.
Xanthine
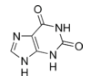
Xanthine (pronounced /ˈzænθiːn/ or /ˈzænθaɪn/), (3,7-dihydro-purine-2,6-dione), is a purine base found in most human body tissues and fluids and in other organisms. A number of mild stimulants are derived from xanthine, including caffeine and theobromine.[1]
Xanthine is a product on the pathway of purine degradation.
* It is created from guanine by guanine deaminase.
* It is created from hypoxanthine by xanthine oxidoreductase.
Xanthine is subsequently converted to uric acid by the action of the xanthine oxidase enzyme.
Pathology
People with the rare genetic disorder xanthinuria lack sufficient xanthine oxidase and cannot convert xanthine to uric acid.
Clinical significance of xanthine derivatives
Derivatives of xanthine, known collectively as xanthines, are a group of alkaloids commonly used for their effects as mild stimulants and as bronchodilators, notably in treating the symptoms of asthma. In contrast to other, more potent stimulants, they only inhibit the actions of sleepiness-inducing adenosine, making them somewhat less effective as stimulants than sympathomimetic amines. Due to widespread effects, the therapeutic range of xanthines is narrow, making them merely a second-line asthma treatment. The therapeutic level is 10-20 micrograms/mL blood; signs of toxicity include tremor, nausea, nervousness, and tachycardia/arrhythmia.
Methylxanthines affect not only the airways but stimulates heart rate, force of contraction, cardiac arrhythmias at high concentrations. In the CNS they increase alertness, stimulate the respiratory centre, and are used for treatment of infantile apnea. In high doses they can lead to convulsions that are resistant to anticonvulsants. Methylxanthines induce acid and pepsin secretions in the GI tract. Methylxanthines are metabolized by Cytochrome P450 in the liver. Methylated xanthines include caffeine, aminophylline, IBMX, paraxanthine, pentoxifylline [2], theobromine, and theophylline. These drugs act as both
1. competitive nonselective phosphodiesterase inhibitors [3] which raise intracellular cAMP, activate PKA, inhibit TNF-alpha [2] [4] and leukotriene [5] synthesis, and reduce inflammation and innate immunity [5] and
2. nonselective adenosine receptor antagonists [6] which inhibit sleepiness-inducing adenosine.
But different analogues show varying potency at the numerous subtypes, and a wide range of synthetic xanthines (some nonmethylated) have been developed searching for compounds with greater selectivity for phosphodiesterase enzyme or adenosine receptor subtypes.[7][8][9][10][11][12][13][14][15][16][17][18][19] Xanthines are also found very rarely as constituents of nucleic acids.
Caffeine: R1 = R2 = R3 = CH3
Theobromine: R1 = H, R2 = R3 = CH3
Theophylline: R1 = R2 = CH3, R3 = H
Selected Xanthines Name↓ R1↓ R2↓ R3↓ IUPAC nomenclature↓ Found In↓
Caffeine CH3 CH3 CH3 1,3,7-trimethyl-1H-purine-2,6(3H,7H)-dione Coffee, Guarana, Yerba mate, Tea, Kola
Theobromine H CH3 CH3 3,7-dihydro-3,7-dimethyl-1H-purine-2,6-dione Chocolate, Yerba mate
Theophylline CH3 CH3 H 1,3-dimethyl-7H-purine-2,6-dione Tea, Yerba mate
Xanthine H H H 3,7-dihydro-purine-2,6-dione plants, animals
See also
* Murchison meteorite
* Xanthene
* Xanthydrol
* Xanthone
References
1. ^ Spiller, Gene A. (1998). Caffeine. Boca Raton: CRC Press. ISBN 0-8493-2647-8.
2. ^ a b Deree J, Martins JO, Melbostad H, Loomis WH, Coimbra R. (2008). "Insights into the regulation of TNF-alpha production in human mononuclear cells: the effects of non-specific phosphodiesterase inhibition.". Clinics (Sao Paulo). 63 (3): 321–8. doi:10.1590/S1807-59322008000300006. PMID 18568240.
3. ^ Essayan DM. (2001). "Cyclic nucleotide phosphodiesterases.". J Allergy Clin Immunol. 108 (5): 671–80. doi:10.1067/mai.2001.119555. PMID 11692087.
4. ^ Marques LJ, Zheng L, Poulakis N, Guzman J, Costabel U (February 1999). "Pentoxifylline inhibits TNF-alpha production from human alveolar macrophages". Am. J. Respir. Crit. Care Med. 159 (2): 508–11. PMID 9927365. http://ajrccm.atsjournals.org/cgi/pmidlookup?view=long&pmid=9927365.
5. ^ a b Peters-Golden M, Canetti C, Mancuso P, Coffey MJ. (2005). "Leukotrienes: underappreciated mediators of innate immune responses.". J Immunol. 174 (2): 589–94. PMID 15634873. http://www.jimmunol.org/cgi/content/full/174/2/589.
6. ^ Daly JW, Jacobson KA, Ukena D. (1987). "Adenosine receptors: development of selective agonists and antagonists.". Prog Clin Biol Res. 230 (1): :41–63. PMID 3588607.
7. ^ MacCorquodale DW. THE SYNTHESIS OF SOME ALKYLXANTHINES. Journal of the American Chemical Society. 1929 July;51(7):2245–2251. DOI: 10.1021/ja01382a042
8. ^ WO patent 1985002540, Sunshine A, Laska EM, Siegel CE, "ANALGESIC AND ANTI-INFLAMMATORY COMPOSITIONS COMPRISING XANTHINES AND METHODS OF USING SAME", granted 1989-03-22 , assigned to RICHARDSON-VICKS, INC.
9. ^ Constantin Koulbanis, Claude Bouillon, Patrick Darmenton,"Cosmetic compositions having a slimming action", US patent 4288433, granted 1981-09-04 , assigned to L'Oreal
10. ^ Daly JW, Padgett WL, Shamim MT (July 1986). "Analogues of caffeine and theophylline: effect of structural alterations on affinity at adenosine receptors". Journal of Medicinal Chemistry 29 (7): 1305–8. doi:10.1021/jm00157a035. PMID 3806581.
11. ^ Daly JW, Jacobson KA, Ukena D (1987). "Adenosine receptors: development of selective agonists and antagonists". Progress in Clinical and Biological Research 230: 41–63. PMID 3588607.
12. ^ Choi OH, Shamim MT, Padgett WL, Daly JW (1988). "Caffeine and theophylline analogues: correlation of behavioral effects with activity as adenosine receptor antagonists and as phosphodiesterase inhibitors". Life Sciences 43 (5): 387–98. doi:10.1016/0024-3205(88)90517-6. PMID 2456442.
13. ^ Shamim MT, Ukena D, Padgett WL, Daly JW (June 1989). "Effects of 8-phenyl and 8-cycloalkyl substituents on the activity of mono-, di-, and trisubstituted alkylxanthines with substitution at the 1-, 3-, and 7-positions". Journal of Medicinal Chemistry 32 (6): 1231–7. doi:10.1021/jm00126a014. PMID 2724296.
14. ^ Daly JW, Hide I, Müller CE, Shamim M (1991). "Caffeine analogs: structure-activity relationships at adenosine receptors". Pharmacology 42 (6): 309–21. doi:10.1159/000138813. PMID 1658821.
15. ^ Ukena D, Schudt C, Sybrecht GW (February 1993). "Adenosine receptor-blocking xanthines as inhibitors of phosphodiesterase isozymes". Biochemical Pharmacology 45 (4): 847–51. doi:10.1016/0006-2952(93)90168-V. PMID 7680859. http://linkinghub.elsevier.com/retrieve/pii/0006-2952(93)90168-V.
16. ^ Daly JW (July 2000). "Alkylxanthines as research tools". Journal of the Autonomic Nervous System 81 (1-3): 44–52. doi:10.1016/S0165-1838(00)00110-7. PMID 10869699. http://linkinghub.elsevier.com/retrieve/pii/S0165183800001107.
17. ^ Daly JW (August 2007). "Caffeine analogs: biomedical impact". Cellular and Molecular Life Sciences : CMLS 64 (16): 2153–69. doi:10.1007/s00018-007-7051-9. PMID 17514358.
18. ^ González MP, Terán C, Teijeira M (May 2008). "Search for new antagonist ligands for adenosine receptors from QSAR point of view. How close are we?". Medicinal Research Reviews 28 (3): 329–71. doi:10.1002/med.20108. PMID 17668454.
19. ^ Baraldi PG, Tabrizi MA, Gessi S, Borea PA (January 2008). "Adenosine receptor antagonists: translating medicinal chemistry and pharmacology into clinical utility". Chemical Reviews 108 (1): 238–63. doi:10.1021/cr0682195. PMID 18181659.
Retrieved from "http://en.wikipedia.org/"
All text is available under the terms of the GNU Free Documentation License


