In molecular biology, DNA ligase is a special type of ligase, which is basically an enzyme (EC 6.5.1.1) that in the cell repairs single-stranded discontinuities in double stranded DNA molecules, in simple words strands that have double-strand break (a break in both complementary strands of DNA). Purified DNA ligase is used in gene cloning to join DNA molecules together. The alternative, a single-strand break, is fixed by a different type of DNA ligase using the complementary strand as a template but still requires DNA ligase to create the final phosphodiester bond to fully repair the DNA. DNA ligase has applications in both DNA repair and DNA replication (see Mammalian ligases). In addition, DNA ligase has extensive use in molecular biology laboratories for Genetic recombination experiments (see Applications in molecular biology research).
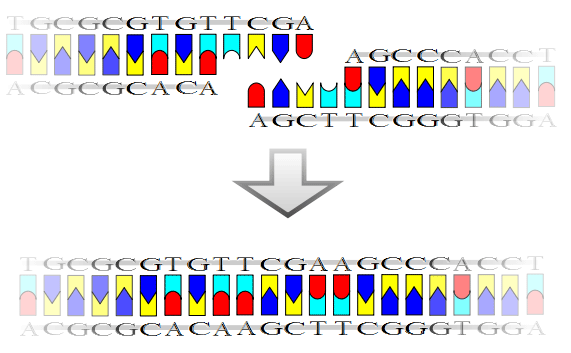
The mechanism of DNA ligase is to form two covalent phosphodiester bonds between 3' hydroxyl ends of one nucleotide with the 5' phosphate end of another. ATP is required for the ligase reaction. A pictorial example of how a ligase works (with sticky ends):
DNA ligation with a TCGA (Taq I) sticky end. The sequence is taken from human hemoglobin alpha subunit gene. (*) Ligase will also work with blunt ends, although higher enzyme concentrations and different reaction conditions are required. Mammalian ligases In mammals, there are four specific types of ligase. * DNA ligase I: ligates the nascent DNA of the lagging strand after the DNA polymerase I has removed the RNA primer from the Okazaki fragments. Some forms of DNA ligase present in bacteria (usually larger) may require NAD to act as a co-factor, whereas other forms of DNA ligases (usually present in E.coli, and usually smaller) may require ATP to react. Also, a number of other structures present in the DNA ligase are the AMP and lysine, both of which are important in the ligation process since they create an intermediate enzyme. Applications in molecular biology research DNA ligases have become an indispensable tool in modern molecular biology research for generating recombinant DNA sequences. For example, DNA ligases are used with restriction enzymes to insert DNA fragments, often genes, into plasmids. One vital, and often tricky, aspect to performing successful recombination experiments involving the ligation of cohesive-ended fragments is controlling the optimal temperature. Most experiments use T4 DNA Ligase (isolated from bacteriophage T4), which is most active at 25°C. However, in order to perform successful ligations with cohesive-ended fragments ("sticky ends"), the optimal enzyme temperature needs to be balanced with the melting temperature Tm (also the annealing temperature) of the DNA fragments being ligated.[1] If the ambient temperature exceeds Tm, homologous pairing of the sticky ends will not occur because the high temperature disrupts hydrogen bonding. The shorter the DNA fragments the lower the Tm. Since blunt-ended DNA fragments have no cohesive ends to anneal, controlling the optimal temperature becomes much less important. The most efficient ligation temperature will be the temperature at which T4 DNA ligase functions optimally (T4 DNA ligase is the only commercially-available DNA ligase to anneal blunt ends).[1] Therefore, the majority of blunt-ended ligations are carried out at 20-25°C. The common commercially available DNA ligases were originally discovered in bacteriophage T4, E. coli and other bacteria. History The first DNA ligase was purified and characterized in 1967.[2] See also * DNA end
1. ^ a b Tabor, Stanley. DNA ligases. Chapter in: Current Protocols in Molecular Biology, Book 1. 2001: Wiley Interscience.
* DNA Ligase: PDB molecule of the month Retrieved from "http://en.wikipedia.org/"
|