Black Hills, S. D.
i. Mustela frenata arizonensis, ♀ ad., 1886, A. M. N. H., type, S. F. Forest, Ariz.
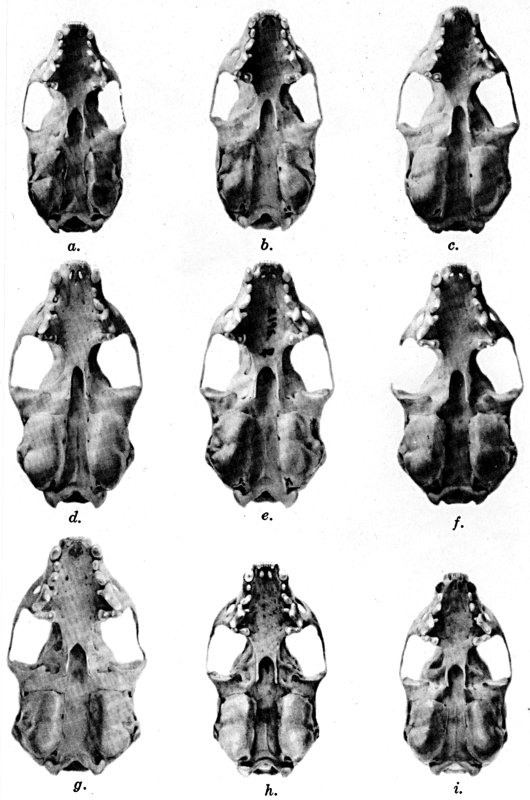
Plate 32. Photos, retouched, of skulls of 9 subspecies of M. frenata, × 1.
a. Mustela frenata noveboracensis, ♀ ad., 64687, U. S. N. M., Wilmington, Mass.
b. Mustela frenata occisor, ♀ ad., 9101, Mus. Comp. Zool., Bucksport, Maine.
c. Mustela frenata primulina, ♀ ad., 3638, U. K. M. N. H., 7 mi. SW Lawrence, Kans.
d. Mustela frenata olivacea, ♀ ad., 41024, Mus. Vert. Zoöl., Sinkola Plantation, Ga.
e. Mustela frenata spadix, ♀ ad., 188410, U. S. Nat. Mus., Elk River, Minn.
f. Mustela f. longicauda, ♀ ad., 75483, U. S. Nat. Mus., Wingard, Sask.
g. Mustela frenata oribasus, ♀ ad., 9058, M. C. Z., type, source of Kettle Riv., B. C.
h. Mustela frenata alleni, ♀?, ad., 7441, A. M. N. H., Black Hills, S. D.
i. Mustela frenata arizonensis, ♀ ad., 1886, A. M. N. H., type, S. F. Forest, Ariz.
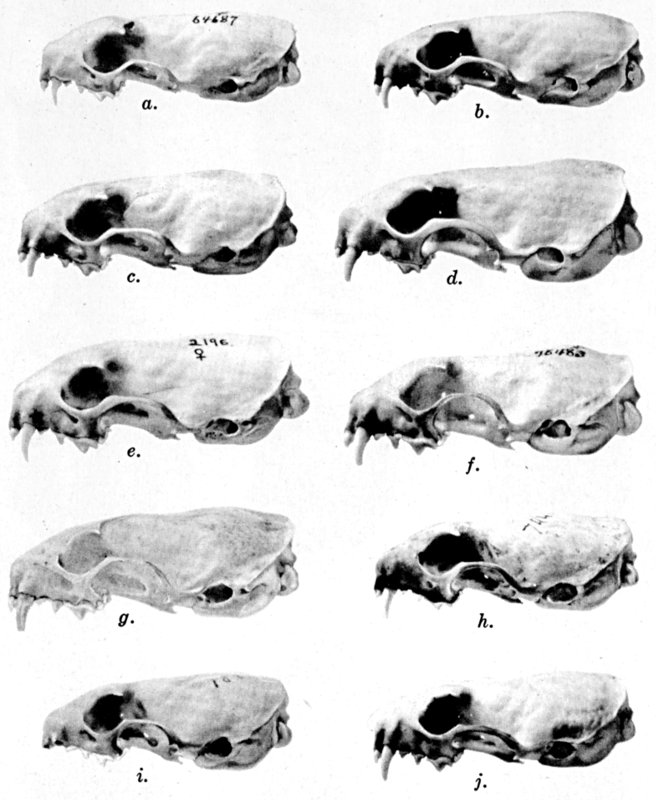
Plate 33. Photographs, retouched, of skulls in lateral view of ten subspecies of Mustela frenata. Natural size.
a. Mustela frenata noveboracensis, ♀ ad., 64687, U. S. N. M., Wilmington, Mass.
b. Mustela frenata occisor, ♀ ad., 9101, Mus. Comp. Zool., Bucksport, Maine.
c. Mustela frenata primulina, ♀ ad., 3638, Univ. Kansas Mus. Nat. Hist., 7 mi. SW Lawrence, Kansas.
d. Mustela frenata olivacea, ♀ ad., 41024, Mus. Vert. Zoöl., Sinkola Plantation, Ga.
e. Mustela frenata spadix, ♀ ad., 188410 (2196), U. S. Nat. Mus., Elk River, Minn.
f. Mustela frenata longicauda, ♀ ad., 75483, U. S. Nat. Mus., Wingard, Sask.
g. Mustela frenata oribasus, ♀ ad., 9058, Mus. Comp. Zool., type, source of Kettle River, British Columbia.
h. Mustela frenata alleni, ♀?, ad., 7441, Amer. Mus. N. H., Black Hills, S. D.
i. Mustela frenata arizonensis, ♀ ad., 1886, Amer. Mus. Nat. Hist., type, San Francisco, Forest, Arizona.
j. Mustela frenata nevadensis, ♀ ad., 41503, M. V. Z., type, 3 mi. E Baker, Nev.
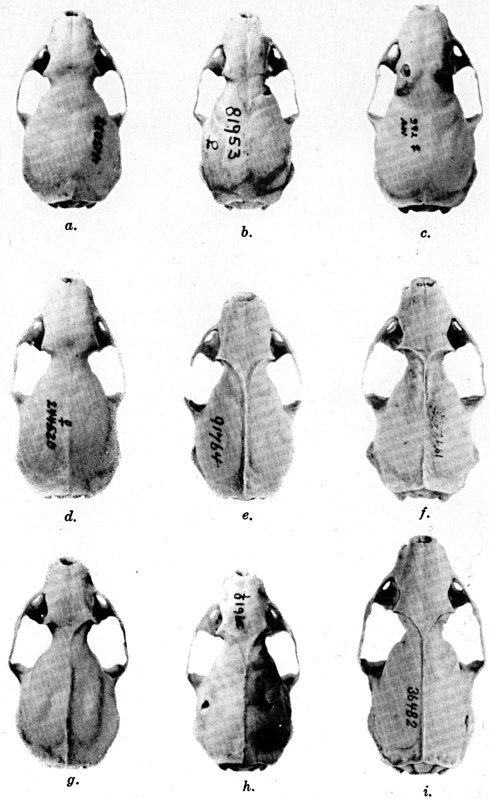
Plate 34. Photographs, retouched, of skulls in dorsal view, of eight subspecies of Mustela frenata. Natural size.
a. Mustela frenata nevadensis, ♀ ad., 41503, M. V. Z., type, 3 mi. E Baker, Nevada.
b. Mustela frenata washingtoni, ♀ sad., 81953, U. S. N. M., Trout Lake, Wash.
c. Mustela frenata altifrontalis, ♀ ad., 392, coll. of Alex Walker, Blaine, Oregon.
d. Mustela frenata oregonensis, ♀ ad., 244520, U. S. Nat. Mus., Medford, Oregon.
e. Mustela frenata munda, ♀ ad., 91764, U. S. Nat. Mus., Point Reyes, California.
f. Mustela frenata munda, ♀ ad., 19723, Mus. Vert. Zoöl., Point Arena, California.
g. Mustela frenata xanthogenys, ♀ ad., 2626, coll. of W. E. Snyder, Selma, California.
h. Mustela frenata nigriauris, ♀ ad., 3761, M. V. Z., San Francisco, California.
i. Mustela frenata neomexicana, ♀ ad., 36482, U. S. N. M., Tombstone, Arizona.
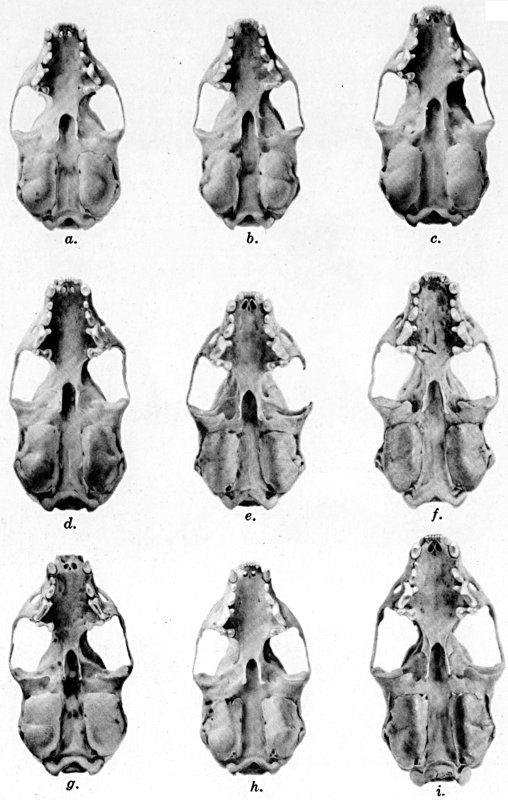
Plate 35. Photographs, retouched, of skulls in ventral view, of eight subspecies of Mustela frenata. Natural size.
a. Mustela frenata nevadensis, ♀ ad., 41503, M. V. Z., type, 3 mi. E Baker, Nevada.
b. Mustela frenata washingtoni, ♀ sad., 81953, U. S. N. M., Trout Lake, Wash.
c. Mustela frenata altifrontalis, ♀ ad., 392, coll. of Alex Walker, Blaine, Oregon.
d. Mustela frenata oregonensis, ♀ ad., 244520, U. S. Nat. Mus., Medford, Oregon.
e. Mustela frenata munda, ♀ ad., 91764, U. S. N. M., Point Reyes, California.
f. Mustela frenata munda, ♀ ad., 19723, M. V. Z., Point Arena, California.
g. Mustela frenata xanthogenys, ♀ ad., 2626, coll. of W. E. Snyder, Selma, Calif.
h. Mustela frenata nigriauris, ♀ ad., 3761, M. V. Z., San Francisco, California.
i. Mustela frenata neomexicana, ♀ ad., 36482, U. S. N. M., Tombstone, Arizona.
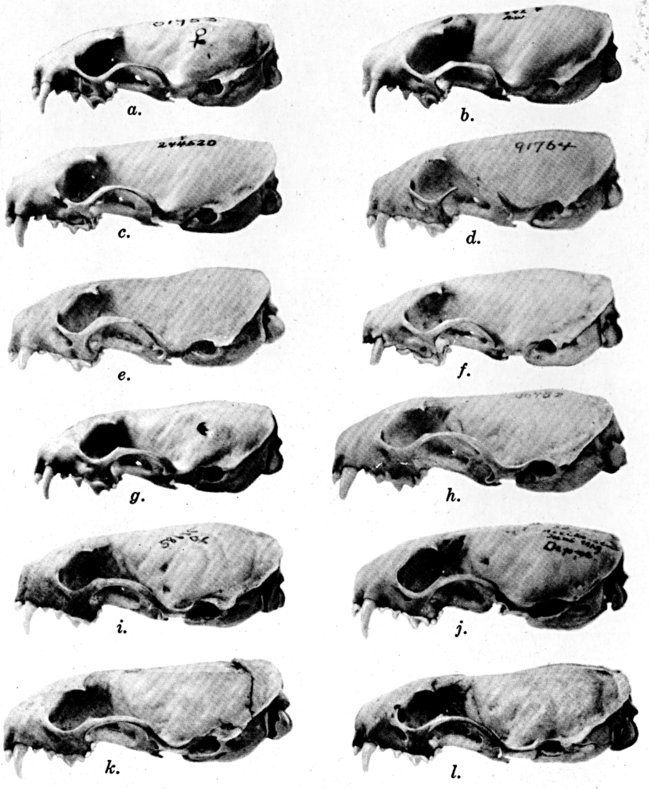
Plate 36. Photographs, retouched, of skulls in lateral view, of eight subspecies of Mustela frenata. Natural size.
a. Mustela frenata washingtoni, ♀ sad., 81953, U. S. N. M., Trout Lake, Wash.
b. Mustela frenata altifrontalis, ♀ ad., 392, coll. of Alex Walker, Blaine, Oregon.
c. Mustela frenata oregonensis, ♀ ad., 244520, U. S. N. M., Medford, Oregon.
d. Mustela frenata munda, ♀ ad., 91764, U. S. N. M., Point Reyes, California.
e. Mustela frenata munda, ♀ ad., 19723, Mus. Vert. Zoöl., Point Arena, Calif.
f. Mustela frenata xanthogenys, ♀ ad., 2626, coll. of W. E. Snyder, Selma, Calif.
g. Mustela frenata nigriauris, ♀ ad., 3761, Mus. Vert. Zoöl., San Francisco, Calif.
h. Mustela frenata neomexicana, ♀ ad., 36482, U. S. N. M., Tombstone, Ariz.
i. Mustela frenata frenata, ♀ ad., 58685, U. S. Nat. Mus., Brownsville, Texas.
j. Mustela frenata frenata, ♀ ad., 991, Berlin Zool. Mus., type, México City, D. F.
k. Mustela frenata leucoparia, ♀ ad., 26153, Amer. Mus. N. H., Artenkiki, Jalisco.
l. Mustela frenata perotae, ♀ ad., 54278, U. S. Nat. Mus., type, 12500 ft., Cofre de Perote, Veracruz.
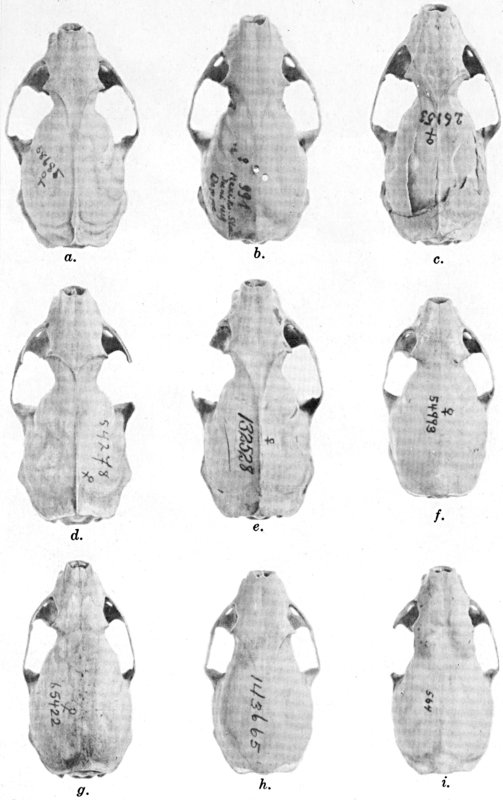
Plate 37. Photos, retouched, of skulls of 8 subspecies of Mustela frenata, × 1.
a. Mustela frenata frenata, ♀ ad., 58685, U. S. Nat. Mus., Brownsville, Texas.
b. Mustela frenata frenata, ♀ ad., 991, Berlin Zool. Mus., type, México City, D. F.
c. Mustela frenata leucoparia, ♀ ad., 26153, Amer. Mus. Nat. Hist., Artenkiki, Jalisco.
d. Mustela f. perotae, ♀ ad., 54278, U. S. N. M., type, Cofre de Perote, Veracruz.
e. Mustela frenata macrophonius, ♀ ad., 132528, U. S. Nat. Mus., Pérez, Veracruz.
f. Mustela frenata tropicalis, ♀ ad., 54993, U. S. Nat. Mus., Jico, Veracruz.
g. Mustela frenata perda, ♀ sad., 65422, U. S. Nat. Mus., Catemaco, Veracruz.
h. Mustela frenata meridana, ♀ ad., 143665, U. S. N. M., Mérida, Venezuela.
i. Mustela f. macrura, ♀ ad., 564, M. P. H. N., type of Mustela jelskii, Cutervo, Perú.
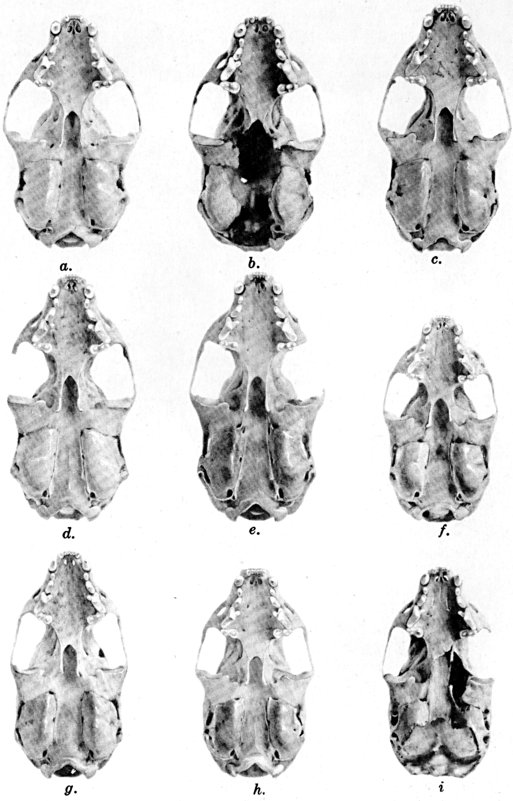
Plate 38. Photos, retouched, skulls in ventral view, 8 subspecies of M. frenata, × 1.
a. Mustela frenata frenata, ♀ ad., 58685, U. S. Nat. Mus., Brownsville, Texas.
b. Mustela frenata frenata, ♀ ad., 991, Berlin Zool. Mus., type, México City, D. F.
c. Mustela frenata leucoparia, ♀ ad., 26153, Amer. Mus. Nat. Hist., Artenkiki, Jalisco.
d. Mustela f. perotae, ♀ ad., 54278, U. S. N. M., type, Cofre de Perote, Veracruz.
e. Mustela frenata macrophonius, ♀ ad., 132528, U. S. Nat. Mus., Pérez, Veracruz.
f. Mustela frenata tropicalis, ♀ ad., 54993, U. S. Nat. Mus., Jico, Veracruz.
g. Mustela frenata perda, ♀ sad., 65422, U. S. Nat. Mus., Catemaco, Veracruz.
h. Mustela frenata meridana, ♀ ad., 143665, U. S. N. M., Mérida, Venezuela.
i. Mustela f. macrura, ♀ ad., 564, M. P. H. N., type of Mustela jelskii, Cutervo, Perú.
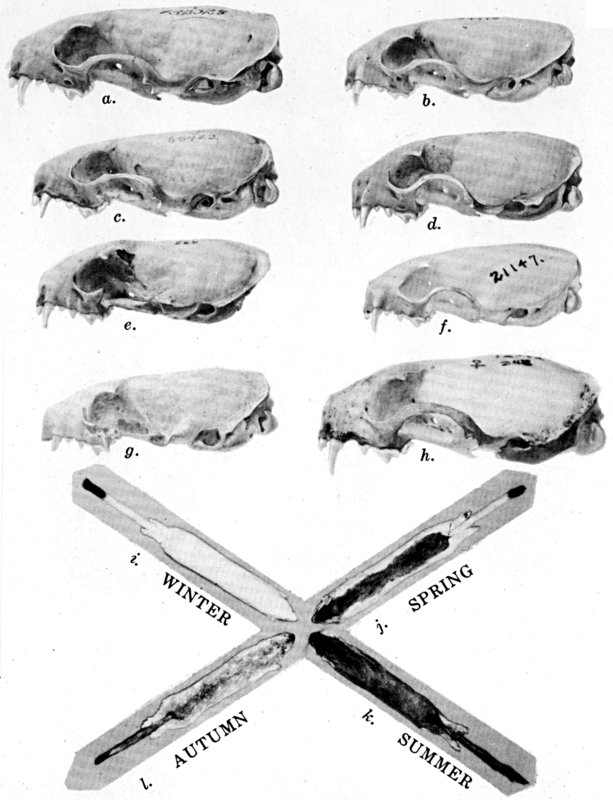
Plate 39. Figs. a-h. Photos, retouched, of 10 kinds Mustela, × 1.
a. Mustela frenata macrophonius, ♀ ad., 132528, U. S. Nat. Mus., Pérez, Veracruz.
b. Mustela frenata tropicalis, ♀ ad., 54993, U. S. Nat. Mus., Jico, Veracruz.
c. Mustela frenata perda, ♀ sad., 65422, U. S. Nat. Mus., Catemaco, Veracruz.
d. Mustela frenata meridana, ♀ ad., 143665, U. S. Nat. Mus., Mérida, Venezuela.
e. Mustela f. macrura, ♀ ad., 564, M. P. H. N., type of Mustela jelskii, Cutervo, Perú.
f. Mustela frenata agilis, ♀ sad., 21147, Field Mus. Nat. Hist., Macate, Perú.
g. Mustela frenata gracilis, ♀?, ad., 12431, Amer. Mus. Nat. Hist., type, Conard Fissure, Arkansas, Pleistocene.
h. Mustela a. stolzmanni, ♀ sad., 24.12.12.24, Brit. M. N. H., Myobamba, Perú.
Figs. i-l. Mustela frenata nevadensis, all males, from Colorado, showing seasonal change in color, × approximately 1/9. Note the sharply marked molt line in the pelage of spring and the absence of any definite molt line in autumn.
i. No. 151415 U. S. Nat. Mus., Coventry, December 27, 1907.
j. No. 202741 U. S. Nat. Mus., Pierce Place, April 18, 1913.
k. No. 201681 U. S. Nat. Mus., Jefferson, June 23, 1913.
l. No. 41997 Amer. Mus. Nat. Hist., Navajo River, October 29, 1913.
Plate 40. Photos, retouched, of skulls and lower jaws of Mustela, × 1.
a. Mustela frenata agilis, ♀ sad., 21147, Field Mus. Nat. Hist., Macate, Perú.
b. Mustela frenata gracilis, ♀?, ad., type, 12431, A. M. N. H., Conard Fiss., Ark.
c. Mustela africana stolzmanni, ♀ sad., 24.12.12.24, Brit. M. N. H., Myobamba, Perú.
d. Mustela frenata agilis, ♀ sad., 21147, Field Mus. Nat. Hist., Macate, Perú.
e. Mustela frenata gracilis, ♀?, ad., type, 12431, A. M. N. H., Conard Fiss., Ark.
f. Mustela africana stolzmanni, ♀ sad., 24.12.12.24, Brit. M. N. H., Myobamba, Perú.
g. Mustela africana stolzmanni, ♀ ad., 563, Mus. Polonais d' Hist. Nat., type, Yurimaguas, Perú. The palate is broken longitudinally and the two maxillae are slightly out of normal position.
h. Mustela frenata oribasus, ♀ ad., 9058, M. C. Z., type, source of Kettle Riv., B. C.
i. Mustela frenata munda, ♀ ad., 91764, U. S. Nat. Mus., Point Reyes, Calif.
j. Mustela frenata frenata, ♀ ad., 991, Berlin Z. M., type, México City, D. F.
k. Mustela f. macrura, ♀ ad., 564, Mus. Polonais d' Hist. Nat., type of Mustela jelskii.
l. Mustela africana stolzmanni, ♀ ad., 563, Mus. Polonais d' Hist. Nat., type, Yurimaguas, Perú. Right half of lower jaw reversed.

Plate 41. Photographs, approximately 1/2, of stuffed study-skins of the four species of American weasels. For each pair the male is at the left and the female at the right. Photo. by W. C. Matthews.
Mustela erminea arctica, both in U. S. B. S., from Mts. near Eagle, Alaska, ♂ 131256 and ♀ 131245.
Mustela erminea invicta, both in U. S. B. S., from Tungsten Mine, Washington, ♂ 235236 and ♀ 235235.
Mustela erminea muricus, both in M. V. Z., from Baker Creek, 8675 ft., Nevada, ♂ 41501 and ♀ 41502.
Mustela frenata nigriauris, both in M. V. Z., from California, ♂ 51666 from Concord and ♀ 73109 from Berkeley.
Mustela africana africana, ♂ 37475 A. M. N. H., from Pará, Brazil.
Mustela rixosa allegheniensis, both in M. Z. U. M., from Michigan, ♂ 83260 Swan Creek Farm and ♀ 88079 from Unadilla.
INDEX TO VOLUME 4
Principal references are in boldface type
aequatorialis,
Mustela, 75
Putorius, 75, 341, 387
aestuarina, Mustela, 82
affinis,
Mustela, 75, 375, 379, 384, 398, 409
Putorius, 372, 375, 379, 384
africana, Mustela, 73, 406, 409
agilis,
Mustela, 74, 393
Putorius, 74, 222
alascensis,
Mustela, 75, 131
Putorius, 75, 131
albigula, Neotoma, 208
allegheniensis,
Mustela, 77, 187
Putorius, 77, 187
alleni,
Mustela, 76, 274
Putorius, 76, 274
altifrontalis, Mustela, 79, 300
americana, 74, 75
americanus, Lepus, 93, 201, 210, 212, 216
anguinae, Mustela, 79, 145
angustidens,
Mustela, 78, 165
Putorius, 78, 165
Annelida, 93
arctica, Mustela, 76, 96
arcticus, Putorius, 76
arizonensis,
Mustela, 75, 276, 280, 291, 323
Putorius, 75, 276, 280
armatus, Citellus, 200
arthuri, Mustela, 78, 241
audax,
Mustela, 77
Putorius, 77
aureoventris, Mustela, 74, 387, 398
Aves, 93
bachmani, Sylvilagus, 213
bangsi, Mustela, 80, 124
barn owl, 173
Belding ground squirrel, 205
beldingi, Citellus, 205
big jumping mouse, 210
birds,
small, 216
wild, 93, 213
Blarina, 91, 205, 209, 210
brevicauda, 205
bobwhite, 213
boccamela, Mustela, 170
boliviensis, Mustela, 79, 402
bottae, Thomomys, 207
boylii, Lampropeltis, 213
brasiliensis,
Mustela, 73, 341, 372, 375
Putorius, 280, 300, 315, 319, 323, 384, 387, 398, 409
brevicauda, Blarina, 91, 205
brush rabbit, 213
bull snake, 213
Bunaelurus, 11
campestris, Mustela, 78, 190
cat, domestic, 174
celenda, Mustela, 80, 139
chicken, 93, 213, 216
chipmunk, 92, 196, 201, 206, 210, 216
cicognanii,
Mustela, 73, 110, 118, 124, 222
Putorius, 118, 124, 128, 145, 155, 161
cinereus, Sorex, 205
Citellus, 216
armatus, 200
beldingi, 205
franklini, 205
lateralis, 206
richardsonii, 205
townsendii, 205
tridecemlineatus, 205
Clethrionomys, 92
gapperi, 175, 181
Colaptes, 213
comadreja, 7
Condylura, 210
costaricensis, Mustela, 78, 372, 379, 387
cotton rat, 208
cottontail, 202, 203, 205, 212
Mearns, 208
coyote, 200
Cratogeomys, 65, 207
culbertsoni,
Mustela, 75
Putorius, 75
deer mouse, 208, 209, 216
domestic cat, 174
donnola, 7
[462] drummondi, Microtus, 175
earthworm, 93
effera, Mustela, 79, 291
energumenos, Mustela, 82
ermine, 87
erminea,
Mustela, 71
Putorius, 222
eskimo,
Mustela, 77, 181
Putorius, 77, 181
evagor, Mustela, 82
evergladensis, Mustela, 82
Evotomys, 175
fallenda, Mustela, 80, 148
Felis, 9
ferrets, 43
fish, 91, 93
flickers, 213
floridana, Neotoma, 208
flying squirrel, 206, 216
Franklin ground squirrel, 205
franklini, Citellus, 205
frenata, Mustela, 73, 232, 252, 280, 309, 338, 341
frenatus, Putorius, 280, 300, 315, 319, 323, 341, 351, 363, 366, 372, 384, 398
frog, leopard, 93
fulvus, Vulpes, 200
furo, Mustela, 43
fusca, Mustela, 222
fuscipes, Neotoma, 208
fuscus, Putorius, 74, 222
Gallus, 93, 213
gapperi, Clethrionomys, 175, 181
Geomyidae, 216
getulus, Lampropeltis, 213
Glaucomys, 206, 216
golden-mantled ground squirrel, 206
goldmani,
Mustela, 76, 355
Putorius, 76, 355
gracilis,
Mustela, 78, 404
Putorius, 78, 404
Grammogale, 407
grasshopper, 208
grasshopper mouse, 207, 216
great-horned owl, 173
ground squirrel, 210, 216
Belding, 205
Franklin, 205
golden-mantled, 206
Richardson, 205
thirteen-lined, 205
Townsend, 205
gulosa, Mustela, 80, 159
haidarum,
Mustela, 76, 142
Putorius, 76, 142
hare, varying, 210, 216
harvest mouse, 208, 216
helleri, Mustela, 79, 391
hispidus, Sigmodon, 208
horned lark, 209
house mouse, 216
hyemalis, Junco, 213
imperii, Putorius, 77, 110
ingens, Mustela, 82
initis, Mustela, 80, 136
insects, 176, 209, 216
intergrades, 45
invicta, Mustela, 80, 128
inyoensis, Mustela, 79, 331
javonica, Mustela, 72
jelskii, Mustela, 75, 398
jumping mouse, 216
big, 210
Junco hyemalis, 213
kadiacensis,
Mustela, 76, 108
Putorius, 76, 108
king-snake, 213
labiata, Mustela, 79
lacustris, Mustela, 82
Lampropeltis,
boylii, 213
getulus, 213
lark, horned, 209
lateralis, Citellus, 205
latimanus, Scapanus, 205
latirostra, Mustela, 79, 323
least weasel, 168, 209
lemming, 92
Lemmus, 92
Leopard frog, 93
lepida, Neotoma, 208
lepta, Mustela, 161
leptus,
Mustela, 161
Putorius, 78, 161
Lepus americanus, 93, 201, 210, 211, 212, 216
letifera, Mustela, 82
leucoparia,
Mustela, 76, 347
Putorius, 76, 347
lizards, 216
longicauda,
Mustela, 73, 232, 252, 262
Putorius, 280
long-tailed weasel, 193
lutensis, Mustela, 82
Lutra minor, 170
Lutreola, 84
lutreola, Mustela, 170
[463] Lyncodon, 407
macrodon, Mustela, 82
macrophonius,
Mustela, 78, 360
Putorius, 78, 360
macrura, Mustela, 75, 387, 393, 398, 402
macrurus, Putorius, 379
maniculatus, Peromyscus, 175
Martinogale, 11
meadow mice, 92, 208, 209, 216
Mearns cottontail, 208
melampelus, Mustela, 82
melodia, Melospiza, 213
Melospiza melodia, 213
meridana, Mustela, 78, 379
Mexican pocket gopher, 207
mexicanus, Putorius, 341
mice, meadow, 92, 208, 209, 216
microtis,
Mustela, 110
Putorius, 77, 110
Microtus, 65, 91, 92, 174, 179, 181, 208, 209, 210, 216, 220
drummondi, 175
minor, 175
montanus, 209
ochrogaster, 179
pennsylvanicus, 175
minimus, Tamias, 206
mink, Mustela, 82
minor,
Lutra, 170
Mustela, 169
minor, Zapus, 210
minuta,
Mustela, 169
Palaeogale, 169
minutus, Putorius, 169
Miomustela, 11
mole, 210
moles, 216
montanus, Microtus, 209
mortigena, Mustela, 110
mouse,
deer, 208, 209, 216
grasshopper, 207, 216
harvest, 208, 216
house, 208, 216
jumping, 210
meadow, 208, 209, 216
red-backed, 92
munda, Mustela, 77, 304, 309
mundus, Putorius, 77, 309
murica, Mustela, 161
muricus,
Mustela, 77, 161
Putorius, 77, 161
Mus, 216
muskrat, 216
Mustela, 83
aequatorialis, 75
aestuarina, 82
affinis, 75, 375, 379, 384, 398, 409
africana, 73, 406, 409
agilis, 74, 393
alascensis, 75, 131
allegheniensis, 77, 187
alleni, 76, 274
altifrontalis, 79, 300
americana, 74, 75
anguinae, 79, 145
angustidens, 78, 165
arctica, 76, 96
arizonensis, 75, 276, 280, 291, 323
arthuri, 78, 241
audax, 77
aureoventris, 74, 387, 398
bangsi, 80, 124
boccamela, 170
boliviensis, 79, 402
brasiliensis, 73, 341, 372, 375
campestris, 78, 190
celenda, 80, 139
cicognanii, 73, 118, 222
cigognanii, 118
costaricensis, 78, 372, 379, 387
culbertsoni, 75
digna, 100
effera, 79, 291
energumenos, 82
erminea, 72, 87, 103
eskimo, 77, 181
evagor, 82
evergladensis, 82
fallenda, 80, 148
furo, 43
frenata, 73, 193, 232, 252, 280, 309, 338, 341
fusca, 224
goldmani, 76, 355
gracilis, 78, 404
gulosa, 80, 159
haidarum, 76, 142
helleri, 79, 391
ingens, 82
initis, 80, 136
invicta, 80, 128
inyoensis, 79, 331
javonica, 72
jelskii, 75, 398
kadiacensis, 76, 108
kaneii, 99
labiata, 79, 105
lacustris, 82
latirostra, 79, 323
lepta, 161
leptus, 161
letifera, 82
leucoparia, 76, 347
longicauda, 73, 232, 252, 262
lutensis, 82
lutreola, 170
macrodon, 82
[464] macrophonius, 78, 360
macrura, 75, 387, 393, 398, 402
melampelus, 82
meridana, 78, 379
microtus, 77, 110
mink, 82
minor, 169
minuta, 169
munda, 77, 304, 309
murica, 161
muricus, 77, 161
neomexicana, 76, 333
neomexicanus, 333
nesolestes, 82
nevadensis, 79, 280
nicaraguae, 78, 370
nigriauris, 79, 319
nigripes, 74
nivalis, 72
notius, 77
noveboracensis, 74, 222, 252
numidica, 170
occisor, 77, 230
olivacea, 78, 244
olympica, 80, 153
oregonensis, 76, 304
oribasa, 270
oribasus, 77, 270
orientalis, 100
panamensis, 78, 375
paraensis, 76, 409
peninsulae, 75, 250
perda, 77, 366
perotae, 79, 351
polaris, 77, 103
primulina, 78, 232
pulchra, 79, 328
pusilla, 74, 118
putorius, 43
richardsonii, 73, 110
rixosa, 76, 153, 155, 161, 168, 184
salva, 80, 135
saturata, 76, 297
seclusa, 80, 141
semplei, 78, 105
spadix, 76, 252
stolzmanni, 75, 409, 413
streatori, 76, 155
texensis, 79, 338
tropicalis, 76, 363, 367
vison, 82
vulgivaga, 82
washingtoni, 76, 294
xanthogenys, 74, 315, 331
neomexicana, Mustela, 76, 333
neomexicanus,
Mustela, 333
Putorius, 76, 333
Neotamias, 206
Neotoma, 208, 216
albigula, 208
floridana, 208
fuscipes, 208
lepida, 208
nesolestes, Mustela, 82
nevadensis, Mustela, 79, 280
nicaraguae, Mustela, 78, 370
nigriauris, Mustela, 79, 319
nigripes,
Mustela, 74
Putorius, 74
nivalis, Mustela, 72, 169
northern pocket gopher, 206
notius, Putorius, 77, 222
noveboracensis,
Mustela, 74, 222, 252
Putorius, 74, 222
numidica, Mustela, 170
numidicus, Putorius, 170
occisor,
Mustela, 77, 230
Putorius, 77
Ochotona, 92
ochrogaster, Microtus, 179
olivacea, Mustela, 78, 244
olympica, Mustela, 80, 153
Ondatra, 210, 216
Onychomys, 207, 216
oregonensis,
Mustela, 304
Putorius, 304
oribasa, Mustela, 270
oribasus,
Mustela, 77, 270
Putorius, 77
Orthogeomys, 65
owl,
barn, 173
great-horned, 173
snowy, 90
Palaeogale, 11
minuta, 169
panamensis, Mustela, 78, 375
paraensis,
Mustela, 76, 409
Putorius, 76, 409
peninsulae,
Mustela, 75, 250
Putorius, 75, 250
pennsylvanicus, Microtus, 175
perda, Mustela, 77, 366
perdus, Putorius, 77, 366
Peromyscus, 91, 92, 173, 196, 208, 209, 210, 216
maniculatus, 175
perotae, Mustela, 79, 351
pheasant, 209
ring-necked, 202
pigmy weasel, 216
pika, 92
pikas, 201
[465] pipiens, Rana, 93
Pisces, 93
Pituophis sayi sayi, 213
Pliogale, 11
pocket gopher, 209, 216
Mexican, 207
northern, 206
Shaw, 208
polaris,
Mustela, 77, 103
Putorius, 77
polecats, 43
porcupine, quills of, 200
primulina, Mustela, 78, 233
pulchra, Mustela, 79, 323
pusilla, Mustela, 74, 118
pusillus, Putorius, 118, 155, 184, 190
Putorius, 76, 294, 300
aequatorialis, 75, 341, 387
affinis, 372, 375, 379, 384
agilis, 74, 222
alascensis, 75, 96, 131
allegheniensis, 77, 187
alleni, 76, 274
angustidens, 76, 165
arcticus, 76, 96
arizonensis, 75, 276, 280
audax, 77, 96
brasiliensis, 280, 300, 315, 319, 323, 384, 387, 398, 409
cicognanii, 96, 110, 118, 124, 128, 145, 155, 161
culbertsoni, 75
erminea, 96, 110, 222
eskimo, 77, 181
frenatus, 280, 300, 315, 319, 323, 341, 347, 351, 363, 366, 372, 384, 398
fuscus, 74, 222
goldmani, 76, 355
gracilis, 78, 404
haidarum, 76, 142
imperii, 110
kadiacensis, 76, 96, 108
kaneii, 100
leptus, 78, 161
leucoparia, 76, 347
longicauda, 262, 270, 280
macrophonius, 78, 360
macrotis, 77
macrurus, 370
mexicanus, 341
microtus, 100
minutus, 169
mundus, 77, 309
muricus, 77, 161
neomexicanus, 76, 333
nigripes, 74
notius, 77, 222
noveboracensis, 74, 222, 232
numidicus, 170
occisor, 77, 230
oregonensis, 304
oribasus, 77, 270
paraensis, 76, 409
peninsulae, 75, 250
perdus, 77, 366
polaris, 77, 103
pusillus, 118, 155, 184, 190
richardsonii, 96, 110
rixosus, 76, 181, 184
saturatus, 76, 294, 300
spadix, 76, 252
stolzmanni, 145, 148, 155, 159
streatori, 76
tropicalis, 76, 363, 370
vulgaris, 118, 155, 181, 184
washingtoni, 76, 294
xanthogenys, 315, 319, 323, 331
putorius, Mustela, 43
quail, 209, 213
rabbit,
brush, 213
snowshoe, 93, 201
racer, red, 213
Rana pipiens, 93
rat,
brown, 202
cotton, 208, 216
wood, 208
Rattus, 91, 92, 210, 216
rattlesnake, 200
red-backed mouse, 92
red
racer, 213
squirrel, 206
Reithrodontomys, 175, 216
reptiles, 213
Richardson ground squirrel, 205
richardsonii,
Citellus, 205
Mustela, 73, 110
Putorius, 222
ring-necked pheasant, 202
rixosa, Mustela, 76, 153, 155, 161, 168, 184
rixosus, Putorius, 76, 181, 184
Rodentia, 216
russet-backed thrush, 204
salva, Mustela, 80, 135
saturata, Mustela, 76, 297
sayi, Pituophis, 213
Scalopus, 209
Scapanus latimanus, 205
Schönthierlein, 7
Sciurus, 210
seclusa, Mustela, 80, 141
[466] semplei, Mustela, 78, 105
short-tailed shrew, 208, 209
short-tailed weasel, 87
shrews, 216
Sigmodon, 216
Sigmodon hispidus, 208
slate-colored junco, 213
snake,
bull, 213
king, 213
snakes, 216
snowshoe rabbit, 93, 210
snowy owl, 90
song sparrow, 201, 213
Sorex, 91, 209
cinereus, 205
Soricidae, 216
spadix,
Mustela, 76, 252
Putorius, 76, 252
sparrow,
song, 201, 213
tree, 208, 209
spermophile, Uinta, 200
squirrel,
flying, 206
red, 206
tree, 216
stolzmanni, Mustela, 75, 409, 413
streatori,
Mustela, 76, 155
Putorius, 76, 145, 148, 155, 159
striatus, Tamias, 206
Sylvilagus, 91, 93, 209, 210, 216
bachmani, 213
Talpidae, 216
talpoides, Thomomys, 207
Tamias, 91, 92, 206, 210, 216
minimus, 206
striatus, 206
Tamiasciurus, 206, 216
texensis, Mustela, 79, 338
thirteen-lined ground squirrel, 205
Thomomys, 206, 219
bottae, 207
talpoides, 207
thrush, russet-backed, 204
towhee, 201
Townsend ground squirrel, 205
townsendii, Citellus, 205
tree sparrow, 208, 209
tridecemlineatus, Citellus, 205
tropical weasel, 406
tropicalis,
Mustela, 76, 363, 367
Putorius, 76, 363, 370
Uinta spermophile, 200
varying hare, 210, 216
vison,
Mustela, 82
Lutreola, 84
vulgaris,
Mustela, 73
Putorius, 118, 155, 181, 184
vulgivaga, Mustela, 82
Vulpes fulvus, 200
washingtoni,
Mustela, 76, 294
Putorius, 76, 294
weasel,
least, 168, 209
long-tailed, 193
Pigmy, 216
short-tailed, 87
tropical, 406
wild birds, 93, 213
wood rat, 208, 210, 216, 219
xanthogenys,
Mustela, 74, 315, 331
Putorius, 315, 319, 323, 331
Zapus, 216
minor, 210
FOOTNOTES:
[1] Acad. Nat. Sciences of Philadelphia
[2] American Mus. Nat. History
[3] Baylor University
[4] Berlin Zoological Museum
[5] Boston Society of Natural History
[6] Brigham Young University
[7] British Museum of Natural History
[8] California Academy of Sciences
[9] Carnegie Museum
[10] Charles R. Conner Museum
[11] Charleston Museum
[12] Coe College
[13] Collection of A. H. Miller
[14] Collection of Alex Walker
[15] Collection of Arthur Peake
[16] Collection of D. D. Stone
[17] Collection of Donald V. Hemphill
[18] Collection of E. J. Koestner
[19] Collection of Edward R. Warren
[20] Collection of Frank Stephens
[21] Collection of Ian McTaggart-Cowan
[22] Collection of J. A. Munro
[23] Collection of J. Arnold
[24] Collection of J. E. Law
[25] Collection of J. M. Edson
[26] Collection of Jack C vonBloeker
[27] Collection of Joe and Dean Thiriot
[28] Collection of John Cushing
[29] Collection of John Fitzgerald, Jr.
[30] Collection of John Tyler
[31] Collection of Kenneth Racey
[32] Collection of L. M. Huey
[33] Collection of Lloye H. Miller
[34] Collection of Mr. Green
[35] Collection of Myron H. Swenk
[36] Collection of O. J. Murie
[37] Collection of O. P. Silliman
[38] Collection of R. D. Moore
[39] Collection of R. H. Coleman
[40] Collection of R. W. Jackson
[41] Collection of Ralph Ellis
[42] Collection of Robert T. Orr
[43] Collection of Rollin H. Baker
[44] Collection of Ross Hardy
[45] Collection of Stanley C. Arthur
[46] Collection of Stanley G. Jewett
[47] Collection of Stuart Criddle
[48] Collection of T. C. Stephens
[49] Collection of Victor B. Scheffer
[50] Collection of W. E. Snyder
[51] Collection of Walter W. Dalquest
[52] Collection of William B. Davis
[53] Collection of William B. Richardson
[54] Collection of William Bebb
[55] Collection of William T. Shaw
[56] Collection Rocky Mt. Spotted Fever Lab.
[57] Colorado Museum of Natural History
[58] Cornell University
[59] Donald R. Dickey Collection
[60] Field Museum of Natural History
[61] Florida State Museum
[62] Fresno State Junior College
[63] Humboldt State Teachers College
[64] Illinois Natural History Survey
[65] Iowa State College
[66] Iowa Wesleyan College
[67] Kansas State Agric. College
[68] Leland Stanford Junior University
[70] Los Angeles Mus. Hist. Art and Sci.
[71] Louisiana State University
[72] Mt. Rainier Nat'l Park Collection
[73] Mus. Polonais d'Hist. Nat., Warsaw
[74] Mus. Vert. Zoöl., Univ. California
[75] Museum of Comparative Zoölogy
[76] Museum of Zoölogy, Univ. Michigan
[77] National Museum of Canada
[78] Naturhistoriska Ricksmuseum, Sweden
[80] New York State Museum
[81] Ohio State Museum
[82] Oklahoma Agric. and Mech. College
[83] Ottawa University, Kansas
[84] Paris Museum
[85] Provincial Museum of British Columbia
[86] Royal Ontario Museum of Zoölogy
[87] San Diego Society of Natural History
[88] State Hist. and Nat. Hist. Soc. Colo.
[89] State Normal School, Cheney, Wash.
[90] Texas Cooperative Research Collection
[91] United States National Museum
[92] Univ. California Mus. Palaeo.
[93] Univ. Kansas Mus. Nat. History
[94] Univ. Washington Museum of Zoölogy
[95] Univ. Zool. Mus., Copenhagen
[96] University of Arkansas
[97] University of Idaho
[98] University of Minnesota
[99] University of Notre Dame
[100] University of Oklahoma
[101] University of Oregon
[102] University of South Dakota
[103] University of Utah
[104] University of Wisconsin
