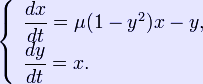In the mathematical theory of bifurcations, a Hopf or Poincaré–Andronov–Hopf bifurcation, named after Henri Poincaré, Eberhard Hopf, and Aleksandr Andronov, is a local bifurcation in which a fixed point of a dynamical system loses stability as a pair of complex conjugate eigenvalues of the linearization around the fixed point cross the imaginary axis of the complex plane. Under reasonably generic assumptions about the dynamical system, we can expect to see a small-amplitude limit cycle branching from the fixed point. For a more general survey on Hopf bifurcation and dynamical systems in general, see [1][2][3][4][5].
Supercritical / subcritical Hopf bifurcations The limit cycle is orbitally stable if a certain quantity called the first Lyapunov coefficient is negative, and the bifurcation is supercritical. Otherwise it is unstable and the bifurcation is subcritical. The normal form of a Hopf bifurcation is: where z, b are both complex and λ is a parameter. Write
The number α is called the first Lyapunov coefficient. * If α is negative then there is a stable limit cycle for λ > 0: where The bifurcation is then called supercritical. * If α is positive then there is an unstable limit cycle for λ < 0. The bifurcation is called subcritical.
The "smallest chemical reaction with Hopf bifurcation" was found 1995 in Berlin, Germany[6]. The same biochemical system has been used in order to investigate how the existence of a Hopf bifurcation influences our ability to reverse-engineer dynamical systems[7]. Under some general hypothesis, in the neighborhood of a Hopf bifurcation, a stable steady point of the system gives birth to a small stable limit cycle. Remark that looking for Hopf bifurcation is not equivalent to looking for stable limit cycles. First, some Hopf bifurcations (e.g. subcritical ones) do not imply the existence of stable limit cycles; second, there may exist limit cycles not related to Hopf bifurcations. Example
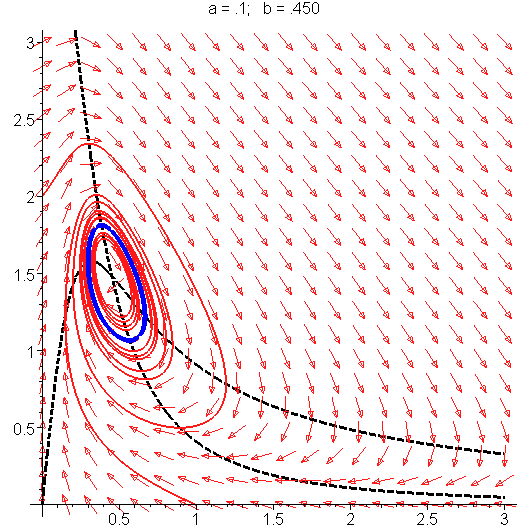
The Hopf bifurcation in the Selkov system (see article). As the parameters change, a limit cycle (in blue) appears out of an unstable equilibrium. Hopf bifurcations occur in the Hodgkin–Huxley model for nerve membrane, the Selkov model of glycolysis, the Belousov–Zhabotinsky reaction, the Lorenz attractor and in the following simpler chemical system called the Brusselator as the parameter B changes:
The Selkov model is
The phase portrait illustrating the Hopf bifurcation in the Selkov model shown above. See [1], page 205 for detailed derivation. Definition of a Hopf bifurcation The appearance or the disappearance of a periodic orbit through a local change in the stability properties of a steady point is known as the Hopf bifurcation. The following theorem works with steady points with one conjugate nonzero purely imaginary eigenvalues. It tells the conditions under which this bifurcation phenomena occurs. Theorem (see section 11.2 of [3]). Let J0 be the Jacobian of a continuous parametric dynamical system evaluated at a steady point Ze of it. Suppose that all eigenvalues of J0 have negative real parts except one conjugate nonzero purely imaginary pair Routh–Hurwitz criterion Routh–Hurwitz criterion (section I.13 of [5]) gives necessary conditions so that a Hopf bifurcation occurs. Let us see how one can use concretely this idea[8]. Sturm series Let
The coefficients ci,0 for i in Propositions Proposition 1. If all the Hurwitz determinants ci,0 are positive, apart perhaps ck,0 then the associated Jacobian has no pure imaginary eigenvalues. Proposition 2. If all Hurwitz determinants ci,0 (for all i in The conditions that we are looking for so that a Hopf bifurcation occurs (see theorem above) for a parametric continuous dynamical system are given by this last proposition. Example Let us consider the classical Van der Pol oscillator written with ordinary differential equations: The Jacobian matrix associated to this system follows: The characteristic polynomial (in λ) is equal to: P = λ2 − μλ + 1. The above proposition 2 tells that one must have: c0,0 = 1 > 0,c1,0 = − μ = 0,c0,1 = − 1 < 0. Because 1 > 0 and −1 < 0 are obvious, one can conclude that a Hopf bifurcation may occur for Van der Pol oscillator if μ = 0. References 1. ^ a b Strogatz, Steven H. (1994). Nonlinear Dynamics and Chaos. Addison Wesley publishing company.
Retrieved from "http://en.wikipedia.org/" |
|